Vol6, No 1, January - March 1997 Progestogen-AndrogenCombination
l5
Progestogen-Androgen
Combination:
Results of Indonesian
Clinical Trials
Kiagus Muhammad Arsyad
Abstrak
Makalah ini menyaikan hasil dua penelitian klinis WHo yang dilaksanakan di Indonesia dalam upaya mendapatkan kombinasi Pro9estoïen-androgen yang cocok sebagai kontrasepsi pia dan mengidenrifi,kasi masa!,ah untuk penelitian dan pengimbangan di masa datang. Penelitian pertama menggunakan kombinasi DMPA danTE yang dibeikan secara injelcsi intramuskular à"ngon interval satu
bulan
250sentra
u ko250
m
TEmingg
24. Hasilyang menggembirakanyang didapat dari dua penelitian ini memberikan harapan untuk mengembangkan suatu kontrasepsi yang arnan, efektif dai cocok untuk'prià Ind.onesia. Walaipun masih diperlukan penelitian dan pengembangan untuk mendapatkan hormon yang dapat diberikan lebih pàktis dan degan dosis yang lebih rendah.Abstract
This article summarizes the results of
two
recent WHO supported clinical studiesin
Indonesiato
assess the potential of progestogen-androgen combinations as adevelopment, TheJirst study was carried ou and at two dose levels: 100 mg DMPA ptus is carried out in Indonesia, in which male v
I9-NT. In both cases, 250 mg DMPA was injected IM at week 0, 6, the first seven weekls and then at 3-weekly intervals until week 24.
g a safe and effective hormonal contracep gh levels of hormones that needed to be i c urre n t ly av ai lab le c o mp o unds.
Keywords: Male hormonal contraceptives, progesrogen-testosterone regimen, Indonesian men.
Compared
with
the extensive resezrch that has been carriedout on
methodsfor
women, the amount of attention that has been givento
the developmentof
contraceptive methods for men is inadequate. The con_ dom and coitus interruptus are theonly
reversible methodsof
contraception currently available to men. Bothare
on the initiative of the couple andoften
edical advice. There is clearly a needfo
ent in the number and type ofcontrac
n
orderto
expandfamily
duals and couples.The
In
n
Population andDevelo
EgYPt,called
ongovernments to "increase the participation and sharing
Department of Medical Biology riw ijaya U nivers ity, p a le mb ang,
of responsibility of men in the actual practice of family planning."r Research
in
thisfield ihould
be mulri-faceted with the goal of developing a range of fertility regulation methods to meet the needsof
menof
dif-ferent social, cultural, and ethnic backgrounds.
Male fertility has been described as being regulated by
three main facrors:2
(i)
the
hormonai- deierminanr which regulates steroidogenesis and spermatogenesis;(ii)
the male accessory glands and organswhich
are under strict hormonal control and which are respon_ sible for the producrion of the seminal plasma;(iii)
the psychogenic determinant which controls masculine behavior and male Iibido.The hormonal
suppressionof spermatogenesis
t6 Arsyad
of various hormonal contraceptive agents for men have been supported
by
national and internationalagen-cies.3'4
ih"
ug"ntt that have been tested in such trialsinclude:
andiogens alone or in combination withprogestogens; danazol;
GnRH
agonists and anta-gonists,ind
the data obtainedin
these studies have Ë"en reuiewed recently.5'6'7The main advantages of contraception that have that they involve the use
duced and; (v) the relatively high cost ofthese reagents (e.g. GnRH antagonist).
An
ideal male
contraceptive methodwould be
a preparation that would not inhibit masculine behaviour ànOmut" libido,
but
would completelyinhibit
the productionof spermatozoa
-
or inhibit the fertilizing lapucityof
speimatozoa-
for the desired period ofcontraception.
It
is with this goal in mind, that the WHO Task Force on Methodsfor
the Regulationof
Male Fertility has been conducting researches to develop safe, effective, reversible and acceptable methods offertility
regula-tion for men since l972.Theneed for research on male methodsof
contraception andfor
a greaterinvolve-ment
of
men
in
reproductive health
have been promotedby many national and international agencies' women's health groups and governments, including that of Indonesia.'This article summarizes the results of two WHO sup-ported clinical trials that have been carried out recently
in
Indonesiato
assess the potentialof
combined progestogen-androgen injections as an approach to the regulationof
malefertility.
Both studies were phar-macokinetic/pharmacodynamic studiesto
assess the effect of the treatment on the subjects'spermatogenesrs and biochemistry; they did not involve assessmentof
the contraceptive effect of the treatment bydetermin-Med J Indones
ing the pregnacy rates
in
the partnersof
the treated mèn.In
addition, these studies identified some areas for future research and development.PROGESTOGEN ANDROGEN
COMBINATION
AND
TIIEIR
EFFECT ONMALE FERTILITY
Depot
Medroxy
ProgesteroneAcetate
and
Tes-tosterone EnanthateIn the first study done by Pangkahila,8 a combination acetate
as given
ls and at
ived 100 mg DMPAplus 100 mg TE and a second group of men 1nl t O1 received 200 mg DMPA plus 250 mg TE (Table 1).
Table
l.
The résumé of hormonal method clinical trials in IndonesiaNo. Variables Results of studies
Pangkahila
Multicentresl'
Hormones2.
Dosages (mg)3.
InterventionDMPA + TE
100 + 100 200 +250
IM-monthly 4 months (18 weeks) 4 2.5 months (20 weeks) IOOVo t00% 2.0 months
DMPA + TE DMPA + l9-NT 250 +200 250 + 200
TE or 19-NT: IM-weekly-7 weeksÂM + 3 weekly-to week24 DMPA: IM-6 weeklY-to week l8
24 weeks
20 weeks
95.6% 97.8Vo 6.5 months t4 4. 5 6. 7. (Suppression Time) Number of Injection Time to Achieving Azoospermia
7o Azoospermia
Recovery Time
A total of twenty men aged
fromZ'l
to 38 years were recruited.All
but one were married and had fathered at least onechild.
At
entry
into
study,their
anthro-pometric data, mean (SEM), were as follows: height
165.8 (1.0) cm, weight
e.2QJ)
kg, body surface area 1 .7 1 (0.003) m2, body mass index 23 -3(0.l)kglm2
and [image:2.595.298.545.325.651.2]Vol6, No I, January - March 1997
The side effects of treatment were considered minimal
and none
of
the subjects discontinued the study.All
men reported an increase in libido and sexual frequen-cy during the treatment periods. Five volunteers noteda
fall in
their
libido
1-2 months after cessationof
injections, but libido was restored and normal sexualfunction
returned
during
the
subsequent three months.Most of the subjects (19120) reporred weight gun (3.7
+
0.5kg,
range 0-9kg)
at the endof
the treatmentperiod, which remained above the pre-treatment level
BASELINE I
Progestogen-Androgen
Combination
I7at the end of the study (67.4 + 2.0 kg). Facial acne \ryas
noted
in
one man and another complainedof
non-specific weakness and sleepiness which did notinter-fere with his daily work.
The
proportionof
menwith
azoospermiain
both groups is illustrated in Figurel.
The rate and degreeof
reduction
in
sperm concentrationsin
the two groups were similar and the combined meansperm-concentra-tion
is
illustrated
in
Figure
2.
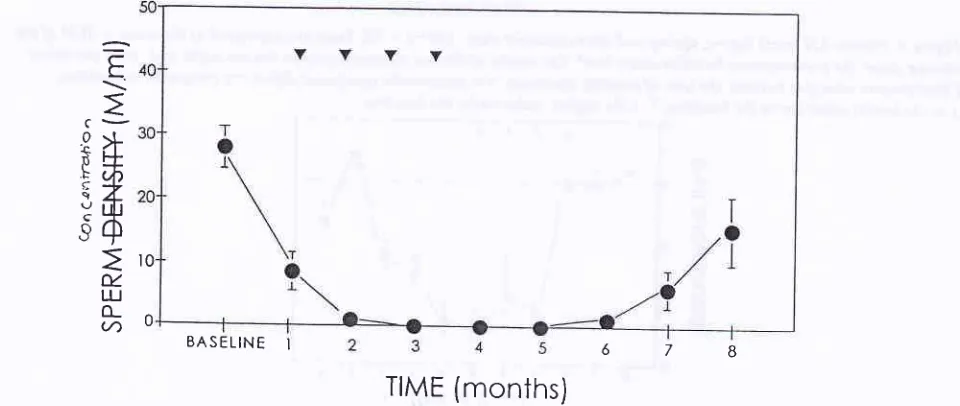
Azoospermia was achieved at a median time of 2.5 months from the startof injections and sperm reappeared in the ejaculate at a median time of 2 months after cessation of injections.
o_o
HtcHDosEo_ o
LowDosE 100\
\
8uo
u.
LU
A40
o
g20
\o
[image:3.595.59.556.168.440.2] [image:3.595.69.549.483.686.2]TIME
(months)
Figure 1. Proportion of azoospermic men during and after tfeatment with high dose (200 ng DMpA
+
250 mg TE,fiIIed circles) and low-dose ( 100 mg DMPA + 100 mg TE, open cicles). Filled inverse triingles indicate tie time of monthly
injection.
t0
0
E
\
c-8>l
Êfi
,<z
Yub
ô+
u.
LU o_
(/)
TIME
(months)
Figure 2. Mean sperm concentration before, during and after treatment with DMpA two treatment groups
did
notdffir
and are combined (mean=solid circres,t
sEM). monthly injections, M/ml = miltiott/mt.678
l8 Arsyad
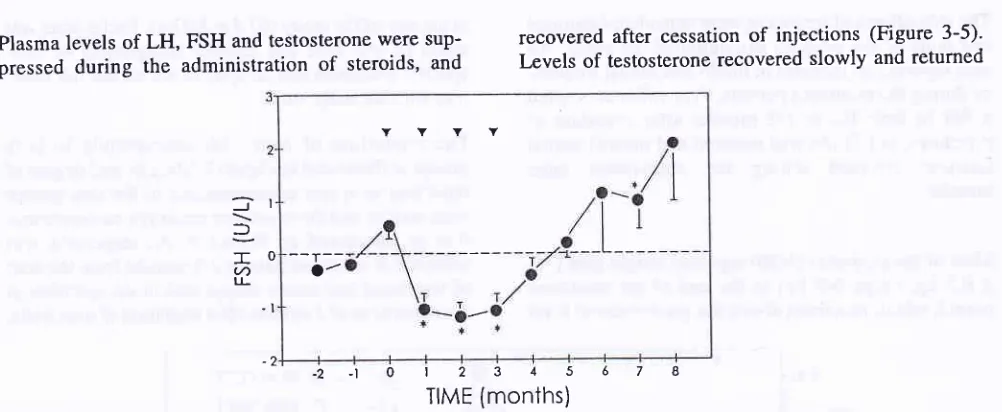
Plasma levels of LH, pressed
during
theFSH and testosterone were suP-administration
of
steroids, andMed J Indones
recovered after cessation
of
injections (Figure 3-5)' Levels of testosterone recovered slowly and returnedJ
=
-U) LL
I
0
-l
-2-1012345678
[image:4.595.58.559.74.280.2] [image:4.595.67.557.300.492.2] [image:4.595.88.556.492.688.2]TIME (monThs)
Figure 3.
PIas
,
ed as the mean + sEM of thechange
from
e
differ and were combined'Filled
inverse
t
compared to baseline'I
= the lowest value above the baseline,I
= the highest value under the baseline.VYVV
TIME (months)
Figure 4. Plasma-LH levels before, during and after treatmenl with DMPA + TE. Data are expressed as the mean + SEM of tlrc chànge from rhe pre-treattnent baseline-mean level. The resulrs of the two lrealment Sroups did not differ and were conbined-Filleâ itiverse triangles indicate the time of monthly injections, * = statistically signifrcant dffirence compared to baseline,
I
= rhe lowest value above the baseline, T = the highest value under the baseline.TIME (months)
Figure 5. Plas,no-testosterone levels before, during and after treatmenl with DMPA + TE. Data are expressed as the mean + SEM of tlri clnnge from the pre-treatntenl bascline-ntean level. The resuhs of rhe wo treatnrcnt Broups did not difrer and were
combined-Fillet! inverse triangles indicate tlrc time of monthll, injectiorrs, + -- statistically sigttificant difference compared lo baseline,
I
= the lowest value above the baseline, F = tlrc highcst value under the baseline'6
4
2
J
?
I .J
YYYY
/
--T
a
Vol6, No l, January - March 1997
to baseline levels at the fourth post-treatment month. The levels of LH and FSH exhibited a rebound
eleva-tion from three to five months after cessation of
injec-tions.
The treatment was
associatedwith
smallbut
sig-nificant
increasein
the
level
of
plasma glucose(<l 0 Vo), bi I irub i n (<l Vo) and total protein (< 5Vo) whil e albumin level showed a decrease (<5Vo) and returned to baseline by the end of the study. Plasma level of total
cholesterol was increased
by
less thanl\Vo
at
Thefourth
andfifth
post-treatment month and remainedlower than baseline values, at the end
of
the study. There were no significant changes in the level ofhigh-density lipoprotein cholesterol, serum
glutamate-oxa-late
transaminase,
serum
glutamate-pyruvatetransaminase or calcium.
Depot
Medroxy
ProgesteroneAcetate and
Tes-tosterone Enanthate
or
19-Nortestosterone
hexyloxyphenilpropionate
In the second study,g male volunteers received either a combination
of
DMPA plus TE or a combinationof
DMPA plus
l9-nortestosteronehexyloxyphenilpro-pionate (19-NT).
In
both casesDMpA
(250 mg) was injected at week0,6,
12, and 18, and the TE (200 mg)or 19-NT (200 mg) was given weekly for rhe first seven
weeks and then at three weekly intervals until week 24. Twenty healthy male volunteers from each
of
five Indonesian centrcs (Palembang, Jakarta, Semarang, Surabaya and Denpasar) were randomized using sealed envelopes to one of the two treatment groups.Progestogen-Androgen
Combination
19Suppression
of
sperm production was measured by semen analysis at three week intervals and clinical chemistry, hematology, and levelsof
gonadotrophin hormones were monitored at six week intervals. Therecovery was assessed monthly until normal sperm concentration (220
x
lOo/ml) was reached.A
totalof 96
men were recruited to the study. Their anthropometric data, mean (SD),on entry were
as follows: mean age was 34.7 (5.8) years, height 162.6(5.1) cm, weight 57.6 (7.6)kg, BodyMass Index 21.8
(2.8)kglm',
testicular volume 22.4 (5.3) ml.The baseline semen characteristics varied from center [o center. However, these factors were balanced across
the two treatment groups, with the exception of sperm
concentration, which was slightly higher
in
the TE group (P=0.05, two-wayANOVA
adjustedfor
be-tween center differences). The median sperm con-centrations (5th and 95th percentiles) were 46.3 (22.7, 123.2)
x
l0o/ml in allmen and40J (21.8, 109.4) and 55.7 (23.0,141.5) x 10"/ml in the l9-NT and TE groups respectively. The mean (SD) levels among all men forhemoglobin, white blood cells, and platelet count were
145 (12) gA,6.4 (1.3)
x
10efl, and 241 (47)x
l0e/1,respectively, and did not differ between the two treat-ment groups.
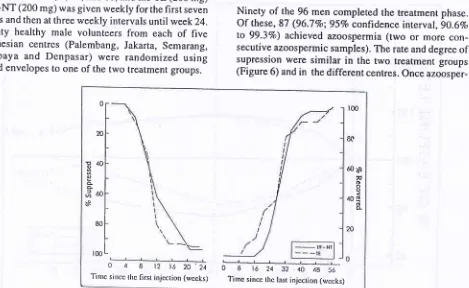
Ninety of the 96 men completed the treatment phase. Of these, 87 (96.7Vo;95Vo confidence inrerval, 9O.6Vo
to
99.3Vo) achieved azoospermia (twoor more
con-secutive azoospermic samples). The rate and degreeof
supression were similar
in
the two treatment groups (Figure 6) andin
the different centres. Onceazoosper-Ëll
:t
60 ëa
v
oo o 40qô
è-0 4 I t2 t6 20.24
[image:5.595.80.549.407.695.2]Tinrc sincc thc first injcction (rvccks) 0 8 16243240 4856Time sincc thc last injcction (wccks)
20 Arsyad
mia
in
two
samples was achieved, a subsequent in-onlY one man,îl;îi'"ii:
6/ml; after thelast injection was 6.5 months
in
those men who had achieved azoospermia (Figure 6). Two of the three menwho
failed
to
achieve consistent azoospermia hadlowest spenn concentration
of
0.1 and 0.8x
106/ml, respectively, and the third subject, only a single samplerilas azoospermic. These
men were
similar
in
anthropometric characteristics to the other men.
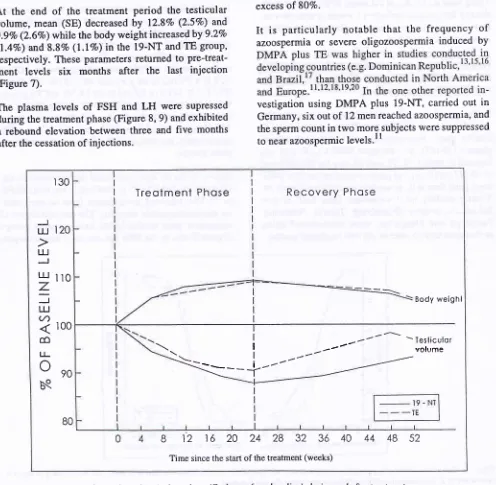
At
the
endof
the
treatmentperiod the
testicular volume, mean (SE) decreasedby
12.87o (2.5Vo) and 9.9Vo (2.67o) while the body weight increased by 9.2Vo(l.4Vo) and 8.87o
(l.IEo)
in the 19-NT and TE grouP'respectively. These Parameters returned to
Pre-treat-ment levels
six
months
after the last
injection(Figure 7).
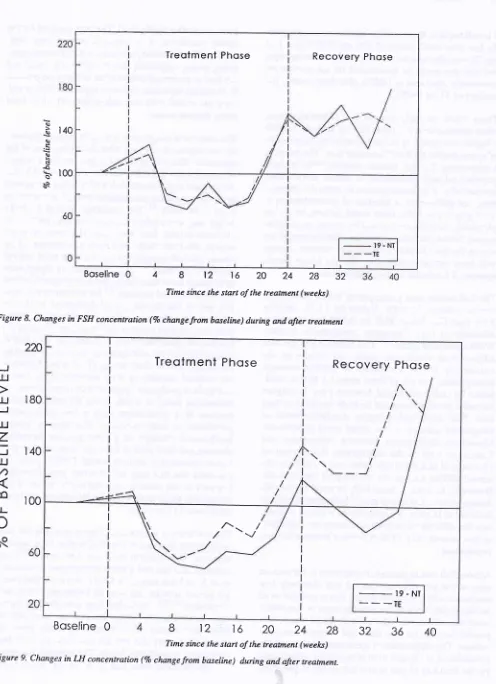
The plasma levels
of
FSH andLH
were supressed during the treatment phase (Figure 8, 9) and exhibiteda rebound elevation between three and
five
months after the cessation of injections.Med J Indones
COMPARISON BETWEEN VARIOUS
DMPA.
ANDROGEN REGIMENSThese two studies demonstrate that the combination of progestogen
with
an androgenis
highly effective ininducing azoospermia in normal Indonesian men.
In
studies of DMPA plus TE carried out previously,needed to achieve a cumulative rate of azoospermia in excess o180Vo.
It
is
particularly
notable
that the
frequency
of
azoospermiaor
severe oligozoospermia induced byvestigation using DMPA plus 19-NT, carried out in
Germany, six out of
l2
men reached azoospermia, and the sperm count in two more.subjects were suppressed to n"u, azoospermic levels.l tr30
Treolment
Phose
Recovery
Phose
J
l.rl
120ul
J
Lu
Ito
z
J
LU
(/)
4
100co
u-oeo
bq
_-_-\
Body weighl
I
-'---
-TesliculorI --' r,^r,,É6
I
_--'-
volume\---
\---+
| ---'---
'----"-'
BO
B 12 1620242832
36Time since the start of the treatment (weeks)
[image:6.595.62.558.241.726.2]40 44
48
52Vol6, No I, January - March 1997
Boseline
48t21620242832
[image:7.595.58.554.61.745.2]Time since the starl of the treatment (weeks)
Figure 8. Changes in FSH concentration (Vo change from baseline) during and after treatment
220
180
140
100
Boseline
0812t6202428
Time since the start of the treatment (weel<s)
P ro g e s to g enAndro g en C ombination 2t
c)
s
t40 q)s
EJq
s .ô
t,
100èa
-J LU
LU
LU
z
=
LU(/)
æ
lJ-o
b.q36 40
Treotment
PhoseRecovery
PhoseTreotment
Phose
Recovery
Phose
_
19 - Nr_ _
_ïE
-Â-. /
\
/
-\./
\\t
Figure 9' Changes in IJI concentration (Vo changefrom baseline) duing and after lreatment.
22 Arsyad
It is striking that, in the studies in Indonesian men' even the low doie combination
of
100 mg DMPA plus 100 mg TE was effective in inducing uniform azoospermiaand that this could be maintained
for
six months by six-weekly injections of DMPA plus three-weekly in-jections of TE or 19-NT.These results strongly suggest that Indonesian men,
when administered the same dose of steroids, exhibit
reason
for
the variabilitywithin
and between centreswill
have crucial significancefor
the futuredevel-opment of hormonal male-contraception.
The Indonesian men participating
in
this study werearea. This could lead
to
higher circulating levels of exogenoussteroid
in
the
small-sized population'Moieover,
differences
betweenIndonesian
andCaucasian
men
in
the
absorption,distribution
orclearence
of
steroids rnight exist, and explain theob-served differences
in
the efficacy
of
the
steroids' However, as evena
three-fold- increasein
steroid-dosage given to Caucasian *"n13 fails to overcome the
differences in rates
of
azoospermia,it
seems unlikelythat the differences in body size alone are important,
as this showed only
l5
Vo difference between the twopopulations.
Although the mean sperm-concentration in Indonesian
men
in
the pre-treatment period was relatively lowcompared with Caucasian norms, this is unlikely to be an important factor in the interpretation of this study,
since the volunteers were normal, healthy men with
proven
fertility
(except one)and
normal testicular volume. The suppression of spermatogenesis wasfully
reversible after the cessation of injections, as indicated
by the recovery
of
sperm concentrations topre-treat-Med J Indones
ment or putative fertile level. The time required for the
were associated
with
few side-effects which did notcause di scontinuation.
The increase in weight seen during the treatment phase,
as the slow
1
rloted ina
previous
in
bodY*"ight
rnuY
cted lateelevation
in
n earlierstudies, the total cholesterol level was unchanged by
similar steroid regimen even though the body weight
remained elevatù.13'ls'16'22 Changes
in
lipoprotein subfraction have been identified as a progestin effectin similar steroidal regiment.22 An altemative
explana-the comparatively short-acting TE.
It
is not knownif
the isolated increase
in
total cholesterol, without achange in high-density lipoprotein cholesterol level, in Indonesian men,o
is
arisk
factorfor
cardiovasculardisease
in
a
population
with
a
low
backgroundprevalence
of
artherosclerosis. The other reversiblebiochemical changes
in
plasma glucose, bilirubin, albuminvious
sregimen
ferences
Indonesian study design (using 20 subjects analysed
serial over
l1
timepoints).The low level of testosterone, four weeks after the last
testosterone injection in the DMPA plus TE treatmen[
regimen,
is
cLnsistentwith
anothèr report.2o Thisdemonstrates that endogenous testosterone production
must have been suppressed
fully,
while by that time,exoqenous testosterone was no longer present
in
thecircilation.2s'26'27
At
earlier time after the lastinjec-tion,
however, circulating testosterone level wouldhave been higher.
The
changesin
libido
reportedduring this study
did not
always correspondto
thecirculating testosterone levels. The reported transient
Vol6, No l, January - March 1997
most subjects, was probably
a
placebo reaction as normal circulating testosterone level would have beenmaintained. Conversely, the decline in
libido
reportedby a minority of subjects following cessation
ofiniec-tions, may be attributed to the prolonged suppression
of
endogenous testosterone induced byDMpA,_rela-tive to the short duration rf the effects ôf TE.2o'2s'26,27
Recent WHO-sponsored studies have demonstrated,
for the
first
time, that hormone-induced azoospermia can be sustained reliably and is highly effective as malecontraceptiv".20 Th" prêsent findings suggest that
hor-monal suppression
of
spermatogenesis may be anef-fective approach
to
male contraceptionin
Indonesiaand possibly other countries. A major limitation of the
treatment regimen used in the multicentre study is the
frequency
of
injections. Intramuscular injectionsof
androgen weekly
for
seven weeks andthen
at threeweek intervals was chosen as the simplest prototype regimen because of its safety, reversibility, ind
efiet-tiveness
in
inducing
azoospermia.
The
phar-macokinetic properties of androgen preparations seem
to be of critical importance in achieving azoospermia
in
men.The
frequent complaintsof
lossof
lUiAo during treatment withl9-NT
compared with TE couldreflect differences either in the efficacy
of
19-NT andTE
or
in dose equivalence of the two regimens usedfor
androgen replacement. Both androgens act uponthe
same androgenreceptor
andexlibit
similarandrogenic and anabolic effects, as demonstrated by
the
similarity
of
their
effecton
androgen-sensitive variables in the second study (body weiglit, testis size,hemoglobin,
LH
and FSH). Nevertheleis, differencesin biological action are possible since
l9-NT
isnon-aromatizable whereas
the
aromatizationof
TE
toestradiol
is
involvedin
some effectsof
T
on brainfunction.
It
remains controversial whether 19-NT isPl-" J.". support sexual function
only
partially28 orfully"
in orchidectomizedr
onkeys.Alternativjy,
theefficacy
of
the 19-NT regimenfor
androgen,"plu""-ment therapy, when administered at three-week inter_
vals (as during maintenance phase in the second study),
is less well established than that of TE,30 and could be less adequate.
Although very high rates ^of azoospermia was achieved
in the multicentre study,e a small minority
of
men (3of
90,3.3Vo) failed to reach consistently azoospermiclevel. The low concentrations of residuàl sperm, how_
ever, suggest that these men may also be rendered
sufficiently infertile to equate reli able contraception.
Indeed,
the functional
capacityof
residual spermduring
steroid-induced supression appearsto
bemarkedly decreased as judged by rheir i^mpaired ability
to fuse with zona-free ham;ter oocyte.3l
Progestogen-Androgen
Combination
23CONCLUSION
The encouraging results obtained in these two studies
offer a good prospect for developing a safe and
effec-tive hormonal contracepeffec-tive method suitable for use by
Indonesian men. However, the frequency of injection
and the high levels
of
hormones currently available that need to be injected would make the dËvelopmentof
such a methoddifficult.
To solve these problems,longer-acting androgens are being developed
to
be used either alone or in combination with a long-actingprogestogens.
It
is
planned that these preparations,which might be given at three-monthly intervals,
will
be assessed
in
Indonesian volunteersfor
safety, ef-frcacy, reversibility and acceptability. These studieswill
include assessment of effects on behavior as wellas the longitudinal impact on health status of the volun-teers.
Appropriate study
instruments
will
need
to
bedeveloped to assess the acceptability of male hormonal contraceptive methods and to determine the preferred contraceptive methods for couples of reproductive age
in lndonesia.
REFERENCES
l.
WHO. Progress in Human Reproduction Research, Special Programme of Research, Development and Research Train_ ing in Human Reproduction. Geneva: WHO, 1994. 2. Homonnai TZ,Paz FG. Methods for evaluating contracep_tive technique. In: Cunningham GR, Schill WB, Hafez ESE,
editors. Regulation of male fertility. The Haque: Martinus Nijhoff 1980;41-50.
3. Patanelli DJ, editor. Hormonal control of male fertility. Washington: US Department of Health, Education and Wel_
fare, 1978.
4. Schearer SB, Alvarez-sanchez F, Anselmo J, Brenner pF, Coutinho E, Latham-Faundes A, et al. Hormonal contracep_
tion for men. Int J Androl 1978: suppl 2:6gO_712.
5. Nieschlag E, Behre HE, WeinbauerGH. Hormonal methods
for the regulation of male fertility. In: Serio M, editor. Perspective in Andrology. Serono Symposia publications volume 53. New York: Raven press, l9g9:517_29. 6. Swerdloff RS, Palacios A. Male contraception: Clinical as_
sessment of chronic administration of testosterone enan_
thate. Int J Androl 1978: Suppl 2:731-47.
7. Waites GMH. Male fertility regulation : The challenges for
the year 2000. Br Med J 1993;49:2lO_21
8. Pangkahila W. Reversible azoospermia induced by an
androgen-progestin combination regimen
in
Inclonesianmen. Int J Androl 199l;14:248-56.
9. World Health Organization. Comparison of testosterone
enanthate plus DMPA and lg-nortestosterone hexoxyphenyl
propionate plus DMPA for male contraception. Fertil Steril
1 993;60(6):1062-8.
10. Knuth UA, Behre H, Belkein
L,
Bents H, Nieschlag E.24
ArsyadGI, Goldsmith A, Spieler IM, Sciarra JJ, editors' Male con-traception, advances and future prospects. Philadelphia: Harper and Row, 1986:320-8.
ll.
Knuth UA, Yeung CH, Nieschlag E' Combination of19-nortestosterone hexyloxyphenylpropionate (Anadur) and
depot-medroxyprogesterone acetate (Clinovir) for male
con-traception. Fertil Steril 1989;5 I : I 0l l-8.
12. Brenner PF, Mishell DR, Bemstein GS, Ortiz A' Study of
anthate as
-91.
13.
P.Attain-ment and maintenance
of
azoospermia with combinedmonthly injection of depot medroxy progesterone acetate and testosterone enanthate. Contraception 1977 ;15:.63548' 14. Paulsen CA, BremnerWJ, Leonard JM. Male contraception
clinical trials. In: Mishell DR Jr, editor' Advances in fertility research. New York: Raven Press, 1982:15'l-70'
15. Alvarez-Sanchez F, Brache V,LeonP, Faundes A' Inhibition of spermatogenesis with monthly injection of depot medroxy
progesterone acetate and low dose testosterone enanthate' Int
J Androl 1979;2:136-49.
16. Faundes A, Brache V, Leon P, Schmidt F, Alvarez-Sanchez
t7.
men with monthly injection
of
medroxy-progesteroneacetate and testosterone enanthate' Contraception
1977;15:627-34.
I 8. Frick J, B artsch G, Weiske WH. The effect of monthly depot
terone acetate and testosterone on human
s.
I.
Uniform dosage level' Contraception 1977;15:649-68.I 9. Frick J, Bartsch G, Weiske Vy'H. The effect of monthly depot medroxyprogesterone acetate and testosterone on human spermatôgenesis.
I.
Highinitial
dose. Contraception 1977;15:669-77.20. Hedman M, Gortleib C, Svanborg K, Bygdeman M, de la Torre B. Endocrine, seminal and peripheral effects of depot
medroxyprogesterone acetate and testosterone enanthate in
Men. Int J Androl 1988;1 l:265-76.
Med J Indones
21. World Health Organization. Contraceptive Efficacy of
tes-tosterone induced azoospermia in normal men. Lancet 1990;
336:955-9.
22.Wallace EM, Wu FC. Effects of medroxy-progesterone
acetate and testosterone enanthate on serum lipoprotein in Men. ContracePtion 1990;41 :63-71.
23. Oppenheim DS, Greenspan SL, Zervas NT, Schoenfeld DA' Klibanski A. gevated serum lipids in hypogonadal men with
and without hyperprolactinemia. Annals
of
InternalMedicine. 1989;l I l:288-92.
24. Kasperk CH, Wergedal JE, Farley JR, Linkhafi TA, Turner
RT, Baylink DJ. Androgen directly stimulate proliferation ofbone cells in vitro. Endocrinology 1989;124:1576-8' 25. Schulte-Beerbuhl M, Neischlag E. Comparison
of
tes-tosterone, dihydrotestes-tosterone, luteinizing hormone, and follicle-stimulating hormone in serum after injection of tes-tosterone enanthate or testosterone cypionate. Fertil Steril
1980;33:201-3.
26. Sokol RZ, Palacios A, Campheld LA, Saul C, Swerdloff RS' Comparison of the kinetic of injectable testosterone in
eugonadal and hypogonadal men. Fertil Steril
1982;37:425-30.
27. Schumeyer T, Neischlag E. Comparative pharmacokinetics
of testosterone enanthate and testosterone cyclohexanecar-boxylate as assessed by serum salivary testosterone levels in
normal men. Int J Androl 1984;7:18l-7.
28. Michael RP, Bonsall RW, Zumpe D. The behavioural
thresholds of testosterone in castrated male rhesus monkeys (Maccaca mulatta). Horm B ehav I 984; I 8 : I 6 I -76'
29. Weinbauer GF, Jaekwert B, Yoon YD, Behre HM, Yeung CH, Nieschlag E. Pharmacokinetics and pharmacodynamics
of testosterone enathate and dihydrotestosterone enanthate
in non-human primates' Acta Endocrinol (Copenhagen) 1990;122:432-42.
30. Snyder PJ, Lawrence DA. Treatment of male hypogonadism
with testosterone enanthate, J Clin Endocrinol Metab
1 980;5 l: I 335-9.