The Effects
of
W'estern
Diet on
Sperm
Quantity
and
Quatity
and
Serum
Hormonal
Concentration
in
Cynomologus
Monkey
(Macaca
fascicularis)
Injected
with
Testosterone
Enanthate
(TE) plus
Depot
Medroxy
Progesterone
Acetâte
(DVfn4;+)
Sutyarsox, N. Suhana**, N.
Moeloek**,
o. Soeradi**, S. Sri Sukmaniah**x,
JSupriatna****
Abstrak
Tujuan penelitian ini ialah untuk mengetahui pengaruh diit negara barat atau Eropa terhadap jumlahdan kualitas spermatozoa dan konsentrasi hormon dalam serum padn sekelctmpok monyet (Macaca fascicularis) yang diinjeksi dengan kombinasi testosteron enantat (TE) dan depotmedrory progesteron asetat(DMPA). Dua puluh monyet jantan dibagi menjadi dua kelompok tiap kelompok l0 ekor; kelompok pertama
(I)
diberi makan standar "monkey chow" yang susunannnya adalah: 9Vo lemak, 13Vo proteindin
78Vo karbohidrat, sedang kelompok kedua (ll) diberi makanan diit model negara barat yang susunannya adalah: 35Vo lemak, 25Va protein dan 40Vo karbohidrat. Tiga bulan setelah masa adaptasi kedua kelompok kera tersebut diinjeksi dengan 20 mg TE dan 25 mg DMpAdimulai pada minggu ke 0 sampai minggu ke 18, sedangkan TE dilanjutkan sampai minggu ke 24. Plàsma semen dilanmpulkai dengan menggunakan teknik elektro ejakulasi yang dilakukan tiap
j
minggu, jumlah dan kualitas spermcrtozoa ditetapkan berdasarkan meiode WHO. Pengambilan darah untuk pemeriksaan hormon dilakukan tinp 6 minggu sekali. Pada kelompok I terjadi azoospermia l00Vo, sedangkan pada kelompokII hanya mencapai
70Vo azoospermia. Pada kelompok I spermatoToa mulai ditemukan kembali pada minggu ke 33, tetapi sampai akhir penelitian (mingguke
39) hanya mencapai ke keadaan oligospermia berat, bahkan dua individu tetap azoospermia. Pada kelompokII meningkatnya konsentrasi spermatoloa
juga
dimulai pada minggu ke 33 dan mencapai keadaan normospermia pada akhir penelitian (minggu ke 39). Pada kelompok I penurunan kualitas spermatozoa lebih besar daripada ktelompkII.
Kadar hormon menurun pada kelompokI
lebih cepat daripada kelompokII,
sedangkan pemulihannya lebih lambat dari pada kelompokII. Dari hasil penelitian tersebut dapat disimpulkan
bahwa, perbedaannutrisi dapat
menyebabkan perbedaan terjadinya penurunanjumlah dan kualitas spermatozoa maupun penurunan hormon, pada M. fascicularis yang diinjeksi dengan TE dan Dsebutlebih cepat dan lebih besar pada kelompok I daripada pada kelompok II.
Abstract
The aim of this study is to elucidate the effects of western diet on sperm quantity and quality and serum hormone in cynomologus monkeys (Macacafasciculnris) injected with testosterone enanthate (TE) plus depotmedrory progesterone acetate (DM4A). The twenty male monkeys are divided into two groups ten animals each. The Group (l) was fed with the "monkey chow" (9Vo fat, l3Vo protein and 78Vo carbohydrate); the second group (ll)
wasfedwith
"western" diet ( 35Vofat, 25Vo protein and40Vo carbohydrate) Three months after adaptation period, both groups were injected with 20 mg TE and 25 mg DMPAfor 18 weeks, white TE injections continuedfor
another6week.
Thesemenforexaminationof Spermdensityandqualityusingelectro-ejaculationwasdoie inceeverythreewe)ks. and determinedby WHO methods. The blood serumfor hormone assaywas taken once every sixweeks. In groupI azoospermia reached
l00Vo'
while in the groupII
reached only 70Vo. In both groups the sperrnatozoal count were recovered at week 33, but until this investigation terminated onweek 39 the level of spermatoToa of group I reached only severe oligozoospermia, and tyvo animals remained severe azoospermic: while group II has returned to normospermia. The quality of sperm during and after treatment in group II was better than group I. Hormone concentration decreased more rapidLy in group I than in group II, while th" ,"rornry periodwàs slower in group I compared to groupII.
This investigation concludedthat dffirent
diet formula causeddffirent
resulti in decreasing the level of sperm quantity and quality, and hormone concentration of M.fascicularis injected with TE in combinationwith DMpA, ùeing
Iower in animals fed with monkey chow diet than in animals fed with western diet.Keywords: Contraception, Testosterone Enanthate & Depot Medrory Progesterone Acetate, Sperm quality & quantity, Hormonal Iev
el,
Macaca fascicularis.+)
*)
Supported by The Project of ResearchTeam "URGE" No: 023/ HTPP/URGE/1995. Department of Edacation & Culture, Directorate Generul of Higher Educatbn.
Department of Biology, Faculty o.f Mathematics & Natural Science, University of Lampung/Biomed.ical P ost Cruduute Program, University of Indonesia, Jakarta, Indonesia
**)
Department of Bioktgy, Fuculty of Medicine, University o.f Indone sia, J akar ta, Indone sia***)
Department of NuÛition, Fdculty of Medicine, tJniversitytf
I ndo nesiu, I akarta, I ndone s ia2OO
Sutyarso et al.INTRODUCTION
There
aredifferences
in
the efectivenessof
injectable
testosterone enanthate
(TE)
or
in
combination with
depotmedroxy progesterone acetate
(DMPA)
onachieving azoospermic state between
Indonesian,
Chinese, and Caucasian.
The injection
of
these
hor-mones
in Indonesian
men
achieved
azoospermiaof
almost
lO\Vo,r'2
in
Chinese
( injected by TE only
)over
9OVo,3whereas
in Caucasian
less than
70Vo.+This strongly
suggests that Indonesian
menexhibit
ahigher susceptibility to
steroidal-induced
suppressionof
spermatogenesis compared to Caucasian men, whenthe
same dosesof
steroids were
administered.
Poten-tial
explanations
for
the
greater
susceptibility
to
steroid-induced azoospermia
include
population-based (ethnic) differences
in
steroid metabolism,
aninsensitivity
to
steroidal
suppression ordifferences
in
the kinetics
of
spermatogenesis. Such
differences
could
in
turn be
due
to
physiological variations in
genetic
andenvironmental
factors
including nutrition
Ërdiet.l'2
Western diet is high
in
fat and
protein
, whereasAsian
diet is
low
in
fat
and protein buthigh in
carbohydrates. Nevertheless, therole of nutrien
onreproductive
func-tion
in
men
atthe moment
is still poorly
understood'
Nutritional
statusis
alsopossible
as a factorinfluenc-ing steroid metabolism that affects fertility'
Lermite
and
Terqui
reported
thatnutritional
status seems to bean additional factor regulating sex steroid binding
protein
(SBP)
level
which may alter
the percentageof
free
testosterone available
for
negative
feedback
Med J Indones
mechanism.
The
decreaseof
SBP, results
in an
in-creaseof free
androgenconcentration in
the
,"ro-.5
Street found that
nonesterified
fatty
acids modlfy
binding affinities
of
sexsteroid hormones to SBP.b
The aim
of
the study
is to
elucidate the effects
of
thewestern
diet (fat
35Vo,protein25Vo,
and carbohydrate40Vo)
on
sperm
levels
in
cynomologus
monkeys
in-jected
with TE
plus
DMPA.
METHODS
The cynomologus monkey ( Macaca
fascicularis)
used
in
this
study were obtainedfrom
thePrimate
Centerof
Bogor
Agriculture
Institute and originated
from
Tinjil
Island, West Jawa, Indonesia. Twenty adult
males (bodyweight4.40to 5.20kg,
and 5 to 9 yearsold)
werehoused
individually
in
cagessized23
xZlxl2
inches,at
roomtemparature
of
20o-29oc.
Thetwenty
malecynomologus
monkeys
was
divided into two
groupsof
10animals.
Group
I
wasfed
with
"monkey chow"
(9Vo
fat,
l3Vo
protein
and
carbohydrate 787o
of
calorie),
while
Group
II
was
fed
with
western diet
(fat35Vo,protein
257o and carbohydrate 4O7o ).This investigation
wasdivided into
threeperiods:
a. Adaptation
period
(3months: -12 to
thecommen-cement
of
treatment/week
0).b.
Treatment period
(6months: week
0to week
24).c.
Recoveryperiod
(3,5months: week24
to week 39).See Table 1.
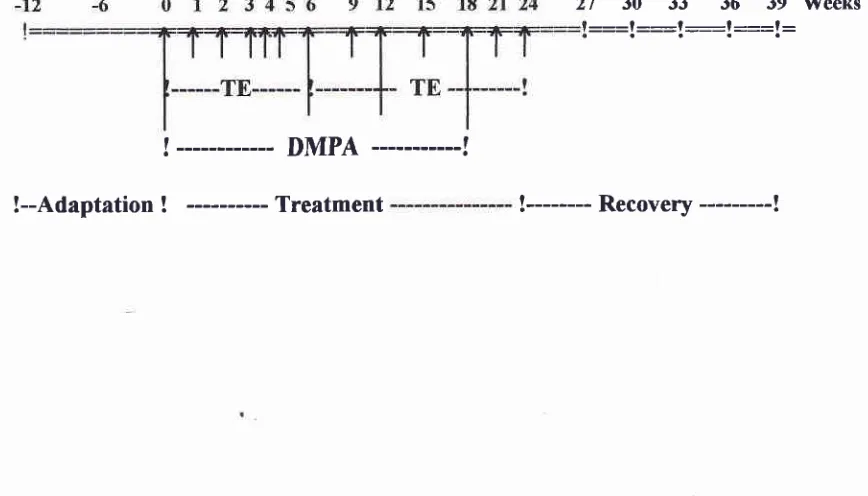
Table
l.
Chart showing the periods of adaptation (-12 to 0), treatment (0 to 24), and recovery (24 to 39) weeks.27 30 33 36
39 TVeeks::=l:__-l-__-l =::l:
!
---
DMPA
---!
[image:2.595.79.513.565.813.2]Three
months after adaptation period,
eachgroup
wasinjected with
20
mgTE
every
weekbegining
arweek
zero
up to
the
week
6
andcontinued every 3
weeksthere
after
until
week
24,
combined
with 25 mg
DMPA injection,
starting
atweek zero
andcontinued
every
6 weeks upto
the
week
18.The
semencollection
using
electro-ejaculation
tech-nique
werecarried
out
every
three weeks,begining
at
week
zerobefore adaptation period continued
until
therecovery
period
wasterminated;
spermcount
andquality
of
sperm
were
determined
by
the
WHO
methods.'
The electro-ejaculator
usedin this
study is
shown
in Figure
1.Blood
sample for the examination
of
FSH,
LH,
total
testosterone
(TT),
free testosterone(FT),
and estradiol(E2)
levels using
RIA
technique
were taken from
femoral
vein
andperformed
at 6week
intervals.
Before
the
electroejaculation was performed all
monkeys were
anesthesized
with
ketamin-HCl.
The semenwas
collected
in
gradual
centrifuge
tubes and
the
approximate
volume
was
recorded.EIn this
study
the statistical
analysis
of
SPSS "release-6 program"
was used.
RESULTS
The results
of
parameters evaluated, ars asfollows:
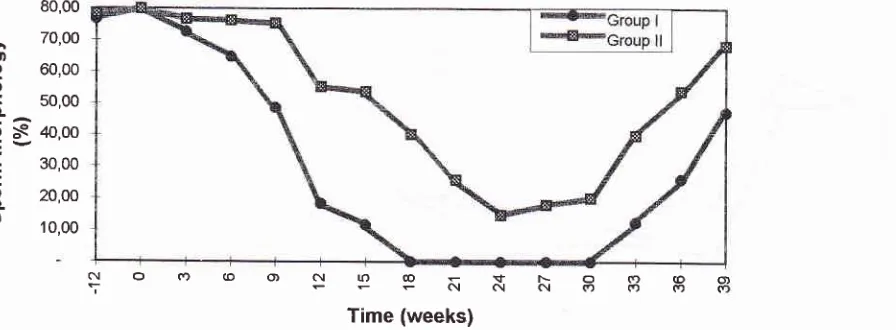
Sperm density
Sperm
count
aswell
as
spermquality
of
all
animalsbefore and during adaptation period were
normal,
using
WHO standard for
human.
The
spermdensity
during:
adaptation, treatment
and recovery periods aredepicted
in
Figure
2.
Sperm
density
in
the group
I
declined sharply
3 weeks after the
first injection, all
animalsbecome
azoospermic (lÙOVo) at week 18,per-sisted
up to week 30
or
6
weeks
after
TE injection
withdrawal.
In Group
I,
the spermreappeared at very
late stage
of recovery period (week 33), and
whenthis
investigation terminated
at
week 39 the
mean
con-centration
of
sperm
reached
the
level
of
severeoligozoospermia
only. More
over
two
animals
of
this
group remained azoospermic when
this
investigation
terminated. On
theother
handin
group
2
only
70Voaf
animals reached
the
azoospermic
level
in
24thweek
(see
Figure 3),
andthe
lowest
meanconcentration
of
sperm was 1.42
million/ml
at week 30,
after
that
theconcentration
increased
to
normozoospermia
at
the endof this
study (week
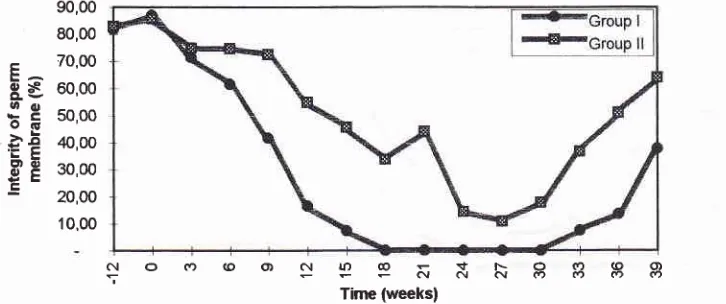
39).Sperm
quality
The results
of
spermquality evaluation
i.e. thepercent-age
of:
sperm
viability,
sperm
motility,
sperm
mor-phology, and
sperm membrane
integrity
(HOS-test)
aredepicted
in
Figures:
4,5,6,
and 7.In
generalit
wasfound
that the spermquality
parametersin
GroupI
waslower
than theGroup
II,
reaching
thelowest at
l8th
to
30th week
(in
accordancewith
azoospermic
level of
theGroup
I). In
Group
I
thequality of
spermdeclined
more
rapidly
comparedto the Group
II,
the
improve-ment
of sperm
quality
in
the Group
I
occured
very
slowly, while
in
the Group
II
the improvement of
spermquality
occured faster.S erum H
ormonal
C onc entration
Serum hormone
concentration of FSH
,LH (mlU/ml),
total
testosterone(TT) (ng/ml),
free
testosterone(FT)
andestradiol
(E2) (pglml),
aredepicted
in
Figures:
8,9,
10,
1|
and
12respectively.
The
concentrations
of
FSH
and
LH
were
decreased
significantly
in both
groups(I
andII)
from
week 6
through
week
24.
TheFSH
and
LH
levels were
increased
more
rapidly in
Group
II
compared to theGroup
I.
The meancocentra-tion
of serum
TT showed
no
significant
difference
between
groups,
both groups
showed
increment
atweek 6, then decreased again
atweek
12up
to
week
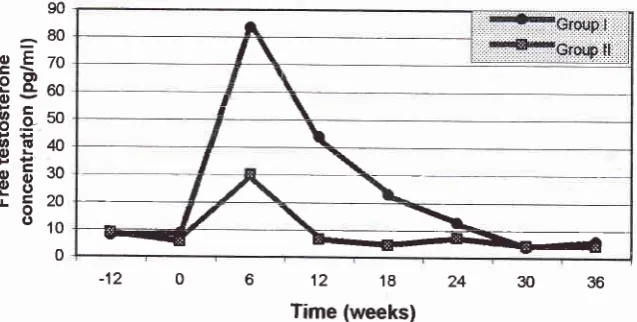
36. The mean serumFT was different
between Groups,being higher
in
theGroup
I
comparedto
theGroup
II.
Thus,at
week6
FT in Group I
was 83.54 + 7 .17pglml
while
in
Group
II
was 30.29
+
9.18 pg/ml. The
dif-ference continued
from
6th
week
to 24th week
(see alsoFigure
11). There was nodifference concentration
ofestradiol
in both
groups;the
cocentration
increased to thehighest
level
at week 6;
but
decreased again atweek
12 and after.DISCUSSION
This
study hasshown that
theinjection of TE in
com-bination with
DMPA
induced
azoospermiain 2
groupsof
cynomologus monkeys (Macaca
fascicularis) fed
with different
diet
formula;
theGroup
I
wasfed
with
monkey
chow
diet
(low
fat
and protein,
high
car-bohydrate)
while
Group
II was
fed with
western diet
(high
fat
and
protein,
low
carbohydrare). This
phenomenon
is
very
understandable,
because
ex-ogenous testosterone
(TE)
and progestogene(DMpA)
supressed
the production
of LH
and FSH
by
202 Sutyarso et al.
Figure 1. The electro-ejaculator used
for
semen collection, the body instument (top) and rectal probe (bottom)Med J Indones
E
€
E Ëtûc
oE
o CL
ah
250,00
200,00
150,00
100,00
50,00
g^ ' r"l <o o) S g p N N \ g E E
E [image:4.595.46.351.84.242.2] [image:4.595.166.544.272.443.2] [image:4.595.54.491.489.690.2]Time (weeks)
Figure
2.
The sperm densityofM.fascicularisinjectedwithTEplusDMPAat dffirent
periods: adaptation (-12 to 0 weeks); treatment (0 to 24 weeks); recoyery Q4 to 39 weeks).Group I, monkey-chow diet; Group II, western diet.
fr.ilffrré1fwebfr):' t'ti:,,:" t' t' t :, :,:,:,:.:.:.,,. t.:,:t'.'.
Figure 3. The percentage of azoospermic levels of M. fascicularis injected with TE plus DMPA at
dffirent
periods: adaptatiorl (.12 to 0 weeks); treatment (0 to 24 weeks); recovery Q4 to 39).Group I, monkey-chow diet; Group II, western diet.
s
Y
.= .cl
,g
tr
Lo
CL
o
90,00 80,00 70,00 60,00 50,00 40,00 30,00 20,00 10,00
(oo)NtO@rSÈ-O(rr(oO)
c!Nôt(orr(D(r,
[image:5.595.35.371.89.250.2]Time (weeks)
Figure 4. The sperm viability of M. fasciculnris injected with TE plus DMPA at different
periods: adaptation (-12 to 0 weeks); treatment (0 to 24 weeks); recovery Q4 to
j9
weeks),Group I, monkey-chow diet; Group
II,
western diet.NO(f)
s
Ë
o
E ts
o
CL
o
90,00
80,00
70,00
60,0,0
50,00
40,00
s0,00
20,00
10,00
C.l O
(Y) (O o) C.l r,
@ r
$
F. O
G) (o
O)iGtclNôrr6ô
[image:5.595.143.534.304.474.2]Time (weeks)
Figure 5.The spermmotility of M.fascicularis injected
with
TEplus
DMPAat dffirent
periods:adaptation (-12 to 0 weeks); treatment (0 to 24 weelcs); recovery Q4 to 30 weelcs).
Group I, monkey-chow diet; Group
II,
western diet.q) .9
o
CL
o
t
E
o
ÉL 3n
80,00
70,00
60,00
50,00
S
ao,oo30,00
20,00
10,00
C{ O (f, (O O) C{ lO @ r rit }.- c| É) (c) c
ï(\c\c{(7)(rj{r)a
Time (weeks)
Figure 6. The sperm morphology of M. fascicularis injected with TE plus DMPA at
dffirent
periods:adaptation (-12 to 0 weeks); treahnent (0 to 24 weel<s); recovery Q4 to 39 weeks).
[image:5.595.41.489.522.687.2]204
Sutyarso et al.90,00 80,00 70,00 60,00 50,00 40,00 30,00 20,00 10,00
c{ O r, (O O) C',1 ln @ t F- O (., (9 q, îNNNo(Y)oo
[image:6.595.46.409.86.238.2] [image:6.595.220.548.303.479.2] [image:6.595.54.374.545.681.2]Time (weeksl
Figure 7. The integrity of sperm membrane of M. fascicularis injected with TE plus DMPA
at different periods: adaptation (-12 to 0 weeks); treatment (0 to 24 weeks); recovery
(24 to 39 weeks). Group I, monkey-chow diet; Group II, western diet'
Med
I
IndonesE
9.5
OO
bF
àE
bE
!rc Ê
tr
3'5gr
âE
2'5(J=
OE
o
.:
1,5o1
IL0
-12
Time (weeks)
Figure 8. The FSH levels of M. fascicularis injected with TE plus DMPA at
dffirent
periods: adaptation (-12 to 0 weelcs); tïeatment (0 to 24 weeks); recovery Q4 to 36 weeks).Group I, monkey-chow diet; Group
II,
western diet.c
2.5o
.F(E^ )
ËE
85
r,s89
1IJ
0,5 0-'12 0
6
12 18 24 æ
36Time
(weeks)
Figure 9. The LH levels of M. fascicularis injected with TE plus DMPA at
dffirent
periods: aiaptation (-12 to 0 weeks); çeafinent (0 to 24 weelcs); recovery (24 to 36 weeks).diel-25
g20
I
(l)
t;
oÈ
115
E B,O
o
ô5
F
0
-'t2
0
6
12
.t8
24
30
36Time (weeks)
Figure 10. The total teslosterone levels of M. fascicularis injected with TE plus DMPA at
dffirent
periods: adaptation (- 12 to 0 weel<s); treatment (0 to 24 weeks); recovery Q4 to 36 weeks). Group I, monkey-chow diet; Group II, western diet.-12061218243036
Time (weeks)
Figure 12. The estradiol levels of M. fasciculais injected with TE plus DMPA at different periods: adaptation (-12 to 0 weeks); treatment (0 to 24 weeks);
recovery (24 to 36 weeks). Group I, monkey-chow diet; Group II, western diet.
l.
il
:llt\
Er:lll/l
\
It
\
\
T
\
-f
-12 0
6
't2
18 24 3)
36 [image:7.595.38.429.84.231.2] [image:7.595.209.528.307.468.2]Time (weeks)
Figure I
I.
The free testosterone levels of M. fascicularis injected with rE plus DMpAat
dffirent peiods:
adaptation (-12 to 0 weeks); treatment (0 to 24 weeks); recovery (24 to 36 weeks). Group I, monkey-chow diet; Group II, western diet.90
=80
PE
zo ;CDËgoo
E,Ë
'o
an6a
g.:
Ë8,
tr-
5
20o10
0
25
c20
_.o
.9Fars
tu-\o5.o10
llJc-
o
o5
206 Sutyarso et al.
since there was a
difference
of
theeffectiveness of TE
plus
DMPA injectionto
induce azoospermic levels, i.e.highly
effective
in
Group
I
(l00Vo
azoospermic),while
in
Group
II
less
effective
(707o
azoospermic),this
phenomenon
should be
elaborated
further. As
aconsequence
of TE
andDMPA injections
it
is natural
that the
FSH
andLH
levels drop simultaneusly (in this
study
6
weeks after the
first
injection), and
serumtestosterone increases (see
Figures:
8,9,
10,and
1 1).Total
testosterone aswell
as free testosteronelevels
in
both groups
increased
to
the highest level 6
weeksafter the
first
injection, but
the concentration
of
freetestosterone
in
Group
I
is
much higher then the
con-centration
of
free
testosteronein
Group
II.
This
situa-tion
explained
why
TE plus
DMPA injection
induced
azoospermic
levels
in
Group I
moreeffectively
thanin
Group
II.
This
explanation also
hold
true
for
the
dif-ference of the sperm
quality
wherein
general the spermquality
of the GroupII
is better compared to thequality
of
theGroup I.
Why
free
testosterone
in
Group
I
is
higher then in
Group
II?
This situation,
may be related to theproduc-tion of
sexhormone
binding globulin
(SHBG). As
hasbeen mentioned earlier
(see
the Introduction),
that
nutritional
status
is
also possible
to
be a factor
in-fluencing
steroid metabolism
that
affects fertility.
Lermite and
Terqui
reported that nutritional
statusseems to be an
additional factor
regulating
sex steroid
binding protein
(SBP)
level which
may alter the
per-centage
of
free
testosterone available
for
negative
feedback mechanism.
The decreaseof
SBP, resultsin
an
increase
of
free
androgen
concentration
in
the,"ru-.5
Street
found that nonesterified
fatty
acidsmodify binding affinities
of
steroid
sexhormones to
SBP.6
Several experimental
studieshave
suggestedthat diet
can
alter
theprgduction
andmetabolism
of
steroid andSHBG
in
men.e
Reed etal. recently
demonstratedthat
a
low fat diet
administered
to normal
men
decreasedthe SHBG levels
and increased
the
free-testosteroneconcentrationr.l0
Th"r"
studies
suggestthat
diet
hasan
effect
on steroid secretion ormetabolism.
In anotherstudy, western
diet
(40Voof
calories
from fat) fed
to
vegetarian men
increased
the urinary excretions of
estrogens and
androgens
while
such excretion
decreased
when omnivorous
men
were
fed
with
vegetarian
diet.ll'12
Thus, nutritional
or
diet
statusseems
to be
an
additional factor regulating SHBG
or
SBP levels
which may alter the
percentage
of
freetestosterone
available
for
negative feedback,
andMed J Indones
excretion
or
metabolism
of
steroid
hormones.
Epidemiological
studies showedthat SHBG
wasposi-tively
correlated
to
total
testosterone
andnegatively
correlated to
insulin
hormone.13 Streetfound
thatnon-esterified fatty
acidsmodlfy binding affinities
of
sexsteroid
hormonesto
SBP.6This
studies showed thattotal
testosterone
was the sinthe Group
I
was
higher;
soit
is
reasonableto believe
that the
level
of
SHBG
washigher
in
Group
II
or may
be
the
level
of
SHBG
was
the
samebut
the
binding
affinities
waslower in
GroupI,
or may be thecombina-tion of two
factors.
This
problems need further
clarifications in
another studies.CONCLUSION
This
investigation concluded
that
different diet
for-mula
causeddifferent
resultsin
decreasing thelevel
of
sperm
quantity
and
quality,
and hormone
concentra-tion
of M.fascicularis
injected
with
TE
incombination
with
DMPA,
being
lower
in
animals
fed with
themonkey chow
diet thanin
animals fedwith
the westerndiet.
REFERENCES
1. Pangkahila
W.
Reversible azoospermia inducedby
anandrogen-progestin combination regimen
in
Indonesian men. Int J Androl 1991; 14: 248-56.2. Moeloek
N.
Comparisonof
two
androgens plus depot-medroxyprogesterone acetate for suppression toazoosper-mia in Indonesian men. Fertil Steril 1993;60: 1062-8. 3. Gui-yuan Z. Contraceptive efficacy of testosterone induced
azoospermia
in
normal men. The Lancet 1990; 336:. 955-9.4. Arsyad
KM.
Progestogen-Androgen combination: Resultsof Indonesian clinical trials. Med J
Indon
1997; 6: 15-24. 5. Lermite V, TerquiM.
Plasma sex steroid binding protein inmature heifers: Effect
of
the reproductive status, nutritionallevels, and porcine growth hormone and estradiol treatment. BioI Reprod 1991; 44: 864-70.
6. Street C, Herry RJS, Al-Othman S, Chard
T.
Inhibition ofbinding
of
gonadal steroidto
serum binding protein by non-esterified fatty acids: The influence of chain length anddegree of unsaturation. Acta Endocrinol 1989; 120:. 17 5-9 .
7. World Health Organization (WHO). Penuntun laboratorium
untuk pemeriksaan semen manusia dan interaksi semen
getah servik. Translations by Tadjudin
MK,
Balai PenerbitFKUI Jakarta 1990.
8. Lipshultz LI, V/itt MA, Grantmyre
JE.
Electroejaculation. Infertil Reprod Med Clin 19921'3:455-66.9. Belanger A, Locong A, Noel C, et
al.
influence of diet on plasma steroidand
sex plasma binding globulin levels in adult men. J Steroid Biochem 1989;32:829-33.10. Reed MJ, Cheng RW, Simmonds M, Richmond W, James
ll.
Hill
P, Wynder EL, Garbaczewski L, Walker ARP. Effectsof
diet on plasma and urinary hormonesin
South African Black Men with prostastic cancer. Cancer Res 1982; 42: 3864-9.12. Gates JR, Parpia B, Campbell TC, Junshi C. Association of
dietary factors and selected plasma variables with SHBG in rural Chinese women. Am J Clin Nutr 1996; 63: 22-31.
Hautanen A, Sarna S, Pelkonen R, Adlercreutz
H.
Serum sex-hormone binding g.lobulin (SHBG), cardiovascular risk factors, and adrenal cortisol responses to dexametasone and corticotropin. Metabolism 1993; 42: 87 0-4.