Risk Factors of anemia in head and neck cancer patients undergoing
chemotherapy with high-dose cisplatin
Johan Kurnianda1, Nugroho Wiyadi2, Wahyu Wulaningsih3
Abstrak
Anemia merupakan salah satu efek samping yang paling sering dialami pasien kanker yang diterapi dengan cisplatin dosis tinggi. Penelitian ini bertujuan untuk mengamati perkembangan anemia dan menentukan faktor-faktor yang berpengaruh terhadap anemia pada pasien yang diterapi cisplatin. Dilakukan pengumpulan data pada pasien kanker kepala dan leher yang menjalani kemoterapi berbasis cisplatin antara Desember 2002 hingga Desember 2005. Insidensi dan faktor risiko anemia dianalisis dengan mencakup faktor usia, jenis kelamin, kadar Hb awal, klirens kreatinin awal, dan metastasis jauh. Stratiikasi menurut usia dan jenis kelamin dilakukan terhadap kadar Hb awal dan CrCl awal. Analisis multivariat digunakan untuk mengidentiikasi prediktor independen anemia. Dari 86 pasien, 26 (30,2%) mengalami anemia, ditandai kadar hemoglobin < 11 g/dL. Kadar hemoglobin turun secara signiikan setelah siklus pertama, dan terus menurun. Usia > 55 tahun (RR = 2.2, 95% CI, 1.2-4.0), jenis kelamin perempuan (RR = 2.0, 95% CI, 1.2-3.8), kadar Hb awal ≤ 13 g/dL (RR = 4.2, 95% CI, 1.9-9.4) dan CrCl awal < 50 mL/menit (RR = 2.9, 95% CI, 1.7-5.1) berkorelasi dengan insidensi anemia (P < 0.05). Pada analisis multivariat, kadar hemoglobin awal dan klirens kreatinin awal merupakan faktor risiko independen anemia. Akan tetapi, terdapat efek perancu pada klirens kreatinin awal pada stratiikasi menurut usia (aRR = 2.2, 95% CI, 1.1-4.7). Kadar hemoglobin awal merupakan prediktor terkuat dari anemia. Kadar hemoglobin awal ≤ 13 g/dL ke bawah dan klirens kreatinin awal < 50 g/dL merupakan prediktor independen anemia akibat cisplatin, sehingga keduanya bernilai penting terhadap upaya prevensi anemia. (Med J Indones 2008; 17: 248-54)
Abstracts
Cisplatin is well-known for its effectiveness against cancer, as well as its toxicity to human tissues. Of several documented side effects, anemia was reported to have signiicant association with decreased quality of life. This study was conducted to investigate development of cisplatin-induced anemia, and to identify independent factors contributing to anemia. Clinical data from head and neck cancer patients treated with high-dose cisplatin between December 2002 and December 2005 were obtained in this study. Incidence and risk factors of anemia were assessed in a model including age, sex, baseline hemoglobin level, baseline creatinine clearance, and occurrence of distant metastases. Multivariate logistic regression was used to deine independent predictors of anemia. Among 86 eligible patients, 26 (30.2%) developed anemia, deined as Hb level lower than 11 g/dL. Age > 55 years old (RR = 2.2, 95% CI, 1.2-4.0), female sex (RR = 2.0, 95% CI, 1.2-3.8), baseline Hb ≤ 13 g/dL (RR = 4.2, 95% CI, 1.9-9.4) and baseline CrCl < 50 mL/min (RR = 2.9, 95% CI, 1.7-5.1) were signiicantly correlated with incidence of anemia (P < 0.05). In multivariate analysis, baseline Hb and baseline CrCl were identiied as independent risk factors for anemia. However, considerable confounding was observed in baseline CrCl after stratiied by age (aRR = 2.2, 95% CI, 1.1-4.7). Thus, baseline Hb level was the strongest predictor of anemia. The indings suggested that baseline Hb and CrCl were useful to recognize cisplatin-treated patients at risk for anemia who might beneits from preventive measures. (Med J Indones 2008; 17: 248-54)
Keywords: anemia, cisplatin, chemotherapy, hemoglobin, creatinine clearance
Anemia is one of the most frequent side effects experienced by cancer patients undergoing chemo therapy with cisplatin.1 Severe ciplatininduced anemia
correlates signiicantly with decreased overall survival
and reduced quality of life.2,3 Several patients will need blood transfusions to correct their anemia.4
1 Division of Hematology-Oncology, Department of Internal Medicine, Faculty of Medicine, Sardjito Hospital, Gadjah Mada University, Jogjakarta, Indonesia
2 Department of Public Health, Faculty of Medicine, Gadjah Mada University, Jogjakarta, Indonesia
In cancer patients, hemoglobin level declined proggressively after administration of cisplatin, but medullary erythroblastic cellularity was reported normal.5 Decline of hemoglobin level correlates with decrease of creatinine clearance, indicating an association between the development of anemia and deterioration of kidney
tubular function. Those indings implied the etiology of cisplatin-induced anemia as an erythropoietin deiciency
state secondary to cisplatininduced nephrotoxicity.6,7
Erythropoietin administration is able to correct the resultant anemia and increase quality of life. However, 2040% patients still need to be transfused.89 Therefore, the use of erythropoietin as a preventive measure, i.e. erythropoietin administration to patients at high risk of developing anemia during chemotherapy, was
considered at higher value. Identiication of high-risk
patients is essential to attain costeffective measures.
Besides, identiication of anemia risk factors in
patients undergoing chemotherapy with cisplatin has a prognostic value for the need of blood transfusions in near future.2
Previous studies have deined several factors
contri-buting in development of anemia in chemotherapy patients,10,11 but there is still lack of evidence providing risk adjustment for potential confounding comorbid. The present study aims to investigate the development of anemia in patients treated with cisplatinbased chemotherapy, as well as to analize in a comprehensive fashion the interaction between factors contributing to
the risk of anemia, and to deine independent risks of
cisplatininduced anemia.
METHODS
This study included 86 nasopharyngeal cancer patients admitted to Oncology Unit, Sardjito Hospital, Jogja karta, Indonesia between December 2002 and December 2005. Data was obtained from two previous studies conducted in Sardjito Hospital.12,13 There were 139 subjects at initial selection, 135 among them have evaluable clinical data. Only 91 patients completing at least three chemotherapy courses were included in the study. Among them, 86 subjects met the inclusion criteria, i.e. patients who did not have anemia at the
beginning of chemotherapy, deined as hemoglobin
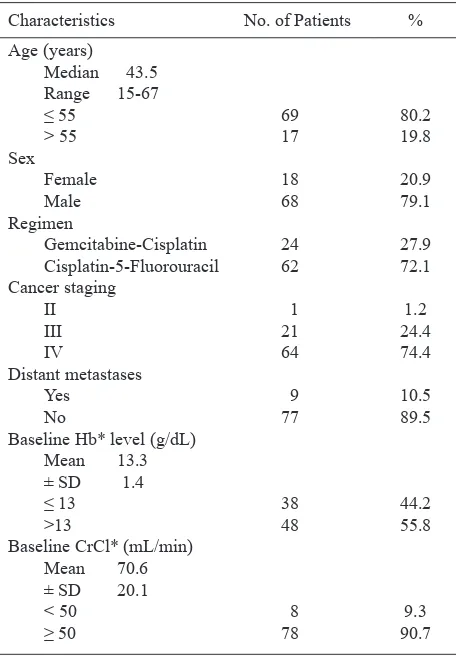
level less than 11 g/dL. Their characteristics are shown in table 1.
Table 1. Characteristics of patients*
Characteristics No. of Patients %
Age (years)
* Hb = hemoglobin; CrCl = creatinine clearance
Two regimens containing highdose cisplatin, i.e. single dose more than 50 mg/m2, were included in this study. Those regimens are gemcitabinecisplatin and cisplatin
5-luorouracil. A cycle of gemcitabine-cisplatin regimen
consisted of administration of cisplatin intravenously in 80 mg/m2 dose on day 1, and gemcitabine 1250 mg/m2 was given on day 1 and 8 every 3 weeks.12 In cisplatin 5FU regimen, cisplatin was given in 100 mg/m2 dose on day 1, and 5FU was given on day 1 and 5 every 3 weeks.13
In this cohort study,14 patients’ data were obtained from a case report form of two previous studies conducted in Sardjito Hospital, Jogjakarta, Indonesia. The outcome
was incidence of anemia, deined as decrease in blood
hemoglobin to a level less than 11 g/dL on any course
among the irst three observed. Every patient was only
counted once. Considered as exposures were age > 55
years old, female sex, baseline hemoglobin level ≤ 13
Data was analyzed with EpiInfo version 6. Hemoglobin level was observed in every cycle, and paired ttest
was used to determine the signiicant decline in mean
hemoglobin levels. Univariate analysis for anemia risk factors was performed with chi2 test. To eliminate confounding factors, baseline hemoglobin level and
baseline creatinine clearance level were stratiied for
patients’ age and sex, using Mantel Haenszel chi2 test. A multivariate logistic regression was performed to
deine independent predictors of anemia. P value < 0.05 was considered signiicant.
RESULTS
At the beginning of chemotherapy, all patients have hemoglobin level above 11 g/dL, therefore no anemia
was observed. After irst course, 8 (9.3%) patients
became anemic, no moderatesevere anemia according
to WHO classiication, i.e. hemoglobin level < 9.5 g/dL,
was observed. Hence either incidence and prevalence
on irst cycle was 9.3%. Prevalence of anemia increased
on second course (17.4%) and third course (27.9%). Moderatesevere anemia was experienced by 3 (3.5%) patients on second course, and 5 (5.8%) patients on third course of chemotherapy. Overall incidence of anemia during three chemotherapy courses was 26 (30.2%). Of 26 patients, 9 (34.6%) had onset of anemia on third course, 9 (34.6%) on the second. Thus incidence of
anemia in this study was 9.3% on irst course, 11.5% on
second course, and 13.1% on third course. Prevalence
and incidence of anemia are shown in igure 1.
Figure 1. Prevalence and incidence of anemia in patients under going chemotherapy with high-dose cisplatin. At initial encounter, none of the patient was anemic, deined as hemoglobin level less than 11 g/dL. The incidence and prevalence of anemia rose proggressively after irst course of cisplatin-based chemotherapy.
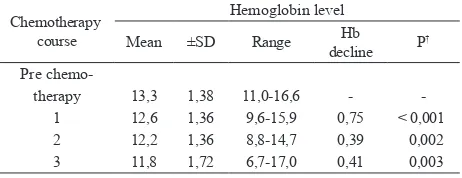
To understand the development of anemia, the alterations of mean hemoglobin levels before and after every chemotherapy course were observed. Paired ttest results are shown in table 2. Before chemotherapy, mean hemoglobin level was 13.3 g/dL, with the range of 11.0 to 16.6 g/dL. This level decline accumulatively
during subsequent courses, as shown in igure 2. Overall decrease was signiicant (P < 0.001) with a
difference 1.55 g/dL from baseline level. Decrease of
hemoglobin was signiicant for course 1, 2, and 3, with most prominent decline on irst course, which differed
0.75 g/dL from baseline.
Figure 2. Mean hemoglobin levels before chemotherapy and after each course of chemotherapy observed (1st, 2nd, and 3rd course). There was a signiicant decrease overall and after every course of chemotherapy with cisplatin. Greatest hemoglobin decline was observed after irst course of chemotherapy with cisplatin.
Table 2. Hemoglobin levels before and during chemotherapy*
Chemotherapy course
Hemoglobin level
Mean ±SD Range Hb decline P
†
Pre chemo therapy
1 2 3
13,3 12,6 12,2 11,8
1,38 1,36 1,36 1,72
11,016,6 9,615,9 8,814,7 6,717,0
0,75 0,39 0,41
< 0,001 0,002 0,003
* Hb = hemoglobin; Hb decline was calculated by substracting hemoglobin level of previous course with hemoglobin level of the next course.
† Signiicant for P value < 0,05
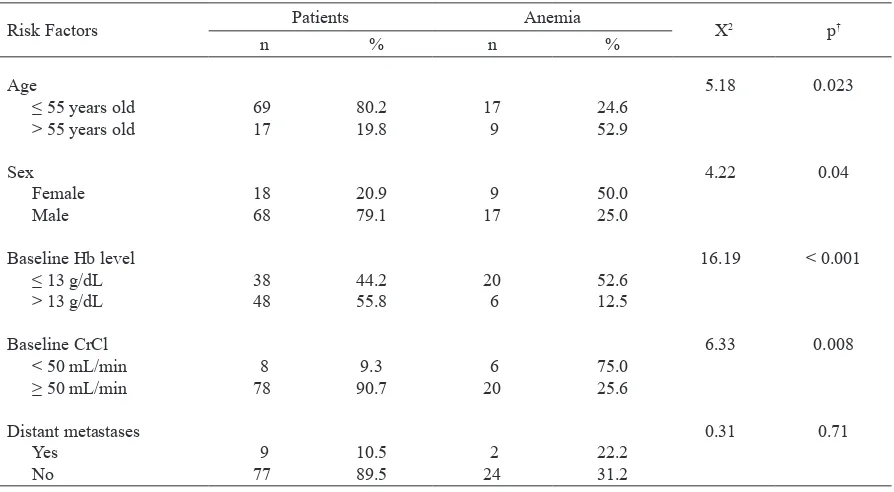
From univariate analysis results as shown in table 3, age > 55 years old (RR = 2.2, 95% CI, 1.24.0), female sex (RR = 2.0, 95% CI, 1.23.8), baseline Hb level
≤ 13 g/dL (RR = 4.2, 95% CI, 1.9-9.4) and baseline
CrCl < 50 mL/menit (RR = 2.9, 95% CI, 1.75.1) have
0 5 10 15 20 25 30
%
o
f
p
a
ti
e
n
ts
0 1 2 3
Chemotherapy course
signiicant correlation with the incidence of anemia (P < 0.05). P value was not signiicant (> 0,05) for
presence of distant metastases in this study.
Age and sex were wellknown as factors associated with baseline hemoglobin and baseline creatinine
clearance.16,17 Therefore, stratiication was performed on baseline hemoglobin level and baseline creatinine clearance for patients’ age and sex to eliminate confounding effects and to calculate the actual risk. The results are shown in table 4.
Table 3. Univariate analysis for risk factors of cisplatininduced anemia*
Risk Factors Patients Anemia X2 p†
n % n %
Age
≤ 55 years old > 55 years old
Sex Female Male
Baseline Hb level ≤ 13 g/dL > 13 g/dL
Baseline CrCl < 50 mL/min ≥ 50 mL/min
Distant metastases Yes
No
69 17
18 68
38 48
8 78
9 77
80.2 19.8
20.9 79.1
44.2 55.8
9.3 90.7
10.5 89.5
17 9
9 17
20 6
6 20
2 24
24.6 52.9
50.0 25.0
52.6 12.5
75.0 25.6
22.2 31.2
5.18
4.22
16.19
6.33
0.31
0.023
0.04
< 0.001
0.008
0.71
* Anemia was deined as Hb < 11 g/dL. Hb = hemoglobin; CrCl = creatinine clearance.
Table 4. Stratiication analysis for risk factors of cisplatin-induced anemia*
Variables Crude RR (95% CI) Adjusted RR (95% CI) p†
Age
Baseline Hb level ≤ 13 g/dL > 13 g/dL Baseline CrCl
< 50 mL/min ≥ 50 mL/min
4.2 (1.99.4) 1
2.9 (1.75.1) 1
4.1 (1.98.9) 1
2.2 (1.14.7) 1
< 0.001
0.059
Sex
Baseline Hb level ≤ 13 g/dL > 13 g/dL Baseline CrCl
< 50 mL/min ≥ 50 mL/min
4.2 (1.99.4) 1
2.9 (1.75.1) 1
4.3 (1.711.0) 1
2.6 (1.54.4) 1
< 0.001
0.015
On stratiication based on patients’ sex, baseline
creatinine clearance less than 50 mL/min still
signiicantly correlated with the risk of anemia,
however, there is a difference in risk calculated after
stratiication, implying some degree of confounding. On stratiication based on patients’ age, there was a
substantial confounding observed, indicated by more than 10% difference between adjusted risk and the initially calculated risk, thus the correlation between baseline creatinine clearance with incidence of anemia
became insigniicant. Patients’ age and sex confound
the relationship between baseline creatinine clearance and incidence of anemia, therefore baseline creatinine clearance less than 50 mL/min was a weak risk factor for cisplatininduced anemia.
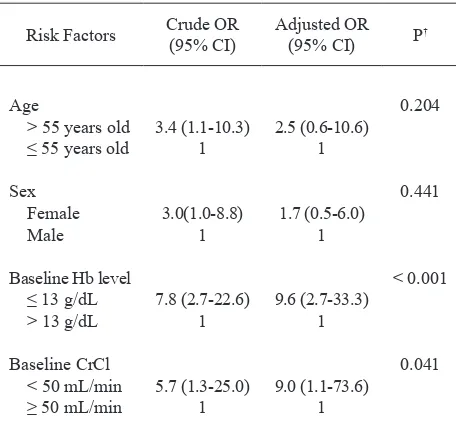
In multivariate analysis as shown in table 5, among age, sex, baseline hemoglobin level and baseline creatinine clearance, only two were recognized as independent risk factors of anemia, those were baseline hemoglobin level 13 g/dL and less (P < 0.001) and baseline creatinine clearance less than 50 mL/min (P < 0.05).
Baseline hemoglobin level ≤ 13 g/dL was the strongest
risk factor for cisplatininduced anemia.
Table 5. Multivariate analysis for risk factors of cisplatininduced anemia
Risk Factors Crude OR (95% CI)
Adjusted OR
(95% CI) P
†
Age
> 55 years old ≤ 55 years old
Sex Female Male
Baseline Hb level ≤ 13 g/dL > 13 g/dL
Baseline CrCl < 50 mL/min ≥ 50 mL/min
3.4 (1.110.3) 1
3.0(1.08.8) 1
7.8 (2.722.6) 1
5.7 (1.325.0) 1
2.5 (0.610.6) 1
1.7 (0.56.0) 1
9.6 (2.733.3) 1
9.0 (1.173.6) 1
0.204
0.441
< 0.001
0.041
* Baseline Hb level = baseline hemoglobin level; baseline CrCl = baseline creatinine clearance; OR = odds ratio; a value of 1 is a reference.
† Signiicant for P value < 0,05.
DISCUSSION
This study was intended to identify risk factors of cisplatininduced anemia. In univariate analysis, age > 55 years old, female sex, baseline hemoglobin level
≤ 13 g/dL, and baseline creatinine level < 50 mL/min were correlated signiicantly with incidence of anemia. Occurrence of distant metastases was not signiicantly correlated with anemia. This inding is similar to
previous study which stated that metastases did not
have signiicant correlation with nadir of hemoglobin
post chemotherapy with cisplatin10. On the other side, a recent study16 mentioned that occurrence of metastases was an independent risk factors of severe anemia on patients treated with cytotoxic chemotherapy (OR = 2.8; 95% CI = 1.2–7.1). Those difference may be caused by different characteristics of subjects included. In this study, the most prominent decline of hemoglobin
occurred after irst course of chemotherapy, while in
previous study, hemoglobin level fell most markedly after third course of cisplatinbased chemotherapy.6
Baseline hemoglobin level 13 g/dL and less was reported to correlate with risks of severe chemotherapy associated anemia.16 Similar inding was noted in this
study, where hemoglobin level ≤ 13 g/dL was deined
as risk factor for anemia (RR = 4.2; 95% CI = 1.99.4). Several other studies also establish baseline hemoglobin level as risk factor for anemia with different cutoffs, such as hemoglobin level less than 12 g/dL18 and low normal hemoglobin level, i.e. 12.013.4 g/dL for male and 12.012.9 g/dL for female,19 as independent risk factor of chemotherapyinduced anemia.
Patients with age older than 55 years have greater risk of developing anemia (RR = 2.2; 95% CI = 1.24.0).
The results conirmed that advanced age was a risk
factor for cisplatininduced anemia, as stated by some previous studies. In a study evaluating risk factors of cisplatinassociated anemia,10 it was reported that the nadir of hemoglobin throughout chemotherapy
with cisplatin was signiicantly lower in patients
with age older than 60 years old (P < 0.05), while in National Health And Nutrition Examination Survey III
(NHANES III), prevalence of anemia is signiicantly
higher in age population more than 65 years old.20 Another study18 reported no association between severe anemia requiring transfusions (SAART) with age in patients undergoing cytotoxic chemotherapy.
signiicant correlation with severe anemia (P < 0.05), but was not identiied as independent risk factor. The present study has similar inding, female sex was
correlated with anemia (RR = 2.0; 95% CI = 1.13.8) but was not an independent predictor in multivariate
analysis. Another study observed no signiicant
relationship between sex and nadir of hemoglobin after
irst course of chemotherapy with cisplatin.10
Baseline creatinine clearance less than 50 mL/min
was correlated signiicantly with incidence of anemia
(RR = 2.9; 95% CI = 1.75.1). As a comparison, from the data obtained through NHANES III survey, it was reported16 that at creatinine clearance level below 50
mL/min, a signiicant decreace in hemoglobin level was
observed in both males and females, only in males this fall of hemoglobin level started at creatinine clearance
level less than 70 mL/min. This inding suggested
a correlation between sex and baseline creatinine clearance as risk factor of anemia.
In regards of previous studies reporting a relationship between age and sex with baseline hemoglobin level and baseline creatinine clearance,16,17 stratiication analysis were performed to eliminate confounding factors. As the results, both age and sex of patients have confounding effects towards the correlations between baseline hemoglobin level with anemia, and baseline creatinine clearance with anemia as well. For baseline hemoglobin, confounding were only slightly observed, that the adjusted risk calculated was not notably different from initial risk (aRR = 4.1; 95% CI = 1.710.0) and
the correlation with anemia remained signiicant (P <
0.001). In contrast, adjusted risk for baseline creatinine clearance was considerably lower than initial risk (aRR = 2.7; 95% CI = 1.07.6) that the correlation with
anemia became insigniicant (P > 0.05). This implied
creatinine clearance as a weak risk factor.
In multivariate analysis with logistic regression, only
baseline hemoglobin level ≤ 13 g/dL (OR = 9.,6; 95%
CI = 2.733.3) and baseline creatinine clearance < 50
mL/min (OR = 9.0; 95% CI = 1.1-73.6) were identiied
as independent predictors, with baseline hemoglobin level as predominant risk factor (P < 0.001). These
results were equivalent with prior indings10 that reported baseline hemoglobin level as independent
predictor of nadir of hemoglobin after irst course of
cisplatinbased chemotherapy, and the recent cohort11 that denoted baseline hemoglobin level less than 13 g/dL as independent risk factor of chemotherapy
associated anemia (OR = 0.55; 95% CI 0.39–0.77). With different cutoffs, other study also reported that
hemoglobin level was identiied as independent risk
factor of severe anemia following chemotherapy (OR = 14.0; 95% CI = 7.030.0). A recent survey in Australia19 also stated that low normal hemoglobin level was a risk factor of anemia in chemoterapy patients (OR = 5.4; 95% CI = 2.7–10.9).
Most recent study indicated that maintenance of hemoglobin level between 11 to 12 mg/dL is
signiicantly correlated with increased quality of life in
cancer patients.21 This inding gave rise to preventive efforts in order to maintain the level of hemoglobin in
desired range. Identiication of risk factors is thereby important for high-risk patients who would beneit
most from prophylaxis for anemia.9
There were limitations in this study, such as the predicted value instead of measured actual creatinine clearance, the elimination of risk from both combining
cytotoxic agents and dose difference, and the deinition
of anemia that was limited to decrease of hemoglobin level instead of patients’ functional state. For consideration, earlier study reported that there was a
signiicant correlation between the rise of hemoglobin
level and improvement of quality of life in cancer patients,22 hence identiication of risk factor of anemia will indirectly promote the preservation of quality of life by maintaining hemoglobin level in desired range, i.e. above 11 g/dL.21
It is concluded that hemoglobin level in cancer patients undergoing cisplatinbased chemotherapy fell
signiicantly after irst course of chemotherapy, and
continue to decrease afterward. Baseline hemoglobin level of 13 g/dL and less and baseline creatinine clearance less than 50 mL/min are independent
predictors of cisplatin-induced anemia. These indings
suggest that both hemoglobin level and creatinine clearance are useful to recognize highrisk patients who
might need speciic treatment for anemia in the future.
By contrary, prevention of cisplatininduced anemia may be accomplished by modifying these baseline characteristic of patients. However, futher studies are needed to establish particular management of cisplatin based anemia based on individual risk assessment.
REFERENCES
Miller CB, Platanias LC, Mills SR, Zahurak ML, Ratain 1.
the treatment of cisplatinassociated anemia. J Natl Cancer Inst 1992; 84: 98103.
Groopman JE, Itri LM. Chemotherapyinduced anemia in 2.
adults: Incidence and treatment. J Natl Cancer Inst 1999; 91: 161634.
Pivot X, Guardiola E, Etienne M, Thyss A, Foa C, Otto J, 3.
et al. An analysis of potential factors allowing an individual prediction of cisplatininduced anaemia [abstract]. Eur J Cancer 2000; 36: 8527.
Locatelli MC, Tedeschi L, Clerici M, Romanelli A, 4.
D’Antona A, Labianca R, et al. Cisplatinassociated anaemia in patients with solid tumours: A retrospective evaluation and considerations relative to erythropoietin administration. Support Care Cancer 1996; 4: 2189. Dufour P, Bergerat JP, Eber M, Renaud P, Karcher V, Giron 5.
C, et al. Cisplatininduced anemia: a potential interference with iron metabolism at erythroid progenitors level [abstract]. Anticancer Drugs 1990; 1: 4954.
Wood PA, Hrushesky WJM. Cisplatinassociated anemia: 6.
An erythropoietin deiciency syndrome. J Clin Invest 1995; 95: 16509.
Lee SJ, Kwon JH, Jung CW. Erythropoietin response is 7.
inadequate in cancer patients receiving chemotherapy. Int J Hematol 2001; 74: 41620.
Chu E, Einhorn LH, Lefebvre P. Clinical beneits of once-8.
weekly epoetin alfa in anemic patients with colorectal cancer receiving chemotherapy. J Support Oncol 2006; 4: 24350.
Del Mastro L, Venturini M. Strategies for the use of epoetin 9.
alfa in breast cancer patients. The Oncologist 1998; 3: 3148.
Okamoto H, Saijo N, Shinkai T, Eguchi K, Sasaki Y, 10.
Tamura T, et al. Chemotherapyinduced anemia in patients with primary lung cancer [abstract]. Annl Oncol 1992; 3: 81924.
Juan O, Albert A, Almenar D, Olmos S, Vidal J, Molins C, 11.
et al. Risk model for severe anemia in patients with non hematologic cancer receiving conventional chemotherapy: Results from a multicenter prospective cohort study [abstract]. J Clin Oncol 2005; 23(Pt.I): 8121.
Mann CJ. Observational research methods. Research design 12.
II: cohort, cross sectional, and casecontrol studies. Emerg Med J 2003; 20: 5460.
Kurnianda J, Pardjono E, Adiwijono A, et al. Combination of 13.
gemcitabine and cisplatin (GC) chemotherapy for advanced or recurrent nasopharyngeal cancer (NPC): A phase II study. J Clin Oncol 2004; 22(14 Suppl): 5577. Presented on the 2004 ASCO Annual Meeting, New Orleans, 2004.
Widayati K, Kurnianda J, Purwanto I, et al. Evaluation 14.
of nasopharyngeal cancer neoadjuvant standing treatment protocol 4 cycles of cisplatin5 FU followed by EBRT at dr Sardjito Hospital Yogyakarta Indonesia. Presented on the East West Symposium on Nasopharyngeal Cancer, Toronto, June 1618, 2005.
Cockcroft DW, Gault MH. Prediction of creatinine clearance 15.
from serum creatinine. Nephron 1976; 16: 3141.
Hsu CY, McCulloch CE, Curhan GC. Epidemiology of 16.
anemia associated with chronic renal insuficiency among adults in the United States: Results from the Third National Health and Nutrition Examination Survey. J Am Soc Nephrol 2002; 13: 50410.
Guralnik JM, Eisenstaedt RS, Ferrucci L, et al. Prevalence 17.
of anemia in persons 65 years and older in the United States: Evidence for a high rate of unexplained anemia. Blood 2004; 104: 22638.
RayCoquard I, Le Cesne A, Rubio MT, et al. Risk model 18.
for severe anemia requiring red blood cell transfusion after cytotoxic conventional chemotherapy regimens. J Clin Oncol 1999; 17: 28406.
Seshadri T, Prince HM, Bell DR, et al The Australian 19.
Cancer Anaemia Survey: A snapshot of anaemia in adult patients with cancer. MJA 2005; 182: 4537.
Balducci L. Anemia, cancer, and aging. Cancer Control 20.
2003; 10: 47886.
NCCN Clinical Practice Guidelines in Oncology: Cancer 21.
and TreatmentRelated Anemia [monograph online]. Rodgers GM, Cella D, ChananKhan A, et al. National Comprehensive Cancer Network, Inc, producers. Version 2.2006. Utah: NCCN; 2006 [cited 2006 Dec 15]. Available from: URL:http://www.nccn.org.
Sabbatini P. The relationship between anemia and quality 22.