JABATAN PELAJARAN MELAKA JABATAN PELAJARAN MELAKA BUKIT BARU, MELAKA
BUKIT BARU, MELAKA
PEPERIKSAAN AKHIR TAHUN
PEPERIKSAAN AKHIR TAHUN
TINGKATAN
TINGKATAN 4
4 2011
2011
CHEMISTRY CHEMISTRY Kertas 1 Kertas 1 Oktober 2011 Oktober 20114541/1
4541/1
11 ¼ ¼ jamjam Satu jam lima belas minitSatu jam lima belas minit
JANGAN BUKA KERTAS SOALAN INI SEHINGGA DIBERITAHU JANGAN BUKA KERTAS SOALAN INI SEHINGGA DIBERITAHU
1.
1. Kertas soalan ini Kertas soalan ini mengandungmengandungii 50 50 soalan.soalan. 2.
2. Jawab Jawab semua semua soalan.soalan. 3.
3. Jawab deng Jawab dengan menghitamkan an menghitamkan ruangan yang ruangan yang betul pada kerbetul pada kertas jawapan.tas jawapan. 4.
4. Hitamkan Hitamkan satu satu ruangan sahaja bagi setiap soalan.ruangan sahaja bagi setiap soalan. 5.
5. Rajah tidak dilukis me Rajah tidak dilukis mengikut skalangikut skala kecuali kecuali dinyatakandinyatakan 6.
6. Anda dibenAnda dibenarkan mengarkan menggunakan kgunakan kalkulator saintifik yalkulator saintifik yang tidak bang tidak boleh diprograoleh diprogramkanmkan
Kertas soalan ini mengandungi 25 halaman bercetak Kertas soalan ini mengandungi 25 halaman bercetak
INFORMATION FOR CANDIDATES INFORMATION FOR CANDIDATES 1.
1. This question paper consists of 50 This question paper consists of 50 questions.questions. 2.
2. Answer Answer allall questionsquestions.. 3.
3. Answer eac Answer each question by blach question by blackening the corkening the correct space on rect space on the answer shethe answer sheet et .. 4.
4. Blacken only Blacken only oneone space for each question.space for each question. 5.
5. If you wish to chan If you wish to change your ansge your answer, erase wer, erase the blackened mthe blackened mark that you havark that you have made.e made. Then blacken the space for the new
Then blacken the space for the new answer.answer. 6.
6. The diagrams in the questions provided are not The diagrams in the questions provided are not drawn to scale unless stated drawn to scale unless stated .. 7.
7. You may use a You may use a non-programmnon-programmable scientific calculator.able scientific calculator.
MAKLUMAT UNTUK CALON MAKLUMAT UNTUK CALON 1.
1. Kertas soalan ini mengandungi 50 soalanKertas soalan ini mengandungi 50 soalan .. 2.
2. Jawab Jawab semuasemua soalansoalan 3.
3. Jawab denga Jawab dengan menghitamkan rn menghitamkan ruangan yang uangan yang betul pada kertas betul pada kertas jawapanjawapan .. 4.
4. Hitamkan Hitamkan satusatu ruangan sahaja bagi setiap soalanruangan sahaja bagi setiap soalan.. 5.
5. Sekiranya anda hendak menukarkan jawapan, padamkan tanda yang telah Sekiranya anda hendak menukarkan jawapan, padamkan tanda yang telah dibuat.dibuat. Kemudian hitamkan jawapaan yang baru.
Kemudian hitamkan jawapaan yang baru. 6.
6. Rajah yang me Rajah yang mengiringi soalan tidak dngiringi soalan tidak dilukis mengikut skilukis mengikut skala kecuali dinyatakala kecuali dinyatakan.an. 7.
7. Anda dibenar Anda dibenarkan menggunakan menggunakan kalkulator skan kalkulator saintifik yang tidak boleh aintifik yang tidak boleh diprogramkandiprogramkan
Question 1
Question 1 toto Question 50Question 50 are followed by four optionsare followed by four options A, B, CA, B, C or or D.D. Choose the best option for
Choose the best option for each question and blackened the corresponding space oneach question and blackened the corresponding space on the objective answer sheet.
the objective answer sheet. Bagi
Bagi Soalan 1Soalan 1 hinggahingga Soalan 50Soalan 50,, tiap-tiap soalan diikuti oleh empat pilihan jawapantiap-tiap soalan diikuti oleh empat pilihan jawapan A, B,A, B, C
C dandan DD.. Pilih satu jawapan yang terbaik bagi Pilih satu jawapan yang terbaik bagi tiap-tiap soalan dan hitamkan ruangan yangtiap-tiap soalan dan hitamkan ruangan yang sepadan pada kertas jawapan objektif anda
INFORMATION FOR CANDIDATES INFORMATION FOR CANDIDATES 1.
1. This question paper consists of 50 This question paper consists of 50 questions.questions. 2.
2. Answer Answer allall questionsquestions.. 3.
3. Answer eac Answer each question by blach question by blackening the corkening the correct space on rect space on the answer shethe answer sheet et .. 4.
4. Blacken only Blacken only oneone space for each question.space for each question. 5.
5. If you wish to chan If you wish to change your ansge your answer, erase wer, erase the blackened mthe blackened mark that you havark that you have made.e made. Then blacken the space for the new
Then blacken the space for the new answer.answer. 6.
6. The diagrams in the questions provided are not The diagrams in the questions provided are not drawn to scale unless stated drawn to scale unless stated .. 7.
7. You may use a You may use a non-programmnon-programmable scientific calculator.able scientific calculator.
MAKLUMAT UNTUK CALON MAKLUMAT UNTUK CALON 1.
1. Kertas soalan ini mengandungi 50 soalanKertas soalan ini mengandungi 50 soalan .. 2.
2. Jawab Jawab semuasemua soalansoalan 3.
3. Jawab denga Jawab dengan menghitamkan rn menghitamkan ruangan yang uangan yang betul pada kertas betul pada kertas jawapanjawapan .. 4.
4. Hitamkan Hitamkan satusatu ruangan sahaja bagi setiap soalanruangan sahaja bagi setiap soalan.. 5.
5. Sekiranya anda hendak menukarkan jawapan, padamkan tanda yang telah Sekiranya anda hendak menukarkan jawapan, padamkan tanda yang telah dibuat.dibuat. Kemudian hitamkan jawapaan yang baru.
Kemudian hitamkan jawapaan yang baru. 6.
6. Rajah yang me Rajah yang mengiringi soalan tidak dngiringi soalan tidak dilukis mengikut skilukis mengikut skala kecuali dinyatakala kecuali dinyatakan.an. 7.
7. Anda dibenar Anda dibenarkan menggunakan menggunakan kalkulator skan kalkulator saintifik yang tidak boleh aintifik yang tidak boleh diprogramkandiprogramkan
Question 1
Question 1 toto Question 50Question 50 are followed by four optionsare followed by four options A, B, CA, B, C or or D.D. Choose the best option for
Choose the best option for each question and blackened the corresponding space oneach question and blackened the corresponding space on the objective answer sheet.
the objective answer sheet. Bagi
Bagi Soalan 1Soalan 1 hinggahingga Soalan 50Soalan 50,, tiap-tiap soalan diikuti oleh empat pilihan jawapantiap-tiap soalan diikuti oleh empat pilihan jawapan A, B,A, B, C
C dandan DD.. Pilih satu jawapan yang terbaik bagi Pilih satu jawapan yang terbaik bagi tiap-tiap soalan dan hitamkan ruangan yangtiap-tiap soalan dan hitamkan ruangan yang sepadan pada kertas jawapan objektif anda
1.
1. Which Which of of the the following following statements statements best best describes describes ‘Chemistry’?‘Chemistry’? Antara pernya
Antara pernyataan berikut, yantaan berikut, yang manakah terg manakah terbaik menerangbaik menerangkan ‘kimia’ ?kan ‘kimia’ ? A
A Chemistry is the study Chemistry is the study gained through scientific investigations.gained through scientific investigations. Kimia ialah kajian
Kimia ialah kajian diperoleh melalui penyiasatan saintifik.diperoleh melalui penyiasatan saintifik. B
B Chemistry is a sChemistry is a systemic study of natural phenomena.ystemic study of natural phenomena. Kimia ialah kajian
Kimia ialah kajian sistemik bagi fenomena semulajadi.sistemik bagi fenomena semulajadi. C
C Chemistry is the study of the composition, structure, properties and interactions of Chemistry is the study of the composition, structure, properties and interactions of matter.
matter.
Kimia ialah kajian mengenai komposisi, struktur, sifat-sifat dan interaksi antara Kimia ialah kajian mengenai komposisi, struktur, sifat-sifat dan interaksi antara jirim.
jirim. D
D Chemistry is an art of transforming metals.Chemistry is an art of transforming metals. Kimia ialah satu
Kimia ialah satu seni perubahan logam.seni perubahan logam.
2.
2. There There are are many many industries industries involve involve chemistry chemistry include include …… Terdapat banyak industri melibatkan kimia termasuk … Terdapat banyak industri melibatkan kimia termasuk … I
I Petroleum Petroleum industryindustry Industri petroleum Industri petroleum II
II Nutritional Nutritional industryindustry Industri pemaka Industri pemakanannan III
III Mining industryMining industry Industri perlombon Industri perlombongangan IV
IV Paint Paint making making industryindustry Industri membuat c Industri membuat cat at A
A I and II onlyI and II only I dan II sahaja I dan II sahaja B
B I, II and III onlyI, II and III only I, II dan III sahaja I, II dan III sahaja C
C I, II and IV onlyI, II and IV only I, II dan IV sahaja I, II dan IV sahaja D
D I, II, III and IVI, II, III and IV I, II, III dan IV I, II, III dan IV
3 Which of the following is the best solution for an arteriosclerosis patient?
Antara berikut yang manakah penyelesaian terbaik bagi pesakit arteriosklerosis? A take food rich in calcium
makan makanan yang kaya dengan kalsium B take food rich in iron
makan makanan yang kaya dengan zat besi C reduce fatty food intake
kurangkan mengambil makanan berlemak D reduce sugary food intake
kurangkan mengambil makanan bergula
4 Which of the following substance exists as atoms?
Antara bahan berikut, yang manakah wujud sebagai atom? A Emas Gold C Water Air B Nitrogen Nitrogen D Chlorine Klorin
5 The melting point of a substance X is -2oC and its boiling point is 75oC. What is the physical state of substance X at -5oC and 85oC?
Takat lebur bahan X ialah -2oC dan takat didihnya ialah 75oC. Apakah keadaan fizikal bahan X pada suhu -50C dan 850C?
At -5oC Pada -5oC At 85oC Pada 85oC A Solid Pepejal Gas Gas B Solid Pepejal Liquid Cecair C Liquid Cecair Gas Gas D Gas Gas Solid Pepejal
6 Diagram 1 below shows an atomic model. Rajah 1 menunjukkan suatu model atom.
Diagram 1 Rajah 1 Which of the following is true about Diagram 1?
Antara penyataan berikut yang manakah benar tentang Rajah 6? A The atomic model above was proposed by Neils Bohr.
Model atom di atas dicadangkan oleh Neils Bohr.
B The atom is a sphere of positive charge which contains a few electrons Atom merupakan sfera bercas positif yang mengandungi sedikit elektron. C The electrons in an atom move in shells around the nucleus.
Elektron dalam atom bergerak dalam petala-petala yang mengelilingi nukleus. D The electrons move in a space that is larger than the space occupied by the nucleus.
Electron bergerak dalam ruang yang lebih besar daripada ruang yang ditempati oleh nukleus.
7 This isotope produces gamma radiation to kill cancer cells. What is the isotope?
Isotop ini menghasilkan sinar gamma untuk membunuh sel kanser. Apakah isotop ini? A Iodine-131 Iodin-131 B Cobalt-60 Kobalt-60 C Sodium-24 Natrium-24 D Carbon-12 Karbon-12
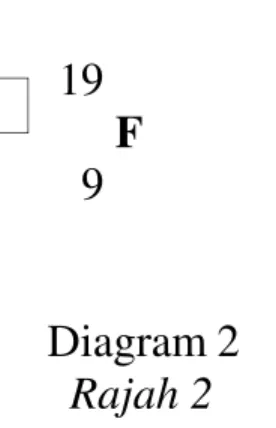
8 Diagram 2 shows the symbol of fluorine atom. Rajah 2 menunjukkan simbol bagi atom fluorin
19 F 9
Diagram 2 Rajah 2
Which of the following is true based on the symbol in Diagram 2.
Antara berikut yang manakah benar berdasarkan simbol dalam Rajah 2.
Proton number Nombor proton Nucleon number Nombor nukleon Number of electron Bilangan elektron A 9 19 9 B 9 19 19 C 9 10 9 D 19 9 9
9 Diagram 3 shows the electron arrangement of atom X. Rajah 3 menunjukkan susunan elektron bagi atom X.
Diagram 3 Rajah 3
How many valence electrons are there in the atom of X?
Berapakah bilangan elektron valens yang terdapat dalam atom X?
A 2 C 5
B 3 D 7
10 Diagram 4 is a graph which shows the cooling curve for gas X.
Rajah 4 ialah satu graf yang menunjukkan lengkung penyejukan bagi gas X.
Diagram 4 Rajah 4
Which of the following statements is true?
Antara pernyataan yang berikut, manakah benar?
A From point P to point Q, kinetic energy increases as temperature decreases. Dari titik P ke titik Q, tenaga kinetik bertambah apabila suhu menurun. B From point Q to point R, forces of attraction between particles become weak
Dari titik Q ke titik R, semua daya tarikan antara zarah-zarah menjadi lemah. C At point R, all the gas X has completely melted
Pada titik R, semua gas X telah melebur keseluruhannya. D From point R to point S , heat is released to the surroundings
Dari titik R ke titik S, haba dibebaskan ke persekitaran
11 The following equation represents the chemical process to extract aluminium from aluminium oxide.
Persamaan dibawah menunjukkan proses kimia untuk mengekstrak aluminium dari aluminium oksida
2 Al2O3 → 4 Al + 3 O2
What is the volume of oxygen gas evolved when 204 g of aluminium oxide is used in the extraction ?
Berapakah isipadu gas oksigen yang terbebas apabila 204 g aluminium oksida digunakan di dalam proses ini?
[ Relative atomic mass ; O = 16 ; Al = 27, 1 mol of gas occupies 24.0 dm3 at room temperature ]
[Jisim atom relatif ; O = 16, Al = 27, 1 mol gas menempati 24.0 dm3 pada suhu bilik.]
A 36 dm C 48 dm
12 Diagram 5 shows the electron arrangement of a molecule of compound XY2. ( Only the valence electrons are shown ).
Rajah 5 menunjukkan susunan elektron bagi molekul sebatian XY 2 (hanya elektronvalens yang ditunjukkan).
Diagram 5 Rajah 5 X and Y could be : X dan Y mungkin : X Y A Carbon Oxygen B Carbon Chlorine C Carbon Hydrogen D Chlorine Carbon
13 Which of the following represents the electron arrangement of a noble gas? Antara berikut, yang manakah mewakili susunan elektron gas adi?
A C B D R R R R
14 Diagram 6 shows the set-up of the apparatus to determine the empirical formula of substance Y.
Rajah 6 menunjukkan susunan radas untuk menentukan formula empirik bahan Y.
Diagram 6
Rajah 6
What could be substance Y ?
Apakah mungkin bahan Y?
I Copper(II) oxide
Kuprum(II) oksida
II Magnesium oxide
Magnesium oksida
III Potassium oxide
Kalium oksida
IV Lead (II) oxide
Plumbum (II) oksida A I and IV only
I and IV sahaja B II and III only
II and III sahaja C III and IV only
III and IV sahaja D I, II, III and IV
I, II, III and IV
15 Table 1 shows the mass of element P and Q which react with each other. Jadual 1 menunjukkan jisim bagi unsur P dan Q yang bertindak balas.
Element Unsur P Q Mass(g) Jisim(g) 0.32 0.36 Table 1 Jadual 1
What is the empirical formula for the substance formed? Apakah formula empirik bagi sebatian yang terbentuk ?
[Relative atomic mass :P = 64 ; Q = 35.5] [ Jisim atom relatif: P =64; Q = 35.5]
A PQ C PQ4 B PQ2 D P2Q3 SubstanceY Bahan Y Hydrogen gas Gas hidrogen
16 2.7 g of element X reacts with 2.4 g element Y to form a substance with empirical formula of X2Y3. What is the atomic mass of Y?
2.7 g unsur X bertindak balas dengan 2.4 g unsur Y untuk membentuk sebatian dengan formula empirik X 2Y 3. Berapakah jisim atom relatif bagi Y?
(Relative atomic mass : X = 27) ( Jisim atom relatif : X = 27)
A 8 C 24
B 16 D 31
17 The chemical symbol of an element T is as shown : Simbol unsur T ditunjukkan seperti di bawah:
What would be the electron arrangement of the ion formed by an atom of T ? Apakah susunan elektron bagi ion yang terbentuk oleh atom T?
A 2.8
B 2.8.2
C 2.8.8
D 2.8.8.8
18 Which of the following elements are located in group 17 of the Periodic Table?
Manakah antara unsur-unsur berikut diletakkan di dalam kumpulan 17 Jadual Berkala? A Neon and Argon
Neon dan Argon
B Lithium and Potassium Litium dan Kalium C Iodine and Bromine
Iodin dan Bromin
D Nitrogen and Phosphorus Nitrogen dan Fosforus
24
T
19 The following statement is about the arrangement of the elements in the Periodic Table of elements.
Pernyataan berikut adalah mengenai susunan unsur di dalam Jadual Berkala Unsur .
Which of the following scientist made the above statement?
Antara saintis berikut siapakah yang membuat pernyataan di atas?
A Newlands
B Meyer
C Mendeleev
D Dobereiner
20 Table 2 shows the proton number and number of electrons of several particles. Which of the following particles is an anion?
Jadual 2 menunjukkan nombor proton dan bilangan elektron bagi beberapa zarah. Manakah di antara berikut merupakan anion?
Particles Zarah Proton Number Nombor Proton Number of electrons Bilangan elektron P 3 2 Q 9 10 R 11 11 S 20 18 Table 2 Jadual 2 A P B Q C R D S
“Elements are arranged in order of increasing proton number in The Periodic Table. Unsur-unsur disusun mengikut nombor proton menaik dalam Jadual Berkala.”
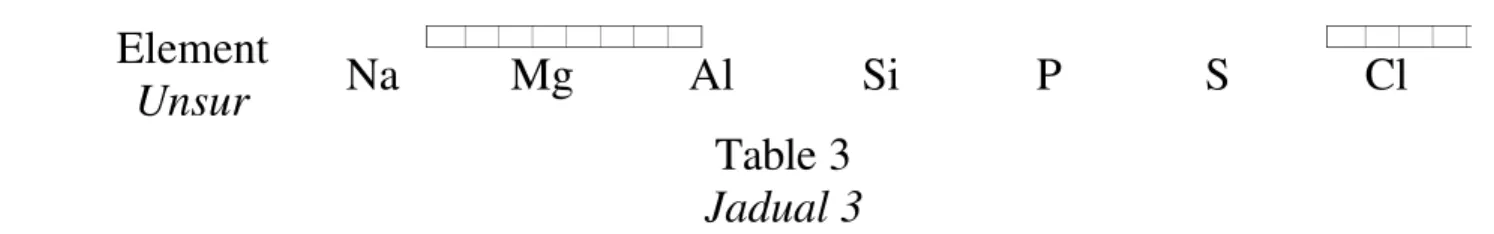
21 Table 3 shows the elements in Period 3 of the Periodic Table of elements. The elements can react with oxygen to form oxides.
Jadual 3 menunjukkan unsur-unsur Kala 3 dalam Jadual Berkala Unsur. Unsur-unsur tersebut bertindak balas dengan oksigen membentuk oksida.
Element
Unsur Na Mg Al Si P S Cl
Table 3 Jadual 3
Which of the following element forms amphoteric oxide? Antara berikut unsur manakah membentuk oksida amfoterik?
A S C Si
B P D Al
22 The number of neutrons of atom X is 20. If the number of electrons of ion X + is 18, what is the relative atomic mass of X?
Bilangan neutron bagi suatu atom X ialah 20. Jika bilangan elektron ion X 2+ ialah 18, berapakah jisim atom relatif X?
A 18 C 38
B 20 D 40
23 Which of the following is true about elements in the Periodic Table?
Antara berikut yang manakah benar tentang unsur-unsur dalam Jadual Berkala? A The metallic properties increases from left to right across a period
Sifat kelogaman bertambah apabila merentasi Jadual Berkala dari kiri ke kanan B Elements of the same group have the same physical properties
Unsur dalam kumpulan yang sama mempunyai sifat fizik yang sama
C Group 18 elements have low melting and boiling points
Unsur kumpulan 18 mempunyai takat lebur dan takat didih yang rendah D Group 17 elements exist naturally as monoatoms
24 What happens to a sodium atom when chlorine combines with sodium?
Apakah yang berlaku kepada atom natrium apabila klorin berpadu dengan natrium? A Accepts an electron
Menerima satu elektron
B Donates an electron Menderma satu elektron C Accepts two electrons
Menerima dua elektron D Shares a pair of electrons
Berkongsi sepasang elektron
25 Sodium and sulphur have the proton number 11 and 16 respectively. They react to form sodium sulphide. Which of the following statements are true?
Natrium dan sulfur masing-masing mempunyai nombor proton 11 dan 16. Mereka bertindakbalas membentuk natrium sulfida. Antara pernyataan berikut, yang mana adalah benar?
I A sodium atom donates an electron to form a sodium ion
Satu atom natrium menderma satu elektron untuk membentuk ion natrium II A sodium atom donates two electrons to the sulphur atom
Satu atom natrium menderma dua elektron kepada atom sulfur III A sulphur atom receives two electrons to form a sulphide ion
Satu atom sulfur akan menerima dua elektron untuk membentuk ion sulfida IV Sodium and sulphur combine to form an ionic compound
Natrium dan sulfur bergabung untuk membentuk sebatian ion.
A I and IV only I dan IV sahaja B I, II and III only
I, II dan III sahaja
C I, III and IV only I, III dan IV sahaja D II, III and IV only
26 Table 4 shows the electron arrangement of atoms G, H, J and K Jadual 4 menunjukkan susunan elektron bagi atom G,H, J dan K
Atom G H J K Electron arrangement Susunan elektron 1 2.5 2.8.2 2.8.8.1 Table 4 Jadual 4
Which of the following pairs of elements can combine to form a covalent compound? Yang manakah antara pasangan unsur-unsur berikut boleh membentuk sebatian kovalen?
A G and H
B H and J
C G and K
D H and K
27 Which of the following set of apparatus is the most suitable to be used in electrolysis of copper(II) sulphate solution?
Antara susunan radas yang berikut, yang manakah paling sesuai digunakan dalam proses elektrolisis larutan kuprum(II) sulfat ?
A B C D Carbon rod Rod karbon Copper(II) sulphate solution Larutan
ku rum(II) sul at Carbon rod
Rod karbon Copper(II)
sulphate solution Larutan
28 Table 5 contains information on the melting points and boiling points of substances P, Q, R and S.
Jadual 5 mengandungi maklumat tentang takat lebur dan takat didih bagi bahan-bahan P,Q,R dan S Substance Bahan Melting point (ºC) Takat lebur (ºC) Boiling point (ºC) Takat didih (ºC) Electrical conductivity in molten state P -30 174 Cannot conduct Q 43 182 Cannot conduct R 119 444 Can conduct S 688 1100 Can conduct Table 5 Jadual 5
Which of the following classifications of substances P, Q, R and S is correct? Antara pengkelasan berikut, yang manakah benar bagi bahan P, Q, R dan S?
Ionic compound Sebatian ion Covalent compound Sebatian kovalen A P and Q R and S B R and S P and Q C P, Q and R S D S P
29 What is the percentage of nitrogen by mass in ammonium nitrate, NH4NO3? Apakah peratus nitrogen dalam ammonium nitrat NH 4 NO3 mengikut jisim?
[ Relative atomic mass: H = 1, N = 14 ,O = 16] [ Jisim atom relatif : H = 1, N = 14, O = 16]
A 17.50 %
B 21.21 %
C 35.00 %
30 Aqueous sodium chloride solution is an electrolyte because it has
Larutan natrium klorida berair ialah sejenis elektrolit kerana ia mempunyai A Free moving ions
Ion yang bebas bergerak B Free moving electrons
Elektron yang bebas bergerak C Free moving atom
Atom yang bebas bergerak D Free moving molecules
Molekul yang bebas bergerak
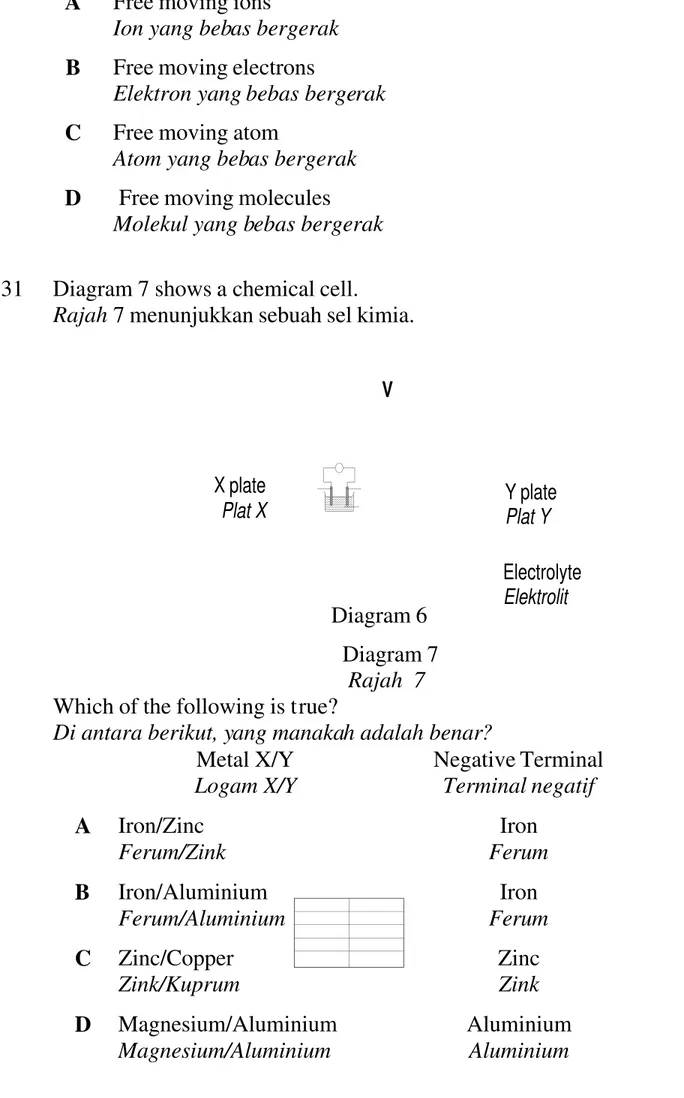
31 Diagram 7 shows a chemical cell.
Rajah 7 menunjukkan sebuah sel kimia.
Diagram 6 Diagram 7 Rajah 7 Which of the following is true?
Di antara berikut, yang manakah adalah benar? Metal X/Y Logam X/Y Negative Terminal Terminal negatif A Iron/Zinc Ferum/Zink Iron Ferum B Iron/Aluminium Ferum/Aluminium Iron Ferum C Zinc/Copper Zink/Kuprum Zinc Zink D Magnesium/Aluminium Magnesium/Aluminium Aluminium Aluminium Y plate Plat Y Electrolyte Elektrolit V X plate Plat X
32 Diagram 8 shows the set–up of apparatus of a chemical cell. Which of the following are not true of a chemical cell? Rajah 8 menunjukkan susunan radas suatu sel kimia.
Antara berikut yang manakah tidak benar mengenai sel kimia?
Diagram 8 Rajah 8 I The electrodes are of the same metal.
Elektrod-elektrod adalah logam yang sama. II The electrodes are of different metals
Elektrod-elektrod adalah logam yang berlainan. III Liquid H is an electrolyte.
Cecair H ialah sejenis elektrolit .
IV Electrical energy change to chemical energy Tenaga elektrik bertukar kepada tenaga kimia A I and III only
B I and IV only C II and IV only D I, III and IV only
33 Which of the following substances is acidic?
Antara bahan berikut yang manakah bersifat asid?
A Ammonia Ammonia B Potassium oxide Kalium oksida C Carbon dioxide Karbon dioksida D Sodium hydroxide Natrium hidroksida Electrodes Elektrod Liquid H Cecair H V
34 It is discovered that a compound with formula AB dissolves in water. What inference can you make about the compound ?
Didapati satu sebatian dengan formula AB larut dalam air. Apakah inferens yang boleh dibuat tentang sebatian itu?
A It is a covalent compound Merupakan sebatian kovalen B The compound ionizes in water
Sebatian mengion dalam air
C The compound will dissolve in an organic solvent Sebatian yang larut dalam pelarut organik
D The intermolecular forces between the particles are very weak Daya antara molekul pada zarah-zarah adalah sangat lemah
35 During the electroplating of an iron key with copper, which of the following should be done?
Semasa menyadurkan kunci besi dengan kuprum, yang manakah antara berikut perlu dilakukan?
I The iron key should be made the cathode Kunci besi perlu dijadikan katod
II Molten copper is used as the electrolyte Kuprum lebur dijadikan elektolit
III An aqueous solution of copper (II) sulphate is made the electrolyte Larutan berair kuprum (II) sulfat dijadikan elektrolit
IV Two copper plates should be used as electrodes Dua kepingan kuprum digunakan sebagai elektrod
A I and III only B II and IV only C I, II and IV only D I, III and IV only
36 Diagram 9 shows the set-up of the apparatus of an electrolytic cell. Rajah 9 menunjukkan susunan radas bagi satu sel elektrolisis.
Which of the following half-equations represents the reaction that occurred at electrode Q? Antara persamaan setengah berikut, yang manakah mewakili tindak balas yang berlaku di
elektrod Q?
A Na+ + e- →Na
B 2H+ + 2e- →H2
C 2Cl- → Cl2 + 2e
-D 4OH- → O2 + 2H2O + 4e
-37 Which of the following equations does not involve neutralisation?
Antara persamaan kimia berikut yang manakah tidak melibatkan peneutralan? A 2HCl (aq) + MgO(s) MgCl2(aq) + H2O(l)
B H2SO4(aq) + BaCl2(aq) BaSO4(s) + 2HCl(aq)
C HNO3(aq) + NaHCO3(s) NaNO3(aq) + CO2(g) + H2O(l) D HCl(aq) + NH3(aq) NH4Cl(aq)
Diagram 9 Rajah 9
38 Diagram 10 below shows the set-up of apparatus for electrolysis. Rajah 10 di bawah menunjukkan susunan radas untuk elektrolisis.
Diagram 10 Rajah 10
Which of the following substance could light up the bulb?
Antara bahan berikut, yang manakah boleh menyalakan mentol? A Potassium chloride solution
Larutan kalium klorida B Glucose solution Larutan glukosa C Magnesium oxide Magnesium oksida D Tetrachloromethane solution Larutan tetraklorometana
39 Table 6 shows the degree of dissociation of four solutions of alkalis which have the same concentration.
Jadual 6 di bawah menunjukkan darjah penceraian empat larutan alkali yang sama kepekatan. Solution Larutan Degree of dissociation Darjah penceraian W Low Rendah X Medium Sederhana Y High Tinggi Z Very high Sangat tinggi Table 6 Jadual 6 Which solution has the highest pH value?
Larutan yang manakah mempunyai nilai pH yang paling tinggi?
A W C Y
B X D Z
Substance bahan
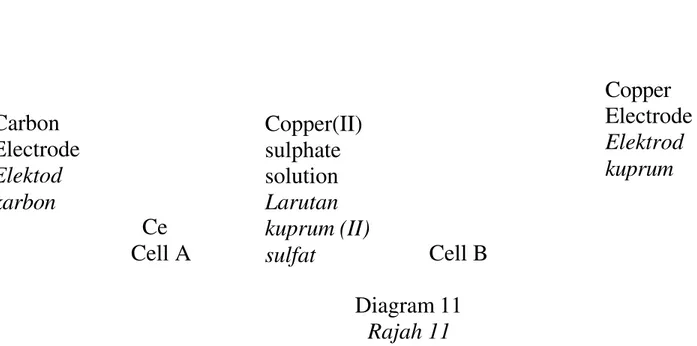
40 Diagram 11 shows two electrolytic cells which use two different type of electrodes, cell A uses carbon electrode while cell B uses copper electrode.
Rajah 11 menunjukkan dua sel elektolisis yang mana menggunakan dua jenis elektrod yang berlainan. Sel A mengunakan elektrod karbon manakala sel B mengunakan elektrod kuprum.
Ce
Cell A Cell B
Diagram 11 Rajah 11
What are the half equation that represented the reaction at anode for both cells?
Apakah setengah persamaan yang mewakili tindak balas di anod pada kedua-dua sel?
Cell A Sel A Cell B Sel B A 4OH-→ 2H2O + O2+ 4e- Cu ++ 2e-→Cu B 4OH-→ 2H2O + O2+ 4e- Cu →Cu ++ 2e -C Cu ++ 2e-→ Cu 4OH-→ 2H2O + O2+ 4e -D Cu →Cu ++ 2e- 4OH-→ 2H2O + O2+ 4e
-41 What is the mass of sodium hydroxide, NaOH which is required to prepare 250 cm sodium hydroxide solutions with a concentration of 100 gdm-3?
[Relative atomic mass: Na = 23; O = 16; H = 1]
Berapakah jisim natrium hidroksida, NaOH yang diperlukan untuk menyediakan 250 cm3 larutan natrium hidroksida dengan kepekatan 100 gdm-3?
[ Jisim atom relatif Na = 23; O = 16; H = 1]
A 25g C 20g B 35g D 40g Copper(II) sulphate solution Larutan kuprum (II) sulfat Copper Electrode Elektrod kuprum Carbon Electrode Elektod karbon
42 50.0 cm of 0.4 moldm- potassium hydroxide solution, KOH is titrated with sulphuric acid, H2SO4. What is the volume of 1.0 moldm-3 sulphuric acid which is needed to neutralise this potassium hydroxide solution?
50.0 cm3 larutan kalium hidroksida, KOH 0.4 moldm-3 dititratkan dengan asid sulfurik, H 2SO4. Berapakah isipadu asid sulfurik 1.0 moldm-3 yang diperlukan untuk meneutralkan
larutan kalium hidroksida ini?
A 10.0 cm C 40.0 cm
B 20.0 cm D 50.0 cm
43 Which of the statements are true about a weak alkali?
Antara pernyataan berikut, yang manakah benar tentang alkali lemah? I It dissociates partially into hydroxide ions in water
Ia terurai separa kepada ion hidroksida di dalam air II Its pH value is higher than that of sodium hydroxide
Nilai pH nya adalah lebih tinggi daripada nilai pH natrium hidroksida III It has low OH- ion concentration
Ia mempunyai kepekatan ion hidroksida yang rendah IV Most of its molecules remain unchanged in the water
Kebanyakan molekulnya kekal tidak berubah di dalam air A I and II only
B I, II and III only C I, III and IV only D II, III and IV only
44 The formula for a sulphate ion is SO4 - and for a nitrate ion is NO3- . If the formula of the sulphate salt of M is MSO4, what is the formula of nitrate salt of M?
Formula bagi sulfat ion adalah SO42- dan bagi nitrat ion adalah NO3-. Jika formula bagi garam M sulfat adalah MSO4 , apakah formula bagi garam M nitrat?
A MNO3
B M2NO3 C M(NO3)2 D M(NO3)3
45 The following is chemical equation for a reaction. Berikut ialah persamaan kimia bagi satu tindak balas.
CaCO3 + 2HNO3 Ca(NO3)2 + H2O + CO2
Which of the following are the products?
Antara berikut, yang manakah hasil tindak balas? A H2O and CO2
B HNO3 and CO2 C CaCO3 and HNO3
D Ca(NO3)2, H2O and CO2
46 Which of the following salts can be prepared by using double decomposition method?
Yang manakah di antara garam-garam berikut boleh disediakan dengan menggunakan kaedah penguraian ganda dua?
I Zinc chloride Zink klorida II Barium sulphate
Barium sulfat III Sodium nitrate
Natrium nitrat
IV Magnesium carbonate Magnesium karbonat A I and III only
B II and IV only C I, II and III only D II, III and IV only
47 Excess magnesium oxide is reacted with 25.0 cm of 2.0 moldm- nitric acid. Calculate the maximum mass of magnesium nitrate salt formed.
[Relative atomic mass: N=14, 0=16, Mg=24]
Magnesium oksida berlebihan bertindak balas dengan 25.0 cm3 asid nitrik berkepekatan 2.0 moldm-3. Kira jisim bagi garam magnesium nitrat yang terbentuk.
[Jisim atom relatif : N=14, 0=16, Mg=24]
A 7.40 g C 3.70 g
48 When heated, solid S gives off a gas that turns lime water milky. What is solid S?
Apabila dipanaskan, pepejal S membebaskan gas yang menukarkan air kapur menjadi keruh. Apakah pepejal S?
I Sodium carbonate Natrium karbonat II Calcium carbonate Kalsium karbonat III Potassium carbonate
Kalium karbonat IV Magnesium carbonate Magnesium karbonat A II only B II and IV only C I and IV only D II, III and IV only
49 The equation shows a reaction between silver nitrate solution and sodium chloride solution. Persamaan di bawah menunjukkan tindak balas di antara larutan argentum nitrat dan larutan natrium klorida
AgNO3(aq) + NaCl(aq)→ NaNO3(aq) + AgCl(s)
Calculate the mass of silver chloride formed when 0.1 mol of silver nirate solution is added to 0.1 mol of sodium chloride solution?
Hitungkan jisim argentum klorida yang terbentuk apabila 0.1 mol larutan argentum nitrat di tambah ke dalam 0.1 mol larutan natrium klorida?
[ Relative atomic mass : Cl = 35.5; Ag = 108] [ Jisim atom relatif : C =,35.5; Ag =108] A 28.70 g
B 43.05g C 21.50 g D 14.35g
50 Which of the following catalyst is used in the Haber process?
Yang manakah di antara mangkin berikut digunakan dalam proses Haber ? A Platinum Platinum B Nickel Nikel C Iron Besi D Vanadium (V) oxide Vanadium (V) oksida
END OF QUESTION PAPER KERTAS SOALAN TAMAT
Nama Kelas
JABATAN PELAJARAN MELAKA BUKIT BARU, MELAKA
PEPERIKSAAN AKHIR TAHUN
TINGKATAN 4 2011
CHEMISTRY Kertas 2
Oktober 2011
4541/2
2 ½ jam Dua jam tiga puluh minit
JANGAN BUKA KERTAS SOALAN INI SEHINGGA DIBERITAHU
1. Kertas soalan ini mengandungi tiga bahagian: Bahagian A, Bahagian B and Bahagian C. 2. Jawab semua soalan dalam Bahagian A. Tulis
jawapan dalam Bahagian A dalam ruangan
yang disediakan..
3. Jawab satu soalan dalam Bahagian B dan satu soalan dalan Bahagian C. Jawab soalan dalam
Bahagian B dan Bahagian C dengan
terperinci. Anda boleh menggunakan
persamaan kimia, rajah, jadual, graf dan kaedah yang bersesuaian untuk menerangkan jawapan anda.
4. Tunjukkan jalan kerja. Ia dapat membantu anda mendapat markah.
5. Rajah dalam soalan tidak dilukis mengikut skala.
6. Markah yang diperuntukan bagi setiap soalan ditunjukkan dalam kurungan.
7. Anda boleh menggunakan kalkulator saintifik yang tidak diprogramkan.
8. Serahkan kertas jawapan pada akhir waktu peperiksaan.
Kertas soalan ini mengandungi 17 halaman bercetak
Untuk kegunaan pemeriksa
Bahagian No. Markah
A 1 2 3 4 5 6 Jumlah B 7 8 C 9 10 Jumlah Jumlah Markah
Section A [60 marks] Answer all questions Jawab semua soalan
1 Table 1 shows the proton number and nucleon number for atoms P, Q , R and S. Jadual 1 menunjukkan nombor proton dan nombor nukleon bagi atom P, Q, R dan S.
Atom Proton number Nucleon Number
P 16 32 Q 17 35 R 17 37 S 18 40 Table 1 Jadual 1 (a) (i) What is meant by proton number?
Apakah yang dimaksudkan dengan nombor proton ?
...………...…... [ 1 mark ] (ii) What is the number of neutrons in atom P?
Apakah bilangan neutron dalam atom P?
...………...…. [ 1 mark ] (b) (i) Which atoms are isotopes? Explain your answer.
Atom-atom yang manakah adalah isotop? Terangkan jawapan anda.
... ... [ 2 marks ] (ii) Write the isotopes in the form of X .
Tuliskan isotop-isotop dalam bentuk X.
... [ 2 marks ] (c) (i) Write the electron arrangement for atom R.
Tuliskan susunan elektron bagi atom R.
... A
Z A Z
dry hydrogen hidrogen kering
(ii) Draw the electron arrangement of atom R. Lukiskan susunan elektron bagi atom R.
[ 2 marks ] (d) S is an inert gas which chemically unreactive
S merupakan gas lengai yang tidak reaktif secara kimia Explain the above statement.
Terangkan kenyataan di atas.
... [ 1 mark ] 2 Diagram 2 shows the apparatus set-up used in an experiment to determine the empirical formula
for an oxide of lead.
Rajah 2 menunjukkan susunan radas yang digunakan dalam eksperimen bagi menentukan formula empirik bagi oksida plumbum.
oxide of lead oksida plumbum
Diagram 2 Rajah 2
The following data was obtained based on the experiment : Data berikut diperolehi berdasarkan suatu ekperimen:
Mass of an oxide of lead = 4.78 g Jisim oksida plumbum
Mass of lead = 4.14 g
(a) What is meant by empirical formula ?
Apakah yang dimaksudkan dengan formula empirik ?
……….………... ………...
[ 1 mark ] (b) Name one substance that can be used to dry the hydrogen gas.
Namakan bahan yang boleh digunakan untuk mengeringkan gas hidrogen.
... [ 1 mark] (c) Why does the hydrogen gas need to be passed through the combustion tube for a few
minutes before heating ?
Mengapakah gas hidrogen dialirkan ke dalam tiub pembakaran beberapa minit sebelum pemanasan ?
... ... [ 1 mark] (d) How to ensure the reaction is complete ?
Bagaimana untuk memastikan tindak balas adalah lengkap?
... ... [ 2 marks] (e) Determine the empirical formula for oxide of lead ?
Tentukan formula empirik bagi oksida plumbum ? [Relative atomic mass for O = 16 and Pb = 207] [Jisim atom relatif bagi O = 16 dan Pb = 207]
(f) Why does the empirical formula of magnesium oxide cannot be determined using the above method ?
Mengapa formula empirik magnesium oksida tidak boleh ditentukan menggunakan kaedah di atas ?
... [ 1 mark] 3 Diagram 3 shows the positions of 6 elements represented by the letters A, B, C, D, E, F in the
Periodic Table of Elements.
Rajah 3 menunjukkan kedudukan 6 unsur yang diwakili oleh huruf A,B,C,D,E, F dalam Jadual Berkala Unsur.
A B C D E
F Diagram 3
Rajah 3 Using these letters, answer the following questions.
Menggunakan huruf-huruf tersebut, jawab soalan-soalan berikut.
(a) State the element which exists as a diatomic molecule under room condition. Nyatakan unsur yang wujud sebagai molekul dwiatom pada keadaan bilik.
………. [ 1 mark ] (b) State one special characteristic of element F .
Nyatakan satu ciri istimewa bagi unsur F.
………... [ 1 mark ] (c) (i) Arrange atoms A, B, C, D and E in decreasing order of atomic size.
Susun atom A, B ,C , D dan E mengikut tertib pengurangan saiz atom.
………... [ 1 mark ] (ii) Explain your answer in (c)(i).
Huraikan jawapan anda dalam (c)(i).
……….….. ………...
(d) Write the chemical formula for the ion formed by element D. Tuliskan formula kimia bagi ion yang terbentuk dari unsur D.
………... [ 1 mark ] (e) Write the chemical equation for the reaction between the oxide of A and water.
Tuliskan persamaan kimia untuk tindakbalas antara oksida A dan air.
………..………... [ 3 marks] 4 Table 4 below shows the electron arrangement of 6 elements T,U,V,W, X and Y.
Jadual 4 menunjukkan susunan elektron bagi unsur-unsur T,U,V,W, X dan Y Elements Proton number Electron arrangement
T 6 2.4 U 17 2.8.7 V 11 2.8.1 W 12 2.8.2 X 2.8.8 Y 19 Table 4 Jadual 4
(a) Based on Table 4, answer the following questions. Berdasarkan Jadual 4, jawab soalan-soalan berikut.
(i) What is the proton number of element X ? Berapakan nombor proton bagi unsur X?
... [ 1 mark ] (ii) What is the electron arrangement for ion Y?
Apakah susunan elektron bagi ion Y?
...………... [ 1 mark ] (b) Element T reacts with element U to form a compound
Unsur T bertindakbalas dengan unsur U untuk membentuk satu sebatian (i) Give the molecular formula of compound formed.
Berikan formula molekul bagi sebatian yang terbentuk.
(ii) State the type of bonding in the compound formed in (b)(i).
Nyatakan jenis ikatan yang terbentuk bagi sebatian yang terbentuk dalam (b)(i). ………...
[ 1 mark ] (iii) Give two physical properties of the compound formed.
Berikan dua sifat fizikal sebatian yang terbentuk.
………... ………... [ 2 marks ] (iv) Illustrate the electron arrangement of the compound formed in (b)(i)
Lukis susunan elektron bagi sebatian yang terbentuk dalam (b)(i)
[ 2 marks ] (c) Element W reacts with element U to form a compound. Write the chemical formula of the
compound formed by U and W.
Unsur W bertindakbalas dengan unsur U membentuk satu sebatian. Tuliskan formula kimia bagi sebatian yang terbentuk oleh U dan W.
………... [ 1 mark ] (d) State an element that can form a cation.
Nyatakan satu unsur yang boleh membentuk kation.
………...
5 Table 5.1 shows incomplete observation for two sets of electrolysis of sodium chloride solution with different concentration using carbon electrode.
Jadual 5.1 menunjukkan pemerhatian yang tidak lengkap bagi dua set elektrolisis larutan natrium klorida dengan kepekatan yang berbeza menggunakan elektrod karbon.
Set Concentration of sodium chloride solution (mol dm-3) Kepekatan larutan natrium klorida (mol dm-3) Observation Pemerhatian Set 1 0.002
Anode Bubbles of colourless gas is released Gas tanpa warna dibebaskan
Cathode Bubbles of colourless gas is released Gas tanpa warna dibebaskan
Set 2 1.0
Anode ...
Cathode ... Table 5.1
Jadual 5.1 (a) (i) Complete Table 5.1
Lengkapkan Jadual 5.1
[ 2 marks ] (ii) Describe a chemical test to identify the product formed at anode in set 2.
Huraikan satu ujian kimia untuk mengenalpasti hasil yang terbentuk di anod pada set 2.
... ... ... [ 2 marks ] (b) Based on experiment in set 1:
Berdasarkan eksperimen dalam set 1:
(i) Name the colourless gas formed at the cathode. Namakan gas tanpa warna yang terbentuk di katod.
... [ 1 mark ]
(ii)
(ii) Write a Write a half half equation equation for the for the formation formation of gaof gas at s at the cthe cathode.athode. Tulis setengah persamaan untuk pembentukan gas di
Tulis setengah persamaan untuk pembentukan gas di katod.katod. ...
... [ 1 mark ] [ 1 mark ] (c)
(c) In anIn another expeother experiment, a riment, a student student using using apparatus apparatus and mateand materials in rials in Table Table 5.2 to 5.2 to produce produce anan electrical energy from chemical energy.
electrical energy from chemical energy. Dalam
Dalam satu satu eksperimen eksperimen lain, lain, seorang seorang pelajar pelajar menggunakmenggunakan an radas radas dan dan bahan bahan dalamdalam Jadual 5.2 un
Jadual 5.2 untuk menghasilkan tetuk menghasilkan tenaga elektrik naga elektrik dari tenaga kimiadari tenaga kimia.. Apparatus
Apparatus Radas Radas
Beaker, connecting wire,
Beaker, connecting wire, galvanometergalvanometer Bikar, wayar
Bikar, wayar penyambung, gpenyambung, galvanometer alvanometer Materials
Materials Bahan Bahan
Zinc , copper strip, copper (II) sulphate solution Zinc , copper strip, copper (II) sulphate solution Zink, kepinga
Zink, kepingan kuprum, larutan n kuprum, larutan kuprum (II) kuprum (II) sulfat sulfat Table 5.2
Table 5.2 Jadual 5.2 Jadual 5.2 (i)
(i) Diagram Diagram below below shows shows incomplete incomplete apparatus apparatus set-up set-up used used in in (c).(c). Rajah
Rajah di di bawah bawah menunjukkan menunjukkan susunan susunan radas radas tidak tidak lengkap lengkap yangyang digunakan pada (c).
digunakan pada (c).
Complete the diagram below. Complete the diagram below. Lengkapka
Lengkapkan rajah di bawah.n rajah di bawah.
[ 2 marks ] [ 2 marks ] (ii)
(ii) State State the the direction direction of of electron electron flow flow in in this this experiment.experiment. Nyatakan ara
Nyatakan arah pergerakah pergerakan elektron dalam n elektron dalam eksperimen ini.eksperimen ini. ...
... [ 1 mark ] [ 1 mark ]
(iii)
(iii) State State the the observation observation in in the the copper copper (II) (II) sulphate sulphate solution.solution. Nyatakan pe
Nyatakan pemerhatian pada larumerhatian pada larutan kuprum(Itan kuprum(II) sulfat I) sulfat .. ...
... [ 1 mark ] [ 1 mark ] 6
6 An An experiment experiment is cis carried arried out to out to find find the the end end point point of of acid-base acid-base titration. 25titration. 25.0 .0 cm cm of of 1.0 1.0 moldmmoldm --sodium hydro
sodium hydroxide solution reacxide solution reacts with hydrochloric ts with hydrochloric acid. acid. Table 6 shTable 6 shows the results ows the results of theof the experiment.
experiment.
Satu eksperimen dijalankan untuk menentukan takat akhir peneutralan antara asid dan alkali. Satu eksperimen dijalankan untuk menentukan takat akhir peneutralan antara asid dan alkali. 25.0 cm
25.0 cm33 bagi 1.0 moldmbagi 1.0 moldm-3-3 larutan natrium hidroksida bertindak balas dengan larutan natrium hidroksida bertindak balas dengan asid hidroklorik.asid hidroklorik. Jadual 6
Jadual 6 menunjukkamenunjukkan keputusan bagi en keputusan bagi eksperimen tersksperimen tersebut.ebut.
Volume
Volume of of hydrochloric hydrochloric acid acid First First readingreading SecondSecond reading reading Third Third reading reading Final
Final burette burette reading/ reading/ cm cm 21.00 21.00 41.60 41.60 25.8025.80 Initial burette reading/ cm
Initial burette reading/ cm 0.00 0.00 21.00 21.00 5.005.00 Volume of hydrochloric acid used/ cm
Volume of hydrochloric acid used/ cm
Table 6 Table 6 Jadual 6 Jadual 6
(a)
(a) Draw Draw the the set-up set-up of aof apparatus pparatus that that is is used used in in the the experiment.experiment. Lukiskan gamb
Lukiskan gambar rajah susunaar rajah susunan alat radas yan alat radas yang digunakan dng digunakan di dalam eksperimei dalam eksperimen ini.n ini.
[ 2 marks ] [ 2 marks ] (b)
(b) (i) (i) State State the the indicator indicator solution solution that that is is suitable suitable to to be be used used in in the the experiment.experiment. Nyatakan larutan
Nyatakan larutan penunjuk yapenunjuk yang sesuai untuk ng sesuai untuk digunakan di dalam digunakan di dalam eksperimen ini.eksperimen ini. ………... ………...
(ii)
(ii) Based Based on the on the indicator solution indicator solution mentioned mentioned in (b)(i), stain (b)(i), state the te the colour ccolour change at hange at thethe end point.
end point. Berdasark
Berdasarkan an larutan larutan penunjuk penunjuk yang yang dinyatakan dinyatakan di di dalam dalam (b)(i), (b)(i), nyatakannyatakan perubahan
perubahan warna pada takawarna pada takat akhir.t akhir.
………... ………... [ 1 mark ] [ 1 mark ] (c)
(c) (i) (i) Complete Complete Table Table 6 6 to to find find the the volume volume of of hydrochloric hydrochloric acid acid used.used. Lengkapka
Lengkapkan n Jadual 6 untuk meJadual 6 untuk menentukan isipadnentukan isipadu asid yang digunu asid yang digunakan.akan.
[ 1 mark ] [ 1 mark ] (ii)
(ii) Calculate Calculate the the average average volume volume of of hydrochloric hydrochloric acid acid used.used. Kira isipadu purata bagi asid hidroklorik yang digunakan. Kira isipadu purata bagi asid hidroklorik yang digunakan.
[ 1 mark ] [ 1 mark ] (d)
(d) Write Write the the chemical chemical equation equation for for this this experiment.experiment. Tulis persamaan kimia bagi eksperimen ini. Tulis persamaan kimia bagi eksperimen ini.
………. ………. [ 1 mark ] [ 1 mark ] (e)
(e) Find Find the the concentration concentration of of hydrochloric hydrochloric acid acid used.used. Tentukan kepekatan bagi asid hidroklorik yang
Tentukan kepekatan bagi asid hidroklorik yang digunakan.digunakan.
[ 2 marks ] [ 2 marks ] (f)
(f) Hydrochloric Hydrochloric acid acid is is classified classified as as a a strong strong and and monoprotic monoprotic acid. acid. State State another another example example of of monoprotic acid.
monoprotic acid. Asid hidroklorik d
Asid hidroklorik dikelaskan sebaikelaskan sebagai asid kuat dan asid mogai asid kuat dan asid monoprotik. noprotik. Nyatakan contoh Nyatakan contoh asid asid monoprotik yang lain.
monoprotik yang lain.
………....……. ………....……. [ 1 mark ] [ 1 mark ]
Section B [20 marks]
Answer any one question from this section Jawab mana-mana satu soalan dari bahagian ini 7 (a) Diagram 7 shows the electron arrangement of a molecule PQ2.
Rajah 7 menunjukkan susunan elektron bagi satu molekul PQ2.
Diagram 7 Rajah 7
Based on Diagram 7, write the electron arrangement for atoms of element P and element Q.
Explain the position of element Q in the Periodic Table.
Berdasarkan Rajah 7, tulis susunan elektron bagi atom unsur P dan unsur Q. Terangkan kedudukan unsur Q dalam Jadual Berkala
[6 marks] (b) Table 7 shows the electron arrangement for atoms W, X and Y.
Jadual 7 menunjukkan susunan elektron bagi atom W,X dan Y. Element Electron arrangement
W 2.4
X 2.8.7
Y 2.8.8.2
Table 7 Jadual 7
Using the information in Table 7, explain how two compounds can be formed from these elements based on their electron arrangements.
The two compounds should have different bond types.
Menggunakan maklumat dalam Jadual 7, terangkan bagaimana dua sebatian dapat terbentuk dari unsur-unsur ini berdasarkan susunan elektron.
Dua sebatian ini mestilah berbeza jenis ikatan.
[10 marks] (c) Compare the physical properties for both chemical compound formed in (b).
Bandingkan sifat-sifat fizikal bagi kedua-dua sebatian yang terbentuk dalam (b).
8 Table 8 shows the information on two type of acids. Jadual 8 menunjukkan maklumat tentang dua jenis asid.
Acid Asid Concentration (moldm-3) Kepekatan (moldm-3 ) pH value Nilai pH Hydrochloric acid, HCl Asid hidroklorik, HCl 0.1 1
Ethanoic acid, CH3COOH
Asid etanoik, CH 3COOH 0.1 4
Table 8 Jadual 8
(a) One of the acids is a strong acid, while the other is a weak acid.
Satu daripada asid tersebut ialah asid kuat dan satu lagi ialah asid lemah. (i) What are the definition for a strong acid and a weak acid?
Apakah takrifan bagi asid kuat dan asid lemah?
[ 2 marks ] (ii) Identify the strong acid and weak acid.
Kenalpasti asid kuat dan asid lemah.
[ 2 marks ] (iii) Explain why the pH values of the two acids are different.
Jelaskan tentang perbezaan nilai pH kedua-dua asid.
[ 4 marks ] (b) (i) What is the definition of molarity?
State the formula and the unit of molarity. Apakah takrifan kemolaran?
Nyatakan formula dan unit bagi kemolaran.
[ 3 marks ] (ii) Explain how you would prepare 100.0 cm of 0.05 moldm- of hydrochloric acid
from 0.50 moldm-3 of hydrochloric acid.
Terangkan bagaimana anda menyediakan 100.0 cm3 , 0.05 moldm-3 asid hidroklorik daripada 0.50 moldm-3asid hidroklorik.
[ 5 marks ] (iii) Calculate the volume of 40.0 gdm- of sodium hydroxide solution required to
neutralise the solution in b (ii).
[Relative atomic mass: H=1; O=16; Na=23]
Kira isipadu bagi 40.0 gdm-3 larutan natrium hidroksida yang diperlukan untuk meneutralkan larutan dalam b(ii).
[Jisim atom relatif: H=1; O=16; Na=23]
Section C [20 marks]
Answer any one question from this section
Jawab mana-mana satu soalan dari bahagian ini
9 (a) State 3 factors that affects the selective discharged of ions during electrolysis .
Nyatakan 3 faktor yang mempengaruhi pemilihan ion untuk nyahcas semasa elektrolisis. [ 3 marks ] (b) Diagram 9 shows the set-up of apparatus of two experiments to investigate the electrolysis
process by using different electrodes.
Rajah 9 menunjukkan susunan radas bagi dua eksperimen untuk mengkaji proses elektrolisis dengan menggunakan elektrod-elektrod yang berlainan.
Experiment
Eksperimen Apparatus set-up
Susunan radas
I Copper electrodes
Elektrod kuprum
Copper(II) sulphate solution
Larutan kuprum(II) sulfat A
II
Diagram 9 Rajah 9
Compare the observation and product formed at the anode for both experiments Bandingkan pemerhatian dan hasil yang terbentuk di anod bagi kedua-dua eksperimen.
Write the half equation at the anode for both experiments.
Tuliskan setengah persamaan di anod bagi kedua-dua eksperimen.
[ 6 marks ]
Copper(II) sulphate solution
Larutan kuprum(II) sulfat
A
Carbon electrodes
(c) Table 9 shows the voltage of chemical cell using metals A, B and C as the electrode.
Jadual 9 menunjukkan nilai voltan bagi sel kimia menggunakan logam A, B dan C sebagai elektrod. Table 9 Jadual 9 Pairs of metal Pasangan logam Voltage (V) Nilai voltan (V) Positive terminal Terminal positif B / C 0.1 C A / C 0.8 C
Based on the information given,
Berdasarkan kepada maklumat yang diberikan,
(i) state the voltage of a chemical cell using metal A and B as the electrodes.
nyatakan nilai voltan yang akan dihasilkan oleh sel kimia yang menggunakan logam A dan B sebagai elektrod.
(ii) state the negative terminal in (c)(i) and explain your answer.
nyatakan terminal negatif pada (c)(i) dan terangkan jawapan anda.
[ 3 marks ] (d) A student has an inattractive ring and it is easy to corrode. To solve the problem, he uses
an electrolysis process in a laboratory.
Seorang pelajar mempunyai sebentuk cincin yang tidak menarik dan mudah berkarat. Untuk mengatasi masalah ini, dia menggunakan proses elektrolisis di dalam makmal.
A student may used the apparatus and chemical as follow:
Pelajar boleh menggunakan radas dan bahan kimia seperti berikut: Beaker, connecting wire and crocodile clip, battery, silver metal, silver nitrate solution,
Bikar, wayar penyambung bersama klip buaya, bateri, kepingan argentum , larutan argentum nitrat,
Describe an experiment on how to solve these problem. In your description include a diagram to show the set-up of apparatus.
Huraikan satu eksperimen bagaimana untuk mengatasi masalah ini. Dalam penerangan anda sertakan gambarajah untuk menunjukkan susunan radas.
10 (a) (i) What is meant by ‘salt’ ?
Apakah yang dimaksudkan dengan ‘garam’?
[ 2 marks ] (ii) Name salt derived from
• Nitric acid
• Hydrochloric acid
Namakan garam yang dihasilkan dari
• Asid nitrik
• Asid hidroklorik
[ 2 marks ] (b) The following are three examples of sulphate salts that can be prepared in the
laboratory.
Berikut adalah tiga contoh garam sulfat yang boleh disediakan di dalam makmal. • Potassium sulphate, K2SO4
Kalium sulfat, K 2SO4 • Lead (II) sulphate, PbSO4
Plumbum (II) sulfat, PbSO4 • Zinc sulphate, ZnSO4
Zink sulfat, ZnSO4
(i) From these examples, identify the soluble and insoluble salts.
Daripada contoh-contoh ini, kenal pasti garam terlarutkan dan garam tak larut. [ 2 marks ] (ii) State the reactant for the preparation of the insoluble salt in 10(b)(i)
Nyatakan bahan tindak balas bagi penyediaan garam tak larut di 10(b)(i).
[ 2 marks ] (c) With the aid of a labelled diagram, describe the procedure of experiment to prepare
the named insoluble salts by using the reactant in 10(b)(ii). Your answer should include the
• list of materials and apparatus • chemical equation
• ionic equation for the reaction.
Dengan bantuan gambarajah berlabel, huraikan langkah-langkah eksperimen untuk menyediakan garam tidak larut yang dinamakan dengan menggunakan bahan
tindak balas dalam 10(b)(ii).
Jawapan anda mestilah mengandungi
• senarai bahan dan alat radas • persamaan kimia
• persamaan ion bagi tindak balas.
[12 marks] END OF QUESTION PAPER
INFORMATION FOR CANDIDATES MAKLUMAT UNTUK CALON
1. This question paper consists of three sections: Section A, Section B and Section C.
Kertas soalan ini mengandungi tiga bahagian: Bahagian A, Bahagian B dan B ahagian C.
2. Answer all questions in Section A. Write your answers for Section A in the spaces provided in the question paper.
Jawab semua soalan dalam Bahagian A. Tuliskan jawapan bagi Bahagian A dalam ruang yang disediakan dalam kertas soalan.
3. Answer one question from Section B and one question from Section C.
Write your answers for Section B and Section C on the `writing paper’ provided by the invigilators.
Answer questions in Section B and Section C in detail.
You may use equations, diagrams, tables, graphs and other suitable methods to explain your answer.
Jawab satu soalan daripada Bahagian B dan satu soalan daripada Bahagian C. Tuliskan jawapan
bagiBahagian B dan Bahagian C pada kertas tulis yang disediakan. Jawab Bahagian B dan
Bahagian C dengan terperinci. Anda boleh menggunakan persamaan, gambar rajah, jadual, graf dan cara lain yang sesuai untuk menjelaskan jawapan anda.
4. Show your working. It may help you to get marks.
Tunjukkan kerja mengira.Ini membantu anda mendapatkan markah.
5. If you wish to change your answer, neatly cross out the answer that you have done. Then write down the new answer.
Sekiranya anda hendak membatalkan sesuatu jawapan, buat garisan di atas jawapan itu.
6. The diagrams in the questions are not drawn to scale unless stated.
Rajah yang mengiringi soalan tidak dilukiskan mengikut skala kecuali dinyatakan.
7. Marks allocated for each question or part question are shown in brackets.
Markah yang diperuntukkan bagi setiap soalan atau ceraian soalan ditunjukkan dalam kurungan.
8. The time suggested to answer Section A is 90 minutes, Section B is 30 minutes and Section C is 30 minutes.
Masa yang dicadangkan untuk menjawab Bahagian A ialah 90 minit, Bahagian B ialah 30 minit dan Bahagian C ialah 30 minit.
9. You may use a non-programmable scientific calculator.
Anda dibenarkan menggunakan kalkulator saintifik yang tidak boleh diprogramkan.
10. Hand in your answer sheets at the end of the examination.
4 3 Li Lithium 7 4 Be Beryllium 9 5 B Boron 11 6 C Carbon 12 7 N Nitrogen 14 8 O Oxygen 16 9 F Fluorine 19 10 Ne Neon 20 11 Na Sodium 23 12 Mg Magnesiu m 24 13 Al Aluminiu m 27 14 Si Silicon 28 15 P Phosphor us 31 16 S Sulphur 32 17 Cl Chlorine 35 18 Ar Argon 40 19 K Potassium 39 20 Ca Calcium 40 21 Sc Scandium 45 22 Ti Titanium 48 23 V Vanadiu m 51 24 Cr Chromium 52 25 Mn Manganes e 55 26 Fe Iron 56 27 Co Cobalt 59 28 Ni Nickel 59 29 Cu Copper 64 30 Zn Zinc 65 31 Ga Gallium 70 32 Ge Germaniu m 73 33 As Arsenic 75 34 Se Selenium 79 35 Br Bromine 80 36 Kr Krypton 84 37 Rb Rubidium 86 38 Sr Strontium 88 39 Y Yttrium 89 40 Zr Zirconium 91 41 Nb Niobium 93 42 Mo Molybden um 96 43 Tc Technetiu m 98 44 Ru Rutheniu m 101 45 Rh Rhodium 103 46 Pd Palladium 106 47 Ag Silver 108 48 Cd Cadmium 112 49 In Indium 115 50 Sn Tin 119 51 Sb Antimony 122 52 Te Tellurium 128 53 I Iodine 127 54 Xe Xenon 131 55 Cs Caesium 133 56 Ba Barium 137 57 La Lanthanu m 139 72 Hf Hafnium 179 73 Ta Tantalum 181 74 W Tungsten 184 75 Re Rhenium 186 76 Os Osmium 190 77 Ir Iridium 192 78 Pt Platinum 195 79 Au Gold 197 80 Hg Mercury 201 81 Tl Thallium 204 82 Pb Lead 207 83 Bi Bismuth 209 84 Po Polonium 210 85 At Astatine 210 86 Rn Radon 222 87 Fr Francium 223 88 Ra Radium 226 89 Ac Actinium 227 104 Unq Unnil− quadium 257 105 Unp Unnil− pentium 260 106 Unh Unnil−hex ium 263 107 Uns Unnilsepti um 262 108 Uno Unnilocti um 265 109 Une Unnilenni um 266 58 Ce Cerium 140 59 Pr Praseo− dymium 141 60 Nd Neodymiu m 144 61 Pm Promethiu m 147 62 Sm Samarium 150 63 Eu Europium 152 64 Gd Gadoliniu m 157 65 Tb Terbium 167 66 Dy Dyprosiu m 163 67 Ho Holmium 165 68 Er Erbium 167 69 Tm Thulium 169 70 Yb Ytterbium 173 71 Lu Lutetium 175 90 Th Thorium 232 91 Pa Protactini um 231 92 U Uranium 238 93 Np Neptuniu m 237 94 Pu Plutonium 244 95 Am Americiu m 243 96 Cm Curium 247 97 Bk Berkelium 247 98 Cf Californiu m 249 99 Es Einsteiniu m 254 100 Fm Fermium 253 101 Md Mendelev ium 256 102 No Nobelium 254 103 Lr Lawrenci um 257
Reference: Chang, Raymond (1991). Chemistry. McGraw− Hill
10 Ne Neon 20 Symbol Name of element Relative atomic mass
CHEMISTRY 2 Oktober
2011
JABATAN PELAJARAN MELAKA BUKIT BARU, MELAKA
____________________________________________________________ PEPERIKSAAN AKHIR TAHUN
TINGKATAN 4 2011
CHEMISTRY 2 Oktober
2011
JABATAN PELAJARAN MELAKA BUKIT BARU, MELAKA
____________________________________________________________ PEPERIKSAAN AKHIR TAHUN
TINGKATAN 4 2011
CHEMISTRY 1 CHEMISTRY 2
PERATURAN PEMARKAHAN
MARKING SCHEME OF CHEMISTRY PAPER 1 1 C 11 D 21 D 31 C 41 A 2 D 12 A 22 D 32 B 42 A 3 C 13 D 23 C 33 C 43 C 4 A 14 A 24 B 34 B 44 C 5 A 15 B 25 C 35 A 45 D 6 D 16 B 26 A 36 D 46 B 7 B 17 A 27 B 37 B 47 C 8 A 18 C 28 B 38 A 48 B 9 C 19 A 29 C 39 D 49 D 10 D 20 B 30 A 40 B 50 C
MARKING SCHEME OF CHEMISTRY PAPER 2
Scheme Marks
Sub-total 1. (a) (i) The number of protons in the nucleus of an atom 1
2
(ii) 16 1
(b) (i) Q and R,
Q and R have same number of protons but different number of neutrons/ different nucleon number 1 1 4 (ii) Q Q 1 1 (c) (i) 2.8.7 1 3 1 Total 10 m (ii) Number of shells(3) Number of electrons 1 1
(d) Because S has 8 valence electrons // has achieved [stable] octet electron arrangement.
1 37 17 35 17 R
2 a Empirical formula is a chemical formula that shows the simplest ratio of the number of atom for each element in the compound
1 1
b Anhydrous Calcium chloride // concentrated sulphuric acid
1 1
c To ensure that the combustion tube has been filled
with hydrogen gas 1 1
d The process of heating, cooling and weighing are repeated
until a constant mass of substance and its container is obtained 1 1 2 e Pb O Mass (g) 4.14 0.64 No of mole n=4.14/207 =0.02 n= 0.64/16 =0.04 Simplest ratio 1 2 Empirical formula PbO2 1 1 1 1 4
f Because Mg is at higher position than H in the
electrochemical series. 1 1
Total 10m
3 a D 1 1
b - has more than one oxidation number // - can act as catalyst//
- can formed complex ion - can formed coloured ion
[ any one ]
1 1
c (i) A,B,C,D,E
(ii) - Across the period, the proton number of each atom increase
- Nuclei charge in the nucleus increase - Force of attraction between nucleus and
outermost shell become stronger // outermost shell are pulled closer to the nucleus 1 1 1 1 4
-- formula of reactant - formula of product - balance equation 1 1 1 3 Total 10 m 4 a (i) 18 1 2 (ii) 2.8.8 1 b (i) TU4 1 6
(ii) Covalent bond 1
(iii) - low melting and boiling point//
- cannot conduct electricity in any state // - insoluble in water // soluble in organic
solvent.
[any two of the above]
1 1
(iv) - correct number of electron in each shell - four atom U combine with one atom T
[ the atom is labelled]
1 1 c WU2 1 1 d V // W // Y [ any one ] 1 1 Total 10m
5 a (i) Anode : Greenish yellow gas is released Cathode : Colourless gas is released
1 1
4 (ii) - Insert a damp blue litmus at [the anode]
- Blue litmus paper turn red and [bleached]
1 1
b (i) Hydrogen 1
2
(ii) 2H+ + 2e →H2 1
c (i) Functional diagram
Labelled diagram – zinc strip, copper strip , copper(II) sulphate solution
1 1
4 (ii) Zinc to copper through connecting wire 1
(iii) Blue turn colourless 1
Total 10m
6 a - Functional diagram with correct shaded solution in conical flask and burette
- label of sodium hydroxide solution in conical flask and hydrochloric acid in the burette
1
1
2
b (i) - Phenolphtalein solution//
[ accept any suitable indicator solution ]
1
2
(ii) pink to colourless 1
c (i) 21.00, 20.60, 20.80 1 1
(iii) =21.00 + 20.60 + 20.80 // 3
= 20.80 cm3 1 1
d NaOH + HCl NaOH + H2O
- correct formula and reactant
1 1 e MaVa = na MbVb nb Max 20.80 = 1 1.0 x 25.0 1 Concentration of HCl, Ma = 1.20 moldm-3 [ accept another suitable method ]
1
1 2
f Nitric acid 1 1
Total 10m
7 a - The electron arrangement of P atom is 2.4 - and Q atom is 2.6.
- Q atom is in group 16 because it - has 6 valence electron and
- it is in period 2 because
- it has 2 shells occupied with electron.
1 1 1 1 1 1 6 b Ionic bond:
- X atom has 7 valence electron and Y atom has 2 valence electron.
- These electron arrangement are unstable.
- To achieve a stable electron arrangement, Y atom donate 2 valence electron and form Y2+ ions
- 2 atom X receive 1 electron each and form X- ions. - Y2+ ion and X- ions are attract to each other
- to form an ionic compound with formula YX2
1 1 1 1 1 1
Covalent bond:
- W atom has 4 valence electron and X atom has 7 valence electron.
- To achieve a stable electron arrangement, one W atom share 4 valence electron with 4 atom X. - They will form 4 single covalent bond
- with a formula WX4 1 1 1 1 10
c Ionic compound Covalent compound
1. soluble in water Insoluble in water 2. insoluble in organic solvent Soluble in organic solvent 3. conduct electricity in
molten and aqueous state
Cannot conduct
electricity in any state 4. high melting and boiling
point
Low melting and boiling point
1 1 1
1 4
Total = 20m
8 a (i). - strong acid is an acid which ionises /dissociates completely in water to produce a high
concentration of hydrogen ions, H+
- weak acid is an acid which ionises / dissociates partially in water to produce a low
concentration of hydrogen ions, H+ (ii). Strong acid - hydrochloric acid / HCl
Weak acid - ethanoic acid / CH3COOH
(iii). - hydrochloric acid ionises completely in water and produced high concentration of hydrogen ions
- the high concentration of hydrogen ions causes the pH value to become lower
- ethanoic acid ionises partially in water and produced lower concentration of hydrogen ions.
- the lower concentration of hydrogen ions causes the pH value to become higher
1 1 1 1 1 1 1 1 2 2 4
b (i). Molarity is the number of moles solute present in 1 dm3 solution.
- Molarity = no of moles of solute (mol) Volume of solution (dm3) - Unit , moldm-3
(ii). - Calculation: M2= 100 X 0.05 0.5 = 10 cm3
- Using pipette, 10.0 cm3of 0.50 moldm-3of hydrochloric acid is transferred to a 100 ml volumetric flask.
- Distilled water is then added to the acid in the volumetric flask until near the calibration mark - Add distilled water dropwise to the volumetric
flask until water level exactly reach its calibration mark
- Stopper the flask and shake well the solution by up side down the flask to ensure thorough mixing (iii). - Molarity of NaOH = 40 gdm-3
23 + 16 + 1 = 1.0 moldm-3 NaOH + HCl NaCl + H2O MaVa = na MbVb nb - 0.05 X 100.0 = 1 1.0 X Vb 1 - Volume of NaOH, Vb = 5.0 cm3 [ Accept another suitable method ]
1 1 1 1 1 1 1 1 1 1 1 1 3 5 4 Total 20m 9 (a) i. concentration of ion
ii. type of electrode
iii. position of ion in electrochemical series
1 1
1 3
(b)
I II
Anode becomes thinner Gas bubbles are released Copper (II) ion Oxygen
Cu Cu ++ 2e 4OH- O + 2H O + 4e
1+1 1+1
(c) (i) 0.8 - 0.1 = 0.7V // 0.7V 1
3 (ii) - Electrode A,
- A is more electropositive than B
1 1 (d) Procedure of experiment:
i. [50-200 cm3] dilute silver nitrate solution is poured into a beaker.
ii. An iron ring is cleaned using sand paper. iii. Iron ring is connected to negative terminal of
battery // silver metal is connected to positive terminal of battery
iv. Iron ring and silver metal is immersed into the silver nitrate solution
v. The circuit is completed using connecting wire vi . Using small current/turn the ring steadily vii. Diagram:
- Functional diagram contain two different metals as electrode ( no need with correct electrode) and the solution is shaded correctly.
- iron spoon as cathode, silver as anode and silver nitrate solution are labelled.
1 1 1 1 1 1 1 1 8 Total = 20m 10 a b c
(i). - An ionic compound formed when the - hydrogen ion in an acid is replaced by the
metal ion or ammonium ion
(ii)● sodium nitrate / potassium nitrate etc ● silver chloride / sodium chloride etc
(i) Soluble salts : potassium sulphate and zinc sulphate
Insoluble salts : lead (II) sulphate (ii) Lead (II) nitrate and sodium sulphate.
[ any suitable and correct answer ] - Alternative Diagram :
(i) Process of mixing the solution, contains: • Diagram shows the mixture of lead (II)
nitrate in and sodium sulphate in a beaker with glass rod
• shade the solution / mixture correctly
1 1 1 1 1 1 1+1 1 1 4 4
OR
(ii) Diagram shows the filtration process, contains:
● Labelled;
Lead (II) sulphate in filter paper (as residue), Sodium nitrate solution in the beaker (as filtrate and shaded )
● The set of apparatus contain filter paper,
filter funnel ( clamped at retort stand) and beaker.
- Materials and apparatus:
[0.1 – 1.0moldm-3] sodium sulphate solution, [0.1 – 1.0 moldm-3] lead (II) nitrate solution, 50 ml beaker, 50.0 ml measuring cylinder, rod, filter paper, filter funnel, conical flask. - Procedure:
i. [ 50.0 cm3 of 0.5 moldm-3] of sodium sulphate solution is measured using 50.0 ml measuring cylinder and pour into a beaker, A.
ii. [50.0 cm3 of 0.5 moldm-3] of lead (II ) nitrate solution is measured using 50.0 ml measuring cylinder and pour into another beaker, B. iii. Solution in beaker A is poured into the
solution in beaker B.
iv. Stir with glass rod, precipitate formed.
v. The precipitate is filtered into a conical flask using filter paper and filter funnel and rinsed the residue with distilled water
vi. Dry the residue by pressing between two filter papers.
- Chemical equation:
Na2SO4 + Pb(NO3)2 PbSO4 + 2NaNO3 - Correct reactant and product
- Balance equation - Ionic equation: Pb2+ + SO42- PbSO4 OR 1 1 1 1 1 1 1 1 1 1 1 1 12 Total 20