Metallurgy and Materials Engineering Department
University of Indonesia
2007
Corrosion Resistant Steel
(Stainless Steel)
Dr.-Ing. Bambang Suharno
Dr. Ir. Sri Harjanto
Kuliah Baja Paduan & Super Alloy
University of Indonesia
Metallurgy and Materials Engineering Department UI
Stainless Steel, Why Stainless?
Stainless steels =
Cr containing
steel alloys
Cr content is
min. 10.5% and
max 30%
Cr makes the steel
'stainless'
=
improved corrosion resistance,
due to a
chromium oxide film
that is formed on the
steel
surface
This extremely thin layer is
also
self-repairing
in the
presence of oxygen
and damage by abrasion,
cutting or machining is
quickly repaired
C : < 0.03 % - 1,2%
0
Cor
ro
si
on Rat
e
(
m
m/
year)
% Chromium
5
10
0
0.1
0.2
University of IndonesiaChromium Oxide Film
Fig. 1 - In any normal oxidising environment a protective coating
of
passive chromium rich oxide film
is
automatically formed
on
stainless steel.
Fig. 2 - When
scratched, damaged or machined
this protective film
is denuded exposing the steel to the atmosphere.
Fig. 3 - The protective coating is
quickly restored
through the rapid
self-repairing quality
of the chromium rich film.
University of Indonesia
Pasivitas Pada Stainless Steels
Pasivitas dikarenakan
oleh adanya lapisan
oksida yang bersifat
self-repairing
dengan
karakteristik :
Kompak
, lapisan
kontinyu memerlukan ~
11wt% Cr.
Pasivitas
meningkat
dengan meningkatnya
Cr hingga ~17wt%
Umumnya stainless
steels mengandung
17-18wt% Cr
Pada Duplex SS Cr 22-27%
Ketahanan korosi
tergantung pada
kestabilan lapisan oksida
Untuk lingkungan yang
berbeda dioptimalkan oleh
alloying
dengan unsur lain
Contoh; Ni, Mo, N,
Cu
University of Indonesia
Metallurgy and Materials Engineering Department UI
Produk Stainless Steel
Wrought Product
Long Product: Pipa,
Batangan, Profil
Flat Product: Lembaran,
sheet, Pelat
Casting Product
Impeller, Flange, Valve
University of Indonesia
Metallurgy and Materials Engineering Department UI
Pengelompokan Stainless Steels
Berdasarkan
mikrostruktur
Stainless steels (SS)
dapat dikelompokkan atas:
•
Feritik
SS
•
Austenitik
SS
•
Duplex
(Feritik-Austenitik) SS
•
Martensitik
SS
•
Precipitation Hardening
(PH) SS
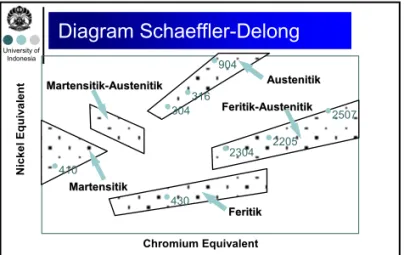
Mikrostruktur stainless steels (sangat tergantung dari
komposisi) dapat diprediksi menggunakan diagram
Schaeffler-Delong
University of IndonesiaSchaefler Diagram
University of IndonesiaUniversity of Indonesia
Metallurgy and Materials Engineering Department UI
Diagram Schaeffler-Delong
Chromium Equivalent = %Cr + 1.5%Si + %Mo
Nickel Equivalent = %Ni + 30(%C + %N) + 0.5(%Mn + %Cu + %Co)
Feritik
Feritik
Feritik
Feritik
-
-
Austenitik
Austenitik
Martensitik
Martensitik
Martensitik
Martensitik
-
-
Austenitik
Austenitik
Austenitik
Austenitik
Chromium Equivalent
Ni
ckel
Equiv
a
le
nt
•
430
•
410
•
304
•
316
•
904
•
2304
•
2205
•
2507
University of IndonesiaMetallurgy and Materials Engineering Department UI
Classification of Stainless Steel
University of Indonesia
Strength and Ductility of
Stainless Steel
University ofIndonesia
University of Indonesia
Metallurgy and Materials Engineering Department UI
Family
of SS
University of Indonesia
Metallurgy and Materials Engineering Department UI
Effect of Alloying Elements
Unsur paduan berkontribusi terhadap
pembentukan fasa
ferrite-austenite
Ferrite stabilizer (misal: Cr, Mo, W, V)
Austenite stabilizer (misal: C, Cu, Ni, Mn, N)
pembentukan
fasa kedua (precipitate)
yang melibatkan
unsur Cr, Mo, W, Cu, N
Sigma phase
Chi phase
Sangat penting untuk mengetahui pengaruh elemen
paduan terhadap ‘complex metallurgical system”
University of Indonesia
Effect of Alloying on SS Properties
-√ -X X X Cold Workability √ -√ -X -√ X -X X Weldability -√ -√ -√ -X X Machinability √ -√ -X √ √ -High Temperature Resistance
√ -√ √ √ √ √ -√ √ Mechanical Properties -√ -√ -X √ √ -Corrosion Resistance Ti or Nb Se Mo Cu P Si Mn S Ni Cr C Property University of Indonesia
Effect of Carbon
Iron + carbon =
increasing the hardness
and
strength
of iron.
In austenitic and ferritic stainless steels
a high carbon content is
undesirable
,
especially for
welding
Æ
carbide precipitation
University of Indonesia
Metallurgy and Materials Engineering Department UI
Effect of Chromium
Chrom :
To increase
resistance to oxidation
.
This resistance increases as
more chromium is added
.
Duplex Stainless Steel
Cr =
ferrite former
and
sigma phase
(carbide former)
Cr > 22%
increase in
pitting and crevice
corrosion resistance
Cr < 27 % in order to
retain
ductility, toughness and corrosion resistance
University of Indonesia
Metallurgy and Materials Engineering Department UI
Effect of Chromium on oxidation
resistance
University of Indonesia
Effect of Nickel
Ni =
austenite former
(austenite promoting element)
To
balance
the microstructure to
ferrite/ austenite
ratio
Affects the
corrosion
and
mechanical
properties
Excessive Ni:
increase in austenite content
Promoting a greater conc. of
ferrite stabilizer element
(Cr, Mo) in the remaining ferrite
(not change to the
precipitation of sigma phase)
University of Indonesia
Effect of Mo and N
Molybdenum (Mo):
Strong
ferrite former
, similar effect as Cr does on
properties
when added to austenitic stainless steels
improves
resistance to pitting and crevice
corrosion
especially in
Cl and S
containing environments
Nitrogen (N):
N =
austenite forming
element
increasing the
austenite stability
Yield strength
is greatly improved without sensitization
(e.g. carbon)
University of Indonesia
Metallurgy and Materials Engineering Department UI
Effect of Mn and Cu
Manganese (Mn):
to
improve hot working properties
and
increase strength
,
toughness
and
hardenability
.
Mn = austenite forming element
used as a substitute for nickel
in Austenitic SS
e.g. AISI 202 as a substitute for AISI 304
Copper (Cu):
Cu = normally present as
a residual element
in a few alloys to
produce precipitation hardening properties
or to
enhance corrosion resistance
University of Indonesia
Metallurgy and Materials Engineering Department UI
Effect of W
W =
minor elements
improving
corrosion resistance
The addition of W causes
easy to form inter metallic phase compare with W-free
duplex SS
W =
like Cr and Mo promotes
sigma phase
formation
promote of
Chi phase
University of Indonesia
Properties of Stainless Steel
Low Low Low Low Martensitic Low Low High Medium Ferritic High Medium Low Medium Duplex Very High Very High Very High Very High Austenitic Weldability Low Temperature Resistance3 High Temperature Resistance Ductility Alloy Group Age Harden Medium Medium Yes Precipitation Hardening Quench & Temper Medium Medium Yes Martensitic No Medium Medium Yes Ferritic No Very High Medium Yes Duplex By Cold Work High Very High Generally No Austenitic Hardenable Corrosion Resistance2 Work Hardening Rate Magnetic Response1 Alloy Group University of Indonesia
University of Indonesia
Metallurgy and Materials Engineering Department UI
Metallurgy and Materials Engineering Department
University of Indonesia
2007
Mekanisme Penggetasan
(Brittleness) pada Stainless Steel
University of Indonesia
Mekanisme Penggetasan pada
Stainless Steel
Stainless Steel peka terhadap
Embrittlement
(Kehilangan ductility/
toughness)
Penyebab
:
Sensitasi
475°C Embrittlement (350°C
-550°C).
Sigma Phasa (
σ
phase)
University of Indonesia
Sensitasi pada Stainless Steel
Austenitic SS peka terhadap
intergranular
corrosion
jk berada pada temp
480–815
OC
Umumnya akibat:
Welding
Service condition
Terjadi karena terbentuk endapan
M
23C
6(Cr
3Fe)
23C
6pada batas butir
Pencegahan
:
Kurangi Kadar C (0.015 – 0.02%), substitusi
dengan N
University of Indonesia
Metallurgy and Materials Engineering Department UI
475°C Embrittlement
Paduan dengan Cr tinggi, cenderung untuk
Brittle
,
terutama jika ditahan atau pendinginan lambat pada
400 – 550
OC
475°C Embrittlement
menyebabkan
:
UTS, Hardness naik
Ductility turun
Ketangguhan turun
Corrosion resistance turun
Penyebab
475°C Embrittlement:
Terbentuk second phase (carbides, nitrides,
oxides, phosphides)
Pembentukan Fe3Cr, FeCr, FeCr3, mirip sigma
phase hanya saja pada temp rendah
Kecenderungan Brittle
jika:
Kandungan Cr tinggi
Kandungan Carbide former tinggi (Mo, V, Ti, Nb)
Pengerjaan pada temp 475
OC
University of Indonesia
Metallurgy and Materials Engineering Department UI
Sigma Phasa (
σ
) Embrittlement
Pembentukan
FeCr Intermetallic
yang
keras, brittle (68 HRC)
Terbentuk jika temperatur proses sekitar
565 – 980
OC
dan berlangsung lama, hal ini
dapat menyebabkan fracture
Semua elemen paduan
penstabil ferrite
dapat men promote pembentukan sigma
phase
Cr yang tinggi mem promote sigma
phase
C yang tinggi pembentukan sigma phase
dikurangi sebab terbentuk Cr-Carbide
University of Indonesia