A new strategy to stabilize oxytocin in aqueous solutions: i. the effects of divalentt metal ion and citrate buffer.
Bebas
7
0
0
Teks penuh
Gambar
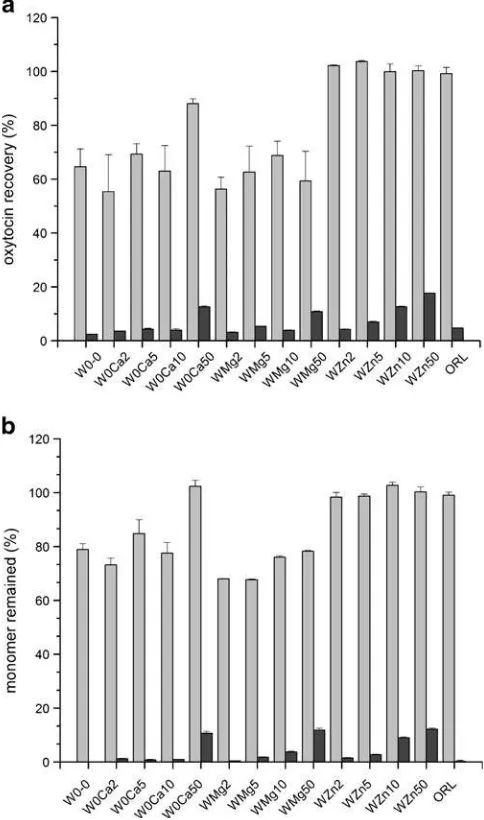
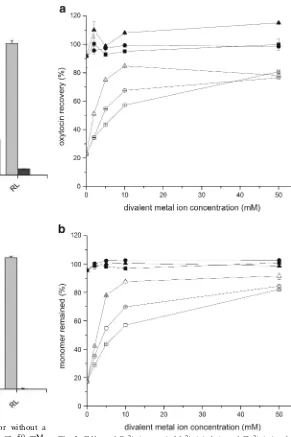
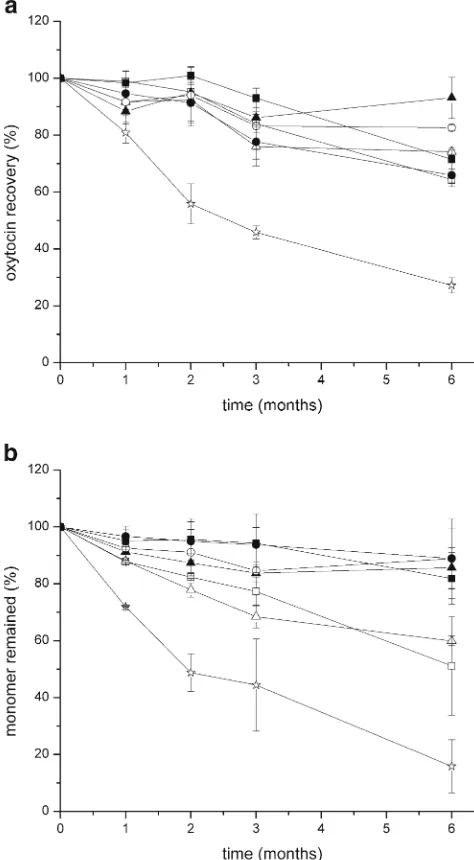
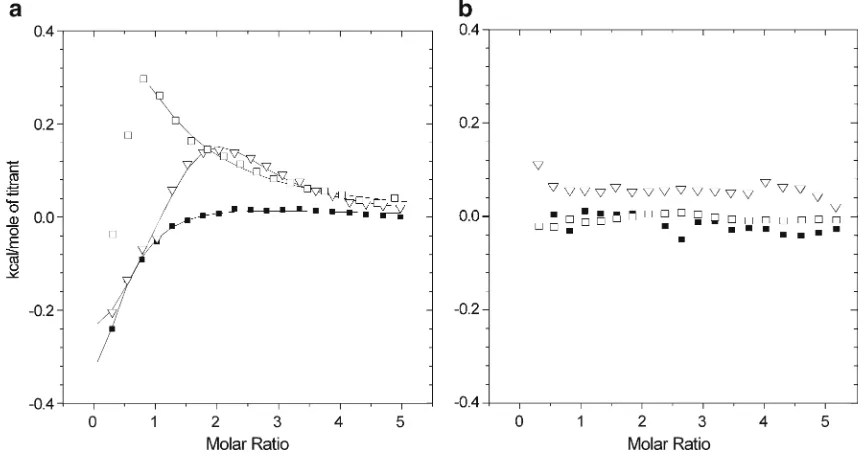
Dokumen terkait