CORAL REEF HEALTH CONDITION
BASED ON HERBIVOROUS FISH DENSITY
IN
PULAU TIGA SUBDISTRICT,
NATUNA DISTRICT
DEDY DAMHUDY
POST GRGDUATE SCHOOL
BOGOR AGFUCULTURAL UNIVERSITY
BOGOR
THESIS STATEMENT AND INFORMATION SOURCE
ABSTRACT
DEDY DAMHUDY. Coral Reef Health Condition based on Herbivorous Fish Density in Pulau Tiga Subdistrict, Natuna District. Under direction of
M.
MUKHLISKAMAL, and YUNIZAR ERNAWATI
The research was conducted in Pdau Tiga Subdistrict, Natuna District, Riau islands Province from April to August 2009. Pulau Tiga is an area dominated by a sea area with marine resource potential, particularly coral reefs. Around the area of coral reefs, fishing activities have been canied out intensively by fishermen using explosives Oombs) and toxic (potassium). Consequently, aquatic ecosystems have been exploited, take time to make a natural recovery in order to maintain and restore the quality and quantity of available resources. Especially for coral reef ecosystem, one of the factors that influence the resilience rate is the availability of hard substrate in a bottom waters as a settlement for coral larvae. Herbivorous animal has a major influence in determining the rate of coral reef animal larval settlement on a substrate because it can prevent the occurrence of a macro-algae excessive closure of hard substrate. This study was aimed to know the relationship between the conditions of the abundance of herbivorous fish with coral reef recovery rate. The methods used were the square transect for determining the condition of coral reefs, growth of juvenile corals and algae cover (DCA), whereas for the determination of herbivorous fish community structure using modification of Line Intercept Transect (LIT), Quadrat Method (QM) and
Underwater Fish Visual Census (UVC). The analysis used was standard
ecological analysis, correlation test, linier regression and multivariate analysis to find the relationship between the abundance of herbivorous fish, algae (DCA) and coral reefs. The results showed that coral reefs ecosystem in the area are still in good condition with live coral cover average of 63.17%, and shown an indication of an increase in the percentage of coral cover which previously
R5%.
The result of multivariate analysis can be stated that the higher the abundance of herbivorous fish, then live coral cover and growth of juvenile corals increasing, and decreasing algae cover on coral reef ecosystems. The results of correlation test, multivariate analysis and linear regression (t-Student test), show that from 24 species of herbivorous fish recorded then obtained three species of herbivorous fish that play a role in herbivory in stabilizing coral reefs ecosystem in Pulau Tiga Subdistrict, they are Chlorurus microrhinos, Scarus rivulatus and Siganus doliatus.SUMMARY
DEDY DAMHUDY. Coral Reef Health Condition based on Herbivorous Fish Density in Pulau Tiga Subdistrict, Natuna District. Under direction of M. MUKHLIS
KAMAL, and YONIZAR ERNAWATI.
Pulau Tiga as one of subdistricts in Natuna District is the area dominated by sea areas with very potential marine resource, especially coral reef. Around the coral reefs areas, fishing activities have been carried out intensively by fishermen using explosives (bombs) and toxic (potassium). Consequently, aquatic ecosystems have been exploited at the area, take time to make natural recovery in order to maintain and restore the quality and quantity of available resources. Especially for coral reef ecosystem, one of the factor that influence the resilience rate is the availability of hard substrate in a bottom waters as a settlement for coral larvae.
Herbivorous animal has a major influence in determining the rate of coral reef animal larval settlement on a substrate because it can prevent the occurrence of a macro-algae excessive closure of hard substrate. Among herbivorous fish, three families are considered as the indicator of marine ecosystem health, namely Siganidae, Scaridae, and Acanthuridae.
The aim of this study is determining the abundance and diversity of herbivorous fish species, determining the relationship between benthic structure and fish community structure (non-herbivorous and herbivorous reef fish), determining the relationship between the conditions of herbivorous fish species abundance with coral reef resilience level, and formulating recommendations for development of coral reef ecosystem management base on ecology in an integrated and sustainable. The results of this study would be an expected as reference in development of sustainable management of coral reef ecology and reef fish and the concept of sustainable fishing to local fishemen.
The research was conducted in Pulau Tiga Subdistrict waters, Natuna District of Riau Kepulauan Province. Time of research implementation was carried out during five months from April to August 2009.
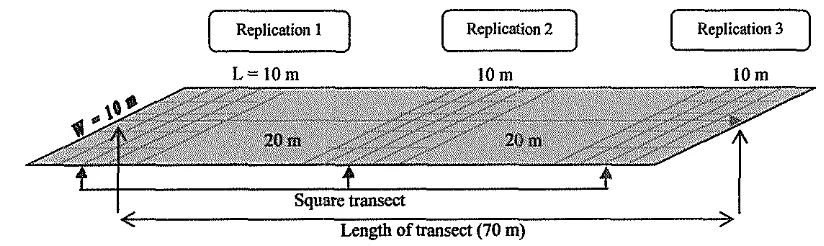
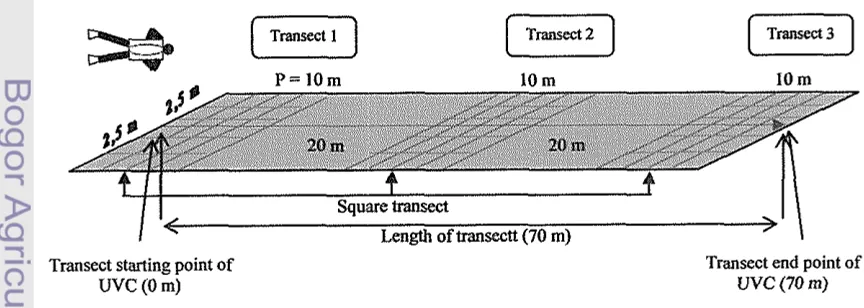
Data collection method that used in the research is survey method for collecting primary data, and desk study for secondary data. The method used for determining coral reefs condition, growth of juvenile corals and algae cover (DCA) was square transect (Quadrat Method) with quotation of observation area width of 10 m x 10 m, modified by line intercept fransect (LIT) method along 70 meters parallel with coastline. Observation of corals was also measured to determine growth rate of new coral (recruitment) with calculating the spread ofnew coral colonies from species of Acropora spp. (diameters ranged fiom 1 to 30 cm). Whereas for the determination of herbivorous fish c o m d t y structure was used modification of Line Intercept Transect (LIT), Quadrat Method (QM) and Underwater Fish Visual Census (UVC).
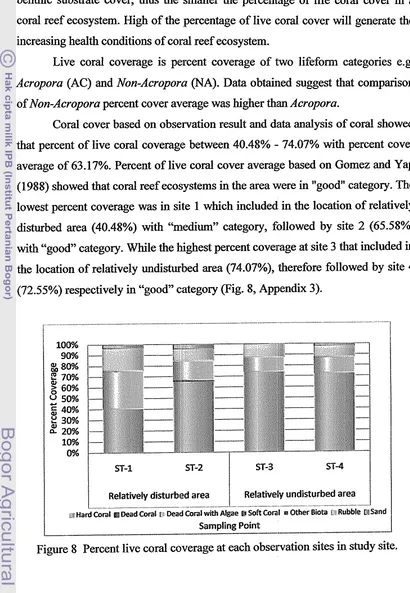
The results showed that the lowest percent coverage was in site 1 which included in the location of relatively disturbed area (40.48%), and the highest percent coverage at site 3 that included in the location of relatively undisturbed area (74.07%) with percent cover average of 63.17% or in "good" category. Growth of juvenile coral colonies was obtained the average of diameter length of the colony ranged from 16.00 to 17.26 cm with a percent coverage ranged from 2.772% to 4.250% and the average number of colonies per transect (100 m-2) ranged from 100 to 200 colonies. Conversely, the highest algae (DCA) cover was found in site 1 (34.830%) and the lowest at site 3 (13.279%).
The number of herbivorous fish species obtained during the study was amounted to 24 species. Individuals of herbivorous fish recorded was amounted to 203 individuals, with the total average of abundance of herbivorous fish in the entire sites of 1,450 individuals per hectare or 5.01% of the total abundance of reef fish, the highest abundance was Scaridae (864 individuals ham') or 57.35%, followed by Siganidae (350 individuals haM') or 23.22%, and Acanthuridae (236 individuals ha-') or 15.64%.
The result of multivariate analysis of the relationship between herbivorous fish abundance, coral reef and alga cover (DCA) showed that algae negatively correlated to the abundance of herbivorous fish, live coral cover and juvenile coral growth. Meanwhile, based on results of correlation test, linear regression and multivariate analysis were selected three species of fish that have a significant relationship of the abundance to live coral cover with the positive correlation, i.e.
C. microrhinos (Gm (2.813)> tmb (2.228), P = 0.018), S. rivulatus (& (2.418)> t a (2.228), P = 0.036), and S. doliatus (h, (3.846p t* (2.228), P = 0.003). Then, the abundance of three species of fish also has a significant relationship to the algae cover (DCA) with a negative correlation, i.e. C. microrhinos (5bt (2.288)> Lb
(2.228), P = 0.045), S. rivulatus (& (2.349)> t* (2.228), P = 0.041) and S. doliatus
(&, (2.813)> t* (2.228), P = 0.018). Implication for coral reef management in Pulau Tiga Subdistrict may be done with a few steps of strategy, which include: a) prohibition of destructive fishing practices, b) maintain water quality that supports the health and growth of coral reef, c) maintain and improve the abundance and diversity of herbivorous fishes, d) prohibition of taking the live coral and dead coral, and, e) environmental engineering.
O
Copyrighted by Institut Petanian Bogor, 2009
Copyright protected by Indonesia Legislation
Copying parts or overall of this paper is prohibited without any permission from the author. Citation is tolerated as long as source is referenced Citation is limited for education purpose, research and analysis, paper writing, report writing, criticism, or cross reference for problem solving, and the citation does not implies negative impact into IPB.
CORAL REEF HEALTH CONDITION
BASED ON HERBIVOROUS FISH DENSITY
IN PULAU TIGA SUBDISTRICT,
NATUNA DISTRICT
DEDY DAMHUDY
Thesis
As one of the requirement for the degree of Master of Science in
Study Program of Coastal and Marine Resources Management Science
POST GRADUATE SCHOOL
BOGOR AGRICULTURAL UNIVERSITY
BOGOR
Title : Coral Reef Health Condition based on Herbivorous Fish Density in Pulau Tiga Subdistrict, Natuna District
Name : Dedy Damhudy
SRN : C252070304
Approved by
Advisory Board
Dr. Ir. M. Mukhlis Kamal M.Sc Dr. Ir. Yunizar Ernawati, MS
Supervisor Co-Supervisor
Acknowledged by
Head of Study Program of Coastal and Marine Resources
Management Science
'---
>
Prof Dr. Ir. Mennofatria Boer, DEA
PREFACE
Thanks to God Almighty (Allah S.W.T) for all blessing and guidance, thus
the Thesis with entitled "Coral Reef Health Condition based on Herbivorous
Fish Density in Pulau Tiga Subdistrict, Natuna District" has been successfully
completed. This Thesis is as one of requirement on Post Graduate title
acomplishment on Study Program of Coastal and Marine Resources Management
Science -Post Graduate School, Bogor Agriculture University.
I
would like to thanks to all parties who have provided assistance, support and guidance to all the time, especially:1. Dr. 11. M. Mukhlis Kamal, M.Sc. as Supervisor and Dr. Ir. Yunizar Ernawati,
MS as Co-Supervisor who has devoted much time and thought for their
direction and suggestion during the writing period.
2. Prof. Dr. Ir. Khairil Anwar Notodiputro, M.S as Dean of Post Graduate
School, Bogor Agriculture University and staff and Prof. Dr. Ir. Mennofatria
Boer, DEA as Head of Study Program of Coastal and Marine Resources
Management Science who have given maximum administrative services.
3. Ministry of Marine Affairs and Fisheries Republic of Indonesia, which has
initiated educational program at Post Graduate School by Coral Reef
Rehabilitation and Management Program - Phase I1 (COREMAP 11-ADB),
during this two years.
4. Drs. H. Daeng Rusnadi, M.Si as Head of Natuna Regency who has given the
o p p o h t y for me to participate in educational program at Post Graduate
School during this two years.
5. My dearest Father (Harsono) and Mother (Supiah) who always giving
support, interest and prayer for me.
6. My dearest Wife (Eka Risanti), and my lovely Children (Fathur Andyka
Putra, Aqillah Dewanti and M Wahyudyka Nurashri) who give me the
motivation, patience and prayer during this educational program.
7. My Old-Brother (Joko Setyo, S.Pi) who has given moral and spiritual supports
to me, and my Old-Sister (Sri Rezeki), my young sisters (Widi Hastuti
8. All colleagues (Mas ArieJ; Dodon, Jojo, Ichal, ni and Mu 'in) for tremendous
support and contribution during the research.
9. My colleagues in Coastal and Marine Resources Management Program 2007,
Sandwich Program COREMAP I1 DKP (Amehr Hakim, Ahmad Jaelani, Budi
Hartono, Cici Kurniawati, Dedy Eka Syaputra, Dini, Endar Maraskuranto,
Febrizal, Hadi Suryanto, Harlim Maharbakti, Hemat Sirait, Ilham, Jimmy,
Jusak Wiraharja, Lida Aro Ndhuru, M. Riza Kurnia Lubis, Partini,
Raymundus Ngajo, Simon Sinaga, Syarviddint Alustco, Tenny Apriliani,
Tema Aro Ndhuru, Wan Irham, Zulfkar).
10. My best-friend as my brothers; l h m as a travelling and studying companion
in the spiritual path of Islam, and Hemat Sirait who always together and
provides happiness in the integrity of friendship.
I realize that this Thesis is still found any imperfection. Therefore, I
welcome constructive suggestion and recommendation from various persons.
Hopefully this Thesis can give sense and benefit to the reader.
Bogor, November 2009
CURRICULUM
VITAE
Dedy Damhudy was born in Tanjungpinang, Riau Kepulauan Province at
December 31S', 2009 with Harsono as father and Supiah as mother. He is the third
child of five children.
In 1994, I graduated from Senior High School (SMUN 1) of
Tanjungpinang and did my baclielor degree at Department of Aquatic Resources
Management, Riau University, Pekanbaru and graduated in 1999.
I have been working for Marine and Fisheries Services (DKP), Natuna
Regency since 2002. In 2007, I have had an opportunity to continue the education
at Post Graduate School, Bogor Agricultural University, Study Program of
TABLE OF CONTENTS
Page
LIST OF TABLES
...
xiiiLIST OF FIGURES
...
xivLIST OF APPENDICES
...
xvi1 INTRODUCTION
1.1 Background
...
1 1.2 Problem Statement...
3 1.3 Objectives and Purpose...
4...
1.4 Logical Framework 4
2 LITERATURE REVIEW
2.1 Coral Reef
...
6...
2.1.1 Biology of Coral 6
...
2.1.2 Characteristics of Coral Reef S
2.1.3 Threat, Limitation Factors of Growth and Phase Shift of 10
. .
...
2.1.4 Coral Reef Resilience
2.2 Characteristic of algae in coral reef ecosystem
...
2.3 Characteristic of Herbivorous Fish as Reef Fish...
2.4 Relationship between Herbivorous Fish Community, Coral Reef and...
Algae
3 MATERIALS AND METHODS
...
3.1 Time and Location
...
3.2 Material and Tools
...
3.3 Data Collection
3.3.1 Primary Data
...
3.3.2 Secondary Data...
3.4 Data Analysis...
4 STUDY AREA OVERVIEW
. .
4.1 Local Geographical Condition
...
4.2 Social and Demographic Condition...
4.3 Community Local Wisdom...
...
4.4 Description of Research Location and Site
5 RESULTS AND DISCUSSION
5.1 Coral Reef Health Rate based on Waters Physical and Chemical
. .
...
Condiaon
5.3 Coral Reef Health Rate based on Fish Community Structure
...
5.3.1 Composition of Species and Family of Reef Fishes...
5.3.2 Density of Species and Family of Herbivorous Fish...
5.3.3 The Presence Relative Frequency of Herbivorous FishSpecies
...
5.3.4 Diversity, Evenness and Dominance of Herbivorous Fish...
5.4Relationship between Benthic Structure and Fish CommunityStructure
...
5.4.1 Benthic Structure of Coral Reef Ecosystem...
5.4.2 Fish Community Structure (Non-Herbivorous and HerbivorousReef Fishes)
...
5.5 Relationship between Herbivorous Fish Density, Coral Reef and...
Algae Cover (DCA)
5.6 Implication for Coral Reef Management
...
xii6 CONCLUSION AND RECOMMENDATION
6.1 Conclusion
...
786.2 Recommendation
...
78REFERENCES
...
SOLIST OF
TABLES
Page
1 Criteria of coral reef assessment based on percent coral coverage (Gomez and Yap 1988)
...
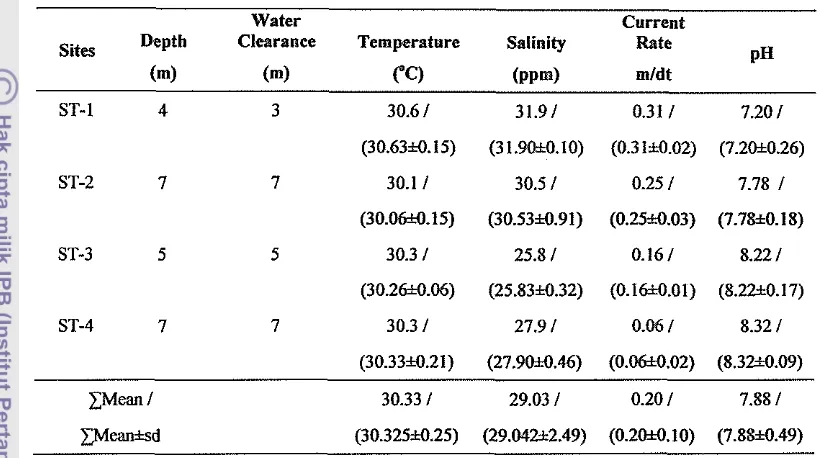
302 Water quality in the average (or meanksd) which measured of each
...
sites of research in Pulau Tiga Subdistrict 43
3 Number and percent coverage of growth of juvenile coral colonies from genus of Acropora at each site in study site
...
514 Six species with the highest reef fish density in study sites
...
575 The highest value of presence relative frequency of herbivorous fish species of the whole observation site in study site
...
626 Correlation and t-student test between density of herbivorous fish species, percent live coral coverage and algae cover @CA)
...
(n=12) 72
LIST OF FIG
S
Page
The flow chart of research framework..
...
5Map of study site
...
25Scheme of square transect plot placement in line intercept transect (English et al. 1994)
...
27Scheme of transect and underwater fish visual census of herbivorous fishes (English et al. 1994)
...
28Photo of waters location in each research sites: (a) site 1 (ST-1); @) site
2 (ST-2); (c) site 3 (ST-3); and (d) site 4 (ST-4)
...
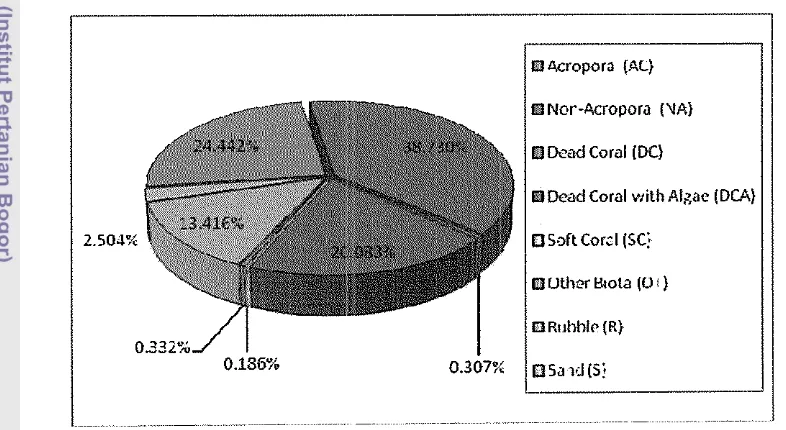
38 Mean of percent benthic coverage of biota and substrate categories in...
the study sites 44
Variation of benthic substrate structure of biotic categorize and other biota (a) and abiotic @) in study site
...
45Percent live coral coverage at each observation sites in study site
...
46Some corals species of genus Acropora, Porifes and Montipora in study site
...
48Values of diversity index
(H'),
evenness index Q and dominance index (C) of coral reef at each observation sites in study site...
50Average of percent coverage of growth of juvenile coral colonies from genus of Acropora at each site in study site
...
52Several lifeform of growth of juvenile coral colonies from genus of Acropora (colonies diameter of 1- 30 cm) in study site
...
53Percent cover of dead cord with algae @CA) at each site in study site
...
55Number of forms of dead coral with algae (DCA) were found in study site..
...
56Values of diversity index (H'), Evenness index Q and dominance index (C) of overall reef fishes (non-herbivorous and herbivorous) at
.
.
each observation sltes m study site...
58Density and dominance of fish from family of Pomacentridae that found in study site
...
59Percent average of herbivorous fish of the whole observation sites in study site
...
61Some of herbivorous fish species h m the family of Scaridae, Acanthuridae, and Siganidae with the highest presence relative frequency in study site
...
62Values of diversity index
(H'),
evenness index (E) and dominance index (C) of herbivorous fish at each observation sites in study site...
63The dendrogram of cluster analysis of biota and substrate characteristic in study site. (T = transect, ST = site)
...
64MDS of biota and substrate characteristic in study site. (T=transect, ST=site, Cl=cluster 1, C2=cluster 2, C3=cluster 3)
...
65PCA-plot of biota and substrate category in study site. (T=transect, ST=site)
...
66The dendrogram of cluster analysis based on individual number of reef fish families (non-herbivorous and herbivorous) in study site. (T =transect, ST=site)
...
67MDS of fish distribution based on individual number of reef fish families (non-herbivorous and herbivorous) in study site. (T=transect,
ST=site. C 1 =cluster 1. C2=cluster 2. C3=cluster 3, C4=cluster
PCA-plot of fish distribution based on individual number of reef fish family (non-herbivorous and herbivorous) in study site. (T=transect,
...
ST=site) 69
The graph of correlation between herbivorous fish density (Ks) toward
live coral cover (LC), junile coral growth (CJ), and algae
...
(DCA) 71
The graph of correlation between density of herbivorous fish species (C. microrhinos,
S.
rivulatus and S. dolintus) to live coral cover(LC), juvenile coral growth (CJ) and algae
...
LIST OF APPE1\TI)ICES
Page
Number of coral species at each study site
...
91Percent coral coverage of each genus in study site
...
93Percent cover of biota and substrate at study site
...
96Species number (S), individuaI number
0,
diversity index(H3,
evenness index (E) and dominance index (C) of coral reef at each site in study site...
97Growth of juvenile coral colonies at site 1
...
98Growth of juvenile coral colonies at site 2
...
99Growth ofjuvenile coral colonies at site 3
...
100Growth of juvenile coral colonies at site 4
...
101Density of species and family of overall reef fishes in study site
...
102Species number (S), individual number
0,
diversity index (H'), evenness index Q and dominance index (C) of overall reef fishes in study site...
105Species number (S), individual number
0,
diversity index (H'), evenness index Q and dominance index (C) of herbivorous fish in study site...
106Correlation and PCA between Percent Live Coral Coverage (LC), dead coral with algae (DCA), juvenile coral (CJ) and herbivorous fish (Ks)
...
107Correlation and PCA between percent live coral coverage (LC), dead coral with algae (DCA), juvenile coral (CJ) and herbivorous fish (Ks) &om species of C. mirrorhinos, S. rivulatm and S. doliatus
...
108Lmear regression between percent live coral coverage (Y1) and DCA
(Yz)
of herbivorous fish species (C. microrhinos (Xi), S. rivulafus (X,), and S. doliatus (X3))...
109Example of coral species diversity inside 1 (one) squae transect with measurement of l m x l m
was
obtained from underwater camera in study site...
1121 INTRODUCTION
1.1 Background
Coral reef ecosystem as one of coastal area ecosystem has a very
important role, both ecologically and economically. The former, coral reef
ecosystem is an area of various associated organisms providing nursery ground,
feeding ground and spawning ground. The latter, it is as a potential fishing
ground, especially for traditional fishermen.
Generally, coral reef ecosystem is very sensitive to environmental impacts
related to human activity on coastal and marine areas. Human impacts, direct and
indirect, have been recognized as a higher threat than the natural disturbances.
The major causes of coral reef degradation in Indonesia are blast fishing, sewage,
industrial pollution and cyanide fishing (Suharsono 1998).
Pulau Tiga as one of subdistricts in Natuna District is the area dominated
by sea areas with very potential marine resource, especially coral reef. Around the
coral reefs areas, fishing activities have been carried out intensively by fishermen
using explosives (bombs) and toxic (potassium). The exertion of the bomb had
been long enough since 1970s, but had almost no longer exists since 2007.
Meanwhile, the use of potassium began to develop in early 1990s. Destructive and
poisonous fishing has been banned since 2007 as the area was selected to be the
COREMAP site.
Nowadays, in the context of community life in Pulau Tiga Subdistrict,
sustainability of community economic activities will be greatly affected by the
waters ecosystem health condition because most of populations are concentrated
in coastal areas and their livelihood depend on the results of marine resources,
particularly fishermen. Consequently, aquatic ecosystems have been exploited at
the area, take time to make natural recovery in order to maintain and restore the
quality and quantity of available resources.
One indication of a good ecosystem health is coral reef ecosystem
resilience rate, explicitly the ability of an ecosystem to recover from a damaged
condition to be better (Salm 2002). Resilience rate of coral reef ecosystem is one
ecosystem. This information is usually needed in assessing vulnerable rate of
coral reef &om disturbances and estimate the length of the process of restoration
and rehabilitation of coral reefs ecosystem (Grimsditch & Salm 2006), especially
for coral reef ecosystem, one of the factor that influence the resilience rate is the
availability of hard substrate in
a
bottom waters as a settlement for coral larvae (Grimsditch & Salm 2006; Salm 2002).Herbivorous animal has a major influence in determining the rate of coral
reef animal larval settlement on a substrate because it can prevent the occurrence
of a macro-algae excessive closure of hard substrate. This excessive closure can
inhibit coral animal larval settlement on the substrate which in turn will reduce the
ability of coral reefs to recover soon. Herbivorous fish feed on a variety of macro-
algae, therefore substrates will always be in clean condition (Marshal &
Schuttenberg 2006).
Four groups of the main herbivorous fish that live in Indonesia coral reefs
are the family of Pomacentridae (damseljsh), Siganidae (rabbitfish),
Acanthuridae (surgeonzsh) and Scaridae (parroflsh). In many tropical areas,
these fish families cover 15-25% of the biomass and diversity of coral species
(Tomascik 1997).
Among herbivorous fish, three families are considered as the indicator of
marine ecosystem health, namely Siganidae, Scaridae, and Acanthuridae (Russ
1984a). In this fish indicates 35-90% composition of diet is algae (Ferreira &
Goncalves 2006). In Lizard Island, Great Barrier Reef, and surrounding, the
abundance of three major herbivorous fish, respectively Achanthuridae (54%),
Scaridae (31%) and Siganidae (14%) (Meekan and Choat 1997). Example of
Siganidae is Siganus spp., Scaridae (Scarus spp.), and Acanthuridae (Acanthurus
spp.). Those Siganids have relatively high economic value.
Thus, to determine coral reef ecosystem health will require a study to
notice the condition of abundance and composition of groups of herbivorous fish
species. Indirectly, the condition can be a bioindicator of coral reef ecosystem
1.2 Problem Statement
Pulau Tiga is one of subdistricts formed from the expansion of the
Bunguran Barat Subdistrict, Natuna District in 2006. In these areas the coastal
waters have the potential resources of coral reefs, which supporting to
surrounding community livelihood. Iluman activities that utilize aquatic resources
by destructive fishing has lasted a long time still exists even today, and has put
pressures and disturbances of its existence, especially the health of coral reefs.
Coral reefs are under pressure and disturbance will create natural
resilience to restore ecosystem functions. The ability of coral reef to perform the
recovery process must be maintained by a variety of ecological factors. One of
these factors is biological factors, especially herbivorous fish. Herbivorous fish is
indicated for health as bio-indicator of reef health also has an economic value.
This phenomenon is an issue for local government and community as
stakeholders to be more serious in managing coral reef resources, because the
recovery rate of coral reef ecosystem is a vulnerability level assessment of
disturbance and the basic for management of coral reef ecosystem.
According to the facts above, subsequently the main problems in this
research are:
1. Dependence of community of Pulau Tiga Subdistrict to coral reef resource
existence as supporting community livelihood.
2. Utilization of aquatic resources by destructive fishing practices which affecting
coral reef health.
3. Existence of reef fishes, especially herbivorous fish indicated as bio-indicator
of reef health even have economic value and tends to decrease.
4. Management of coral reef ecosystem that have not been directed due to lack of
seriousness by the stakeholders in the region.
There are many aspects related to marine ecosystem health problems,
associated with the presence of herbivorous fishes. However, in this study, the
problem is limited and defined in terms of question, as follows: "How is the
prospect of coral reef health and resilience level if viewed from the conditions of
density and community structure of herbivorous fishes in Pulau Tiga Subdistrict
1.3 Objectives and Purposes
The aim of this study is:
1. Determining the abundance and diversity of herbivorous fish species.
2. Determining the relationship between benthic structure and fish community
structure (non-herbivorous and herbivorous reef fish).
3. Determining the relationship between the conditions of herbivorous fish
species abundance with coral reef resilience level.
4. Formulating recommendations for development of coral reef ecosystem
management base on ecology in an integrated and sustainable.
The results of this study would be an expected as reference in development
of sustainable management of coral reef ecology and reef fish and the concept of
sustainable fishing to local fishermen.
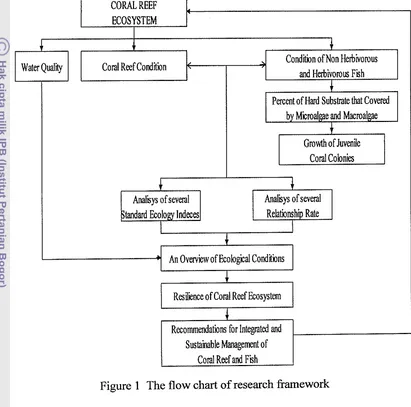
1.4 Logical Framework
Ecologically, relationship between coral reef and herbivorous fish are
supporting each other in the food chain. If stress occurs to coral reef, then it will
affect to herbivorous fish and vice versa.
Basically, there is a strong dynamic between the dynamic presence of
herbivorous fish, macro-algae cover, and coral larvae settlement in the substrate
(Albert et al. 2007). The relationship is consistent as long as measured from the
same habitat type (Russ 1984). Coral animal larvae and macro-algae are
competing to obtain a suitable substrate for their life. Nevertheless, the presence
of herbivorous fish in a certain amount will lead competition victory more of coral
animal larvae (Sluka & Miller 2001). On the basis of these ideas, then the
recovery rate of coral reef ecosystem assessed based on approach the condition of
herbivorous fish abundance.
Conditions of herbivorous fish abundant will reduce macro-algae percent
coverage in a hard substrate because of its existence likely will be grazed by
herbivorous fish. It will provide the opportunity to coral animal larvae add to
settlement rate in suitable substrate. This condition will facilitate a coral reef
Hence, it is necessary to manage the development pattern of coral reef and
the relation to reef fish ecologically, so that the balance between conservation-
based utilization of ecosystem can be maintained.
CORAL REEF ECOSYSTEM
t
C o n d k n ofNon Hedi~omus Water Quality Coral Reef Condition
and Habivomur; Fih
t
I
Percent of Hard Subsink that CoveredI
by Mimalgaeand
M a c m a bGrowth of Jwenii
/
Relabyhip RateI
t
Reskrce of Coml Reef Ecosystem
[image:22.595.92.503.166.573.2]2
LITERATURE REVIEW
2.1 Coral Reef
2.1.1 Biology of Coral
Corals are the animals belong to Phylum Coelenterate (Goreau el al.
1982). Corals consist of polyps that can live in colony and solitary. According to
Boaden and Seed (1985) the corals component comprise: coral skeleton, coral
animal, and zooxhantellae. These components have the interaction between one
coral to another. Likewise, the change of coral animal biomass (polyp) is
determined by the availability of zooxanthellae photosystesis product (quality and
quantity). While, the presence of zooxanthellae is affected by amount of nutrients
or C02 can be reciprocal translocation by coral animal and zooxanthellae.
Coral polyps are composed of soft part and hard part shaped calcareous
skeleton. The mouth is at the top that also functions as an anus. Food is digested
by the mesenterial filaments and leftovers out through the mouth. Coral tissue
composed of ectoderm, mesoglia and endoderm. Ectoderm is the outer tissue and
equipped with cilia, the mucus and a number of nematocyst. Mesoglia is a tissue
that lies between the ectoderm and endoderm, shaped like jelly. Endoderm is the
innermost tissue and contains most of zooxanthellae (Nybakken 1997; Suharsono
1984). The formation of coral skeleton is generally interpreted as an increase in
calcareous skeleton mass of corals, where the live tissue of coral animals covered
by a skeleton composed of calcium carbonate in the form of aragonite (CaC03
crystal fibers) and calcite (a common crystal form of CaC03) (Goreau et al.
1982).
Coral skeleton is structured of calcium carbonate (CaC03) which is
secreted by the epidermis in the middle below the polyp. Secretion process
produces a calcareous skeleton as cup-shaped which settled polyp. A cup is
known as the calyx; wall surrounding called theka; floor of the cup is called the
basal-plate). On the floor occured a septa made of a calcareous radiating septa.
Besides functionate as a place of polyps, skeleton (shell) also provides a shelter
According to Goreau (1959), Calcification increases with decreasing
concentration of C 9 due to photosynthesis activity of zooxanthellae and
influence of carbonic anhidrase enzymes. Reduced C02 will increase the value of
acidity level (pH) becomes more alkaline and thus more easy precipitation of
calcium carbonate. Calcium ions are actively transported by the calcium
bicarbonate through gastrodermis and combined with bicarbonate ions. At first,
forming the dissolved calcium bicarbonate then formed eksoskeleton crystal of
CaC03.
Coral performed both sexual and asexual reproduction. Asexual
reproduction can occur by fragmentation, the release of polyp from the skeleton
and asexual production of larvae. Such reproduction is limited geographically by
the origin of coral, lifeform of colony and growth. In sexual reproduction, gamete
cells will be settled to the mesenterials that usually occurs in every year, seasonal,
monthly or uncertain time. In hermaphrodite or gonochoris coral, spawning events
may take place through external fertilization while the brooding can occurs
through internal fertilazation, both will produce planula which is teleplanic or
philopatric (Veron 1995).
Planula that had been released, will swim toward the light, then swim back
to the bottom, if favorable conditions, they will be attached and form a new
colony. The colonies become sexually mature at the minimum size. Massive coral
of Favia dor-eyensis has sexually mature at 8-years-old colony with a diameter of
10 cm. Some of the branching corals like species of Acropora, Pocillipora, and
Stylophora, reach sexual maturity at a younger age (Barnes & Hughes 1999).
Lifeform of stony coral generally a reflection of the surrounding
environmental condition, morphological plasticity provides an o p p o b t y for
coral reef to local adaptation. For example, species of branching corals with a slim
form generally found in areas with low wave energy, coral colonies in areas with
concentrations of low light, generally sprawl or is shaped like a tubulate, and
many coral reefs in turbid areas have a lifeform more vertical (upward) compared
2.1.2 Characteristics of Coral Reef
Coral reef is a colony of solid formation and composed of solid sediment
skeleton of benthic organisms that live in warm sea waters with a depth of
sufficient light, is formed on physiographic construction of tropical waters, and
mainly consists of calcareous skeleton constructed by a hermatypic coral building
reef (Levinton 1988). Reef or calcareous benthic building is a combination of the
all numbers of biota that are continuous binding of calcium and carbonate ions
from seawater to produce a skeleton (Odum 1971). Then Nybakken (1997) stated
that the reefs are essentially massive deposits of calcium carbonate produced
primarily by corals (phylum Cnidaria, class Anthozoa, order Madreporaria =
Scleractinia) with minor additions from calcareous algae and other organisms that
secrete calcium carbonate.
Coral has some significance, but usually only a general name given to the
order Scleractinia, which the components have a hard calcareous skeleton. Order
scleractinia divided into 2 (two) building groups i.e. reef building and non-reef
building. In the first group known as hermatypic that needed sunlight for survival,
and the second group as ahermatypik, normally, the life does not depend on
sunlight (Veron 1986). The distinguishing feature between the two is that most
hermatypic corals have in their tissues small symbiotic (living together) plant cells
called zooxanthellae, whereas most ahermatypic corals do not have these cells.
Furthermore, hermatypic corals are those that produce reefs, while ahermatypic
corals do not form reefs (Nybakken 1997).
Coral reefs are an ecosystem in a tropical sea that have a high productivity
(Sukarno et al. 1983), the productivity amount of coral reef due to the recycIing of
nutrients through biological processes (Longhurs & Pauly 1987).
Based on lifeform, coral is classified into two groups specifically, stony
coral and soft coral. Stony coral is the common name of the order Scleractinia
which has a hard calcareous skeleton, and is the most important structure in
building reef and red algae, which functions in the growth of the physical
structure of coral reefs, especially for coral reefs facing the sea. While soft coral
better known as Alcyonaria is one of the Coelenterata has an important role in the
limber and not easily broken. Alcyonaria flabby body, but is supported by a large
number of the spines that sturdy, small in size, arranged in such a way that the
flexible body Alcyonaria and not easily broken. These spines contain calcium
carbonate called spicula (Manuputty 1986).
Based on the type, coral reef can be classified into three categories
(Sukarno et al. 1983; Nontji 1993; Nybakken 1997), explicitly:
1. Atoll: this coral reef form is a circle like a ring, around the lagoon with depth
between 40 to 100 meters, as well as the barrier reef, atoll can be lived on the
maximum depth of coral reefs which coral reef can still be alive.
2.
Fringing Reef ov Shore Reej this coral reef lies on the coastline and reachesdepths of less than 40 meters. Fine growth of this reef is in the areas with large
waves, with growth upwards the surface (vertical) and to the open seaward
(horizontal), this coral reef is almost not found on the coast which has many
wide rivers.
3. Barrier Reej This coral reef is located some distance from the coast, separated
by lagoon with a depth that reaches 40 to 70 meters, this coral reef is generally
elongated shape along the coastline as if such a barrier.
Furthermore, according to Sukarno (1993) stated that coral reef
ecosystems can be found basically three kinds of the floor surface which is a
zonation of coral reef ecosystems are:
1. ReefFlat: The floor surface shape of coral reef with flat in the shallow waters
is more influenced by tides and waves of seawater, so that these habitats have
a variety of environmental condition and very large fluctuated. In the tidal
condition, many parts suffer from drought and the type of coastal coral reef
gets a lot of sediment influence, fresh water and the additional nutrient from
the ground from the land.
2. Reef Slope: the floor surface form that sloping can be divided into two reef
slopes. Reef slope with slant to a deeper area, out of reef flat towards the open
sea is called fore reefslope and reef slope towards the lagoon is called back
reef slope or lagoon slope. The state of fore reef slope is opened facing the
wave and opened sea, while the slope of lagoon fairly protected from the
a depth of 15-20 m to surface, coral growth is lush and diverse, dominated by
the branching arms of the Acropora.
3. Lagoon Floor or Sub-Marine-Terrace: form of floor surface that flat in a deep
area has more varied condition than at the lagoon floor which is usually the
area of sediment. The floor of sub-marine-terrrace that shallow is a coral reef
community or benthic community that solid when sufficient of current and
waves that can prevent the accumulating organism brought from shallow water
areas. According to Nybakken (1997), that condition inside the lagoon, with the
small wave and circulation, and greater sediment is not good for coral growth.
Lagoon floor is more overgrown by seaweed (Thalassia, Cynodocea), or green
algae such as Caulerpa and Halimeda.
2.1.3 Threat, Limiting Factors of Growth and Phase Shift of Coral
Causes of coral reef damage are classified into three parts, specifically: (1)
coral damage by biological causes such as the existence of competition, predation,
explosion of phytoplankton population, (2) coral damage by mechanism causes
such as the existence of strong current, sedimentation, volcanism activities,
changes in temperature and salinity, and sunlight penetration, (3) coral damage to
human activities such as oil pollution, chemicals, taking the stony coral for
industrial purposes and building, bombing, a collection of biota and etc.
(Soeharsono 1990).
Illegal fishing with homemade bombs or dynamite is rampant throughout
Southeast Asia and has devastated many coral reefs in the region. In addition to
fish and other organisms being indiscriminately killed, coral skeletons are
shattered by the blasts, leaving fields of broken rubble. This rubble shifts in the
current, abrading or burying any new coral recruits, thereby slowing or preventing
reef recovery (Fox et al. 2003).
Corals 'bleach' (i.e. go pale or snowy-white) as a result of a variety of
stresses, both natural and human-induced, which cause the degeneration and loss
of the coloured zooxanthellae from their tissues. Under normal conditions,
zooxanthellae numbers may fluctuate seasonally as corals adjust to fluctuations in
regular feature in some areas. During a bleaching event, corals may lose 60 - 90% of their zooxanthellae, and the remaining zooxanthellae may lose 50-80% of their
photosynthetic pigments (Glynn, 1996). Once the source of stress is removed,
affected corals may recover, with zooxanthellae levels returning to normal, but
this depends on the duration and severity of the environmental disturbance
(Hoegh-Guldberg, 1999). Prolonged exposure can lead to partial or complete
death of not only individual colonies but also large tracts of coral reef.
Bleaching can also occur in non-reef building organisms such as soft
corals, anemones and certain species of giant clam (Tridacna spp.), which also
have symbiotic algae in their tissues. As with corals, these organisms may also die
if the conditions leading to bleaching are sufficiently severe (Westmacott et al. 2000).
Reefs that are aIready under stress from human activities may be more
susceptible to bleaching when HotSpots develop, since weakened corals may lack
the capacity to cope with the additional stress of increased sea surface
temperature. Furthermore, even after SSTs return to normal, humaninduced
stressful conditions may inhibit the settlement and growth of new corals. Indeed,
reefs that have already been exposed to persistent human disturbances often show
a poor ability to recover (Brown, 1997). On the other hand, a reef that is not
stressed by human activities may have a greater chance of recovery, as
environmental conditions will he closer to those optimal for coral settlement and
growth.
Physical environment play a role in determining the composition of the
coral community, while the biological environment play a role in shaping species
richness. The diversity can occur only after attainment a series of ecological
balance; not only the balance between the coral organisms, but also between the
coral with other organisms, including predator and parasite, and also between
other organisms that have a direct relationship with coral, such as the balance
between herbivorous fishes and macroalgae (Veron 1986).
Temperature is an important parameter of aquatic environment. Increase or
decrease in water temperature due to the powerplant activity which was built
(Suharsono 1990). Optimum temperature of coral growth in the tropical area is 25
- 30°C, extreme temperature will affect the coral animal in the process of
metabolism, reproduction and calcification. Reef-building stony coral requires sea
water temperature is fairly high over 20°C, but will reaches maximum growth at
temperature range of 25 - 30°c (Sumich 1992).
Salinity is another factor limiting the growth of coral reef. The range of
salinity in Indonesian coral growth is between 29 - 33 %o (Coles & Jokiel 1992).
Coral reefs are not found in the waters near the estuary that receive large
freshwater inputs (Sumich 1996). According Nybakken (1997), coral can only live
well in the normal salinity of seawater specifically in the range of 32-35 %, but
stony coral can tolerate high salinity i.e. between 42 960
Sunlight has an important role in the process of structure and growth of
coral, which is to determine the continuity of the photosynthesis process of
calcareous algae symbiotic within coral skeleton (Nybakken 1997). Sunlight is
one of the most important factors limiting the growth of coral reef which light is
needed by zooxanthellae for photosynthesis.
Coral requires clear waters to ensure the availability of light that required
for photosynthesis of zooxanthellae. Each different species of coral have different
tolerance to level of maximum and minimum light availability. This is the main
reason of variation of coral community structure at various depths. Coral reef is
existed in shallow water among 0-50 m with a hard bottom and clear waters
(Veron 1986). Even the reef-building coral can grow at a depth of 80 m on
oceanic islands with clear waters, contrary to the turbid waters, coral habitat found
at a depth of 2 m (Ditlev 1980). Good conditions for coral growth is at depth
between 3-10 m, while at a depth of 10-15 m is a transition area, an area of less
than optimal for the growth of coral reef are at depth between 15-20 m
(Soedharma 1984).
Water movement is a factor in giving a clear water source that carries
oxygen, nutrient and plankton as well to clean the particles that stick to the coral
skin. The absence of wave or current allows the deposition on coral reef, also
reduced an input of clear water that rich of plankton and oxygen (Nybakken
The composition and type of algal colonisation may be important to the
consequences of coral bleaching both in terms of the severity of disturbance and
the potential for future reef recovery. The severity of the disturbance may be
influenced by the effects of the colonising algae on the ability of corals to recover
from bleaching. The composition and successional trajectory of the colonising
algae may also influence the ability of coral populations to re-establish on algal
dominated substratum, either through recolonisation by surviving corals or new
recruitment. The outcome of the competitive intezwtion between corals and
benthic algae may depend on che species of coral and alga involved and the
mechanism mediating the interaction, with consequences for the processes of reef
phase shifts and recovery from disturbances. For example, filamentous turfing
algae delay the regeneration of coral tissue after mechanical damage. In con-
filamentous algae did not affect cord recovery, and in one instance, canopy-
forming Sargassum spp. Beds were found to protect corals from bleaching
damage (Diaz-Pulido & McCook, 2002).
Studies on some facts were done by Nostrom et al. (2009) for an
alternative state of phase shift of coral reef, that during the times, based on
experiments, theories and models indicate that the loss of recovery capacity due to
human intervention, e.g. overfishiig, preceding the phase shift &om coral to
macroalgae, making the system more vulnerable to disturbance. The loss of key
species of herbivorous and increase nutrient contributing to the coral degradation.
Meanwhile, the case shifting to corallimorpharian domination, soft corals and
sponges are triggered by the mass bleaching event and the low tide. In Japan,
particularly coral of Japanese Gardens, chronic oil pollution reduce and prevent
coral resilience. On the other hand, related data suggest that the transition
corallimorpharian domination, sponges, soft corals and sea urchins facilitated by human-influenced environment. However, the relationship was given uncertain,
because the facts did not indicate a causal relationship.
2.1.4 Coral Reef Resilience
Coral reefs are vital ecosystems, providing a source of income, food and
that the goods and services of coral reef provide an annual profit of US $ 30
billion to the economies worldwide (Cesar et al. 2003). Because of this reason,
coral reef health must stay awake, thus has the ability to recover rapidly when
exposed to disturbance both local and regional.
Hughes et al. (2007) defines resilience as the ability of reefs to absorb
recurrent disturbances (e.g., from cyclones, outbreaks of predators, or coral
bleaching events) and rebuild coral-dominated systems. Loss of resilience can
lead to a phase or regime shift to an alternate assemblage that is typically
characterized by hyperabundances of fleshy seaweeds or other opportunistic
species.
The ability of coral reef resilience is the ability of an individual colony or
a coral reef system (including all inhabitants), to defend themselves from
environmental impacts and maintain the ability to recover and grow (Moberg &
Folke 1999). Apparently, the impact is damaging and continuing, slowly reduces
progressively the ability of resilience from these effects. This can interfere with
the recovery of coral reef from a disturbance and probable cause of coral- to algal-
dominated systems (Done 1992, Hughes 1994).
The history of disturbances on a reef contributes to its shvcture because
reefs are naturally dynamic ecosystems. During recovery, species interact and
change their levels of abundance and roles within the community structure. As a
result, reefs may evolve into communities that are substantially different from
those existing prior to the bleaching event, and yet still be diverse and thriving
ecosystems (Westmacott el al. 2000).
A system that tends to return to the same state even after major
perturbations has high resilience, while one that shifts into another state has lower
resilience. Certain factors can increase a coral reefs resilience. They can be
categorised into ecological and spatial resilience factors, with the primary
difference being the scale over which these factors apply (Obura 2005).
Ecological resilience factors are properties present within the spatial boundaries of
the ecosystem. Spatial resilience factors extend beyond ecosystem boundaries and
McClanahan el al. (2002) states that species diversity may have the
capacity to increase ecosystem resilience by ensuring that there is sufficient
informational (species) redundancy to guard against the risks associated with
environmental disturbance.
An important spatial factor for coral reef resilience is the connectivity
among and within coral reefs. Coral's large populations and discharges of larvae
create high genetic diversity that is crucial for resilience against disturbance
(Nystrom and Folke 2001).
Therefore upstream, larval-exporting 'source' reefs with diverse
populations of healthy adult corals are crucial to maintain the genetic diversity
and resilience of downstream, larval-importing 'sink' reefs. Unfortunately, large-
scale mortality on a coral reef reduces its capacity to self-seed, so it is important
that healthy corals produce abundant and robust larvae that reach the degraded
reefs and then settle and grow. It is thus important to identify and protect source
reefs and the ocean currents connecting them to sink reefs (Nystrom et a1 2000).
On the other hand, other field studies show that larvae of reef organisms can
disperse large distances and replenish populations on distant coral reefs
(Grimsditch & Salm 2006).
Furthermore, the mode of reproduction of the corals also determines the
range within which they can repopulate other reefs: asexually-reproducing corals
(from coral fragments) disperse locally while sexually-reproducing corals (from
larvae) can disperse over much larger distances (Nystrom & Folke 2001).
Although not strictly a functional group, dead coral and stable rubble
provide habitat complexity and a substratum for recruits (Grimsditch & Salm
2006). Richmond (1993) stated that in addition to a good connectivity, appropriate
substrates are also crucial for larval settlement. A framework of dead coral or
encrusting calcareous algae provides the best substrate for coral recruitment.
Good substrates should be stable and surrounded by calm waters with salinity
levels between 32%0 and 40%0, and should have adequate light, a lack of
macroalgae, appropriate grazing levels and limited sedimentation. Encrusting
coralline algae that aid settlement and growth of coral recruits also contribute to
types, the varying connectivity, the varying environmental conditions and the
different species involved, coral reef recovery from disturbances through
recolonisation and regrowth can vary greatly spatially and temporally (Gleason
1996).
Historically, coral reefs have been able to recover &om occasional natural
disturbances (e.g. hurricanes, predator outbreaks, and diseases). It is the persistent,
chronic disturbances from human activities that are more damaging today. This
underscores the importance of removing all direct, negative human impacts that
we can, to give reefs the best chance for recovery in the face of bleaching
(Westmacott et al. 2000).
2.2 Algae Characteristic of Coral Reef Ecosystem
Algae can be major space competitors with coral on reefs, invading and
establishing themselves more quickly than corals. Certain algae also bore into
coral skeletons, thus, they break down reef structure. But not all of the algae in the
coral reef ecosystem harm coral existence (Nybakken 1997).
First, the encrusting red coralline algae, such as Liihothamnion, maintain the integrity of the reef by constantly cementing together various pieces of
calcium carbonate, thus, they reinforce the reef against destruction by wave action
and prevent individual pieces fiom being carried off the reef. The algal ridge is
further responsible for breaking the velocity of the waves and producing calmer
conditions that allow growth of other organisms in the reef flat behind it. The
second group of calcareous algae are the greens, dominated by the genus
Halimeda. These algae are significant contributors to the sand found in reefs, particularly in lagoon area. They, therefore, create a special habitat. Algae also
create habitats in and around tliemselves and may furnish necessary shade from
the hot tropical sun for certain organisms. Algal are important as primary
producers in the reef system and as food for various herbivores (Nybakken 1997).
Foliose and turf-forming macroalgae form morphologically distinct
assemblages on shallow reefs worldwide and frequently coexist in a patch mosaic.
Dense mats of low-lying algae commonly referred to as algal turfs, and looser
Turfs are typically dominated by densely packed filamentous and finely branched
algae, and may avoid invasion by taller foliose and coarsely branched algae by
inhibiting their recruitment. The turf growth form may vary in morphology and
species composition and is thought to be a response or adaptation to stress,
including grazing, sedimentation, physical disturbance, and, in the intertidal,
desiccation (Miller et al. 2009).
Based on the generally positive correlation between canopy height and
production, one might expect foliose assemblages to be more productive than
turfs. Nonetheless, algal turfs are highly productive on tropical reefs, supporting
diverse and abundant assemblages of grazers (Miller et al. 2009).
Bleached coral tissue may either regain its zooxanthellae and recover, or
may die, in which case it is generally rapidly colonised by benthic algae. Large-
scale mass-bleaching events may result in massive algal overgrowth of the newly
available substratum provided by the dead coral skeleton. These changes may thus
amount to a partial or extensive phase shift, in which abundant benthic algae
replace abundant corals, potentially amounting to or contributing to long-term reef
degradation just as severe as that caused by diseases, eutrophication or over-
fishing (Diaz-Pulido & McCook 2002).
2.3 Characteristic of Herbivorous Fish as Reef Fish
Reef fish is fish associated with coral reef ecosystem as a habitat. Reef fish
is a fish species that usually live or relatively sedentary and the movement is
relatively easy to reach. Types of substrate to be used as habitat are usually on live
coral, dead coral, rubble and soft coral (Suharti 2005). Some groups of fish in the
refuge and explore coral reefs includes butane fish (herbivores), and groups of
carnivores such as snapper and grouper (Adrim 1983). Group of carnivorous fish
in the coral reef around 50-70%, and almost cover the fish species in this area.
Group of corallivorous and herbivorous fish are about 15%. The fishes of this
group depend on the health of reef to develop their population. Planktivorous and
omnivorous fish group founds only in very small quantities (Choat & Bellwood
Based on the feeding active period, reef fish can be classified as a fish
feeding at night (nocturnal), during the day (diurnal) and some at dusk
(crepuscular). According to Adrim (1983) and TERANGI (2004), 3 (three)
groups of reef fishes are as follows:
1. Nocturnal fish (active at night), approximately 10% of reef fish species are
nocturnal, this fish is hiding in the crevices of coral reef or caves during the
day and will come to the surface of the water to find food at night. For
example, the fish from the family of Holocentridae, Apogonidae, Humilidae,
Priacanthidae (bigeyes), Muranidae (eels), Serranidae (iewjsh) and several
family of Mullidae (goatjishes).
2. Diurnal fish (active when the daytime), 75% of fish that live in the coral reef
and some of these fish are very attractive color and generally very closely
related to coral reefs, for example, the fish from the family of Labridae
(wrasses), Chaetodontidae (butterflyjshes), Pomacentridae (danzselJishes),
Scaridae @arro@shes), Acanthuridae (surgeonjshes), Monacanthidae,
Ostracionthidae (boxjshes), Etraodonthidae, Canthigasteridae and some
Mullidae (goafjshes).
3. Crepuscular fish (active in the morning or the afternoon until early evening).
For example, the fish from the family of Sphyraenidae (baracudas),
Serranidae (groupers), Carangidae (iacks), Scorpaenidae (lionjshes),
Synodontidae (lizardjshes), Carcharhinidae, Larnnidae, Spymidae (shark)
and some of the Muraenidae (eels).
Reef fish has an important role in coral reef ecosystem, thus reef fish can
be classified based on their role. The first role as the target fish, the fish is a target
for fishing or better known as an important economic fish or consumption fish
such as Serranidae, Lutjanidae, Kyphosidae, Lethrinidae, Acanthuridae, Mullidae,
Siganidae, Labridae (Chelinus, Himigymnus, and Choerodon) and Haemulidae.
Second, as a determining fish (indicator) for coral reef because the fish is closely
associated with the fertility of coral reef, the fish family of Chaetodontidae
(Ehrlich 1975; Kuiter 1992). Reef fish species commonly found in large amounts
(major) and used as seawater aquarium fish is of the type (Pomacentridae,
Herbivorous fish is the main perpetrator of grazer or herbivory, in addition
to sea urchins (Echinoidae). Among the herbivorous animals, sea urchin
(Diadema spp.) and the fish of Scarus spp. and Siganus spp. are the major
herbivore groups (Bachtiar 2008).
Russ (1984a) who conducted a survey of herbivory in nine coral reefs, in
the Great Barrier Reef has limited herbivorous fish on the family of
Achanthuridae, Scaridae and Siganidae. At Lizard Island, GBR, and surroundings,
an abundance of the three main herbivorous fish, respectively Achanthuridae
(54%), Scaridae (31%) and Siganidae (14%) (Meekan & Choat 1997). In the San
Blas Islands, Panama, also showed a similar pattern, although there is one location
where Kyphosidae shows the proportion of comparable abundance of
Achanthuridae, Scaridae and Siganidae. In the Ambergris Caye, Belize,
herbivorous fish biomass composition was different from Lizard Island and the
San Blas Islands with Scaridae (65.4%) as the most dominant followed by
Acanthuridae (30.1%) and Pomacentridae (4.5%) (Williams et al. 2001).
Herbivory by Pomacentridae is specific because territorial, so not discussed
together with herbivory by Scaridae, Acanthuridae and Siganidae.
Variation of ecological fimction can be shown by the fish from the family
of Scaridae. In Australia GBR, Scarid herbivorous fishes have been classified
based on myology and osteology of oral and pharyngeal jaws into three functional
groups, i.e., as excavators (Bolbometopon muricatum, Cetoscarus bicolor and
Chlorurus spp.), scrupers (Hipposcarzr spp. and Scarus spp.), and croppers
(Calotomus spp., and Leptoscarus vaigiensis) (Bellwood & Choat 1990; Bellwood
1994). Excavators and scrapers fish consume macroalgae and all at once coral
polyps. Both groups open a settlement space for coral larvae and macroalgae
spore. Croppers Fish only eats macroalgae, thus coral juvenile can grow better.
Parrotfishes (scarids) and the echinoid are the dominant external
bioeroders. Both, Eclunoids and Scarids erode substratum by removing calcareous
material as they graze the epilithic algal matrix. Scarid has long been recognized
as a distinctive group of bioeroders, with numerous studies documenting their
erosional activities. This designation was based largely on their feeding mode,
large proportion of finely ground carbonate. Scarids have been long regarded as a
relatively uniform group of grazing or scraping herbivores that erode the
substratum during feeding (Sale 2002). However, based on observations of
functional morphology, ecology, and feeding behavior of scarids, Bellwood and
Choat (1990) suggested that only a minority of Indo-Pacific scarid species are
significant bioeroders. A comparable pattern is seen in the Caribbean. In both the
Caribbean and the Indo-Pacific, scarid erosion is dependent on a few species, with
local patterns dependent on the abundance, size, and behavior of this key species.
In the Indo-Pacific, one of the primary eroders, Chlorurus microrhinos (formerly
Chlorurus gibbus), has very spesific feeding patterns, with most erosion being
from convex epilithic algal-covered substrata in shallow water, near the reef edge.
Herbivorous fish based on the abundance is varies among the reef-shelves.
In the Central Section GBR, Russ (1984a) reported that the abundance of
herbivorous fishes, Acanthuridae and Scaridae were diverse among the reef-
shelves, while Siganidae was not more changed. Number of species and
abundance of Acanthuridae and Scaridae populations are generally higher in the
midshelf and outershelf, even as in the innershelf most dominated by Siganidae.
In the Northern Section GBR, Scaridae community showed a high abundance and
low biomass in the innershelf, whereas Scaridae showed the low abundance with a
high biomass in the outershelf (Hoey & Bellwood 2008). These results also reflect
a difference in body size and composition of Scaridae fish community
intershelves. Fish Bolbometapon mztricaturn in a large size were found on the
outershelf and became the major bioeroders of calcareous erosion (87%) and
corallivors (99%), while Scarus rivztlatus fish was the major herbivory in the
innershelf (70%) (Hoey & Bellwood 2008).
On a scale of tens or hundreds kilometers, herbivores that play an
important role in herbivory may vary. On coral reef in the Nymph Island and the
Turtle Group, GBR, Scarus rivulatus was reported
as
the most important herbivore (Hoey & Bellwood 2008), while Siganus canaliculatus was reported asan important herbivorous fish on coral reef in the Pioneer Bay, Orpheus Island
2.4 Relationship between Herbivorous Fish Community, Coral Reef and Algae
Relationship between the reef fish and reefs in a similar ecology of the
area is complex, for example of a special relationship that occurs of consumer
species of benthic sessile and small invertebrate. This resulted in most diversity must be identified. Substratum complexity as shelter is more characteristic of the
ecological characteristic of reef fish population than the substratum as a source of
feed (Choat & Bellwood 1991). Diversities and densities of high reef fish caused by the many variation of habitat found in coral reefs. These fish have a narrower
ecological niche, so that more species can only move in a particular area. As a
result of this state, the fish are confined of localized in certain areas on coral reef.
In addition to, among the fish that can immigrate and even some species of
sheltered their area (Nybakken 1997).
Habitat associations can be used to explain the distribution patterns of reef
fish and many species have large geographic distribution. Fish groups are always
associated with coral will reach a very high abundance in a habitat that has a large
geographic range. This association may be used as an