Postharvest Biology and Technology 20 (2000) 203 – 206
Short communication
Long term, low temperature storage injures kangaroo paw
cut flowers
Daryl C. Joyce
a,*, Anthony J. Shorter
baPosthar6est Group,Cranfield Uni6ersity,Silsoe,Bedfordshire,MK45 4DT,UK bCSIRO Plant Industry,120Meiers Road,Long Pocket,Qld.4068,Australia
Received 6 March 2000; accepted 9 May 2000
Abstract
Cut kangaroo paw (Anigozanthosspp.) inflorescences are regarded as having poor storage potential. Storage for up to 4 weeks at temperatures between 0 and 10°C was investigated. Post-storage vase life and chlorophyll fluorescence data collected for the hybrids ‘H1’ and ‘Bush Dawn’ showed that this crop is chilling sensitive. There was a marked drop in quality at B2.0°C. Between 2 and 5°C is a relatively safe storage temperature range for cut kangaroo paw
flowers. © 2000 Elsevier Science B.V. All rights reserved.
Keywords:Anigozanthosspp.; Chilling injury; Hybrids
www.elsevier.com/locate/postharvbio
1. Introduction
Cut kangaroo paw (Anigozanthosspp.) inflores-cences are produced in Africa, the Americas, Aus-tralasia and Europe and are marketed around the world. Holding cut flowers at low temperature for long periods is attractive for both storage and transport, as long-term storage offers greater flex-ibility in terms of selling time. Extended transport periods also permit export by sea, which can be a less expensive alternative to exporting by air.
Kangaroo paw flowers, however, are not re-garded as having good storage potential (Joyce et al., 1993a). In storage experiments running for up to 5 weeks at 0 – 2°C, the vase life of the kangaroo paw cut flower varieties ‘Red’ (A. rufus) and ‘Yellow’ (A. pulcherrimus) and of the hybrids ‘Gold Fever’ (A.pulcherrimusxA.fla6idus), ‘Bush
Harmony’ (A. pulcherrimus x A. fla6idus), ‘Bush Dawn’ (A. pulcherrimus x A. fla6idus) and ‘Ruby
Delight’ (A. fla6idus x A. rufus) were reduced
substantially with increasing time (Seaton and Joyce, 1989; Jones and Faragher, 1991; Teagle et al., 1991). Moreover, storage for weeks at such low temperatures results in a marked loss in vase * Corresponding author. Fax: +44-1525-863277.
E-mail address:[email protected] (D.C. Joyce).
D.C.Joyce,A.J.Shorter/Posthar6est Biology and Technology20 (2000) 203 – 206
204
life compared with other cut flowers (Seaton and Joyce, 1989; Jones and Faragher, 1991). This rela-tively dramatic loss in vase life upon protracted storage has led to the suggestion that kangaroo paw flowers are chilling sensitive (Joyce et al., 1993b).
To investigate the hypothesis that kangaroo paw flowers are indeed chilling sensitive, storage experiments were conducted with the commercial cut flower kangaroo paw hybrids ‘H1’ (A.mangle
-sii x A. fla6idus) with red flowers, and ‘Bush
Dawn’ (A. pulcherrimusxA.fla6idus) with yellow
flowers.
2. Materials and methods
‘H1’ and ‘Bush Dawn’ inflorescences, with one to three open flowers, were harvested during the spring flowering season (September/October) from a cut flower farm near Gatton (27°33% S,
152°17% E), Queensland. These were dipped in
Rovral fungicide (1 g l−1; a.i. iprodione) and allowed to air dry. For storage, individual infl-orescences were wrapped in newsprint moistened with deionized water and then over-wrapped with a loosely sealed 35-mm thick plastic sleeve. The inflorescences were stored in research coldrooms belonging to the Department of Primary Indus-tries (Hamilton, Qld.).
Identical experiments were initially conducted with each of the hybrids. The treatments were control (non-stored) and storage for 4 weeks at 0, 2.5, 5, 7.5 or 10°C. In a third experiment, treat-ments for ‘Bush Dawn’ flowers were control (non-stored) and storage for 1, 2, 3 or 4 weeks at 0, 1, 2, 4 or 8°C.
Stem ends were trimmed prior to vase life as-sessment in a 22°C controlled environment room. Chlorine (25 mg l−1
) was used as the vase solu-tion. Vase life was assessed as days to each of pronounced wilting and discoloration. The aver-age number of flowers open per day over the first 6 days of vase life (days 0 – 5 inclusive) and the chlorophyll fluorescence ratio on day 0 of vase life assessment were recorded as additional measures of flower condition. Minimum fluorescence (F0)
and maximum fluorescence (Fm) parameters were measured with a Walz PAM fluorometer (Effet-trich, Germany) following dark acclimation for 30 min. Relative fluorescence orFv/Fmwas calculated as (Fm−F0)/Fm.
Experiments were arranged in a completely ran-domized design. Single stem replication was 10-fold for the paired experiments with ‘H1’ and ‘Bush Dawn’ and 5-fold for the additional experi-ment with ‘Bush Dawn’ (see above). Treatexperi-ment effects data were evaluated by one way analysis of variance and least significant difference values (LSD; P=0.05) calculated for comparisons of treatment means.
3. Results and discussion
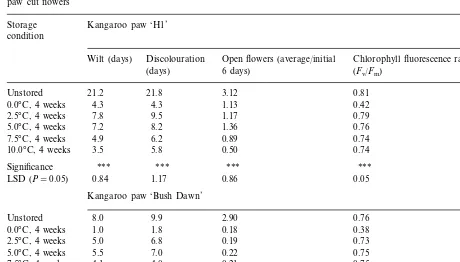
Trends in the results were generally similar for both kangaroo paw cut flower hybrids. Storage for 4 weeks markedly reduced vase life compared to non-stored control flowers (Table 1). Storage at 0°C consistently gave reduced vase life in terms of time both to wilt and to discolour compared to the higher storage temperatures of 2.5, 5 and 7.5°C.
The adverse effect of the 0°C storage treatment was visibly evident upon removal of the flowers from storage. Stems of inflorescences that suffered chilling injury collapsed in the region immediately below the flowers, particularly for hybrid ‘H1’. Moreover, flowers on inflorescences from 0°C were twisted in orientation. Deterioration also occurred at the highest storage temperature of 10°C, but was usually not so dramatic as at 0°C. This deterioration was attributed to development and senescence during storage at higher temperatures.
Flower opening was notably reduced by long term, low temperature storage (Table 1). How-ever, the degree of reduction in flower opening was generally similar across all storage tempera-tures. Hybrid ‘H1’ appeared to maintain a pro-portionally greater degree of flower opening following storage than ‘Bush Dawn’.
D.C.Joyce,A.J.Shorter/Posthar6est Biology and Technology20 (2000) 203 – 206 205
Table 1
Time to wilting and to discolouration, average number of open flowers during the initial 6 days of vase life and chlorophyll fluorescence ratio on day 0 for non-stored (control) and stored (0, 2.5, 5, 7.5 or 10°C for 4 weeks) ‘H1’ and ‘Bush Dawn’ kangaroo paw cut flowers
Storage Kangaroo paw ‘H1’ condition
Discolouration Open flowers (average/initial
Wilt (days) Chlorophyll fluorescence ratio on day 0
6 days) (Fv/Fm)
(days)
21.8 3.12
21.2 0.81
Unstored
4.3 1.13
0.0°C, 4 weeks 4.3 0.42
9.5 1.17
7.8 0.79
2.5°C, 4 weeks
8.2 1.36
5.0°C, 4 weeks 7.2 0.76
6.2 0.89
4.9 0.74
7.5°C, 4 weeks
10.0°C, 4 weeks 3.5 5.8 0.50 0.74
***
Significance *** *** ***
0.84
LSD (P=0.05) 1.17 0.86 0.05
Kangaroo paw ‘Bush Dawn’
9.9
Unstored 8.0 2.90 0.76
1.8
0.0°C, 4 weeks 1.0 0.18 0.38
6.8 0.19
5.0 0.73
2.5°C, 4 weeks
7.0
5.0°C, 4 weeks 5.5 0.22 0.75
4.0 0.21
4.1 0.75
7.5°C, 4 weeks
2.5 0.30
10.0°C, 4 weeks 3.1 0.71
*** ***
*** ***
Significance
LSD (P=0.05) 0.95 1.38 0.65 0.05
by the 0°C storage treatment, where they were almost halved compared to all other treatments (Table 1). This effect implies that a physicochemi-cal lesion resulting in loss in efficiency of utiliza-tion of light energy by chloroplasts is specifically associated with the lowest storage temperature. This damage can be regarded as a form of chilling injury (Raison and Lyons, 1986). Chilling of sus-ceptible plant species is known to cause a drop in
Fv (Smillie, 1979). There was no visible evidence of tissue freezing, such as water soaking (Harden-burg et al., 1986).
The proposition that loss of vase life was due to chilling injury was confirmed by data from the
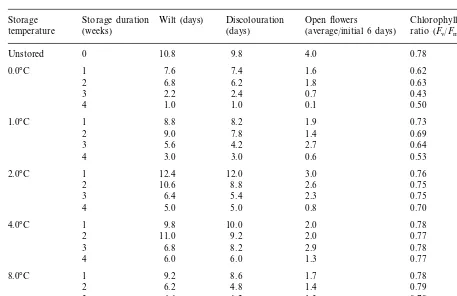
second experiment with ‘Bush Dawn’ (Table 2). Vase life following storage for 4 weeks at 0, 1 and 2°C increased progressively with temperature, as did flower opening and the chlorophyll fluores-cence ratio measured on the day of removal from coldstorage.
D.C.Joyce,A.J.Shorter/Posthar6est Biology and Technology20 (2000) 203 – 206
206
Table 2
Time to wilting and to discolouration, average number of open flowers during the initial 6 days of vase life and chlorophyll fluorescence ratio on day 0 for non-stored (control) and stored (0, 1, 2, 4 or 8°C for 1, 2, 3 or 4 weeks) ‘Bush Dawn’ kangaroo paw cut flowers
Storage Storage duration Wilt (days) Discolouration Open flowers Chlorophyll fluorescence (days) (average/initial 6 days) ratio (Fv/Fm)
(weeks) temperature
Unstored 0 10.8 9.8 4.0 0.78
7.6 7.4
Significance *** *** ***
0.62
LSD (P=0.05) 0.61 0.50 0.02
Acknowledgements
The authors thank Ken Young for donating the flowers used in this study and the Department of Primary Industries, Queensland for access to cold-room facilities.
References
Hardenburg, R.E., Watada, A.E., Yang, C.Y., 1986. The Commercial Storage of Fruits, Vegetables, and Florist and Nursery Stocks. Agricultural Handbook No. 66 (revised). US Department of Agriculture, 130 pp.
Jones, R., Faragher, J., 1991. Cold storage of selected mem-bers of the Proteaceae and Australian native cut flowers. HortScience 26, 1395 – 1397.
Joyce, D., Jones, R., Faragher, J., 1993a. Postharvest
charac-teristics of native Australian flowers. Postharv. News In-form. 4, 61N – 67N.
Joyce, D.C., Joyce, P.A., Miranda, J., Hetherington, S., Shorter, A.J. 1993b. Is kangaroo paw cv. ‘Bush Dawn’ chilling sensitive? Proceedings of the Australasian Posthar-vest Conference, 20 – 24th September, 1993, The University of Queensland, Gatton College, pp. 149 – 152.
Raison, J.K., Lyons, J.M., 1986. Chilling injury: a plea for uniform terminology. Plant Cell Environ. 9, 685 – 686. Seaton, K.A., Joyce, D.C., 1989. Cold storage of Geraldton
wax, kangaroo paw and banskia. Proceedings of the Fifth Australian Agron Conference, September, 1989, The Aus-tralian Society of Agron., p. 532.
Smillie, R.M., 1979. The useful chloroplast: A new approach for investigating chilling stress in plants. In: Low Tempera-ture Stress in Crop Plants. Academic Press, New York, pp. 187 – 202.
Teagle, S., White, J., Sedgley, M., 1991. Post-harvest vase life of cut flowers of three cultivars of kangaroo paws. Sci. Hortic. 48, 277 – 284.