SUPPLEMENTARY MATERIAL
(Tables S1–S3)
Theoretical Study of Reaction of Trifluoromethyl Radical
with Hydroxyl and Hydrogen Radicals
Haruhiko Fukaya, Taizo Ono, and Takashi Abe
Chemistry Department, National Industrial Research Institute of Nagoya
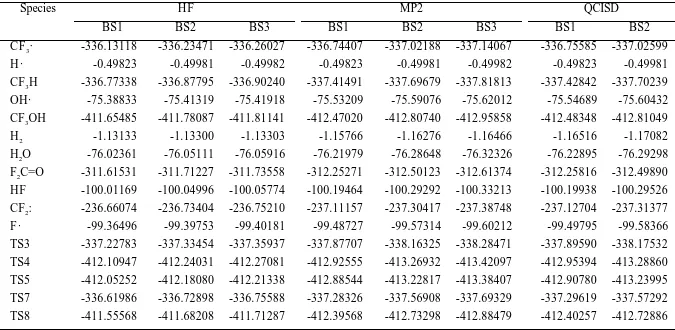
TABLE S1.
The electronic energies calculated by ab initio methods.a
Species HF MP2 QCISD
BS1 BS2 BS3 BS1 BS2 BS3 BS1 BS2
CF3· -336.13118 -336.23471 -336.26027 -336.74407 -337.02188 -337.14067 -336.75585 -337.02599
H · -0.49823 -0.49981 -0.49982 -0.49823 -0.49981 -0.49982 -0.49823 -0.49981
CF3H -336.77338 -336.87795 -336.90240 -337.41491 -337.69679 -337.81813 -337.42842 -337.70239
OH· -75.38833 -75.41319 -75.41918 -75.53209 -75.59076 -75.62012 -75.54689 -75.60432
CF3OH -411.65485 -411.78087 -411.81141 -412.47020 -412.80740 -412.95858 -412.48348 -412.81049
H2 -1.13133 -1.13300 -1.13303 -1.15766 -1.16276 -1.16466 -1.16516 -1.17082
H2O -76.02361 -76.05111 -76.05916 -76.21979 -76.28648 -76.32326 -76.22895 -76.29298
F2C=O -311.61531 -311.71227 -311.73558 -312.25271 -312.50123 -312.61374 -312.25816 -312.49890
HF -100.01169 -100.04996 -100.05774 -100.19464 -100.29292 -100.33213 -100.19938 -100.29526
CF2: -236.66074 -236.73404 -236.75210 -237.11157 -237.30417 -237.38748 -237.12704 -237.31377
F · -99.36496 -99.39753 -99.40181 -99.48727 -99.57314 -99.60212 -99.49795 -99.58366
TS3 -337.22783 -337.33454 -337.35937 -337.87707 -338.16325 -338.28471 -337.89590 -338.17532
TS4 -412.10947 -412.24031 -412.27081 -412.92555 -413.26932 -413.42097 -412.95394 -413.28860
TS5 -412.05252 -412.18080 -412.21338 -412.88544 -413.22817 -413.38407 -412.90780 -413.23995
TS7 -336.61986 -336.72898 -336.75588 -337.28326 -337.56908 -337.69329 -337.29619 -337.57292
TS8 -411.55568 -411.68208 -411.71287 -412.39568 -412.73298 -412.88479 -412.40257 -412.72886 a
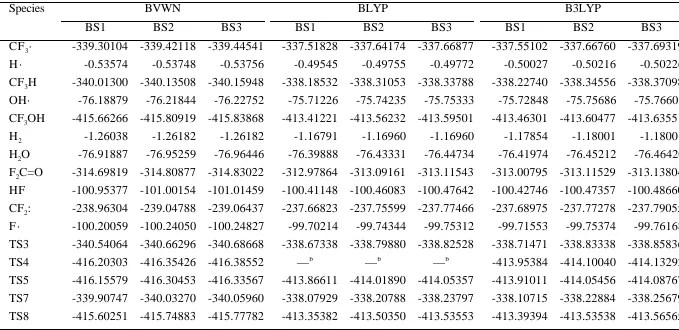
TABLE S2.
The electronic energies calculated by the DFT methods.a
Species BVWN BLYP B3LYP
BS1 BS2 BS3 BS1 BS2 BS3 BS1 BS2 BS3
CF3· -339.30104 -339.42118 -339.44541 -337.51828 -337.64174 -337.66877 -337.55102 -337.66760 -337.69319
H · -0.53574 -0.53748 -0.53756 -0.49545 -0.49755 -0.49772 -0.50027 -0.50216 -0.50226
CF3H -340.01300 -340.13508 -340.15948 -338.18532 -338.31053 -338.33788 -338.22740 -338.34556 -338.37098
OH· -76.18879 -76.21844 -76.22752 -75.71226 -75.74235 -75.75333 -75.72848 -75.75686 -75.76603
CF3OH -415.66266 -415.80919 -415.83868 -413.41221 -413.56232 -413.59501 -413.46301 -413.60477 -413.63551
H2 -1.26038 -1.26182 -1.26182 -1.16791 -1.16960 -1.16960 -1.17854 -1.18001 -1.18001
H2O -76.91887 -76.95259 -76.96446 -76.39888 -76.43331 -76.44734 -76.41974 -76.45212 -76.46420
F2C=O -314.69819 -314.80877 -314.83022 -312.97864 -313.09161 -313.11543 -313.00795 -313.11529 -313.13804
HF -100.95377 -101.00154 -101.01459 -100.41148 -100.46083 -100.47642 -100.42746 -100.47357 -100.48660
CF2: -238.96304 -239.04788 -239.06437 -237.66823 -237.75599 -237.77466 -237.68975 -237.77278 -237.79055
F · -100.20059 -100.24050 -100.24827 -99.70214 -99.74344 -99.75312 -99.71553 -99.75374 -99.76168
TS3 -340.54064 -340.66296 -340.68668 -338.67338 -338.79880 -338.82528 -338.71471 -338.83338 -338.85836
TS4 -416.20303 -416.35426 -416.38552 —b
—b
—b
-413.95384 -414.10040 -414.13292
TS5 -416.15579 -416.30453 -416.33567 -413.86611 -414.01890 -414.05357 -413.91011 -414.05456 -414.08767
TS7 -339.90747 -340.03270 -340.05960 -338.07929 -338.20788 -338.23797 -338.10715 -338.22884 -338.25679
TS8 -415.60251 -415.74883 -415.77782 -413.35382 -413.50350 -413.53553 -413.39394 -413.53538 -413.56565 a
The energies are given in hartree. b
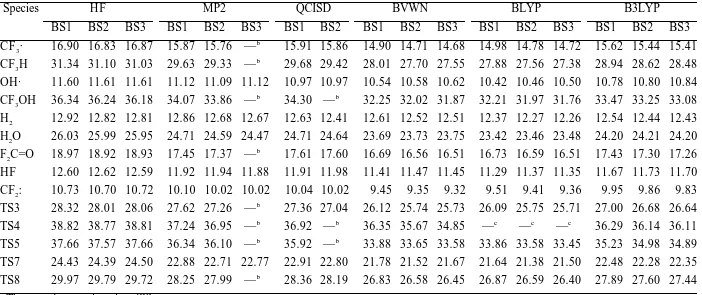
TABLE S3.
The thermal energy corrections at 298.15 K.a
Species HF MP2 QCISD BVWN BLYP B3LYP
BS1 BS2 BS3 BS1 BS2 BS3 BS1 BS2 BS1 BS2 BS3 BS1 BS2 BS3 BS1 BS2 BS3
CF3· 16.90 16.83 16.87 15.87 15.76 —b
15.91 15.86 14.90 14.71 14.68 14.98 14.78 14.72 15.62 15.44 15.41
CF3H 31.34 31.10 31.03 29.63 29.33 —b
29.68 29.42 28.01 27.70 27.55 27.88 27.56 27.38 28.94 28.62 28.48
OH· 11.60 11.61 11.61 11.12 11.09 11.12 10.97 10.97 10.54 10.58 10.62 10.42 10.46 10.50 10.78 10.80 10.84
CF3OH 36.34 36.24 36.18 34.07 33.86 —b
34.30 —b
32.25 32.02 31.87 32.21 31.97 31.76 33.47 33.25 33.08
H2 12.92 12.82 12.81 12.86 12.68 12.67 12.63 12.41 12.61 12.52 12.51 12.37 12.27 12.26 12.54 12.44 12.43
H2O 26.03 25.99 25.95 24.71 24.59 24.47 24.71 24.64 23.69 23.73 23.75 23.42 23.46 23.48 24.20 24.21 24.20
F2C=O 18.97 18.92 18.93 17.45 17.37 —b
17.61 17.60 16.69 16.56 16.51 16.73 16.59 16.51 17.43 17.30 17.26
HF 12.60 12.62 12.59 11.92 11.94 11.88 11.91 11.98 11.41 11.47 11.45 11.29 11.37 11.35 11.67 11.73 11.70
CF2: 10.73 10.70 10.72 10.10 10.02 10.02 10.04 10.02 9.45 9.35 9.32 9.51 9.41 9.36 9.95 9.86 9.83
TS3 28.32 28.01 28.06 27.62 27.26 —b
27.36 27.04 26.12 25.74 25.73 26.09 25.75 25.71 27.00 26.68 26.64
TS4 38.82 38.77 38.81 37.24 36.95 —b
36.92 —b
36.35 35.67 34.85 —c
—c
—c
36.29 36.14 36.11
TS5 37.66 37.57 37.66 36.34 36.10 —b
35.92 —b
33.88 33.65 33.58 33.86 33.58 33.45 35.23 34.98 34.89
TS7 24.43 24.39 24.50 22.88 22.71 22.77 22.91 22.80 21.78 21.52 21.67 21.64 21.38 21.50 22.48 22.28 22.35
TS8 29.97 29.79 29.72 28.25 27.99 —b
28.36 28.19 26.83 26.58 26.45 26.87 26.59 26.40 27.89 27.60 27.44 a
The energies are given in millihartree. b
Could not calculate. c