Journal Information
APAC is devoted to publishing research papers for students, technicians and professors to share advances in Pure and Applied Chemistry. APAC is likely to play an important role in science and engineering in the future. This journal serves as a platform that fosters new applications for all scientists and engineers engaged in research and development in these fast growing fields. All publications are open access in full text and free to download.
Mailing Address
World Science Publisher, New York, NY, United States
Focus and Scope
Topics considered include but are not limited to: x Agrochemistry
x Analytical Chemistry x Astrochemistry
x Atmospheric Chemistry x Biochemistry
x Chemical Biology x Chemical Engineering x Chemo-informatics x Electrochemistry
x Environmental Chemistry x Femtochemistry
x Flavor Chemistry x Flow Chemistry x Geochemistry x Green Chemistry x Histochemistry x History of Chemistry x Hydrogenation Chemistry x Immunochemistry x Inorganic Chemistry x Marine Chemistry x Materials Chemistry x Materials Science x Mathematical Chemistry x Mechanochemistry x Medicinal Chemistry x Molecular Biology x Molecular Mechanics x Nanotechnology
x Neurochemistry x Nuclear Chemistry x Oenology
x Organic Chemistry x Organometallic Chemistry x Petrochemistry
x Pharmacology x Photochemistry x Physical Chemistry
x Physical Organic Chemistry x Phytochemistry
x Polymer Chemistry x Radiochemistry x Solid-state Chemistry x Sonochemistry
x Supramolecular Chemistry x Surface Chemistry
x Synthetic Chemistry x Theoretical Chemistry x Thermochemistry
Peer Review Process
APAC receives more submissions than it can publish. Therefore, we ask peer-reviewers to keep in mind that every paper that is accepted means that another good paper must be rejected. To be published in APAC, a paper should meet four general criteria: Evidence, Novel, Necessity, and Interesting.
In general, a paper should represent an advance to influence thinking in the field to be acceptable. There should be a discernible reason why the work deserves the visibility of publication in this journal rather than the best of the specialist journals.
Open Access Policy
This journal provides immediate open access to its content on the principle that making research freely available to the public supports a greater global exchange of knowledge.
Submission Preparation Checklist
As part of the submission process, authors are required to check off their submission's compliance with all of the following items, and submissions may be returned to authors that do not adhere to these guidelines.
1. The submission has not been previously published, nor is it before another journal for consideration 2. The submission manuscript iswell preparedthat are complete andready for publication.
3. Include or give editor the right to includeat least 3 relatedpapers published in journals of WSP in your references
Copyright Notice
Proposed Creative Commons Copyright Notices
1. Proposed Policy for Journals That Offer Open Access
Authors who publish with this journal agree to the following terms:
1. Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under aCreative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).
Proposed Policy for Journals That Offer Delayed Open Access
Authors who publish with this journal agree to the following terms:
1. Authors retain copyright and grant the journal right of first publication, with the work [SPECIFY PERIOD OF TIME] after publication simultaneously licensed under aCreative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).
Privacy Statement
The names and email addresses entered in this journal site will be used exclusively for the stated purposes of this journal and will not be made available for any other purpose or to any other party.
Author Fees
Fast-Track Review: 50.00 (USD)
With the payment of this fee, the review, editorial decision, and author notification on this manuscript is guaranteed to take place within 1 week.
Article Publication: 0 (USD)
If this paper is accepted for publication, you will be asked to pay an Article Publication Fee to cover publications costs.
Editorial Board
Prof. Noureddine Hidouri
Preparatory Institutefor Engineering Studies of Gafsa, Tunisia
Prof. SAM PACK
Department of Anthropology, Kenyon College,Gambier, OH, USA
Prof. KAARINA MÄÄTTÄ
Educational Psychology, Faculty of Education, University of Lapland,
Finland
Prof. José G. Vargas-Hernández
University Center of Economic and Managerial Sciences, University of
Guadalajara,Mexico
Prof. P Malyadri
Government Degree Colledge, Osmania University, India
Prof. SUKUMAR SENTHILKUMAR
School of Mathematical Sciences, Universiti Sains Malaysia, Malaysia
Prof. OLADOKUN SULAIMAN OLANREWAJU
Prof. ASYRAF HJ AB RAHMAN
Department of Nationhood and Civilization Studies, Universiti Malaysia
Terengganu, Kuala Terengganu, Malaysia
Prof. Orwa Jaber Housheya
Department of Chemistry, Arab American University, Palestine
Prof. Hsu-Yang Kung
Department of Management Information Systems, National Pingtung
University of Science and Technology, Taiwan
Prof. Hassan Fartousi
International Islamic University Malaysia, Malaysia
Prof. Xiaojun Yang
Department of Mathematics and Mechanics, China University of Mining
and Technology, P.R. China
Prof. Guang-Sheng Chen
Dept. of Computer Engineering, Guangxi Modern Vocational Technology
College, P.R. China
Prof. A.S.N.CHAKRAVARTHY
Dept. of Electronics & Computer Engineering, KL UNIVERSITY, India
Prof. Bahman Gorjian
Prof. M. PRIYADHARSHINI
Department of IST, Anna University, India
Prof. K.V.L.N.ACHARYULU
Dept. of Mathematics,Bapatla Engineering College, Andhra pradesh,
INDIA
Prof. Saeed Balochian
Department of Electrical Engineering, Gonabad Branch, Islamic Azad
University, Gonabad, Iran
Prof. Sarbapriya Ray
Shyampur Siddheswari Mahavidyalaya,University of Calcutta, India
Prof. Iraj Montashery
English Literature in Azad University of Ardebil, Iran
Prof. J. Namratha Manohar
EEE Department, Ellenki College Of Engineering and Technology,
Patelguda, Medak, India
Prof. Bing-Zhao Li
School of Mathematics, Beijing Institute of Technology, China
Prof. EL Mahdi Ahmed Haroun
Prof. Mohammad Reza Iravani
Department of Social work, Azad University of Khomeinishahr, Islamic
Azad University, Khomeinishahr branch, Khomeinishahr, Esfahan, Iran
Prof. M. Sudha
School of Information Technology & Engineering, VIT University, TN
India
Research Scientist, Yudong Zhang
Columbia University, New York, USA
Researcher Sonal G. Chonde
Department of Environmental Science, Shivaji University, Kolhapur, India
Dr. Mohammad Saad Alam
Battery Electric Vehicle Program, Magna E-Car Systems of North
America, Detroit, MI, USA
Dr. Alishir Moradikordalivand
Electrical Engineering, University Technology Malaysia, Malaysia
Dr. Chang-Shuai Li
College of management, University of shanghai for science and
technology, China
Dr. Chi-hua Chen
Dr. Xin Zhang
College of Communication and Transport, Shanghai Maritime University,
China
Dr. Mohammad Reza Noruzi
Tarbiat Modarres University, Tehran, Iran
Dr. Kunyuan Qiao
Department of Finance and Department of Applied Economics, Guanghua
School of Management, Peking University, China
Dr. Zeyuan Lu
Institute of Intelligent Machines, Chinese Academy Sciences, Hefei, China
Dr. Farhangi Sajad
Department of Engineering, Islamic Azad University, Ahar Branch, Ahar,
Iran
Dr. Taher Bahrani
Faculty of languages and linguistics, University of Malaya, Malaysia
Dr. Tarig Osman Khider
College of Applied and Industrial Sciences, Department of Pulp Paper,
University of Bahri, Khartoum-Sudan
Dr. P.T. Vijayshree
Dr. S. Amir Ghoreishi
Department of Electrical Engineering, Science and Research Branch,
Islamic Azad University, Iran
Technical Coordinator
M.S. Shadi Khojasteh
Faculty of Modern Languages and Communication, University Putra
Malaysia, Malaysia
M.E. Sneha Raichel Mathew
College of Engineering Guindy, Anna University, Chennai
M.E. Shuihua Wang
Department of Electrical Engineering, City College of New York, New
York, USA
B.Tech Arun Prakash. J
SNS College of Technology, Coimbatore, India
M. E. Xinhao Ji
Zhejiang Vocational College of Commerce, China
M.S. B. Sumana
M.S. Muhammad Naufal Bin Mansor
School of Mechatronic, University Malaysia Perlis , Malaysia
B.S. Hoda Rezaeian
Faculty of Educational Studies, University Putra Malaysia
Interns
Name
Country
Period
Contact Us
z Advertisement
z Chairman
z English Editors
z Editor-in-Chief
z Technician
z Ethical Board
z Book Publishing
z Fund Supporting
z ACSA
z ADMM
z AITS
z AASS
z AMEA
z AAEF
z AEES
z ACMA
z AITM
z ALSA
z APAC
z JALIS
z JES
z JCLC
z AEL
Call for Books
Call for Monographs
World Science Publisher (WSP), a leading independent academic publisher of books, journals, and conferences for students and professionals, establishes a program for assisting scholars and students to publish their academic manuscripts. The program covers the print-on-demand publication of revised dissertations, monographs, textbooks, reference works, brochures, and tracts. For complete descriptions of WSP, please visit the sitewww.worldsciencepublisher.org
Author Rights
Unique ISBN
Conditional Free-of-Charge Publishing Earn Royalties
Free eBook Copies Retain 100% of the rights
Personal Assistance Indexed by academic databases.
Print on Demand
http://en.wikipedia.org/wiki/Print_on_demand
PoD is a printing technology and business process in which new copies of a book (or other document) are not printed until an order has been received While build to order has been an established business model in many other industries, "print on demand" developed only after digital printing began, because it was not economical to print single copies using traditional printing technology such as letterpress and offset printing.
Steps to publish your book
Discuss your proposal with the coordinator of book-publishing sector;
Complete your order, pay the fees (150 USD) or mention the two papers published and paid Sign the agreement, and upload all the necessary information to WSP (title, cover, back cover,
summary, and keywords)
Make final changes to your book, sign off the proof, and choose the retail price.
Discount and Waivers Program
Authors who published and paid one paper in any journal of WSP, can enjoy50%discount of the book publication fee.
Authors who published and paid two papers in any journal of WSP, can publish their books free-or-charge.
Proposal
A brief CV including other publications, related activities, etc. A Cover Letter, describing the book
Table of Contents, including an estimate of the length or number of pages for each chapter. A preface introducing the book to potential readers.
The estimated date of manuscript completion
Contact
Please send proposals to
Sharon, Director of Publishing Sector World Science Publisher LLC
Call for Interns
Company:World Science Publisher LLC
Address: World Science Publisher, New York, NY, USA
Deadline:No Deadline
Business Sector: Advertising
Position:Part-Time, Unpaid
US Citizenship Required: No
Number of Openings: 50
Roles and Responsibilities:
World Science Publisher Advertising Sector is seeking Interns who have a laptop and internet access, to join our team of qualified, diverse individuals. Candidate will assist to publish call-for-papers/editors of WSP on the academic forums of his/her own countries.
Preferred Qualifications:
Candidates with these desired skills will be given preferential consideration: Knowledge of MS office Suite and the internet. Fluent written English is a requirement.
The qualified candidates will be engaged to publish our ads at academic forums and social network for at least two weeks. The candidates need to report the progress to the supervisor every week via emails containing the URLs on which the ads are published.
Rewards:
After the internship period, the candidates will receive an e-certificate of internship with seal and their names will be listed on “Interns” page of the company’s website.
Samples
Samples of E-certificate
[WSP] CALL FOR PAPERS
Dear authors
We invited you to send your paper to World Science Publisher. We have 12 journals covering computer science, electric engineering, intelligent transportation, information science, social science, chemistry, multimedia, expert systems, life science, economics and finance.
Free of Charge
Advances in Intelligent Transportation Systems (ISSN 2167-6399, Free)
Advances in Mechanical Engineering and its Applications (ISSN 2167-6380, Free) Advances in Life Science and its Applications (Free)
Advances in Pure and Applied Chemistry (ISSN 2167-0854, Free) Advances in English Linguistics (Free)
Advances in English Linguistics (Free)
Journal of Comparative Literature and Culture (Free) Journal of Expert Systems (Free)
Journal of Applied Library and Information Science (Free)
75 USD Publication Fee
Advances in Asian Social Science (ISSN 2167-6429, Free)
Advances in Computer Science and its Applications (ISSN 2166-2924) Advances in Information Technology and Management (ISSN 2167-6372) Advances in Computational Mathematics and its Applications (ISSN 2167-6356) Advances in Digital Multimedia (ISSN 2166-2916)
Advances in Applied Economics and Finance (ISSN 2167-6348) Advances in Electrical Engineering Systems (ISSN 2167-633X) Advances in Life Science and its Application (ISSN 2167-0854)
These journals are indexed or will be indexed byOCLC, DOAJ, Ulrich, EBSCO, WorldCat, Google Scholar, GetCited, arXiv, Open Archive, Open J-Gate, New Jour, Index Copernicus, PKP Open Archives Harvester.
Most journals are free of charge. For some journals, there is a nominal 75 US dollars for publication of an accepted manuscript. If you do not have funds to pay such fees, our editors can arrange a registered funder to help you pay the publication fee within one day. We do not want fees to prevent the publication of worthy work.
After the author published and paid at least 2 papers in our journals, our editors will recommend one of his papers toEI-indexed Journal or SCI-indexed Journal.
http://www.worldsciencepublisher.org/journals/
Editor in Chief
Advances in Pure and Applied Chemistry, 2012,
2
(
1
): 81 - 105
Table of Content
Vol.
2
, No.
1
, 2012
Articles
Table of Contents
Articles
Uric Acid Inhibition Activity of
Annona muricata
L Leave
Extract in Hyperuricemia induced Wistar Rat
Sri-Wahjuni, Putra-Manuaba, I. B., Rahayu-Artini, N. P., and Wahyu-Dwijani, S.
Chemistry Department, Faculty of Math and Science, Udayana University,
Bali-Indonesia
81 - 90
Kinetics of Organic Dyes Degradation in Water Using
Vacuum Ultra Violet Radiation
Khaled M. Elsousy
Alaqsa University, Gaza, Gaza strip, Palestinian Territories
91 - 97
Occurrence and prevalence of four viruses infecting tomatoes
in Northern districts of West Bank, Palestinian Territories
Hazem Sawalha
Department of Biology and Biotechnology, Faculty of Arts and Sciences,
Arab American University of Jenin, Palestine
98-101
Study of Liquefied Petroleum Gas Heating Value
A Thermodynamics Approach
Niaz Bahar Chowdhury,
1Dr. Md. Iqbal Hossain
Advances in Pure and Applied Chemistry (APAC) 86 Vol. 2, No. 1, 2012, ISSN 2167-0854
Copyright © World Science Publisher, United States
www.worldsciencepublisher.org
Uric Acid Inhibition Activity of
Annona muricata
L Leave
Extract in Hyperuricemia induced Wistar Rat
Sri-Wahjuni, Putra-Manuaba, I. B., Rahayu-Artini, N. P., and Wahyu-Dwijani, S.
Chemistry Department, Faculty of Math and Science, Udayana University,
Bali-Indonesia
Abstract-This research aims to find a cure of gout, base on the utilization of Annona muricata. The research was
started with descriptive study to explore active components of Annona muricata leaf and followed by an experimental
study to investigate uric acid inhibition activity of the leaf extract in hyperuricemia induced wistar rat. We observed three dominant components, i.e. 2,3-dihidrobenzofuran; 3-ethoxy-1,4,4a,5,6,7,8,8a-octahydroisoquinoline; 2-cyclohexen-1-one, 4-hydroxy-3,5,6-trimethyl-4-(3-oxo-1-butenyl) which were probably active to inhibit uric acid formation in hyperuricemic induced wistar rat. In this study, n-buthnol was applied for partitioning the relatively pure compound. The n-buthanol extract obtained was then applied to cure hyperuricemic rat induced by a mixture of chicken livers and Gnetum gnemon a high purine diet. It was obtained, that the highest extract dose of 400 mg/kgBW was able to inhibit the formation of uric acid in hyperuricemic rat. It can be concluded that Annona muricata leaf extracted with n-buthanol in a dose of 400 mg/kg BW has an ability to inhibit further formation uric acid in hyperuricemic rat. Therefore, this natural plant is potent to develop for hyperuricemic medicine.
Keywords: gout, Annona muricata, hyperuricemic rat, active components
Introduction
Uric acid is a metabolic product of exogenous (brought in with food) or endogenous purine bases. This acid in most physiologic fluids is an end product of purine degradation. The serum urate level in a given patient is determined by the amount of purines synthesized and ingested, the amount of urate produced from purines, and the amount of uric acid excreted by the kidney (and, to a lesser degree, from the gastrointestinal tract).1,2Gout is an inflammatory arthritis caused by the deposition of monosodium urate crystals in tissues.1 This condition typically occurs after years of sustained hyperuricemia. It is estimated to affect 5.1 million people in the United States according to the most recent National Health and Nutrition Examination Survey (NHANES III).2 Gout affects approximately 2% of men older than
30 years and2% of women older than 50 years, and is the
most common form ofinflammatory joint disease in men
older than 40 years. Serum uric levels are, on average,
0.5 to 1.0 mg/dL higher in men than women, making
male sex a risk factor for hyperurisemia and gout. Lower serum uric levels in women are associated with the
presence of estrogen, which is thought to act as an
antihyperuricemic.3 In Indonesia, based on Health Survey
in the year of 2005, there were around 10-20% men and postmonopause women have a higher levels of uric acids than normal person.4 It was proven that, celery seed is
often used in treating this condition, as it possesses many anti-inflammatory compounds. Other helpful herbs include turmeric, boswellia, cayenne, colchicum and hyssop were also potent to treat hyperurisemia.
Clearly, uric acid is produced by purine nucleoside metabolism through hipoxanthin, xanthin, and guanin basic purine. Distortion of this metabolism leads to elevate level of uric acid and known as hyperuricemia.5
Annona muricata L is a traditional plant in Bali known as sirsak, empirically in Balinese traditional medicine was proven as a cure of hyperuricemia. This study was carried in order to investigate the component active of the plant that have an ability to inhibit further uric acid formation in the hyperuricemia wistar rat.
Methods
This study employs two research methods, i.e.
descriptive explorative to determine the active
components of Annona muricata L leaf extracted with
n-buthanol and followed by experimental study to observed their hyperuricemia activity.
Leaf extract was obtained through maseration process using methanol and followed by partition using n-buthanol. Crude extract obtained was the identified by applying GS-MS instrument.
Sri-Wahjuni et al, APAC, Vol. 2, No. 1, pp. 86-90, 2012 87
study and devided into 5 groups. First group is a negative control group in which rat fed with a mixture of 4 g/kg
BW of Gnetum gnemon and 50 mL/kg BW of
chicken-liver juice in ad libitum manner. The second group is simillar to first group instead of delivering an anti-hyperuricemia medicine, allopurinol in a dose of 10 mg/kg BW oraly. The third group is simillar to the first group instead of delivering Annona muricata L extract in a dose of 100 mg/kg BW oraly. The fourth and five groups have similar treatment to the first group, instead of have extract of Annona muricata L in dose of 200 mg/kgBW and 400 mg/kgBW, respectively.
Animal ethical clearance was obtained from a local authority body at Veterinary Faculty Udayana University, Bali-Indonesia. Around 1 mL of blood was taken from rat heart aorta which was anesthesia before proceeding. The blood was then centrifuged for 15 minutes at the rate
of 3.000-3.500 rpm. Uric acid reagent, FS TBHBA
(2,4,6-tribromo-3-hydroxybenzoic acid) was then added to the serum obtained. The mixture was then incubated for 10 minutes at a temperature of 370C. Then, optical density of the mixture was determined using sphectrophotometer at 546 nm of wave number.
ANOVA was performed to determine the different effect amongst treatment with p<0.05 was consider significant.
Results
Descriptive study
Around 1,200 g of Annona muricata L. leaf powder
was macerated with methanol for overnight. From this, a number of 158 g crude extract was obtained. This crude extract was then tested for its antioxidant activity using DPPH test. The tes results was presented in Table 1.
Table 1. Antiozidant Activity Test of Annona muricata L Crude Extract
Sampl e
Time
(minutes) Test
Absorbance A 517 nm % inhibition 497 nm 517 nm 537 nm Crude extract
5 DPPH 0.714 0.785 0.698 0.0790 77.22
% Sampl
e
0.635 0.593 0.515 0.0180
60 DPPH 0.651 0.704 0.613 0.0720 85.42
% Sampl
e
0.527 0.508 0.468 0.0105
The crude extract was the purified by applying partition using petroleum ether, chlroform, n-buthanol, and water. Amongst them, in this research, it was obtained that partiton with n-buhanol produce the highest
anti-oxidant ativity indicates by their DPPH test. Therefore, the n-buthanol extract was then identified Phythochemically using a number of reagent as indicated in Table 2.
Table 2. Phythochemical Test of n-Buthanol Extract
N o.
Compounds Reagent Coloue Changes Resul
ts 1
.
Alkaloid Meyer
Wagner
Yellow - orange (without white precipitate)
Yellow - chocolate (without chocolate precipitate) - - 2 .
Flavonoid Wilstatter
NaOH 10 % H2SO4 concentrated
Bate Smith-Metacalf
Yellow - crimson Yellow - chocolate Yellow - crimson Yellow - red
+ + + + 3 .
Triterpenoid Lieberman-Burchard
H2SO4 10 %
Yellow - chocolate Yellow - chocolate
+ + 4
.
Saponin Hot water + HCl No foam formation -
5 .
Phenolate (tannin)
Hot water + FeCl3 Yellow – greenish black +
6 .
Steroid Lieberman-Burchard
H2SO4 10 %
Yellow - chocolate Yellow - chocolate
-
Remarks:
Sri-Wahjuni et al, APAC, Vol. 2, No. 1, pp. 86-90, 2012 88
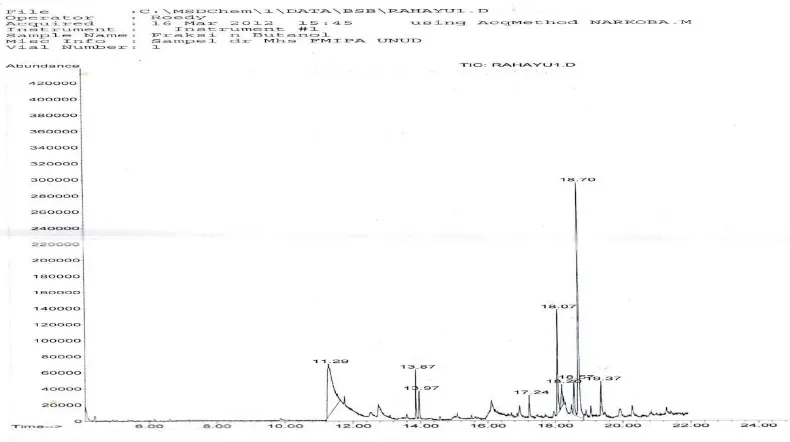
The most active extract was then identified by applying GC-MS, the chromatogram obtained was presented in Figure 1.
Figure 1. GC-MS Chromatogram of The Most Active Extract of Leaf Annona muricata L
Based on library data base of the GC-MS instrument, there were three compound detected as indicated in Table 3.
Table 3. Compound Identified Based on GC-MS Chromatogram
Peaks
Retentiom Time
(tR)
%
Area Compounds identified
Peak 1 11.29minutes 31.4
8
benzofuran,2,3-dihidro
Peak 2 18.07 minutes 11.7
1
3-ethoxy-1,4,4a,5,6,7,8,8a-octahydroisoquinoline
Peak 3 18.70 minutes 30.8
9
2-cyclohexen-1-one, 4-hydroxy-3,5,6-trimethyl-4-(3-oxo-1-butenyl
Experimental study
In this study, increase uric acid in wistar rat was achieved by intake of high purine diet. Rat were fed with a mixture of 4 g/kg BW of Gnetum gnemon with 50
mL/kg BW of chicken liver ad libitum. After
hyperuricemia condition was achieved, the rat then was fed with varies dose of leaf Annona muricata L extract, i.e 100 me/kgBW, 200 mg/kgBW, and 400 mg/kgBW. Other treatments are positive control using allopurinol and negative control. The uric acid concentration of hyperurisemia rat were presented in Table 4.
Table 4. Uric Acid Levels of Hyperurisemia Wistar Rat
Tratrment group Uric acid concentration (mg/dL)
Day-6 Day-9 Day-14 Day-18
Hyperurisemia Control (H)1
Control (H)2
Control (H)3
Control (H)4
Average 3.48 5.54 4.48 4.25 4.44 4.05 5.65 4.62 4.65 4.74 4.35 5.87 5.97 5.03 5.31 4.98 6.07 6.23 5.67 5.74 Allopurinol dose 10 mg/kg BW Control positive 1 Control positive 2 Control positive 3 Control positive 4
Sri-Wahjuni et al, APAC, Vol. 2, No. 1, pp. 86-90, 2012 89
Average 3.91 6.42 4.52 3.93
Extract dose of 100 mg/kg BW Treatment I1
Treatment I2
Treatment I3
Treatment I4
Average 4.81 4.19 4.50 4.92 4.61 7.18 6.11 5.73 6.04 6.27 3.35 3.00 4.19 3.77 3.58 3.08 2.65 3.96 3.50 3.30
Extract dose of 200 mg/kg BW Treatment II1
Treatment II2
Treatment II3
Treatment II4
Average 3.00 3.27 4.11 3.73 3.53 3,38 3,57 4,35 4,11 3,85 2,31 3,00 2,61 3,04 2,74 1,61 1,69 1,69 2,73 1,93
Extract dose of 400 mg/kg BW Treatment III1
Treatment III2
Treatment III3
Treatment III4
Average 3.69 3.81 3.77 4.04 3.83 4.69 5.15 4.19 7.69 5.43 3.00 3.77 2.73 5.19 3.67 3.00 3.00 2.31 3.96 3.09
Anova test indicates there was a significant different between treatment and control groups, indicate by p < 0.05.
Discusions
Discriptive study
As can be seen in Table 2, n-buthanol fraction based on phytochemical test was positively containing flavonoid, triterpenoid, and phenolate indicates by the colour changes for all compounds type tested. This is because of n-buthanol is a polar solvent with 3.9 of polarity index.6 Generally, the present of glucose bind to flavonoid group results in the compound easier to solve on water and polar solvent.7 Tannin group is a phenolate compound, that has a tendency to solve in water and polar solvent. On the other hand, triterpenoid group of compound is a pentacyclic compound tend to solve in nonpolar solvent. GC-MS analysis confirms three important compounds observed as indicates on Table 3. All of these compounds are benzofuran,2,dihidro, 3-ethoxy-1,4,4a,5,6,7,8,8a-octahydroisoquinoline, and 2-cyclohexen-1-one, 4-hydroxy-3,5,6-trimethyl-4-(3-oxo-1-butenyl. The present of triterpenoid is probably due to the present of hydroxide group on the structure. The extract that contain all these thee compound was then tested for their anti-hyperurisemia activity.
Experimental study
A number of 20 wistar rat were adapted in a laboratory condition. Then, all of these rats were fed with high purine diet, i.e. a mixture of 4 g/kg BW of Gnetum gnemon and 50 mL/kg BW of chicken liver and mix with 100 g pelete a standard diet for rat. On day-6 and day-9, about 1 mL of blood were taken from the heart aorta of the rat to determine the increase of uric acid. Before treatment uric acid serum level of the rats were
determined. In this study, uric acid levels of normal rats were in the range of 1.7 – 3.0. After induction with high purine diet the uric acid levels of the rat was increase roughly, in which all rats have uric acid levels above 3 mg/dL, on average of 4.74
r
0.665 mg/dL. It can be said, that all experimental rats are in hyperuricemia condition.Rats induced hyperuricemia were achieved during 9 days after feeding with high purine diet. Then, on day-10 all experimental rats receive treatment for decreasing uric acid levels. Five groups of experiment were carried out as mentioned on the method. The treatment was stopped on the day-18 and uric acid leves were detrmined for all experimental rats.
In this study we obtain that for positive control group treated with allopurinol, there is a 51.93% decrease of uric acid levels, their uric acid levels become 3.93
r
0.995 mg/dL. For the varies extract treatment, i.e. dose of 100, 200, and 400 mg/kg BW, the uric acid decrease levels obtained are 63.98%, 86.29%, and 61.50%, respectively. Therefore, the optimum dose of 200 mg/kg BW produces the highest decrease.Allopurinol was applied in this study as a positive control, since this medicine is a cure for hyperuricemia case. In low dose this compound has an ability to inhibit the formation of xanthine oxidase enzyme.8 Allopurinol dose of 10 mg/kg BW applied is on the basis of Zhao et al, (2005), they obtain this dose was effective to decrease uric acid levels until 125.59
r
1.49 on their mice experimental study.9Conclusion
This study investigates the application of natural plant,
Sri-Wahjuni et al, APAC, Vol. 2, No. 1, pp. 86-90, 2012 90
compounds, i.e. benzofuran,2,3-dihidro,
3-ethoxy-1,4,4a,5,6,7,8,8a-octahydroisoquinoline, and
2-cyclohexen-1-one, 4-hydroxy-3,5,6-trimethyl-4-(3-oxo-1-butenyl) were identified. However, this still need to be further investigated. Our study also gained that the extract leaf of this plant is potent to develop as a cure for hyperuricemia, since we obtain that the dose of 200 mg/kg BW of rat is effective to decrease uric acid levels. This also need to be investigaed further, whether that will give simmilar effect on human.
Akcnowledgment
The authors would like to thank Staff of UPT Lab. Analitik Udayana University for access and aid of their fasilities for managing the research. Thanks also to Mr. Priono from Kristallindo for providing reagents for uric analysis, Mr. Rudy at Forensic Laboratory Police Department for help in assessing the GC-MS data. And special thank to Vetinery Board for providing rat for this experiment.
References
1. Stankov, M., Predrag, D., and Stankov, D. 2003. Determination of uric acid in human serum by an enzymatic method using N-methyl-N -(4-aminophenyl)-3-methoxyaniline reagent. J.Serb.Chem.Soc. 68(8–9)691– 698.
2. Mandell, B. F. 2008. Clinical manifestations of
hyperuricemia and gout. Cleveland Clinic Journal of
Medicine Volume 75, Supplements 5 July 2008.
3. Luk AJ, Simkin PA. Epidemiology of
hyperuricemia and gout. Am J Manag Care. 2005;11 (15
Suppl):S435-S442; quiz S465-S468 [Review].
4. Saraswati-Sylvia. 2009. Diet Sehat Untuk
penyakit Asam Urat, Diabetes, Hipertensi, dan Stroke, Yogyakarta, A-Plus Books.
5. Harn-Shen, C. 2011. Clinical implications of the
metabolic syndrome and hyperuricemia. Journal of the Chinese Medical Association 74 (2011) 527-528.
6. Snyder, C. R., et al, 1997. Practical HPLC
Method Development, Second Edition. New York:John Wiley and Sons, Lnc, Hal 722-723.
7. Markham, K. R., 1988, Cara Mengidentifikasi
Flavonoid, ITB, Bandung, Hal 15-17.
8. Tjay, T.H., dan Raharja., 2002, Obat-Obat
penting, Khasiat, Penggunaan dan Efek-Efek sampingnya, Edisi V, Cetakan ke-2, Penerbit PT. Eleks Media Komputindo Kelompok Gramedia, Jakarta.
9. Zhao, X., Zhu, X. dan Pan, Y., 2005, Effects Of
Cassia Oil On Serum and Hepatic Uric Acid Levels In
Oksonate-Induced Mice and Xantine Dehiydrogenase and
Xantin Oksidase Aktivities In Mouse Liver, Journal Of
Ethnopharmacology, (http:/
Advances in Pure and Applied Chemistry (APAC) 91 Vol. 2, No. 1, 2012, ISSN 2167-0854
Copyright © World Science Publisher, United States www.worldsciencepublisher.org
Kinetics of Organic Dyes Degradation in Water Using
Vacuum Ultra Violet Radiation
Khaled M.
Elsousy
Alaqsa University, Gaza, Gaza strip, Palestinian Territories
Corresponding: Dr. Khaled M. Elsousy, [email protected]
Abstaract: In this study vacuum ultraviolet (VUV) radiation (185 nm wave length) was used in the presence of
atmospheric oxygen as an advanced oxidation technique. Six organic dyes were examined as model pollutants (methyl violet, methyl blue, brilliant green, malachite-green, Remazole blue B and picric acid). Picric acid as the most persistent one was selected for more detailed kinetic investigations. The influence of each of the related main parameters was studied, Radiation time, salinity, pH, temperature and radiation intensity. Kinetics of the oxidation reaction was studied. COD was also followed up. It follows from the results that vacuum-UV radiation of 185 nm in the presence of atmospheric oxygen is an efficient method for the oxidation process. Four of sex examined dyes were degraded in different rates according to persistency of each pollutant dye. The reaction rates were in the order of:
(methyl violet > methyl blue >brilliant green > remazole > malachite green> picric acid). In the case of picric acid; the reaction was promoted by rising the temperature, raising or lowering pH above and below pH 7.0, increasing radiation dosage. The rate was inhibited by increasing salinity and buffering. The present technique was found promising in the elimination of the persistent organic pollutants out of the treated water.
Keywords: Advanced oxidation process - Vacuum ultraviolet - Wastewater treatment, Radiochemistry, Organic pollutants.
1. Introduction
During recent years the Advanced Oxidation
Processes (AOPs) have become an important group of
techniques to the treatment of hazardous water contaminants, with an increasing number of feasible applications. (Ahmed et al. 2011) Normally, the main reason for the use of AOPs is the resistivity and/or the toxicity of pollutants that makes unfeasible the biological treatment. Although AOPs use different reacting systems, all are characterized by the same chemical feature: production of OH free radicals. These radicals are extraordinarily reactive species. (Ahmed et al. 2010) The oxidation reactions involving hydroxyl radical and organic substrates in aqueous solution may be classified with respect to their character to: (Bossmann et. al. 1998) a. Abstraction of hydrogen: .OH +RH R. + H2O
b) Addition reactions: .OH + PhX HOPhX.
c) Electron transfer: .OH +RH [+R-H] + HO-
AOPs are characterized by a free radical mechanism initiated by the interactions of photons of a proper energy level with the molecules of chemical species present in
the solution such as H2O2 or with a catalyst surface such
as TiO2. (US.EPA 1998; Bakardjieva et al 2005;
Bakardjieva et al 2004).
Peroxone is another technique including the oxidation of organic pollutants using ozone with H2O2 was also
reported.(Ben Abdelmelek et al. 2011; Ben Abdelmelek et al. 2010; Chu et al. 2022) Many studies were conducted concerning the elimination of the hardly-oxidized water pollutants by different catalytic methods using hydrogen peroxide as the main source of OH free radicals. Persisting pollutants such as phenol derivatives, (Ben Abdelmelek et al. 2011; Ben Abdelmelek et al. 2010) aromatic pesticides, (Chu et al. 2011; Chu et al. 2010) fuel additives, dyes, and some other pharmaceutical drugs has been treated. (Radjenovic et al. 2009; Gaya et al. 2008)
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 92
reaction rate of compounds of interest with hydroxyl radicals. Aqueous electronsare powerful reducing agents and act as nucleophiles. They react with substrates with one-electron transfer process. One of the most important reactions of aqueous electrons is with halogenated organic compounds resulting elimination of halide anions.
H2O (L) + hȞ (< 190 nm) ĺ H. + .OH ļ H+ + e- + .OH
Carbon centered radicals, formed by their reaction with hydroxyl radicals, may result in the formation of higher molecular weight compounds hindering mineralization and TOC (Total Organic Carbon) reduction. Thisproblem may be surmounted by bubbling oxygen gas. (Oppenländer 2003b)
It was reported that (VUV) radiation produced by an immersed Xe-excimer light source (172 nm) was investigated for the oxidative degradation of organic pollutants in aqueous systems. ( Hashem et al. 1997; Oppenländer and Gliese 1997; Chong et al. 2010) It was shown that the rates of degradation of the substrate decrease in the order of the applied processes, VUV/O3 >
O3 > VUV.
Influence of the oxygen concentration on the rate and reaction pathway of the degradation of organic compounds in aqueous solution by VUV-irradiation (Xe-excimer: 172 nm) was reported. (Heering 2004) The reaction rate was found to be strongly influenced by the concentration of dissolved molecular oxygen in the volume of primary reactions.
In the present research; 185 nm, VUV, radiation from a low pressure mercury lamp in the presence of atmospheric oxygen will be studied as advanced oxidation technique, the process is thought to be simple, cheap and clean where no chemicals are to be used. Six organic dyes will be tested as model pollutants, the method efficiency and the oxidation kinetic parameters will be investigated on the most persisting one of them.
2. Experimental
2.1Materials.
Picric acid; (2,4,6-trinitrophenol) was locally prepared by the nitration of phenol, (Brewster et al. 1977) Remazole blue B commercial grade was purchased from (Brilliant Blue Daystar-LP, India), methyl blue, methyl violet and malachite green, Solid sodium hydroxide and Phosphoric acid 85% ~15M were analytical grade of (Merck-Germany). Distilled water of (TDS 60 ppm) was used in all the experimental activities.
2.2 Instruments
The main kinetic investigation system was a semi-continuous system as shown in (fig. 1, a low pressure mercury, VUV, 185 nm lamp (S415 ROL-Rcan- USA) is contained inside of a sealed stainless steel cell (S300- S2R,OZAP-USA) which is protecting the electrical components of the system. The system also includes air pumpforthe continuous aeration of the reaction media, a thermo-statated water bath, and a circulation system for the treated water.
Spectrophotometer (Spectrum-900-USA) has been used for the spectroscopic measurements. A Hanna pH meter was used to adjust the pH of different solutions.
2.3 Methodology
The reaction kinetic study was conducted to compare similar samples of different pollutants; the samples have been treated identically under the same conditions of pollutant concentration (mg/l), circulating rate (0.65 dm3/ min), aeration rate (6.0 watt pump), working temperature (25.0 Co) and reaction volume (2500 ml).
Substrates depletion rates were followed spectrophotometrically. For each pollutant separately substrate concentration through the reaction term was determined. Each dye was followed at the proper Ȝmax
(picric acid at 355 nm, remazole at 570 nm, methyl blue at 665 nm, methyl violet at 590 nm and malachite green at 625 nm). Absorption bands Ȝmax were obtained
practically, Calibration curves of concentration against absorbance were obtained for the six dyes.
Kinetic investigation was performed such that: in each experimental run, reaction vessels were prepared with the proper volume (500 to 2500ml) of the reaction solution. The (observed reaction half-life) t1/2 was obtained for
each run. The half-life was approved because of the ignorance of the exact reaction order since many reactions are expected to proceed on the same time. Samples of picric acid were read spectrophotometrically with 0.02 mg/L detection limit at 355 nm Ȝmax which was
determined experimentally. Reaction parameters such as salinity (ionic strength), temperature, light intensity and reaction pH) were investigated and the resultant half-lives were plotted against readings (or values) of each parameter to clarify the relation graphically. Phosphoric acid (0.1M; H3PO4), and (0.1M; NaOH) were used to
adjust the reaction pH at the values of (5, 6, 7, 8 and 9).
3. Results and Discussion
3.1 Influence of the organic pollutant resistivity.
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 93
Table 1: Organic pollutants degradation Half-lives (t1/2), (2500 ml total volume, T= 25±0.5 0C, circulating rate, 6.5
dm3/min).
Pollutant die Methyl violet Methyl blue Brilliant green Malachite green Remazole blue B Picric acid
Ȝmax , nm 590 665 523 625 570 355
t1/2 , hrs 1.5 1.83 1.7 3.11 Colloid 4.73
3.2 Effect of salinity
The influence of the treated water salinity on the observed reaction half-life was studied. Salinity of course is mainly proportional to ionic strength. The reaction was conducted under fixed conditions of temperature, circulating power and reaction total volume. In the studied range of [NaCl] the reaction half-life was increasing by increasing [NaCl], direct relation was observed up to 6000 mg/l sodium chloride concentration as shown in (fig. 2), the optimum oxidation rate can be achieved in desalinized treated water. In practical water treatment processes; water salinity is expected to have a negative influence on the rate of degradation. The influence of aqueous NaCl is not clear, wither it is up to free radical scavenging or simply the loss of UV photons by scattering with ions.
3.3 Effect of the volume of the treated water
Degradation rate was lowered by increasing the treated water volume. Reaction half-life was followed. It was increasing directly by increasing the volume of the treated water in the studied range, the relation is plotted in (fig. 3). The conversion rate is decreasing by decreasing the radiation intensity per unit volume of the treated water in the studied range (3.0 to 30.0 Watt per liter).
3.4 Influence of temperature
Increasing temperature has a promotional influence on the oxidation reaction in the range of (25-60 0C). It is clear in (Fig. 4) that an inverse relationship was obtained between ln 1/(t1/2) and 1/T, where a straight-line was
obtained. The result means that the process has an overall Arrhenius type behavior. It was expected according to post studies (Oppenländer et al 2005) that: the radiation intensity is affected by changing the environment temperature. In that case it may affect the UV light intensity and so the reaction kinetics, Arrhenius type relation has been observed in this case, which means a very weak influence of temperature on the radiation intensity under the present reaction conditions.
3.5 Effect of pH
Effect of pH on the oxidation reaction was examined in the pH range (4 to 10). (Fig. 5) shows the values of T1/2 corresponding to each pH value.The oxidation rate is
increasing by increasing each of OH- and H+ concentrations. It means that there are two accelerating roles for each of H+ and OH- according to various
mechanisms. Comparison of t1/2 in buffered and
non-buffered pH 7 reactions which were respectively (14.73 and 4.71 hours) implies that the presence of ions in the treated water again has a retarding influence on the degradation rate.
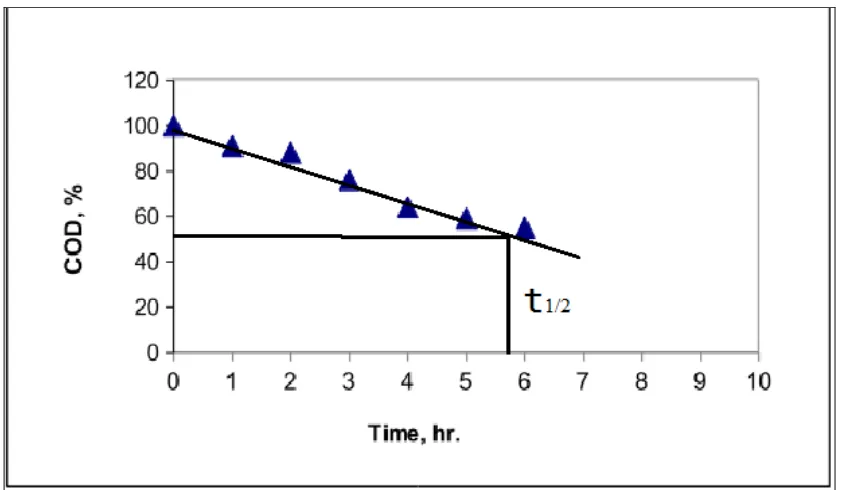
The last results show that the treatment of basic polluted water is easier and faster than the treatment of neutral and acidic polluted water under the same conditions. It is not recommended of course to increase the basicity of the treated water to get a faster process but at least it should be known that if the treated water was basic by chance the process is expected to be faster. 3.6 COD follow up.
Degradation was followed by measuring chemical oxygen demand COD at different time intervals. One model run was performed. Picric acid oxidation derivatives are thought to go further oxidation by time. After the breakage of aromaticity, ketones, aldehydes and carboxylic acids are thought to precede oxidation (Qian-Rong L. 2006). The final expected products are mostly CO2 and H2O. Degradation conditions were (250mg
picric acid in 2500 ml, distilled water, 25oC, 6.5 dm3/min circulating rate). (Fig. 6) insures the degradation process, t1/2 of the COD depletion was 6.5 where the oxidation
process is continuous for the derivatives as well as the starting compound picric acid.
4. Conclusion
The primary goal of this project was to investigate the VUV degradative oxidation of persistent industrial organic pollutants. The method was found efficient for four per six examined pollutants. As expected, the present method is more efficient at higher temperatures and higher intensity of VUV radiation. The present technique can be preferable over other techniques in two ways: it is simple and clean method where no chemicals are needed; only atmospheric oxygen. It can be suggested for practical applications after case study is conducted concerning the real pollutant/s specifically beside the other treated water conditions.
References:
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 94
S. Ahmed, M.G. Rasul, W.N. Martens, R. Brown, M.A. Hashib, Advances in heterogeneous photocatalytic degradation of phenols and dyes in wastewaters: A Review. Water, Air and Soil pollution 215 (2011) 3-29. S. Bakardjieva, J. Subrt, V. Stengl, M. J. Dianez, M. J. Sayagues, Photoactivity of anatase-rutile TiO2nanocrystalline mixtures obtained by heat treatment
of homogeneously precipitated anatase. Applied Catalysis B: Environmental 58 (2005) 193-202.
S. Bekkouche, M. Bouhelassa, N.H. Salah, F.Z.Meghlaoui, Study of adsorption of phenol on titanium oxide (TiO2). Desalination 166 (2004) 355-362.
F. J. Benitez, J. L. Acero, F. J. Real, G. Roldan, Ozonation of pharmaceutical compounds: Rate constants and elimination in various water matrices. Chemosphere 77.1 (2009) 53-59.
F. L. Ben Abdelmelek, E. C. Wert, S. A. Snyder, Evaluation of UV/H2O2 treatment for the oxidation of
pharmaceuticals in wastewater. Water Research 44.5 (2010) 1440-1448.
S. Ben Abdelmelek, J. Greaves, K. P. Ishida, W. J. Cooper, W. Song, Removal of pharmaceutical and personal care products from reverse osmosis retentate using advanced oxidation processes. Environmental Science & Technology 45.8 (2011) 3665-3671.
S. H. Bossmann, E. Oliveros, S. Gob, S. Siegwart, E. Dahlen, L. Payawan, M. Straub, M. Worner, A. Braun, New evidence against hydroxyl radicals as reactive intermediates in the thermal and photochemically enhanced Fenton reactions. J. Phys. Chem 102 (1998) 5542-5550.
R.Q. Brewster, C.A. Vanderwarvf , W.E. Mcewen, Unitized experiments in organic chemistry, 4th edition, 1977, PP 394.
M.N. Chong, B. Jin, C.W.K. Chow, C. Saint, Recent developments in photocatalytic water treatment technology: A review. Water Research 44 (2010) 2997-3027
T.Chu, S.K. Umamaheshwar, A. Mumper, Removal of sulfadiazine, sulfamethizole, sulfamethoxazole, and sulfathiazole from aqueous solution by ozonation. Chemosphere 79.8 (2010) 814-820.
W. Chu, Y.R. Wang, H.F. Leung, Synergy of sulfate and hydroxyl radicals in UV/S2O8-/H2O2 oxidation of
iodinated X-ray contrast medium iopromide. Chemical Engineering Journal 178 (2011) 154-160.
M.C. Dodd, H.P.E. Kohler, U. Von Gunten, Oxidation of Antibacterial compounds by ozone and hydroxyl radical: Elimination of biological activity during aqueous ozonation processes. Environmental Science & Technology 43.7: 2498-2504.
Gaya UI, Abdullaha AH. 2008. Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: A review of fundamentals, progress and problems. Journal of Photochemistry and Photobiology C: Photochemistry Reviews.9 (2009) 1-12.
T.M. Hashem, M. Zirlewagen, A.M. Braun Simultaneous photochemical generation of ozone in the gas phase and
photolysis of aqueous reaction systems using one VUV light source, Water Science and Technology, 35. 4 (1997) 25–30.
W. Heering, UV bources-basics, properties and applications, International ultra violet association, 6. 4 (2004) 7-13.
D. F. Ollis, E. Pelizzetti, N. Serpone, Destruction of water contaminants. Environ. Sci. Technol. 25. 9 (1991) 1523-1529.
T. Oppenländer, Photochemical Purification of water and air, advanced oxidation processes (AOPs): Principles, reaction mechanisms, and reactor concepts. Wiley-VCH, Germany (2003b) 73-91.
T. Oppenländer., S. Gliese, Mineralization of organic micropollutants (homologous alcohols and phenols) in water by vacuum-UV-oxidation (H2O-VUV) with an
incoherent xenon-excimer lamp at 172 nm.
Chemosphere, 40. 1(2000) 15-21
T. Oppenländer, J. Burgbacher, M. Kiermeier, K. Lachner, H.Weinschrottin, Improved vacuum-UV (VUV)-initiated photomineralization of organic compounds in water with a xenon excimer flow-through photoreactor (Xe2* lamp, 172 nm) containing an axially centered ceramic oxygenator. Chemosphere 60.3 (2005) 302-9.
A. B. Prevot, M. Vincenti, A. Bianciotto, E. Pramauro, Photocatalytic and photolytic transformation in aqueous solutions. App. Catal. B: Environ; 22 (1999) 149-158. L. Qian-Rong, G. Cheng-Zhi, D. Yan, y. Hao, Z. Jun-Ying, Photo-degradation of nitrobenzene using 172 nm excimer UV lamp.Materials, 133, Issue: 1. 3 (2006) 68-74
J. Radjenovic, M. Godehardt, M. Petrovic, A. Hein, M. Farre, M. Jekel, D. Barcelo, Evidencing Generation of Persistent Ozonation Products of Antibiotics Roxithromycin and Trimethoprim. Environmental Science & Technology 43.17 (2009) 6808-6815.
Vitae
Dr. Khaled M. Elsousy, was born in Gaza/ Palestinian territories in 1962. He obtained a Bs.C. in chemistry/ physics in 1985, Msc. in physical chemistry from METU-Ankara-Turkey 1988 and Ph.D. degree from ainshams Uni. Cairo Egypt in 2001.
He worked as a chemistry instructor in Alaqsa University/Gaza. His research interest includes Physical chemistry fields such as kinetics, heterogeneous catalysis, advanced oxidation technology, adsorption methods of purification and water treatment methods in general.
He is the peer reviewer of XX. He is a member of Arabian journal of chemistry and Tenside detergents and surfactants.
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 95
Fig. 1. kinetic installation system.
0 5 10 15 20 25 30 35
0 1 2 3 4 5 6 7
[NaCl] x 1000, mg/ L
t
1/
2
,
h
rs
Fig. 2. Effect of [NaCl] on the degradation half-life of picric acid using vacuum UV. (25±0.50C, [picric acid]0=100
mg/l, reaction volume 2500ml, 6.5 dm3/min circulating rate).
0 1 2 3 4 5 6
0 1 2 3 4 5
Reaction volume, L t1/2, hrs.
Fig. 3. The relation between degradation half-life of picric acid and the amount of the treated water, (25±0.5 0C,
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 96
-2 -1.8 -1.6 -1.4 -1.2 -1
2.8 2.9 3 3.1 3.2 3.3 3.4
1/T x 10-3, K-1
ln
1
/(
t1/2
), s
-1
Fig. 4. Effect of temperature change on the VUV oxidative degradation of picric acid, (reaction volume = 2500ml,
[picric acid]0=100 mg/l, 0.65 dm3/min circulating rate).
Fig. 5.
Influence of changing pH on picric acid degradation Half-live (t1/2), (2500ml total volume, T= 25±0.5 0C, 6.5
Khaled M. Elsousy, APAC, Vol. 2, No. 1, pp. 91-97, 2012 97
Fig. 6. COD decay with process time, (2500mL total volume, T= 25±0.50C, circulating rate, 6.5 L/min). [picric
Advances in Pure and Applied Chemistry (APAC) 98 Vol. 2, No. 1, 2012, ISSN 2167-0854
Copyright © World Science Publisher, United States
www.worldsciencepublisher.org
Occurrence and prevalence of four viruses infecting tomatoes
in Northern districts of West Bank, Palestinian Territories
Hazem Sawalha
Department of Biology and Biotechnology, Faculty of Arts and Sciences, Arab American University of Jenin, Palestine
Email: [email protected]
Abstract
–
Studying tomato viruses during 2003/2004 using immunosorbent assays revealed that tomato yellow leafcurl virus (TYLCV) was the most dominant disease infecting tomato plants in the northern districts of the West Bank particularly Jenin and Tobas as its incidence ranged from 28-93%. The maximum disease incidence of 90-93% was recorded in Al-Far'a region of Tobas district. Cucumber mosaic virus (CMV) was the second most prevalent virus of tomato in the studied regions as its incidence ranged from 15-51%. Tomato infection with tomato mosaic virus (TMV) and potato virus Y (PVY) was considered insignificant as these viruses were detected in very few tomato samples.
Keywords – Tomato; Viruses; Palestine.
Introduction
Like many other developing countries, Palestine has an important agricultural sector, which has been considered the backbone of its economy. This was particularly true in the years after instituting its autonomy with the establishment of the National Palestinian Authority in 1994. Since then the agriculture became the largest contributor to the country’s gross product and provided employment to the economically active population (Palestinian Central Bureau of Statistics (PCBS) 2008).
Vegetables are the most important crops grown in the country, occupying 18734.4 hectares, with an annual production of 645157 metric tons. More than 31% of those vegetables are tomatoes, and the remainder include thirty-five other vegetable crops, of which cucumbers are the most important. Tomato is the main greenhouse crop but it is also cultivated in low tunnels and in open fields, comprising a total annual production of 203999 metric tons (PCBS 2008). Several viruses are known worldwide to attack tomatoes causing serious damage and yield reduction. TMV, TYLCV, CMV and PVY are of the ten most viruses affecting the crop elsewhere (Oetting and Yunis, 2004, Agrios, 1997, Pico et. al. 1996, Sawalha. (2009a, 2009b, 2010).
Viral diseases of tomatoes occasionally cause serious damages and large economic losses. The amount of loss depends on the viral disease involved, the strain of the virus, the variety of tomato, the age of the plant at infection time, the presence of other diseases, and the
extent that viruses have spread during the planting of the crop (Walkey 1985).
Tomato production in Palestine, however, has not yet reached its full potential due to many factors related to climate and poor management, and above all, due to the high level of pests and diseases (PCBS 2008). In regard to diseases, viral diseases may be considered an important factor responsible for the recurrent crop failure in the country forcing many farmers to switch their choice of crop production from tomato to other vegetables (Sawalha 2010, Ministry of Agriculture, personal communication).
As tomato is the most important vegetable crop in the country, this research aims to study and identify the viruses affecting this crop and to shed light on their occurrence and prevalence. Northern districts including Jenin and Tobas were selected for this study because tomato production in those regions is 48250 metric tons and they occupy about 47% of the total tomato production in the West Bank part of Palestine (PCBS 2008).
Materials and Methods
Regions of study
Hazem Sawalha., APAC, Vol. 2, No. 1, pp. 98-101, 2012 99
fields located in Jenin were selected in Zababdeih, Al-Jededeih, and Qabatyya regions. Two fields were selected in Tobas; one of them was in Kashda village and the other was selected in Al-Far’a region a few hundred meters away from the town’s midpoint (Fig 1).
Growing season
The experiments were carried out during the summer growing season because this season is considered the main planting one of tomato in the regions of study throughout the year. In this season, seeds are sown during April and May and the seedlings are transplanted from the middle of May until the middle of June.
The pattern of tomato cultivation in the regions of study
Tomato is planted in the studied area in rows with a width of about 1 to 1.2m. The lands are first prepared by hoeing twice and then adding manure. Chemical fertilizers are added in three equal doses; the first when preparing the land, the second after transplanting and the third at fruit set.
Field visits and sample collection
Field visits in the studied regions began in July and continued until the end September. Samples were collected at least one month after the transplanting of tomato seedlings to ensure that tomato had become infected after being exposed for a sufficient period to the viral source. The selected fields were visited weekly in a regular manner and leaf samples were collected randomly from top parts of plants. The samples were labelled and kept frozen for laboratory testing.
Virus identification and incidence
Virus identification was done serologically using virus-specific antibodies purchased from international companies. Thus, indirect enzyme-linked immunosorbent assay (I-ELISA) adopted by Clark et. al (1986) was used to test the plant samples for CMV, PVY and TMV. With respect to the other case, the triple-antibody sandwich enzyme-linked immunosorbent assay (TAS-ELISA) was used to test the TYLCV infection. TAS-ELISA was used according to Macintosh et. al. (1992), Muniyappa et. al.
(1991), Sawalha et. al. (2000: 339) and Sawalha (2000: 24-89). The antibodies of CMV, PVY and TMV and the goat anti-rabbit conjugate were purchased from Bioreba, Inc. The polyclonal and monoclonal antibodies of TYLCV, along with the rabbit anti-mouse conjugate, were purchased from Adgen Ltd (Scotland, UK.). The
results of the ELISA tests were recorded one hour after the substrate incubation took place using the automated ELISA-Reader. The light absorbance was measured for ELISA wells at 405 nanometer (Sawalha (2009c)
Statistical analysis
Statistical analysis of the data was done using the Two-Sample Tests of Proportions (TSTP) to compare virus occurrence in the studied regions. The results were analysed using a level of significance when Ȑ = 0.05 (Lind et. al. 2005).
Results
Virus identification and incidence
ELISA tests showed that the TYLCV, CMV, TMV and PVY parasitize tomato fields in different percentages. ELISA readings recorded for the virus-infected samples were at least eight times greater than the readings recorded for the virus-free samples (Fig 1).
Fig 1: ELISA de te ction of tomato viruse s showing the diffe re nce be twe e n positive and ne gative control sample s
0 0.2 0.4 0.6 0.8 1 1.2 1.4
TYLCV CMV TMV PVY
Tom ato virus
Ab s o rb a n c e a t 4 0 5 n m Positive sample Negative sample
Hazem Sawalha., APAC, Vol. 2, No. 1, pp. 98-101, 2012 100
Fi g 2: Ave rage i n ci de n ce of tom ato vi ruse s i n the stu dy re gi ons i n 2003/2004
0 10 20 30 40 50 60 70 80 90 100
Kashda Al-Far'a Qabatyya Al-Zababdeih Al-Jededeih
Re gion Pe rc e n ta g e o f in fe c ti o n TYLCV CMV TMV PVY
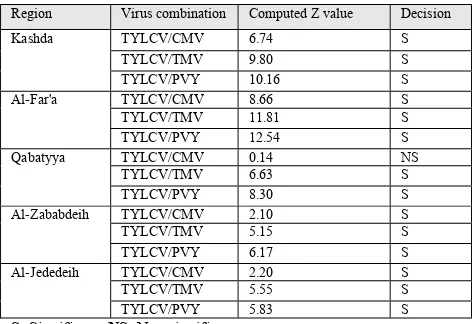
Statistical analysis revealed that the proportions of TYLCV in the studied regions were greater than CMV, TMV and PVY. The maximum computed Z values (Ȑ = 0.05) for the TYLCV compared with the other viruses were recorded in Al-Far'a region followed by Kashda, Qabatyya, Al-Zababdeih and Al-Jededeih. No significant difference was recorded between TYLCV and CMV infecting tomato fields in Qabatyya region as the computed Z value was much less than the critical one (Table 2)
Table 2: Statistical analyses and the Z value of the TSTP. The Z table = 1.65
Region Virus combination Computed Z value Decision
Kashda TYLCV/CMV 6.74 S
TYLCV/TMV 9.80 S
TYLCV/PVY 10.16 S
Al-Far'a TYLCV/CMV 8.66 S
TYLCV/TMV 11.81 S
TYLCV/PVY 12.54 S
Qabatyya TYLCV/CMV 0.14 NS
TYLCV/TMV 6.63 S
TYLCV/PVY 8.30 S
Al-Zababdeih TYLCV/CMV 2.10 S
TYLCV/TMV 5.15 S
TYLCV/PVY 6.17 S
Al-Jededeih TYLCV/CMV 2.20 S
TYLCV/TMV 5.55 S
TYLCV/PVY 5.83 S
S: Significant, NS: Non significant
Discussion
Tomato viruses were identified serologically using I-ELISA for CMV, and TMV, PVY, and TAS-I-ELISA for TYLCV. TYLCV is the most prevalent virus threatening tomato fields in the studied regions. The higher incidence of TYLCV (31-93%) is attributed to the abundance of whiteflies (Bemisia tabaci) and the overlapping that occurs between tomato growing seasons in the regions (Sawalha 2010). The 93% incidence of the virus in Al-Far’a may be attributed also to the warm climate which helps the whiteflies to appear earlier and to reproduce more compared with the other regions (PCBS 2005, Sawalha 2010). In this regards, Nava-Camberos et. al.
(2001) reported that the warm climate is the best condition for the whitefly development, fecundity and survival. Similar results were recorded in Jordan where Al-Musa and Mansour (1983) reported that TYLCV was the predominant virus affecting tomatoes in the Jordan Valley. CMV was detected to be the second-most prevalent virus affecting tomato fields in the studied regions. This infection with CMV may be attributed to allate aphid species which are usually ample in the region, especially the green peach aphid (Myzus persicae
Sulzer) and the melon aphid (Aphis gossypii Glover). The virus has a wide host range (800 species) from which it can be acquired by aphids and transmitted in a non-persistent manner and can also be mechanically transmitted (Oetting and Yunis, 2004, Trigiano et. al.
2004). Another reason for the wide spread occurrence of CMV in the studied regions may be attributed to the intensive and annual culturing of cucurbits including cucumber, squash and melon in the region. According to PCBS (2008), these crops are planted in a wide area with an annual production of 15376 and 9250 metric tons in Jenin and Tobas districts, respectively. These crops are the most suitable hosts for CMV, so their presence in the tomato growing sites makes them the viral source from which the allate aphids transmit the disease to tomato (Oetting and Yunis 2004, Sacristian et. al. 2004). The
Table 1: Number of samples collected from the studied region and the percentages of infection with TYLCV, CMV, TMV and PVY.
Region TYLCV
2003 2004
CMV 2003 2004
TMV 2003 2004
PVY 2003 2004
Total samples collected 2003 2004
NS PI NS PI NS PI NS PI NS PI NS PI NS PI NS PI
Kashda 33 70 35 69 11 23 11 21 3 6 0 0 1 2 0 0 47 51
Al-Far’a 43
93 45 90 14 30 17 34 5 10 3 6 2 4 1 2 46 50
Qabatyya 23
51 23 52 23 51 22 50 4 8 4 9 0 0 0 0 45 44
Al-Zababdeih 16
31 16 33 10 20 9 18 2 4 2 4 0 0 0 0 51 49
Al-Jededeih 17
34 15 28 11 22 8 15 1 2 2 3 1 2 0 0 50 54 TOTAL 132
55 134 54 69 29 67 27 15 6 11 4 4 2 1 0.004 239 248

![Fig. 2. Effect of [NaCl] on the degradation half-life of picric acid using vacuum UV. (25±0.50C, [picric acid]0=100 mg/l, reaction volume 2500ml, 6.5 dm3/min circulating rate)](https://thumb-ap.123doks.com/thumbv2/123dok/708649.284542/30.595.92.508.487.699/effect-degradation-picric-vacuum-picric-reaction-volume-circulating.webp)
![Fig. 4. Effect of temperature change on the VUV oxidative degradation of picric acid, (reaction volume = 2500ml, [picric acid]0=100 mg/l, 0.65 dm3/min circulating rate)](https://thumb-ap.123doks.com/thumbv2/123dok/708649.284542/31.595.76.506.337.558/effect-temperature-oxidative-degradation-reaction-volume-picric-circulating.webp)