www.elsevier.com / locate / bres
Research report
Electrophysiological properties of axons in mice lacking neurofilament
subunit genes: disparity between conduction velocity and axon
diameter in absence of NF-H
a ,1 b b a ,
*
ˇ
Jasna Kriz
, Qinzang Zhu , Jean-Pierre Julien , Ante L. Padjen
a
Department of Pharmacology and Therapeutics, the Montreal General Hospital Research Institute, McGill University,
´
3655 Sir William Osler Promenade (ex-Drummond Street), Montreal, QC H3G 1Y6, Canada
b
Centre for Research in Neuroscience, the Montreal General Hospital Research Institute, McGill University,
´
3655 Sir William Osler Promenade (ex-Drummond Street), Montreal, QC H3G 1Y6, Canada Accepted 22 August 2000
Abstract
Neurofilament proteins (NFs) are made by co-polymerization of three intermediate filament proteins, NF-L, NF-M and NF-H and constitute the most abundant cytoskeletal element in large myelinated axons. NFs have a well-established role as intrinsic determinants of axon caliber with all the functional implications, but the role of each individual NF subunit is much less clear. The aim of our study was to examine functional properties of large myelinated axons with altered morphology from mice bearing a targeted disruption of each NF genes (NF-L2/2, NF-M2/2and NF-H 2/2mice). Membrane properties, action potentials and single axon refractory period were measured in isolated sciatic nerves in vitro, using intra-axonal microelectrode recording in conjunction with current–clamp technique. Some results were obtained from whole nerves by sucrose-gap recording. The NF-knockout mice showed several deficits in physiological properties of low-threshold fibers. In keeping with smaller axon diameter, the conduction velocity was significantly decreased in NF-L
2/2 and NF-M 2/2 transgenic animals (control, 39.961.8 m / s, NF-M 2/2; 23.561.4 m / s, and NF-L2/2; 12.060.7 m / s, mean6S.E.M.; intra-axonal recording; similar ratios obtained by sucrose-gap recording; 22–268C). However, in spite of their preserved caliber, large myelinated axons in NF-H 2/2 mice also showed a significant decrease in conduction velocity (22.861.0 m / s, mean6S.E.M.). Although action potential amplitudes, duration and shape did not differ between control axons and transgenic animals, the refractory period was prolonged in NF-H 2/2 and NF-M 2/2 animals. Intracellular injections of 200 ms depolarizing and hyperpolarizing currents revealed outward and inward rectification in all animal groups. In comparison to control animals, NF-H2/2
mice expressed a significant decrease in outward rectification. Potassium channel blockers (4AP and TEA) and cesium ions were able to block outward and inward rectification in all myelinated axons in qualitatively the same manner. These results suggest that NF-H may have a specific role in modulating ion channel functions in large myelinated fibers. 2000 Elsevier Science B.V. All rights reserved.
Theme: Disorders of the nervous system
Topic: Genetic models
Keywords: Neurofilament; Transgenic mouse; Myelinated axon; Intra-axonal recording; Conduction velocity
1. Introduction ponent in large myelinated axons of the peripheral nervous
system (PNS). The three neurofilaments subunits (NF-L, Neurofilaments represent the major cytoskeletal com- 68 kDa; NF-M, 134 kDa and NF-H, 200 kDa [33,50]) share a conserved domain, common with other inter-mediate filaments (IF), involved in the formation of
*Corresponding author. Tel.: 11-514-398-3603; fax: 11-514-398- coiled–coil structure [23,24,74]. Neurofilaments form het-4449.
eropolymers, which in mice require NF-L with either E-mail address: [email protected] (A.L. Padjen).
1 NF-M or NF-H proteins [48,49].
Present address: Department of Pharmacology, Faculty of Medicine,
University of Rijeka, Rijeka, Croatia. The three neurofilaments are synthesized in the
perikaryon and transported down the axon with the slow structure–function relationship for three isoforms of NF component of axoplasmic transport [33]. Neurofilaments we studied the electrophysiological properties of large seem not to move along the axon as an organized network, myelinated axons in peripheral nerves of mice with as originally considered [44,45] but rather as a dynamic targeted disruption of NF genes. We report here that structure [8,52,53]. There is evidence that subunit ex- changes in expression of NF genes lead to functional change can occur along the axon [57] and that neurofila- changes in peripheral nerves of transgenic mice. Similar ment proteins can be transported in an unpolymerized form conclusion stems from our recent studies of mice
express-along axonal microtubules [76]. ing human NF-H [41,42], as well mice expressing
neuro-It is generally considered that one of the functions of filament-beta-galactosidase fusion protein [40]. Prelimin-neurofilaments is related to the control of radial growth of ary results of the present study have been published in an large myelinated axons. This notion is largely based on the abstract [43].
correlation between the number of neurofilaments and axonal diameter [22,26,30–32]. Direct support for this
hypothesis has been obtained by studies of three animal 2. Material and methods
models: a mutant quail lacking neurofilaments as a result
of a nonsense mutation in the gene coding for the NF-L 2.1. Animals and experimental surgery [56,85,86] a transgenic mouse expressing NF-H /b
-galacto-sidase fusion protein that prevents neurofilament transport The experiments were performed on isolated sciatic into axons [18] and a NF-L knock-out mouse [88]. It has nerves of 3–4-month-old control and transgenic mice (NF-been hypothesized that because the carboxy-terminal do- L2/2, NF-M 2/2and NF-H 2/2). Transgenic animals main of NF-H and NF-M is associated with NF sidearms were produced and housed in the SPF facility at the [28,29], increased phosphorylation at their KSP motif may Montreal General Hospital Research Institute [88]. result in increased negative charges associated with these The animals were decapitated under halothane or ether sidearms and consequently greater electrostatic repulsive anesthesia and the sciatic nerves were quickly dissected forces between neighboring NFs. The higher level of out (proximally at the exit point from the spinal canal, at phosphorylation of NF-H in myelinated (internodal) than the lumbo-sacral level and distally above the ankle). The non-myelinated (e.g. nodal) regions of axons is associated preparations were pinned to the bottom of the wax-coated with a 25–50% greater nearest-neighbor spacing of NFs chamber with insect needles and continuously perfused [17,34,54]. Neurofilaments have also been implicated in with mammalian Ringer saturated with 5% carbon dioxide regulation of several other cellular functions such as and 95% oxygen (pH57.4, temperature 22–258C). The maturation of regenerating myelinated axons [88] and mammalian Ringer solution contained in mM: 127 NaCl, regulation of axoplasmic transport [13,51] in which modu- 1.9 KCl, 2.4 CaCl , 1.2 KH PO , 1.3 MgSO , 26 NaHCO2 2 4 4 3
lation of neurofilament–microtubule interactions and pos- and 10 glucose. sibly microtubule stabilization [75,87], plays a major role.
There is increasing evidence of association between 2.2. Identification of fibers and sampling procedure changes in the normal metabolism of neurofilaments and
disease states [23]. Thus, many human neurodegenerative The intracellular recordings were obtained from 2 to disorders, such as amyotrophic lateral sclerosis (ALS) 3-cm segments of sciatic nerves with the impalement zone [7,11], Parkinson’s disease [27,73] and Alzheimer’s dis- ¯5 mm distal from the spinal roots. The recordings started
ease [73,77] are characterized by NF deposits within after at least 1.5 h incubation of dissected nerves in neurons (so-called spheroids or Lewy bodies). Although it mammalian Ringer solution, a solution that should prompt-is not known what role these deposits play in pathogenesprompt-is ly seal the cut ends of axons [82]. All fibers were impaled of human diseases, recent studies with transgenic mice blindly in the surface layers of the nerves. On the basis of clearly demonstrated that disorganized neurofilaments can previous experience with normal animals (amphibians and cause motor neuron degeneration [13,14,47,83]. Additional mammalian), this approach selects the largest axons in the evidence for neurofilament involvement in human disease population of myelinated axons. This assumption is sup-originates from the discovery of mutant NF-H alleles in ported by the conduction velocity of axons (.30 m / s at some ALS patients [19,39]. Several toxins that disrupt room temperature, control axons) and the low probability neurofilament transport or metabolism result in neuropathy of obtaining stable intra-axonal recordings from axons
[6,25]. ,6–7mm in diameter. At least two stable recordings were
resting membrane potential was more negative than –70 ventral root with the Image-1 software from Universal mV, with overshooting spikes. The microelectrode tech- Imaging Corp. (Pennsylvania) [87,88].
nique used in this study allows recording from undissected
axons; therefore, it is likely that such recordings better 2.6. Data analysis reflect axonal membrane properties in vivo [65,67,68].
Compound action potentials (CAP) were measured from Whenever appropriate results were expressed as sciatic nerves using the sucrose-gap method as previously means6standard error (S.E.M.). Statistical significance
described [61,62]. was assessed by using one-way analysis of variance
(ANOVA) followed by a Tukey HSD test. 2.3. Intra-axonal microelectrode recording technique
Glass microelectrodes were pulled with a Brown-Flam- 3. Results
ing puller (Sutter Instruments Co., San Francisco, CA)
filled with 3 M KCl and had a direct current resistance 3.1. Distribution of axon calibers in peripheral nerves of 60–80 MV. They were selected for their low noise and normal and transgenic mice
their ability to pass #2 nA without rectification. The
microelectrodes were connected to an amplifier with fast To examine the relationship between neurofilament rise time (Axoclamp 2B, Axon Instruments, Foster City, content and the radial growth and functional properties of CA). A bridge circuit allowed simultaneous current in- the peripheral axons, axonal diameters were measured in jection and recording through the same electrode (‘cur- the L5 ventral roots of 3-month-old animals (Fig. 1A). The rent–clamp’ technique). After capacity compensation, the ventral roots of wild-type mice expressed a bimodal rise time of 50 mV voltage calibration pulse was 20–50ms. distribution of axon calibers representing the populations This bandwidth allowed for appropriate bridge balance. of small and large myelinated axons. A bimodal dis-When possible, bridge balance was checked independently tribution with similar axonal diameters was preserved in by ensuring that the action potential overshoot remained NF-H 2/2 mice. However, peripheral nerves of NF-L constant [20]. After differential amplification at 20 kHz, 2/2 and NF-M 2/2 expressed a unimodal distribution following the low pass 8-pole Bessel filter set at 10 kHz, of small axon calibers with the large myelinated axons and a 16 bit A / D converter (ITC 16 Instrutech Corp., NY) shifted to the small category (Fig. 1B). The average caliber with appropriate software (TIDA for Windows) records of myelinated axons in L5 ventral roots of NF-H 2/2 were stored on a computer for further analysis. Sucrose- mice (4.6 mm) did not differ from controls (4.8 mm), gap recording was done with use of a differential amplifier unlike lower average values in NF-M2/2 (3.2 mm) and
[61]. NF-L (2.6 mm).
2.4. Application of potassium channel blockers 3.2. Resting and action potentials of axons in isolated
sciatic nerves
Potassium channel blockers were applied by diffusion
from 3 M KCl recording microelectrodes containing 50 Electrophysiological results described in this study were mM 4-AP and 20 mM TEA (DC resistance ,80 MV). obtained from over 60 stable recordings from large myeli-Previous studies in this laboratory indicate that their effects nated axons in 25–30-mm segments of sciatic nerve. In appear after 10–15 min of recording, reaching approxi- standard medium, intracellular placement of the microelec-mately 80% of maximal values 20–30 min after impale- trode was indicated by a sharp drop in the recorded ment [67]. Cesium ions (CsCl, 3 mM) were added to the potential (usually to255 to270 mV) and the appearance
perfusate. of brief action potentials (,1.5 ms duration and
overshoot-ing). After penetration, it usually required about 10–15
2.5. Histological methods min for the resting potential to stabilize. There was no
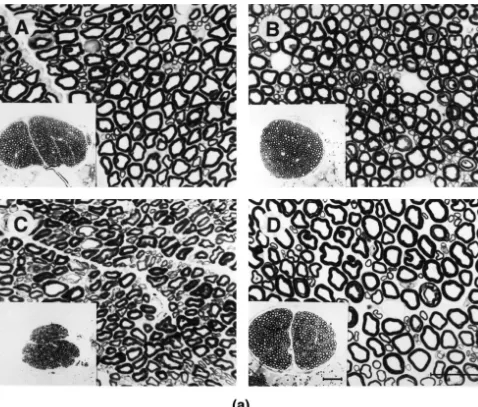
Fig. 1. Light micrographs of L5 ventral roots from normal (A), NF-M2/2(B), NF-L2/2(C), and NF-H2/2(D) mice. The depletion of NF-H2/2
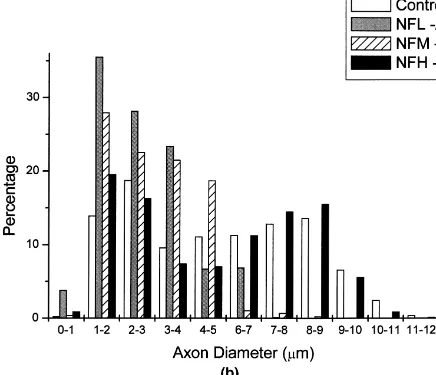
(D) had the least effects on axonal calibers followed by the depletion of NF-M2/2(B). Note the absence of large motor axons in NF-L2/2mice (C). Calibration bar550mm. (B) Comparison of inner axonal diameters from L5 ventral root axons of control, NF-M2/2, NF-L2/2and NF-H2/2mice. Quantitative study of axonal calibers reveals a marked reduction in transgenic sample (NF-L2/2and NF-M2/2) with unimodal distribution showing the absence of large myelinated fibers. Axon diameter is preserved in NF-H2/2mice with apparent maintenance of bimodal distribution as compared to the control. The histogram represents the distribution of inner axonal diameter of axons in L5 ventral roots from 3 to 5 animals per each animal group.
these animals were suitable only for measurements of ms (n525). Two groups of transgenic animals showed a
conduction velocity (CV). significant decrease in CV of their axons (NF-M 2/2:
23.5461.41 m / s n525; NF-L 2/2: 12.0560.69 m / s,
3.3. Conduction velocity n522), as expected from their smaller diameters.
Fig. 1. (continued )
one peak. Fig. 2 shows samples of typical CAPs obtained higher room temperature (268C), which explains their from sciatic nerves of different transgenic animals. The CV faster CV compared to single axons recordings, but the in all transgenic animal groups was significantly slower relative values are essentially identical to those obtained by than controls (control: 48 m / s; NF-H 2/2: 33 m / s; intra-axonal recording. In sciatic nerves from NF-L2/2, NF-M: 28 m / s; NF-L 2/2: 18 m / s for the fastest fiber unlike the CAPs in nerves from wild-type controls, NF-population). These measurements were done at somewhat H2/2 and NF-M2/2mice, low-threshold stimuli
gener-Table 1
a
Functional properties of large myelinated axons of mice with different neurofilament expression
RMP (mV) AP (mV) AP/ 2 (ms) Hyperpolarization Depolarization
Mean6SE n Mean6SE n Mean6SE n Rp (MV) Rss (MV) Rp (MV) Rss (MV) Mean6SE n Mean6SE n Mean6SE n Mean6SE n
Control 275.560.5 31 85.763.0 19 0.6560.02 19 33.562.4 28 25.863.1 28 14.261.0 28 12.161.0 28 NF-M2/2 274.261.0 24 79.063.9 16 0.6860.02 16 31.263.0 11 27.262.7 11 15.561.2 11 12.961.2 11 NF-H2/2 276.160.7 25 85.663.0 16 0.7160.02 16 42.663.9* 14 34.463.4 14 16.861.0 14 15.561.0* 14
a
Results obtained by intra-axonal recording. RMP, resting membrane potential; AP, action potential amplitude; AP/ 2, action potential duration at half-amplitude; R and R , peak and steady state input resistance (measured 15–20 ms after pulse initiation and 190–195 ms after pulse initiation,p ss
ated CAPs with several peaks, suggesting the presence of populations of fibers with similar thresholds but with different CVs. Multiple action potentials have never been observed in intra-axonal recordings from NF-L 2/2 axons, arguing against the alternative possibility that the multiple peaks were due to repetitive activity in these low-threshold fibers.
3.4. Action potential shape and conduction velocity
There was no significant difference in AP amplitude or AP duration between axons from transgenic and control animals, although values recorded from axons of NF-H 2/2 mice showed a tendency for longer APs. Thus, the average AP duration at half-amplitude (AP/ 2) in axons from various mice did not differ (see Table 1; see also samples in Fig. 3). At membrane potentials close to the RMP, action potentials were only occasionally followed by slow depolarizing afterpotentials (DAP, as originally de-scribed in amphibian axons [2] and mammalian axons [16]. In those cases, the DAPs showed voltage dependence by increasing in amplitude with membrane hyperpolariza-tion.
An analysis of the time course of AP (dV / dt of rising and falling phase) revealed no significant differences between the nerves from control and transgenic animals (rising phase: 475634 V/ s in controls, 438629 V/ s in NF-M2/2; 457617 V/ s in NF-H2/2 mice; corre-sponding values of falling phase: 213466 V/ s in control, 2134610 V/ s in NF-M2/2; 213767 in NF-H 2/2 mice, mean6S.E., n513–19).
However, examination of the correlation between the rates of rising and falling phases of AP and CV and / or AP/ 2 revealed significant differences among the tested groups. In control animals the rate of AP rise vs. CV (Fig. 4A), and the rate of AP decline vs. AP duration (Fig. 4B) showed significant positive correlation (P,0.05). In axons from transgenic mice (Fig. 4A,B) only NF-M2/2 mice expressed a significant (positive) correlation between the rate of AP decline and AP duration (Fig. 4B).
3.5. Recovery period of AP generation
Recovery of AP generation (refractory or recovery period) was obtained by double stimulation using supra-maximal stimuli and varying the inter-pulse interval. Recovery of AP generation in axons from NF-M2/2and NF-H2/2 mice was significantly slower than in control animals. In the first 3–5 ms, recovery reached 75–80% of
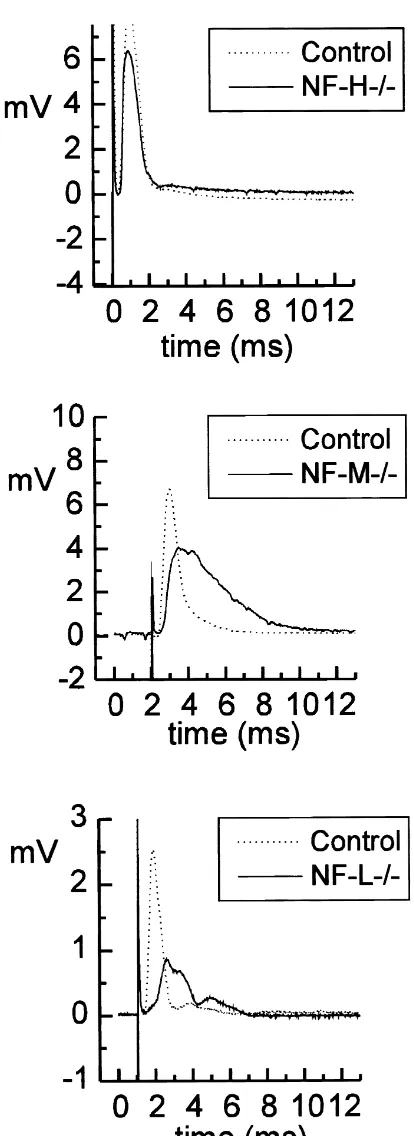
Fig. 2. Compound action potentials (CAPs) were simultaneously
re-the test AP amplitude compared to wild-type mice where
corded from 20 to 25-mm long segments of sciatic nerves isolated from
control and transgenic mice (as labeled). Note that the difference in these values were close to 90% of the initial spike height
delays do not reflect the true difference in CV since the nerve pairs were (Fig. 5). not of the same length (see text for details). Samples of CAPs induced by
supra-maximal stimulation for low-threshold fibers show multiple peaks
3.6. Accommodation
in nerves from NF-L 2/2 mice as compared with control and other knockouts. The conduction velocity obtained from sucrose-gap recordings
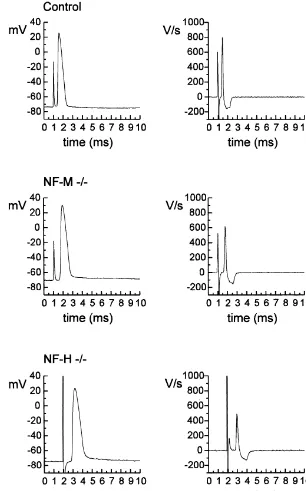
Fig. 3. Samples of typical intra-axonally recorded action potentials with corresponding differentiated values (dV / dt) (control, NF-M2/2and NF-H2/2
mice).
current pulses with only a few action potentials, indicating hyperpolarizing current steps were used to test the ex-the presence of a strong accommodation. This is similar to citability of axons and construct V/I relationships. In all previous observations in other axonal preparations animal groups the voltage responses were characterized by [1,64,67]. Although some of the fibers in this study a fast rise to depolarizing pulses and a noticeably slower showed differences in accommodation no clear pattern response to the hyperpolarizing pulses. The
hyperpolariz-could be established. ing voltage responses featured a ‘sag’, typically observed
Fig. 4. (A) Correlation between dV / dt of rising phase of an action potential and its CV. Note significant positive correlation (P,0.05) for controls, but not for axons from transgenic animal. (B) Correlation between dV / dt of falling phase of action potential and its duration at half-amplitude (AP/ 2). Note significant positive correlation for controls and NF-M2/2(P,0.05), and no correlation for NF-H2/2axons. Correlation coefficients are given above the regression lines. (Control, n518, NF-M2/2, n512, and NF-H2/2, n513).
considerable variety in the expression of inward rectifica- resistance ranged from 1 to 3 MV in responses to tion in the population of axons, but no consistent pattern depolarizing pulses, to 60–70 MV at the peak of
hy-could be established. perpolarizing pulses. There were two regions of linear V /I
To obtain voltage–current (V /I ) curves the membrane relationship below 2100 mV (defined as peak resistance, potential was measured at the peak (R ) and the steadyp R ) and steady-state resistance, R ), the latter identifiedp ss
state (R ) of the voltage displacement. The V /I relation-ss with the activation of inward rectifier. In axons from NF-H ship was strongly non-linear, reflecting the activation of 2/2 mice there was a significant increase in R values inp
subunit genes in order to establish their role in functional properties of axons. As expected, the conduction velocity of axons appeared to be related to their diameter (see Section 3), the smaller average diameters of axons in NF-L 2/2 and NF-M 2/2 being associated with slower conduction in records from whole sciatic nerves. Similar results were obtained at the single axon level using microelectrode recording from large myelinated nerve fibers of NF-L2/2 and NF-M2/2mice. Unexpectedly, this relationship did not hold for the NF-H 2/2 mice where the conduction velocity measured in whole nerves or single axons was significantly decreased in spite of a normal distribution of diameters.
Intra-axonal studies revealed several differences in physiological parameters of peripheral nerves in transgenic mice: (1) the refractory period in NF-M2/2 and NF-H 2/2 mice was prolonged; (2) in contrast to axons from
Fig. 5. Refractory period of action potential (AP) generation is increased
in single sciatic axons of transgenic mice. In intra-axonal recordings wild-type and NF-M2/2, in NF-H2/2 mice there was
samples were obtained by varying delay to second identical stimulus in no correlation between the rate of rise and decay of action dual stimulation protocol (see Section 2). Recovery of AP generation was
potential and conduction velocity (as well as AP duration
measured 3, 5 and 7 ms after first stimulating pulse. Note almost
at half amplitude); (3) changes in rectification in axons of
complete recovery of AP amplitude after delay of 5 ms in axons of
NF-H 2/2 mice (increase in input resistance during
control animals. Recovery of AP in axons of NF-M2/2and NF-H2/2
mice is decreased in first 3–5 ms reaching the control values by 7 ms. hyperpolarization and in the input resistance in the steady
Values represent mean6S.E.; *significantly different from control, P# state period during depolarization). These findings suggest
1
0.05 (n53).
changes in the function of axonal ion channels (K and 1
Na ) and / or possible morphological changes of axons in inward rectification (measured as a ratio of R /R ) weress p transgenic animals that will be discussed separately. similar in all animal groups (0.77, 0.87, 0.81, for control,
NF-M2/2 and NF-H 2/2, respectively)
The shape of the V /I relationship obtained by injection 4.1. Neurofilaments, axonal caliber and conduction of depolarizing currents was highly non-linear, indicating velocity
strong outward rectification of axonal membrane (cf. Refs.
[59,67]). Input resistance was measured at two points; at The present understanding of the role of NFs is that they the voltage displacement 15–20 ms after pulse onset (R )p are major intrinsic determinants of axon caliber in large and at near steady-state point (175 ms later; R ). Theress myelinated nerve fibers [12,46,81,84]. The control of was no significant difference in R , but the axons of NF-Hp axonal caliber has a functional significance because diam-2/2 mice showed a significant increase in Rss resistance eter is the principal determinant of conduction velocity in
(see Table 1). myelinated nerve fibers [30,70,71,78]. The report of axonal
1
The effect of K channel blockade was examined using hypotrophy and reduced conduction velocity in Japanese microelectrodes containing 50 mM 4-AP and 20 mM TEA quail mutant lacking NFs as a result of nonsense mutation (see Section 2). Twenty minutes after impalement there in the NF-L gene [56,72] is particularly strong evidence in was a marked decrease in outward rectification as expected favor of this notion. Reduced axonal calibers accompanied from previous studies [1,67] (data not shown). Approxi- by decreased conduction velocity have also been observed mately 10 min after addition of CsCl (3 mM) to the in transgenic mice expressing NF-H /b-galactosidase fu-perfusate, the impaled axons of control animals lost the sion protein that interferes with neurofilament transport characteristic ‘sag’ in hyperpolarizing potentials. Cesium into axon [18,40]. Reduced axonal diameter (with neurode-did not affect outward rectification in the region positive to generation) has been also observed in various transgenic and just negative to the RMP, suggesting that extracellular mice over-expressing any single neurofilament subunit cesium ions did not affect any conductances activated in [13,14,41,83]. The positive correlation between fiber diam-the region around RMP. TEA (20 mM) depolarized diam-the eter and conduction velocity or between fiber diameter and
fibers 5–10 mV. neurofilament gene expression seems to hold even for
changes following nerve injury or during neuronal disease [30,38,39]. As expected, our study provides evidence in
4. Discussion support of a positive correlation between reduction in the
A plausible view of the mechanism by which NF 4.2. Differences in ion channel functions maintain axon caliber involves the highly phosphorylated
carboxy-tail domain of NF-H that orders axoplasmic The results presented here, and in our recent study of structure through interaction (cross-bridging) between mice expressing human NF-H [41], indicate that mem-adjacent neurofilaments [28,29]. The phosphorylated NF-H brane voltage responses and action potentials recorded in tail may contribute to axonal caliber through a three- axons from normal mice are similar to those obtained by dimensional filament scaffold and / or through interactions intra-axonal recordings from other vertebrate myelinated with other axonal components, such as microtubules, axons. Thus, responses to hyperpolarizing current in cortical actin arrays, and potentially many other com- normal mice axons demonstrate a long charging time ponents. The preserved diameter of large myelinated axons constant, similar in duration to that in amphibian [2,58] in NF-H 2/2 mice could probably be explained by and rat axons [1,5] as well as outward and inward hyperphosphorylation of the NF-H tails that could in- rectifications. In agreement with the findings in rats [1], fluence the nearest inter-filament distance and / or twofold outward rectification is suppressed by 4-AP and TEA, increase in microtubules [69,81,87]. indicating the involvement of at least two
voltage-depen-1
Why the myelinated axons in NF-H 2/2 mice do not dent K internodal conductances [1,9,10,16,70,79,80]; follow the predicted structure–function (i.e., diameter and while the inward rectification is sensitive to cesium, as in CV) relationship? In these mice, the nerve fiber diameter other peripheral axons [1,66,67] and other cells [63]. has the typical bimodal distribution. The fact that the CV Action potentials in mice axons are only occasionally measured by intra-axonal sampling agrees with the CV of followed by a voltage-dependent DAP (increased by the compound action potentials argues against the unrepre- hyperpolarization), as observed in rats [4,5,16], unlike their sentative sampling by the microelectrodes (in any case, common appearance in amphibian axons [2,64]. These intra-axonal recordings appear to preferentially sample the differences in DAPs expression are considered to result
1
largest axons, see Section 2). Preliminary morphological from enhanced activation of K conductances in mam-examination of large myelinated axon from NF-H 2/2 malian axons [16] rather than any difference in the high show that both the G ratio (ratio of axonal diameter vs. conductance pathway through myelin sheets (Barrett–Bar-total myelinated axon diameter [21]; ca. 0.6–0.7) and rett conductance [2,4,70]), as mentioned above.
internodal distances (ca. 1 mm) are similar to those in The upstroke (dV / dt) of the AP in transgenic mice was control mice. Thus, the fibers in NF-H 2/2 mice would not significantly different from the control values, sug-belong to Rushton’s category of ‘dimensionally similar’ gesting that there was no alteration in sodium conductance fibers [71], i.e. one would expect CV to be proportional to activation. However, the refractory period (action potential the axonal diameter. Yet, there is approximately 20% recovery) in transgenic animals was significantly delayed reduction in conduction velocity in axons from NF-H2/2 in the first 3–5 ms after a test AP, suggesting a delay in
1
mice as compared to control fibers suggesting that mem- recovery from Na conductance inactivation. Since several 1
brane characteristics of axons have changed properties. lines of evidence link Na channel phosphorylation and One may also take the differences in correlation between the inactivation process [15,36,55], one may speculate action potential properties and CV (see Section 3) as whether a change in phosphorylation may be involved in another indication of differences in membrane properties in the observed results.
transgenic animals, although for the moment we do not What still remains to be explained are the differences in have a plausible explanation of this phenomenon. correlation between the AP time course on the one hand Axonal diameter has been established as the primary and with the other, conduction velocity and duration of AP. determinant of CV in myelinated axons. Previous theoret- Theoretical studies have identified the rise time of the ical evaluations indicate that CV should be relatively action potential as having an important bearing on the insensitive to nodal parameters, moderately sensitive to conduction velocity in myelinated axons [37]. Thus, at axoplasmic conductance (as well as diameter) and quite physiological temperature the relationship for large myeli-insensitive to internodal parameters [37,70,78]. This early nated axons is found to be approximately 6 m / s per work may not be entirely relevant because it does not take micron of overall fiber diameter [35]. Any deviation from into consideration the recent finding that the internodal this relationship, as was observed in axons from transgenic
1
membrane, with its spectrum of voltage dependent K mice (NF-H 2/2), would suggest a difference in the conductances [79] does participate in the conduction of membrane properties. Preliminary morphological studies impulses via the so-called Barrett–Barrett pathway [2– indicate that the internodal distance in transgenic animals 4,59,70]. Additional evidence in favor of this model of falls within the normal range, suggesting that this mor-axons has recently been obtained in simulation experi- phological parameter cannot be involved. One remaining ments (‘leaky myelin’ model [60]). The present results unchecked parameter that could change (decrease) the CV
1
demonstrate a decrease in internodal conductance (i.e. K is an increase in axoplasmic resistance. At present it is not conductances, see below) in axons of NF-H 2/2 mice. known whether disturbance of the intracellular architecture This decrease may be relevant for the decrease in CV in the transgenic animals may affect the axial resistance.
sclerosis, in: R.A. Smith (Ed.), Handbook of Amyotrophic Lateral
regulatory mechanism(s) linking changes in NF and ion
Sclerosis, Marcel Dekker, New York, 1992, pp. 133–181.
channel functions. However, the existence of a complex
[12] D.W. Cleveland, M.J. Monteiro, P.C. Wong, S.R. Gill, J.D. Gearhart,
and yet unidentified link between NF expression and ion P.N. Hoffman, Involvement of neurofilaments in the radial growth of channel function in myelinated axons is corroborated by axons, J. Cell Sci. Suppl. 15 (1991) 85–95.
our recent study of mice expressing human NF-H [41,42]. [13] J.F. Collard, F. Cote, J.P. Julien, Defective axonal transport in a transgenic mouse model of amyotrophic lateral sclerosis (see
In summary, this study describes some of the basic
comments), Nature 375 (1995) 61–64.
electrophysiological properties of large myelinated fibers
[14] F. Cote, J.F. Collard, J.P. Julien, Progressive neuronopathy in
in mice with different neurofilament subunit expression. transgenic mice expressing the human neurofilament heavy gene: a Our results indicate that myelinated axons of NF-H 2/2 mouse model of amyotrophic lateral sclerosis, Cell 73 (1993) mice, in comparison with control and NF-M2/2 mice, 35–46.
[15] S. Cukierman, Regulation of voltage-dependent sodium channels
express different functional properties than expected from
(Review), J. Membr. Biol. 151 (1996) 203–214.
morphological changes alone (decreased CV without a
[16] G. David, B. Modney, K.A. Scappaticci, J.N. Barrett, E.F. Barrett,
change in axon size), suggesting that NF-H itself or
Electrical and morphological factors influencing the depolarizing
through its interaction with other axonal components after-potential in rat and lizard myelinated axons, J. Physiol. 489 (microtubules, actin or perhaps other cytoskeletal mole- (1995) 141–157.
[17] S.M. De Waegh, V.M.Y. Lee, S.T. Brady, Local modulation of
cules) may have a role in modifying the functional
neurofilament phosphorylation, axonal caliber, and slow axonal
properties of large myelinated fibers. In addition, the
transport by myelinating Schwann cells, Cell 68 (1992) 451–463.
electrical properties defined in this study should help to
[18] J. Eyer, A. Peterson, Neurofilament-deficient axons and perikaryal
generate a computational model that would further com- aggregates in viable transgenic mice expressing a neurofilament-plete our understanding of these complex neural structures. beta-galactosidase fusion protein, Neuron 12 (1994) 389–405.
[19] D.A. Figlewicz, A. Krizus, M.G. Martinoli, V. Meininger, M. Dib, G.A. Rouleau, J.P. Julien, Variants of the heavy neurofilament subunit are associated with the development of amyotrophic lateral
Acknowledgements sclerosis, Hum. Mol. Genet. 3 (1994) 1757–1761.
[20] K. Frank, M.G.F. Fuortes, Stimulation of spinal motoneurons with intracellular electrodes, J. Physiol. (Lond.) 134 (1956) 451–470.
This work has been supported in part by the Canadian
[21] R.L. Friede, R. Bischhausen, How are sheath dimensions affected by
ALS Association, Canadian Diabetes Association and
axon caliber and internode length?, Brain Res. 235 (1982) 335–350.
MRC of Canada. We thank Dr K. Krnjevic for comments. [22] R.L. Friede, T. Samorajski, Axon caliber related to neurofilaments
and microtubules in sciatic nerve fibers of rats and mice, Anat. Rec. 167 (1970) 379–387.
[23] E. Fuchs, D.W. Cleveland, A structural scaffolding of intermediate
References filaments in health and disease. (Review), Science 279 (1998)
514–519.
[1] M. Baker, H. Bostock, P. Grafe, P. Martius, Function and dis- [24] N. Geisler, S. Fischer, J. Vandekerckhove, U. Plessmann, K. Weber, tribution of three types of rectifying channel in rat spinal root Hybrid character of a large neurofilament protein (NF-M): inter-myelinated axons, J. Physiol. (Lond.) 383 (1987) 45–67. mediate filament type sequence followed by a long and acidic [2] E.F. Barrett, J.N. Barrett, Intracellular recording from vertebrate carboxy-terminal extension, EMBO J. 3 (1984) 2701–2706.
myelinated axons: mechanism of depolarizing afterpotential, J. [25] J.W. Griffin, P.N. Hoffman, A.W. Clark, P.T. Carroll, D.L. Price, Physiol. (Lond.) 323 (1982) 117. Slow axonal transport of neurofilament proteins: impairment of [3] A.R. Blight, Computer simulation of action potentials and after- beta,beta9-iminodipropionitrile administration, Science 202 (1978)
potentials in mammalian myelinated axons: the case for a lower 633–635.
resistance myelin sheath, Neuroscience 15 (1985) 13–31. [26] J.W. Griffin, D.F. Watson, Axonal transport in neurological disease. [4] A.R. Blight, S. Someya, Depolarizing afterpotentials in myelinated (Review), Ann. Neurol. 23 (1988) 3–13.
axons of mammalian spinal cord, Neuroscience 15 (1985) 1–12. [27] W.D. Hill, V.M. Lee, H.I. Hurtig, J.M. Murray, J.Q. Trojanowski, [5] C.M. Bowe, J.D. Kocsis, S.G. Waxman, The association of the Epitopes located in spatially separate domains of each neurofilament supernormal period and the depolarizing afterpotential in myelinated subunit are present in Parkinson’s disease Lewy bodies, J. Comp. frog and rat sciatic nerve, Neuroscience 21 (1987) 585–593. Neurol. 309 (1991) 150–160.
[6] M.J. Carden, V.M. Lee, W.W. Schlaepfer, 2,5-Hexanedione neuro- [28] N. Hirokawa, M.A. Glicksman, M.B. Willard, Organization of pathy is associated with the covalent crosslinking of neurofilament mammalian neurofilament polypeptides within the neuronal cyto-proteins, Neurochem. Pathol. 5 (1986) 25–35. skeleton, J. Cell Biol. 98 (1984) 1523–1536.
[7] S. Carpenter, Proximal axonal enlargement in motor neuron disease, [29] N. Hirokawa, S. Takeda, Gene targeting studies begin to reveal the Neurology 18 (1968) 841–851. function of neurofilament proteins, J. Cell Biol. 143 (1998) 1–4. [8] S.S. Chin, R.K. Liem, Transfected rat high-molecular-weight neuro- [30] P.N. Hoffman, D.W. Cleveland, J.W. Griffin, P.W. Landes, N.J.
filament (NF-H) coassembles with vimentin in a predominantly Cowan, D.L. Price, Neurofilament gene expression: a major deter-nonphosphorylated form, J. Neurosci. 10 (1990) 3714–3726. minant of axonal caliber, Proc. Natl. Acad. Sci. USA 84 (1987) [9] S.Y. Chiu, J.M. Ritchie, Evidence for the presence of potassium 3472–3476.
channels in the paranodal region of acutely demyelinated mam- [31] P.N. Hoffman, J.W. Griffin, B.G. Gold, D.L. Price, Slowing of malian single nerve fibres, J. Physiol. (Lond.) 313 (1981) 415–437. neurofilament transport and the radial growth of developing nerve [10] S.Y. Chiu, J.M. Ritchie, On the physiological role of internodal fibers, J. Neurosci. 5 (1985) 2920–2929.
Identification of major structural polypeptides of the axon and their [56] O. Ohara, Y. Gahara, T. Miyake, H. Teraoka, T. Kitamura, Neuro-generality among mammalian neurons, J. Cell Biol. 66 (1975) filament deficiency in quail caused by nonsense mutation in
neuro-351–366. filament-L gene, J. Cell Biol. 121 (1993) 387–395.
[34] S.T. Hsieh, G.J. Kidd, T.O. Crawford, Z. Xu, W.-M. Lin, B.D. [57] S. Okabe, H. Miyasaka, N. Hirokawa, Dynamics of the neuronal Trapp, D.W. Cleveland, J.W. Griffin, Regional modulation of neuro- intermediate filaments, J. Cell Biol. 121 (1993) 375–386. filament organization by myelination in normal axons, J. Neurosci. [58] A.L. Padjen, T. Hashiguchi, Primary afferent depolarization and 14 (1994) 6392–6401. GABA responses: A study using intrafiber recording, Proc. Can. [35] J.B. Hursh, Conduction velocity and diameter of nerve fibers, Am. J. Fed. Biol. Soc. 24 (1981) 240.
Physiol. 127 (1939) 131–139. [59] A.L. Padjen, T. Hashiguchi, Primary afferent depolarization in frog [36] I.I. Ismailov, D.J. Benos, Effects of phosphorylation on ion channel spinal cord is associated with an increase in membrane conductance,
function. (Review), Kidney Int. 48 (1995) 1167–1179. Can. J. Physiol. Pharmacol. 61 (1983) 626–631.
[37] J.J. Jack, Physiology of peripheral nerve fibres in relation to their [60] A.L. Padjen, T. Hashiguchi, A computational model of myelinated size, Br. J. Anaesth. 47 (Suppl.) (1975) 173–182. axons in frog dorsal root, Soc. Neurosci. Abstr. 20 (1994) 1526. [38] J.P. Julien, A role for neurofilaments in the pathogenesis of
[61] A.L. Padjen, P.A. Smith, Specific effect of aD-L-aminoadipic acid on amyotrophic lateral sclerosis. (Review), Biochem. Cell Biol. 73
synaptic transmission in frog spinal cord, Can. J. Physiol. Phar-(1995) 593–597.
macol. 58 (1980) 692–698. [39] J.P. Julien, Neurofilaments and motor neuron disease, Trends Cell
[62] A.L. Padjen, P.A. Smith, The role of the electrogenic sodium pump Biol. 7 (1997) 243–249.
in the glutamate afterhyperpolarization of frog spinal cord, J. ˇ
[40] J. Kriz, J.A. Falchetto, J. Eyer, A. Peterson, A.L. Padjen,
Electro-Physiol. (Lond.) 336 (1983) 433–451. physiological properties of peripheral nerves from mice expressing a
[63] H.C. Pape, Queer current and pacemaker: the hyperpolarization-neurofilament-beta-galactosidase fusion protein, Soc. Neurosci.
activated cation current in neurons, Annu. Rev. Physiol. 58 (1996) Abstr. 22 (1996) 1655.
299–327. ˇ
[41] J. Kriz, J. Mayer, J.P. Julien, A.L. Padjen, Altered ionic
conduct-[64] M.O. Poulter, T. Hashiguchi, A.L. Padjen, Dendrotoxin blocks ances in axons of transgenic mouse expressing the human
neurofila-accommodation in frog myelinated axons, J. Neurophysiol. 62 ment heavy gene: a mouse model of amyotrophic lateral sclerosis,
(1989) 174–184. Exp. Neurol. 163 (2000) 414–421.
[65] M.O. Poulter, T. Hashiguchi, A.L. Padjen, An examination of frog ˇ
[42] J. Kriz, J. Meyer, J.P. Julien, A.L. Padjen, Functional defects in
myelinated axons using intracellular microelectrode recording: the peripheral nerves from transgenic mice with hyperexpression of
role of the voltage dependent and leak conductances on the steady heavy neurofilament, Soc. Neurosci. Abstr. 24 (1998) 481.
state electrical properties, J. Neurophysiol. 70 (1993) 2301–2312. ˇ
[43] J. Kriz, Q. Zhu, J.P. Julien, A.L. Padjen, Functional defects of
[66] M.O. Poulter, A.L. Padjen, Cesium blocks the inward rectifying peripheral nerves from transgenic mice with different neurofilament
conductance in frog sensory axon, Proc. Can. Fed. Biol. Soc. 31 expression, Soc. Neurosci. Abstr. 23 (1997) 1876.
[44] R.J. Lasek, Polymer sliding in axons, J. Cell Sci. Suppl. 5 (1986) (1998) 138.
161–179. [67] M.O. Poulter, A.L. Padjen, Different voltage dependent potassium [45] R.J. Lasek, J.A. Garner, S.T. Brady, Axonal transport of the conductances regulate action potential repolarization and excitability
cytoplasmic matrix, J. Cell Biol. 99 (1984) 212s–221s. in frog myelinated axon, Neuroscience 68 (1995) 497–504. [46] M.K. Lee, D.W. Cleveland, Neurofilament function and dysfunction: [68] M.O. Poulter, A.L. Padjen, T. Hashiguchi, Evidence for a sodium
Involvement in axonal growth and neuronal disease, Curr. Opin. dependent potassium conductance in frog myelinated axon,
Neuro-Cell. Biol 6 (1994) 34–40. science 68 (1995) 487–495.
[47] M.K. Lee, J.R. Marszalek, D.W. Cleveland, A mutant neurofilament [69] M.V. Rao, M.K. Houseweart, T.L. Williamson, T.O. Crawford, J. subunit causes massive, selective motor neuron death: implications Folmer, D.W. Cleveland, Neurofilament-dependent radial growth of for the pathogenesis of human motor neuron disease, Neuron 13 motor axons and axonal organization of neurofilaments does not (1994) 975–988. require the neurofilament heavy subunit (NF-H) or its phosphoryla-[48] M.K. Lee, Z. Xu, P.C. Wong, D.W. Cleveland, Neurofilaments are tion (see comments), J. Cell Biol. 143 (1998) 171–181.
obligate heteropolymers in vivo, J. Cell Biol. 122 (1993) 1337– [70] J.M. Ritchie, Physiology of axons, in: S.G. Waxman, J.D. Kocsis,
1350. P.K. Stys (Eds.), The Axon. Structure, Function and
Pathophysiol-[49] R.K. Liem, Molecular biology of neuronal intermediate filaments, ogy, 1995, pp. 68–96.
Curr. Opin. Cell Biol. 5 (1993) 12–16. [71] W.A.H. Rushton, A theory of the effects of fibre size in medullated [50] R.K. Liem, S.H. Yen, G.D. Salomon, M.L. Shelanski, Intermediate nerve, J. Physiol. (Lond.) 115 (1951) 101–122.
filaments in nervous tissues, J. Cell Biol. 79 (1978) 637–645. [72] T. Sakaguchi, M. Okada, T. Kitamura, K. Kawasaki, Reduced [51] J.R. Marszalek, T.L. Williamson, M.K. Lee, Z. Xu, P.N. Hoffman, diameter and conduction velocity of myelinated fibers in the sciatic M.W. Becher, T.O. Crawford, D.W. Cleveland, Neurofilament nerve of a neurofilament-deficient mutant quail, Neurosci. Lett. 153 subunit NF-H modulates axonal diameter by selectively slowing (1993) 65–68.
neurofilament transport, J. Cell Biol. 135 (1996) 711–724. [73] M.L. Schmidt, J. Murray, V.M. Lee, W.D. Hill, A. Wertkin, J.Q. [52] M.J. Monteiro, D.W. Cleveland, Expression of NF-L and NF-M in Trojanowski, Epitope map of neurofilament protein domains in fibroblasts reveals coassembly of neurofilament and vimentin cortical and peripheral nervous system Lewy bodies, Am. J. Pathol. subunits, J. Cell Biol. 108 (1989) 579–593. 139 (1991) 53–65.
[53] R.A. Nixon, Axonal transport of cytoskeletal proteins, in: R.D. [74] P.M. Steinert, D.R. Roop, Molecular and cellular biology of Burgoyne (Ed.), The Neuronal Cytoskeleton, Wiley-Liss, New York, intermediate filaments, Annu. Rev. Biochem. 57 (1988) 593–625. 1991, pp. 283–307. [75] T. Tashiro, R. Imai, Y. Komiya, Early effects of beta,beta9 -im-[54] R.A. Nixon, P.A. Paskevich, R.K. Sihag, C.Y. Thayer, Phosphoryla- inodipropionitrile on tubulin solubility and neurofilament
[78] S.G. Waxman, Determinants of conduction velocity in myelinated [84] Z. Xu, J.R. Marszalek, M.K. Lee, P.C. Wong, J. Folmer, T.O. nerve fibers, Muscle Nerve 3 (1980) 141–150. Crawford, S.T. Hsieh, J.W. Griffin, D.W. Cleveland, Subunit com-[79] S.G. Waxman, Voltage-gated ion channels in axons: Localization, position of neurofilaments specifies axonal diameter, J. Cell Biol.
function, and development, in: S.G. Waxman, J.D. Kocsis, P.K. Stys 133 (1996) 1061–1069.
(Eds.), The Axon. Structure, Function and Pathophysiology, 1995, [85] H. Yamasaki, G.S. Bennett, C. Itakura, M. Mizutani, Defective pp. 218–243. expression of neurofilament protein subunits in hereditary hypo-[80] S.G. Waxman, J.M. Ritchie, Molecular dissection of the myelinated trophic axonopathy of quail, Lab. Invest. 66 (1992) 734–743.
axon, Ann. Neurol. 33 (1993) 121–136. [86] H. Yamasaki, C. Itakura, M. Mizutani, Hereditary hypotrophic [81] P.C. Wong, J. Marszalek, T.O. Crawford, Z. Xu, S.T. Hsieh, J.W. axonopathy with neurofilament deficiency in a mutant strain of the
Griffin, D.W. Cleveland, Increasing neurofilament subunit NF-M Japanese quail, Acta Neuropathol. (Berl.) 82 (1991) 427–434. expression reduces axonal NF-H, inhibits radial growth, and results 87] Q. Zhu, M. Lindenbaum, F. Levavasseur, H. Jacomy, J.P. Julien, in neurofilamentous accumulation in motor neurons, J. Cell Biol. Disruption of the NF-H gene increases axonal microtubule content 130 (1995) 1413–1422. and velocity of neurofilament transport: relief of axonopathy [82] X.Y. Xie, J.N. Barrett, Membrane resealing in cultured rat septal resulting from the toxin beta,beta9-iminodipropionitrile (see
com-neurons after neurite transection: evidence for enhancement by ments), J. Cell Biol. 143 (1998) 183–193.
Ca(21)-triggered protease activity and cytoskeletal disassembly, J. [88] Q.Z. Zhu, S. Couillard-Despres, J.P. Julien, Delayed maturation of Neurosci. 11 (1991) 3257–3267. regenerating myelinated axons in mice lacking neurofilaments, Exp. [83] Z. Xu, L.C. Cork, J.W. Griffin, D.W. Cleveland, Increased expression Neurol. 148 (1997) 299–316.