CO
2enrichment enhances flag leaf senescence in barley due
to greater grain nitrogen sink capacity
A. Fangmeier
a,*, B. Chrost
b, P. Ho¨gy
a, K. Krupinska
caInstitut fu¨r Pflanzeno¨kologie der Justus-Liebig-Uni
6ersita¨t,Heinrich-Buff-Ring26-32 (IFZ),D-35392Gießen,Germany bBotanisches Institut der Uni
6ersita¨t zu Ko¨ln,Gyrhofstraße15,D-50923Ko¨ln,Germany cBotanisches Institut und Botanischer Garten der Christian-Albrechts-Uni
6ersita¨t,Olshausenstraße40,D-24098Kiel,Germany
Received 21 March 2000; received in revised form 4 July 2000; accepted 4 July 2000
Abstract
Senescence is a highly regulated process which is under genetic control. In monocarpic plants, the onset of fruit development is the most important factor initiating the senescence process. During senescence, a large fraction of plant nutrients is reallocated away from vegetative tissues into generative tissues. Senescence may therefore be regarded as a highly effective salvage mechanism to save nutrients for the offspring. CO2 enrichment, besides
increasing growth and yield of C3plants, has often been shown to accelerate leaf senescence. C3plants grown under
elevated CO2experience alterations in their nutrient relations. In particular their tissue nitrogen concentrations are
always lower after exposure to elevated CO2. We used a monocarpic C3crop — spring barley (Hordeum6ulgarecv.
Alexis) — grown in open-top field chambers to test the effects of CO2enrichment on growth and yield, on nitrogen
acquisition and redistribution, and on the senescence process in flag leaves, at two applications of nitrogen fertilizer. CO2enrichment (650 vs. 366mmol mol−1) caused an increase both in biomass and in grain yield by 38% (average
of the two fertilizer applications) which was due to increased tillering. Total nitrogen uptake of the crops was not affected by CO2treatment but responded solely to the N supply. Nitrogen concentrations in grains and straw were
significantly lower (−33 and −24%) in plants grown at elevated CO2. Phenological development was not altered by
CO2until anthesis. However, progress of flag leaf senescence as assessed by chlorophyll content, protein content and
content of large and small subunit of RubisCO and of cytochrome b559 was enhanced under elevated CO2
concentrations by 4 days. We postulate that CO2enhanced flag leaf senescence in barley crops by increasing the
nitrogen sink capacity of the grains. © 2000 Elsevier Science B.V. All rights reserved.
Keywords:Chlorophyll; Grain-filling;Hordeum6ulgare; Open-top field chambers; Proteins; Redistribution; Yield
www.elsevier.com/locate/envexpbot
1. Introduction
Leaf senescence is a complex and highly coordi-nated developmental process under genetic con-trol (Smart, 1994; Noode´n et al., 1997) which precedes cell death (Gan and Amasino, 1997; * Corresponding author. Tel.:+49-641-9935315; fax:+
49-641-9935309.
E-mail address: [email protected] (A. Fangmeier).
A.Fangmeier et al./En6ironmental and Experimental Botany44 (2000) 151 – 164 152
Chrost et al., 1999). Several attempts have been made to distinguish between different stages dur-ing the leaf senescence process, and somewhat different classifications and numbers of stages have been proposed. It has been generally assumed that regulatory genes are involved in the initiation of senescence. Later on, other senescence-associated genes (SAG’s) are expressed which are encoding proteins involved in breakdown of macromolecules and, thus, in mobilization of nutrients, such as RNAses, proteinases, and lipases (Gan and Amasino, 1997; Thompson et al., 1998). Another class of SAG’s expressed at later stages appears to be related to protective or stress response functions (Bleecker, 1998). Up to now, more than 30 SAG’s have been identified, cloned and characterized (Biswal and Biswal, 1999). Recently, it has also been shown that leaf senescence — at least at late stages — is accompanied by programmed cell death (Yen and Yang, 1998).
One of the most obvious events occurring during early senescence at the cellular level is the transfor-mation of chloroplasts into gerontoplasts (Smart, 1994; Noode´n et al., 1997). The thylakoids are disrupted quite early whereas the plastid envelope remains intact until final stages of senescence (Thompson et al., 1998; Chrost et al., 1999). The disassembly of the photosynthetic apparatus pro-ceeds in a highly regulated manner. In field-grown
barley (Hordeum6ulgare), flag leaf senescence was
first detectable by reduced photosynthetic capacity which was accompanied by decreasing D1 protein
(8 days after anthesis). About 6 days later,
pigment content, photosystem II efficiency, cy-tochrome f and the large subunit of
ribulose-1,5-bis-phosphate carboxylase/oxygenase (RubisCO)
started to decline, whereas the small subunit of RubisCO remained high until 22 days after anthesis (Humbeck et al., 1996).
During dissassembly of the chloroplasts, 70%
of the total nitrogen contained in the cells is remobilized and allocated to sink tissues with high nutrient demand (Smart, 1994; Gan and Amasino, 1997). In monocarpic plants, developing seeds or fruits are the most important sink for nutrients allocated from senescing leaves. In experiments with wheat, we found nitrogen concentrations in flag leaves to decrease by 78% from anthesis to
maturity, and phosphorous concentrations to de-crease by 82%. Correspondingly, at maturity the wheat grains contained 85% of total shoot nitrogen and 94% of total shoot phosphorus, respectively (Fangmeier et al., 1997).
In monocarpic species the onset of fruit develop-ment is the most important factor initiating the senescence process (Noode´n et al., 1997). In addi-tion the onset and progression of senescence is influenced by many external factors such as day-length or insufficient supply of resources (light, water, nutrients) (Smart, 1994; Kleber-Janke and Krupinska, 1997). The molecular mechanism of senescence initiation in relation to fruit develop-ment is not yet clear. Nutrient demand from the developing reproductive tissues might trigger the senescence process in leaves (Kelly and Davies, 1988). Other authors, however, postulate the exis-tence of a ‘death hormone’ initiating the senescence process (Noode´n and Leopold, 1978; Wilson, 1997). Whatever the signal transduction pathway is, senescence (at least in monocarpic species) may be regarded as a highly effective nutrient salvage mechanism for plants to transfer the nutrients to their offspring. It is likely that the nutrient salvage function of senescence is the primary reason for the evolution of such a complicated, multi-factorial process, and that the end of the life-cycle of the organs concerned is just an inevitable by-product (Bleecker, 1998).
Nutrient relations in plants are affected by the
concentration of atmospheric CO2 which
repre-sents the most important plant nutrient in the
biosphere (45% of plant dry matter is carbon).
Due to human activities disturbing the global
carbon cycle, atmospheric CO2 concentrations
have been increasing from 280 mmol mol−1 in
pre-industrial times to more than 360mmol mol−1
today, and a further increase to at least 450 or,
more likely, 700mmol mol−1by the end of the next
century appears inevitable (Anonymous, 1995).
Atmospheric CO2enrichment may be regarded as
a global fertilization of the biosphere with the most abundant plant nutrient. Elevated atmospheric
CO2 concentrations do not only promote growth
and biomass of plants (most effectively in C3
species, Poorter et al., 1996) but have also conse-quences for the demand for other nutrients
ratios in tissues of C3-plants have been shown to increase considerably (Conroy, 1992; Cotrufo et al., 1998). This is not caused by a simple ‘dilution’ due to higher carbohydrate concentra-tions, but is rather due to a decreased demand for
nitrogen in green tissues. Under CO2 enrichment,
optimization of resources within the photosyn-thetic apparatus may occur (Webber et al., 1994) since ribulose-bisphosphate- (RuBP) and
phos-phate- (Pi) regeneration rather than carboxylation
by RubisCO will limit the rate of CO2
assimila-tion (Harley and Sharkey, 1991). Decreased con-tent of RubisCO (Moore et al., 1999) which comprises up to 60% of soluble leaf protein (Ja-cob et al., 1995) will reduce total nitrogen demand of green tissues. Additionally, the depression of the photorespiratory pathway (approximetaly half
at doubled CO2 concentrations, Sharkey, 1988)
will also decrease the leaf nitrogen demand be-cause of smaller contents of enzymes of the glyco-late pathway (Webber et al., 1994; Fangmeier and Ja¨ger, 1998).
In previous studies with cereal crops, we could demonstrate that nitrogen uptake by the crops
was not affected by CO2 enrichment, but was
dependent on nitrogen supply (Fangmeier et al., 1997). At the same time, grain yield was
signifi-cantly increased under CO2 enrichment. We also
observed a faster progress of senescence, and an earlier remobilisation of proteins, in flag leaves of
wheat crops under CO2 enrichment (Vermehren
et al., 1998). Similar observations have been made by Sicher and Bunce (1998).
We assume that enhanced flag leaf senescence
during grain filling in cereals under CO2
enrich-ment may be triggered by the different effects of
elevated [CO2] on grain production (which is
thought to be increased) on the one hand, and on the acquisition and storage of nitrogen in vegeta-tive pools used during grain filling (which are thought to be not affected), on the other hand. By
this means, CO2 enrichment might accelerate
senescence via increased grain nutrient sink capac-ity. We also speculate that the effect of elevated
CO2 on flag leaf senescence will not be mitigated
by additional nitrogen fertilization as additional N will lead to increased biomass and yield rather than to higher nitrogen pools that would be
avail-able per unit grain yield.
To test these hypotheses, we used open-top field
chambers to expose spring barley (H. 6ulgare cv.
Alexis) crops to CO2 enrichment at two nitrogen
supplies and assessed the effects on growth and yield, on nitrogen acquisition and redistribution by the crops, and on the progress of senescence in barley flag leaves.
2. Materials and methods
2.1. Plant culture
On May 3, 1997, (Julian Date (JD) 123) spring
barley (H. 6ulgare L. cv. Alexis) was sown into
circular containers with a volume of 90 l (diame-ter 61 cm, depth 40 cm) which were placed in open-top field chambers (OTC). Sowing density
was 500 plants per m2. One week after seedling
emergence (JD 127) the stands were thinned to
250 plants per m2. Soil was taken from an
agricul-tural field site with vega (fluvisol) as prevailing soil type and mixed with sand 1:1 (vol:vol) to get
a substrate low in organic matter (B1%). The
crops were supplied with two levels of NPK-fertil-izer, also containing micronutrients,
correspond-ing to 140 kg N ha−1
ha−1on JD 140, 148, and 163, respectively, which
corresponded to growth stages (according to Tottman and Broad, 1987) 13, 22 and 34. OTC were equipped with a rain exclusion top. The plants were regularly irrigated using deionized water to avoid any drought stress. Green side shading panels, which reduced diffuse radiation by 50%, were raised as the crop grew. The pots were covered with white isolating panels to avoid high soil temperatures.
2.2. CO2 exposure
Plants were exposed to CO2for 24 h per day in
circular 3.15 m diameter OTC as described by Fangmeier et al. (1992), as soon as seedlings emerged (on JD 127) until canopy maturity (on
JD 231) at final harvest. CO2mean target
A.Fangmeier et al./En6ironmental and Experimental Botany44 (2000) 151 – 164 154
(elevated) mmol mol−1 (see Fig. 1). Air samples
for CO2 monitoring were taken 5 cm above the
top of the canopies. The air sampling lines were moved up as the canopies grew. Microclimatic conditions in OTC were monitored continuously throughout the season and logged as hourly means.
2.3. Assessments
2.3.1. Har6ests for biomass and yield estimations An inner circle of the canopies 50 cm in diame-ter was used for final harvest at maturity to gain biomass and yield data. Samples were separated into main stem ear, tiller ears, and straw (leaves and stems). Because appropriate sampling of roots from cereal crops is extremely time-consum-ing and only restricted man-power was available, roots were not harvested. Straw samples were dried at 70°C, and ears at 35°C, until weight constancy. Ears were threshed to estimate yield, grain number, thousand grain weight, and glume weight, separately for main stem ears and for tiller ears. Nitrogen concentrations were estimated in these samples by Kjeldahl.
2.3.2. Chlorophyll content
Chlorophyll contents of flag leaves were esti-mated non-destructively at the mid position of flag leaf blades using a SPAD 502 chlorophyll meter (Minolta, Japan) on dates when, and just before, flag leaf harvests took place (see below).
2.3.3. Har6ests for estimations of protein
degradation during flag leaf senescence
Flag leaf harvesting and SPAD measurements took place on 7 occasions over a period of 20 days from full maturity of flag leaves (JD 189) until nearly complete senescence (JD 209). Flag leaf ligules were visible on JD 166 (growth stage 39, Tottman and Broad, 1987), and anthesis (growth stage 65) was on JD 178 in all treatments. Twenty two border plants per pot, growing out-side the 50 cm diameter circle, were used for the intermediate harvests. Only plants from high nu-trient supply treatments were analysed. Harvests were done between 1:00 and 2:00 p.m. Then, fresh weight and length of flag leaves were measured immediately and samples were frozen in liquid nitrogen within one minute after harvest. Samples
were stored at −80 °C until analysis.
Data on leaf blade length were used to calculate leaf area by regression analysis of 30 flag leaves harvested from sparse plants in ambient plots.
Best fit (adjusted R2
=0.982) was achieved using
the formula:
y=0.02944+0.2907x+0.03651x2
wherexis the length of flag leaf blade (cm) andy
is the leaf area (cm2).
Proteins were extracted from flag leaf samples as described earlier (Humbeck et al., 1996). Protein concentrations were estimated using the
method of Lowry et al. (1951). Aliquots of 30mg
protein were subjected to SDS – PAGE, and
incu-bated with antibodies directed towards
cy-tochrome b559 (Cyt b559), and large subunit (LSU) and small subunit (SSU) of RubisCO. Gel photographs were analysed for protein contents using a Kodak Electrophoresis Documentation and Analysis System 120 in order to yield relative contents of Cyt b559, LSU, and SSU, based on flag leaf area.
Fig. 1. (A) Daily mean air temperature and cumulative daily global radiation, and (B) CO2concentrations (daily means) in
2.4. Statistical design and data e6aluation
CO2 treatments were carried out in three
repli-cate OTC, respectively. For each nutrient supply, one barley pot was exposed within each OTC. OTC means served as input data for analysis of
variance, usingSPSS 8.0 for Windows (SPSS Inc.,
Chicago). Biomass and nitrogen data represent
CO2 treatment means9standard deviation based
on three chamber replicates.
SPAD measurements were taken both on plants in the inner circle left for final harvest and on border plants. There were no significant differ-ences between these data-sets. SPAD data given for any particular date represent means of eight to 15 single measurements per OTC and each nutri-ent supply. Standard deviation bars shown in the graph were calculated using means of three repli-cate OTC, respectively.
Because of restricted number of border plants available for intermediate flag leaf harvests, no harvests in replicate OTC could be carried out. Rather, at any harvest date four flag leaves taken from all three replicate OTC were combined to yield one sample.
3. Results
3.1. CO2 concentrations and climatic conditions
Seasonal CO2 daily averages measured on top
of the canopy from barley crop emergence (on JD
127) to final harvest (on JD 231) were 36690.9
mmol mol−1 in ambient OTC and 650920 mmol
mol−1 in elevated OTC (Fig. 1). Since no
feed-back control was used in the CO2-dispensing
sys-tem, concentrations in OTC with elevated CO2
were somewhat influenced by ambient wind speed. With high ambient windspeeds, air in OTC
en-riched with CO2 was mixed with intruding
ambi-ent air which caused CO2 concentrations to fall.
Fig. 1 also presents daily averages for air tem-perature and global radiation monitored in OTC. Average temperature during crop growth (JD 127 to 231) was 19.8°C, cumulative temperature (at baseline of 0°C) amounted 2079 day-degrees, and
cumulative radiation sum was 1103 MJ m−2.
Average vapour pressure deficit (VPD) in OTC
during daylight hours (global radiation \50 W
m−2) from crop emergence to final harvest was
15.04 hPa (no VPD data shown in Fig. 1).
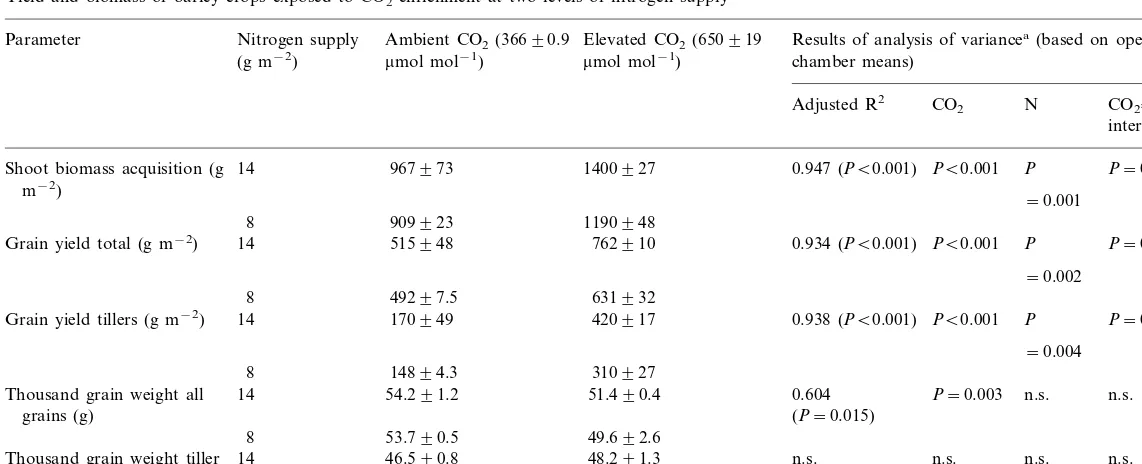
3.2. Treatment effects on biomass and yield
Barley crops grown under CO2 enrichment
ac-quired 38% more shoot biomass than those grown
under ambient CO2 concentrations (Table 1).
Crops fertilized with 14 g N m−2
increased shoot
biomass by 13% compared with 8 g N m−2
. CO2
enrichment and nitrogen supply interacted
posi-tively, i.e. biomass increase due to CO2
enrich-ment was larger at high N supply than at low N supply (45 vs. 30%) and, vice versa, additional nitrogen had greater impact on shoot biomass at
650 mmul mol−1 CO2 than at ambient CO2
(in-crease 18 vs. 7%).
Treatment effects on total grain yield were sim-ilar to shoot biomass responses. Correspondingly, there were no significant treatment effects on
har-vest index. Increases in grain yield due to CO2
enrichment and additional nutrient supply were solely attributable to increased tillering. Neither
CO2 enrichment nor additional fertilization had
any significant effect on grain yield from main stems. However, yield from tiller ears was
en-larged 2.3-fold under 650mmol mol−1CO2.
Addi-tional fertilization increased tiller grain yield by
29%. As for shoot biomass and total yield, CO2
enrichment and nitrogen supply interacted posi-tively on tiller grain yield.
Thousand grain weight (TGW) of main stem
grains was slightly reduced by CO2 enrichment
(−7.5%). This caused also a decline in the
aver-age TGW for all grains. There was no such effect on grains from tiller ears. Fertilization did not affect TGW. Thus, the sink capacity for carbon of one single grain was only slightly affected by the treatments.
3.3. Treatment effects on nitrogen acquisition
In contrast to the effects on shoot biomass and
on yield, CO2enrichment did not affect the
A
.
Fangmeier
et
al
.
/
En
6
ironmental
and
Experimental
Botany
44
(2000)
151
–
164
156
Table 1
Yield and biomass of barley crops exposed to CO2 enrichment at two levels of nitrogen supply
Elevated CO2 (650919
Ambient CO2 (36690.9
Nitrogen supply Results of analysis of variancea(based on open-top field
Parameter
chamber means)
mmol mol−1) mmol mol−1)
(g m−2)
N CO2N Adjusted R2 CO
2
interaction
PB0.001 P 1400927
967973 P=0.021 14
Shoot biomass acquisition (g 0.947 (PB0.001) m−2)
=0.001 8 909923 1190948
P
14 0.934 (PB0.001) PB0.001 P=0.013 Grain yield total (g m−2) 515948 762910
=0.002 49297.5 631932
8
P
Grain yield tillers (g m−2) 14 170949 420917 0.938 (PB0.001) PB0.001 P=0.032
=0.004 8 14894.3 310927
51.490.4 0.604 P=0.003 n.s. n.s. Thousand grain weight all 14 54.291.2
grains (g) (P=0.015) 8 53.790.5 49.692.6
n.s. n.s. n.s. n.s.
Thousand grain weight tiller 14 46.590.8 48.291.3 grains (g)
8 44.692.7 46.592.2
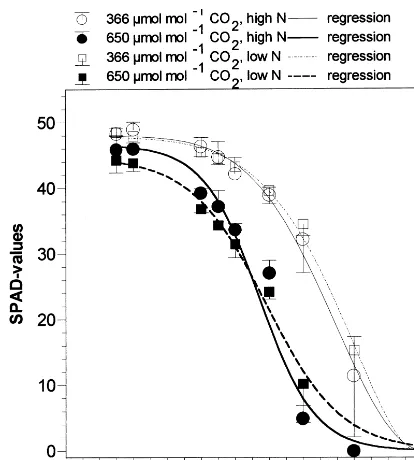
Fig. 2. Chlorophyll breakdown (assessed as SPAD values) during flag leaf senescence in spring barley crops exposed to ambient or elevated CO2 at two levels of nitrogen supply.
Data represent means9standard deviation from three repli-cate OTC. Regression lines were calculated using logistic func-tions.
with 8 g N m−2contained 11.2 g N m−2in their
shoots. Thus, not only nitrogen from fertilization but also from pools in the soil and from mineral-ization served as sources for the crops. Crops
provided with 14 g N m−2contained less nitrogen
in the shoots (13.3 g m−2) than was supplied as
mineral fertilizer. Most of the nitrogen found in the shoots at final harvest was allocated to the grains which contained 77% of total shoot N on average. This nitrogen allocation pattern was not influenced by the treatments (Table 2).
Nitrogen concentrations (expressed as % of dry weight) both in straw and in grain samples were not significantly affected by nutrient supply. This suggests that no nitrogen deficiency occurred in
crops provided with only 8 g N m−2. CO
2
enrich-ment, on the other hand, reduced the nitrogen concentration of the straw by 34% and that of grains by 24%. Since nitrogen concentrations in grains from main stems and from tillers, and treatment effects on it, were nearly identical, only average grain nitrogen concentrations are pre-sented in Table 2. As a consequence of decreased nitrogen concentrations in the tissues, the nitro-gen use efficiency of barley crops was improved
by 35% due to CO2 enrichment.
3.4. Treatment effects on flag leaf senescence
3.4.1. Chlorophyll breakdown
Chlorophyll content was assessed non-destruc-tively during flag-leaf senescence using a Minolta SPAD meter. SPAD values obtained from JD 191 to 209 are shown in Fig. 2. Assessments started right before chlorophyll breakdown began. Before breakdown, chlorophyll content in crops grown
under elevated CO2 were significantly lower than
under ambient CO2(ANOVA results not shown).
However, no significant effects of nutrient supply on chlorophyll degradation were observed. Pro-gress of chlorophyll breakdown was significantly
enhanced by CO2 enrichment. In crops grown at
650 mmol mol−1CO2, chlorophyll degradation to
50% of the maximum values was already achieved on JD 199, whereas it took until JD 203 to get the
same degree of degradation at ambient CO2.
found in barley shoots was 13.2 g m−2at ambient
CO2 and 13.4 g m
−2 at elevated CO
2 when the
plants had high nutrient supply (Table 2). At low
fertilization, the shoots contained 11.2 g m−2
nitrogen (ambient CO2) or 11.3 g m
−2
nitrogen
(elevated CO2). The amount of nitrogen in the
grains of the crops at maturity was 9.83 (ambient
CO2) and 10.7 g m
−2
(elevated CO2) at high N
supply, and 8.60 and 8.75 g m−2at low N supply,
respectively, without any significant impact of
CO2enrichment. However, nitrogen acquisition in
grains on main stems decreased by 26% under
CO2 enrichment corresponding to the decrease in
nitrogen concentration in grains (see below). Ni-trogen acquisition by tiller grains increased by
75% when the plants were grown at 650 mmol
mol−1
CO2 which results both from the 2.3-fold
increase in tiller grain yield, and the 26% decrease
in grain nitrogen concentration, at elevated CO2.
A
.
Fangmeier
et
al
.
/
En
6
ironmental
and
Experimental
Botany
44
(2000)
151
–
164
158
Table 2
Nitrogen acquisition of barley crops exposed to CO2enrichment at two levels of nitrogen supply
Parameter Nitrogen supply Ambient CO2 Elevated CO2 Results of analysis of variancea(based on open-top field
(650919mmol
(36690.9mmol
(g m−2) chamber means)
mol−1)
mol−1)
CO2 N CO2N
Adjusted R2
interaction
n.s. PB0.001 n.s. 13.490.2
14 13.290.4 0.826 (P=0.001) Shoot nitrogen acquisition (g
m−2)
8 11.290.8 11.390.3
0.44090.017 0.787 (P=0.001) PB0.001 n.s. n.s. Straw nitrogen content (% dry 14 0.73790.100
weight)
0.62790.070
8 0.46790.029
PB0.001 n.s. n.s. 0.865 (PB0.001)
14
Main stem grain nitrogen 6.6290.49 4.8990.32 acquisition (g m−2)
8 6.1290.30 4.5390.22
1.4090.04 0.904 (PB0.001) PB0.001 n.s. n.s. Grain nitrogen content (% dry 14 1.9290.10
weight)
8 1.7590.11 1.3990.02
PB0.001 n.s. n.s. 0.932 (PB0.001)
Nitrogen use efficiency (g DM (g 14 73.493.6 104.093.6 N)−1)
8 81.395.6 104.591.4
3.4.2. Time course of soluble protein content
Time-course of total soluble proteins in barley flag leaves (Fig. 3) was very similar to that of chlorophyll. Concentrations were in general lower
when the plants were grown under CO2
enrich-ment. Under ambient CO2, a reduction of protein
contents to 50% of maximum was achieved on JD
197.5, whereas at 650 mmol mol−1 CO2 protein
content was reduced to 50% already on JD 196. Thus, protein breakdown appeared to be
acceler-ated by CO2 enrichment, but was somewhat less
affected than chlorophyll disassembling.
3.4.3. Degradation of RubisCO LSU, SSU, and cytochrom b559
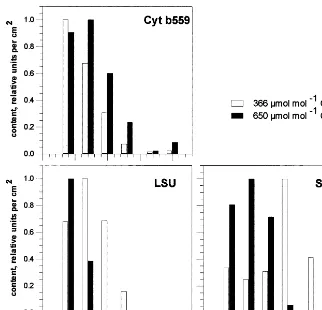
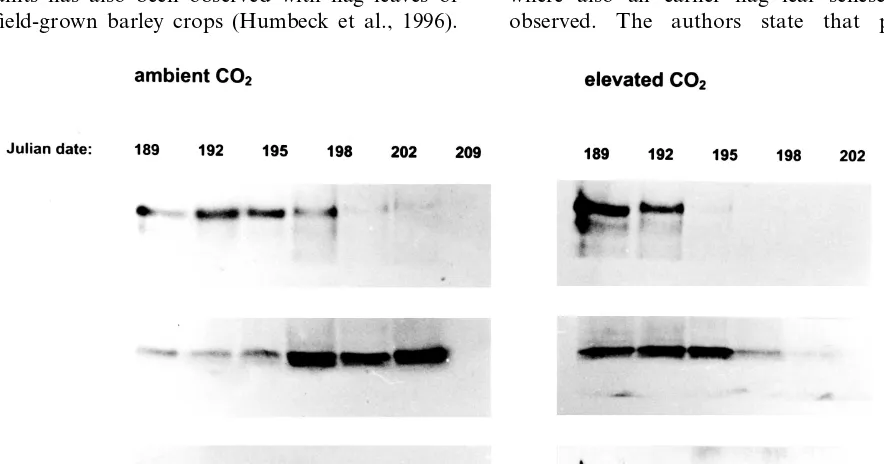
LSU breakdown was clearly accelerated under
CO2enrichment (Figs. 4 and 5). No LSU could be
detected any longer on JD 195, and afterwards, in
plants grown under 650 mmol mol−1 CO2,
whereas flag leaves from plants grown under
am-bient CO2 still had nearly maximum LSU
con-tents at that time. Decline of the SSU level differed signficantly from that of LSU. At
ele-vated CO2 concentrations, degradation of SSU
started only after JD 192. On JD 198, there was
still some SSU (6% of maximum contents)
de-tectable. At ambient CO2, no clear degradation of
SSU towards the end of the sampling period could be observed. Cyt b559 was longer detectable than LSU, but its level started to decline some-what earlier than the level of SSU. Opposite to LSU and SSU, levels of Cyt b559 appeared to
decline somewhat more rapidly at ambient CO2
than at elevated CO2.
4. Discussion
Biomass and yield of barley crops increased by
38% when grown under 650 instead of 366 mmol
mol−1 CO
2 which resulted solely from an
en-hancement of tiller production and tiller survival.
This CO2-fertilization is lower than reported in
previous studies. Poorter et al. (1996) compiled
data from various CO2 enrichment experiments
and cite one study for H. 6ulgare where an
in-crease in biomass of 104% occurred. However, only plants that were not in the reproductive phase were included into that data compilation. Thus, limitations occurring at later stages of de-velopment (nutrients, competition for light) were not included in the evaluation. In a season-long
study under CO2 enrichment, Weigel et al. (1994)
found increases in spring barley yield of up to 89%. However, these authors used plants in small pots with a low plant density. This might cause an
over-estimation of CO2 effects due to a lack of
LAI limitations (Ko¨rner, 1995). The results pre-sented in this study were obtained under condi-tions closely resembling field condicondi-tions. Thus, the 38% increase in yield appears to be realistic.
Plants from the high fertilizer treatment were not subjected to nitrogen limitation, as (1) there was no increase in tissue nitrogen concentrations when nitrogen supply was increased from 8 to 14
g N m−2
(compare Table 2), (2) the crops
fertil-ized with 14 g N m−2did not take up all of the N
applied (total nitrogen amount in the crop at final
harvest: 13.3 g m−2), whereas crops receiving
only 8 g N m−2had a higher nitrogen content on
an area basis (11.2 g m−2) than was applied. In
spite of sufficient nitrogen being available (at least
in the high N fertilizer treatment), CO2
Fig. 3. Concentrations of total soluble protein during flag leaf senescence in spring barley crops exposed to ambient or elevated CO2 and fertilized with 140 kg N ha−1. Data are
A.Fangmeier et al./En6ironmental and Experimental Botany44 (2000) 151 – 164 160
Fig. 4. Contents of different proteins (relative units per cm2leaf area) during flag leaf senescence in spring barley crops exposed to
ambient or elevated CO2and fertilized with 140 kg N ha−
1. Data are from four flag leaves randomly selected from three replicate
OTC at each harvest date, respectively. LSU, large subunit of RubisCO; SSU, small subunit of RubisCO; Cyt b559, cytochrome b559.
enrichment significantly depressed nitrogen con-centrations found in the tissues. In a recent data
compilation, CO2 enrichment has been found to
decrease the nitrogen concentration in green
leaves of non-woody C3 plants by 17%, and in
litter by 9% (Cotrufo et al., 1998). We found even greater reductions (40% in straw at high nitrogen supply, 27% in grains) which compares well to earlier findings in cereals (McKee and Woodward, 1994; Manderscheid et al., 1995; Fangmeier et al., 1997).
Several hypotheses have been proposed to ex-plain the reduced tissue nitrogen concentrations
under CO2 enrichment (Conroy and Hocking,
1993). The most likely candidates to explain these findings are: (1) an optimisation of the
photosyn-thetic apparatus in plants grown under high CO2
concentrations, by which less nitrogen is invested in RubisCO and more nitrogen is allocated to
RuBP-regeneration and to Pi-regeneration (Sage
et al., 1989; Webber et al., 1994; Moore et al., 1999) though this type of acclimation does not always take place and is less obvious in plants well supplied with nitrogen (Theobald et al., 1998; Stitt and Krapp, 1999), and (2) a reduction of the photosynthetic carbon oxidation (PCO) pathway, and thereby a reduction of the requirement for
PCO enzymes, under altered CO2/O2partial
et al., 1999), and of PCO enzymes (Fangmeier and Ja¨ger, 1998) has often been observed in plant
leaves grown at elevated CO2.
The phenological development of the barley crops before grain filling was not responsive to
CO2 enrichment. Dates of plant emergence, of
terminal spikelet formation, of flag leaf appear-ance and of anthesis were identical at either treat-ment. Nevertheless, flag leaf senescence was
significantly affected by CO2 enrichment.
En-hancement of flag leaf senescence was detectable from both estimations of chlorophyll degradation and of the contents of soluble proteins. The latter estimations suggest an earlier protein breakdown and re-allocation away from the leaves. Among the three plastid proteins assessed in our study, the amounts of the LSU of RubisCO declined first. Contents of Cyt b559 clearly declined later than LSU. SSU kept rather high amounts until the end of the harvest period. On JD 205, i.e. 27 days after anthesis, still 31% of maximum SSU
contents were detected at ambient CO2. The lack
of coordination in the decline of RubisCO sub-units has also been observed with flag leaves of field-grown barley crops (Humbeck et al., 1996).
CO2 enrichment accelerated the decline of both
LSU and SSU contents by approximately four days. In contrast, the decline of Cyt b559
ap-peared to be somewhat delayed under CO2
enrich-ment. This might be related to a possible
acclimation effect of CO2enrichment on the
pho-tosynthetic apparatus. However, the data are to scarce to draw further conclusions.
Earlier senescence under CO2 enrichment has
been shown in many other studies with annual C3
crops. Miller et al. (1997) exposed tobacco to 350
or 950 mmol mol−1 CO2 and followed leaf CO2
exchange over the course of leaf development. The authors observed an earlier achievement of
maximum photosynthesis rates at elevated CO2
concentrations, but also a faster progress of devel-opment and an earlier beginning of senescence. From their data, Miller et al. (1997) conclude that
faster leaf development under CO2 enrichment
might explain the often observed photosynthetic
acclimation to elevated CO2. This statement is in
part supported by findings from wheat exposure
to CO2 enrichment (Sicher and Bunce, 1998)
where also an earlier flag leaf senescence was observed. The authors state that premature
A.Fangmeier et al./En6ironmental and Experimental Botany44 (2000) 151 – 164 162
senescence contributed to decreased
photosyn-thetic rates at elevated CO2concentrations. There
is further experimental evidence for a faster de-crease of photosynthetic properties and of en-zymes of the photosynthetic apparatus as a result
of CO2 exposure. Sicher and Bunce (1997) found
decreased RubisCO contents and photosynthesis rates, and an acceleration of senescence, in wheat
and barley leaves grown in elevated CO2, and
Garcia et al. (1998) observed a decline in flag leaf photosynthesis and an earlier senescence at final stages of wheat crop development under free air
CO2 enrichment.
We do not believe that a general enhancement of leaf development and, thus, an earlier onset of leaf senescence can be used as a simple explana-tion of photosynthetic down-regulaexplana-tion due to
CO2 enrichment. Functional and molecular
changes, and the redistribution of nutrients, oc-curring during senescence are too complex for such a simple explanation. Rather, one must search for a signal triggering senescence which is
affected due to growth at elevated CO2
concentrations.
In monocarpic annual species like barley or wheat, a rather rapid transition takes place from a
vegetative ‘green‘ plant acquiring CO2 from the
atmosphere and other nutrients from the soil, to a ‘generative‘ plant which does not acquire soil nutrients at significant amounts any longer be-cause of the breakdown of the root system (Fangmeier et al. 1996), but only redistributes these from vegetative tissues to the generative organs, i.e. to the grains, and which gradually reduces carbon acquisition from the atmosphere.
As stated above, CO2 enrichment reduces the
nitrogen demand of green tissues by several mech-anisms, causing an increase in nitrogen use effi-ciency. In our study (and in hardly any previous
experiment with C3 plants), growth at elevated
CO2 concentrations resulted in higher biomass
(Poorter et al., 1996) and, finally, in greater yield, irrespective of possible down-regulation of the photosynthetic apparatus at leaf level. For the whole plant, yield increase of the barley crops occurred due to increased tillering. Because of
increased nitrogen use efficiency under CO2
en-richment, nitrogen acquisition of the crops did
not keep pace with carbon gain. Rather, crop nitrogen uptake was dependent on nitrogen
sup-ply but did not respond to CO2 treatment. In
cereals, nearly all the nitrogen resources required for grain filling originate from vegetative tissues and there is hardly any further uptake of nitrogen from the soil after anthesis (Van Kraalingen, 1990; Fangmeier et al., 1999). Thus, the grains in
plants grown at elevated CO2 concentrations,
al-though they had 48% more biomass (at 140 kg
ha−1 of N fertilization), had to cope with the
same amount of nitrogen in vegetative pools as
the grains from ambient CO2. There is no reason
to assume that CO2 enrichment reduces the
nitro-gen demand of grains, since none of the mecha-nisms reducing nitrogen demand in green tissues is working in grains. Rather, for optimal grain vitality and (ecological) quality, a certain amount of grain proteins is required and monocarpic plants in particular have been selected to achieve this protein content in order to ensure survival of the population.
Thus, we believe that the higher nitrogen sink capacity of the growing population of grains
un-der CO2 enrichment works as a trigger to induce
nitrogen release from the leaves and can explain the earlier senescence of barley flag leaves ob-served in our study. Nitrogen deficiency has been proved to induce senescence in previous studies (Smart, 1994; Noode´n et al., 1997). In this case,
CO2 enrichment works as a tool to induce
nitro-gen deficiency as ’seen‘ by the grains in late developmental stages, though it does not cause a true deficiency in green tissues, in spite of lowered concentrations, at earlier developmental stages when the leaves act as carbon sources.
5. Conclusions
Our findings explain the acceleration of
senes-cence in annual monocarpic C3plants under CO2
enrichment as a process driven by the differing
effects of CO2on nutrients, in particular nitrogen,
in vegetative and generative tissues. We postulate
that CO2 enhances flag leaf senescence in barley
crops (and probably leaf senescence in
mono-carpic C3 species in general) by a sequence of
several processes induced by CO2: (1) CO2reduces
the nitrogen demand of green tissues by alter-ations of the photosynthetic apparatus, i.e. lower RubisCO contents, lower contents of enzymes of the PCO-cycle. Thus, nitrogen uptake does not keep pace with carbon acquisition during
vegeta-tive growth. (2) CO2 increases the number of
diaspores. Thus, the sink size for nutrients during seed ripening is enlarged. (3) During grain filling (or seed ripening in general) there is a higher demand for nutrient redistribution to the seeds
since CO2 enrichment does not reduce the
nitro-gen demand of the diaspores. Thus, a nitronitro-gen deficiency is induced during the switch from vege-tative to generative growth. (4) This deficiency acts as a trigger to induce leaf senescence in order to release nutrients from vegetative tissues accord-ing to the nutrient salvage function of senescence, although the demand of the seeds can not be accomplished due to restricted nutrient pools.
References
Anonymous, 1995. Climate Change 1995. The Science of Climate Change. Summary for Policymakers. World Mete-orological Organization/United Nations Environment Pro-gramme, pp. 1 – 56.
Biswal, B., Biswal, U.C., 1999. Leaf senescence: physiology and molecular biology. Curr. Sci. 77, 775 – 782.
Bleecker, A.B., 1998. The evolutionary basis of leaf senes-cence: method to the madness? Curr. Opin. Plant Biol. 1, 73 – 78.
Chrost, B., Falk, J., Kernebeck, B., Mo¨lleken, H., Krupinska, K., 1999. Tocopherol biosynthesis in senescing chloroplasts — a mechanism to protect envelope membranes against oxidative stress and a prerequisite for lipid remobilization? In: Argyroudi-Akoyunoglou, J.H., Senger, H. (Eds.), The Chloroplast: from Molecular Biology to Biotechnology. Kluwer Academic Publishers, Dordrecht, pp. 171 – 176.
Conroy, J.P., 1992. Influence of elevated atmospheric CO2
concentrations on plant nutrition. Aust. J. Bot. 40, 445 – 456.
Conroy, J.P., Hocking, P.J., 1993. Nitrogen nutrition of C3
plants at elevated atmospheric CO2concentrations.
Phys-iol. Plant. 89, 570 – 576.
Cotrufo, M.F., Ineson, P., Scott, A., 1998. Elevated CO2
reduces the nitrogen concentration of plant tissues. Global Change Biol. 4, 43 – 54.
Fangmeier, A., Ja¨ger, H.-J., 1998. CO2 enrichment, ozone,
nitrogen fertilizer and wheat: physiological background of growth and yield responses. In: De Kok, L.J., Stulen, I. (Eds.), Responses of Plant Metabolism to Air Pollution and Global Change. Backhuys Publishers, Leiden, pp. 299 – 304.
Fangmeier, A., Stein, W., Ja¨ger, H.-J., 1992. Advantages of an open-top chamber plant exposure system to assess the impact of atmospheric trace gases on vegetation. Angew. Bot. 66, 97 – 105.
Fangmeier, A., Gru¨ters, U., Hertstein, U., Sandhage-Hof-mann, A., Vermehren, B., Ja¨ger, H.-J., 1996. Effects of elevated CO2, nitrogen supply and tropospheric ozone on
spring wheat. I. Growth and yield. Environ. Pollut. 91, 381 – 390.
Fangmeier, A., Gru¨ters, U., Ho¨gy, P., Vermehren, B., Ja¨ger, H.-J., 1997. Effects of elevated CO2, nitrogen supply and
tropospheric ozone on spring wheat — II. Nutrients (N, P, K, S, Ca, Mg, Fe, Mn, Zn). Environ. Pollut. 96, 43 – 59. Fangmeier, A., De Temmerman, L., Mortensen, L., Kemp, K.,
Burke, J.I., Mitchell, R.A.C., Van Oijen, M., Weigel, H.-J., 1999. Effects on nutrients and on grain quality in spring wheat crops grown under elevated CO2concentrations and
stress conditions in the European, multiple-site experiment ‘ESPACE-wheat’. Eur. J. Agron. 10, 215 – 229.
Gan, S., Amasino, R.M., 1997. Making sense of senescence — molecular genetic regulation and manipulation of leaf senescence. Plant Physiol. 113, 313 – 319.
Garcia, R.L., Long, S.P., Wall, G.W., Osborne, C.P., Kimball, B.A., Nie, G.Y., Pinter, P.J., Lamorte, R.L., Wechsung, F., 1998. Photosynthesis and conductance of spring-wheat leaves: field response to continuous free-air atmospheric CO2 enrichment. Plant Cell Environ. 21, 659 – 669.
Harley, P.C., Sharkey, T.D., 1991. An improved model of C3
photosynthesis at high CO2 — reversed O2 sensitivity
explained by a lack of glycerate reentry into the chloro-plast. Photosynth. Res. 27, 169 – 178.
Humbeck, K., Quast, S., Krupinska, K., 1996. Functional and molecular changes in the photosynthetic apparatus during senescence of flag leaves from field-grown barley plants. Plant Cell Environ. 19, 337 – 344.
Jacob, J., Greitner, C., Drake, B.G., 1995. Acclimation of photosynthesis in relation to Rubisco and nonstructural carbohydrate contents and in situ carboxylase activity in Scirpus olneyigrown at elevated CO2in the field. Plant Cell
Environ. 18, 875 – 884.
A.Fangmeier et al./En6ironmental and Experimental Botany44 (2000) 151 – 164 164
Kleber-Janke, T., Krupinska, K., 1997. Isolation of cDNA clones for genes showing enhanced expression in barley leaves during dark-induced senescence as well as during senescence under field conditions. Planta 203, 332 – 340. Ko¨rner, C., 1995. Towards a better experimental basis for
upscaling plant responses to elevated CO2 and climate
warming. Plant Cell Environ. 18, 1101 – 1110.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J., 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265 – 275.
Manderscheid, R., Bender, J., Ja¨ger, H.-J., Weigel, H.-J., 1995. Effects of season long CO2 enrichment on cereals. II.
Nutrient concentrations and grain quality. Agric. Ecosyst. Environ. 54, 175 – 185.
McKee, I.F., Woodward, F.I., 1994. CO2enrichment responses
of wheat: interactions with temperature, nitrate and phos-phate. New Phytol. 127, 447 – 453.
Miller, A., Tsai, C.H., Hemphill, D., Endres, M., Rodermel, S., Spalding, M., 1997. Elevated CO2 effects during leaf
on-togeny — a new perspective on acclimation. Plant Physiol. 115, 1195 – 1200.
Moore, B.D., Cheng, S.-H., Sims, D., Seeman, J.R., 1999. The biochemical and molecular basis for photosynthetic acclima-tion to elevated atmospheric CO2. Plant Cell Environ. 22,
567 – 582.
Noode´n, L.D., Leopold, A.C., 1978. Phytohormones and the endogeneous regulation of senescence and abscission. In: Letham, D.S, Goodwin, P.B., Higgins, T.J.V. (Eds.), Phyto-hormones and Related Compounds: a Comprehensive Trea-tise. Elsevier, Amsterdam, pp. 329 – 369.
Noode´n, L.D., Guiamet, J.J., John, I., 1997. Senescence mech-anisms. Physiol. Plant. 101, 746 – 753.
Poorter, H., Roumet, C., Campbell, B.D., 1996. Interspecific variation in the growth response of plants to elevated CO2:
a search for functional types. In: Ko¨rner, C., Bazzaz, F.A. (Eds.), Carbon Dioxide, Populations, and Communities. Academic Press, San Diego, pp. 375 – 412.
Sage, R.F., Sharkey, T.D., Seemann, J.R., 1989. Acclimation of photosynthesis to elevated CO2 in five C3 species. Plant
Physiol. 89, 590 – 596.
Sharkey, T.D., 1988. Estimating the rate of photorespiration in leaves. Physiol. Plant. 73, 147 – 152.
Sicher, R.C., Bunce, J.A., 1997. Relationship of photosynthetic acclimation to changes of Rubisco activity in field-grown
winter wheat and barley during growth in elevated carbon dioxide. Photosynth. Res. 52, 27 – 38.
Sicher, R.C., Bunce, J.A., 1998. Evidence that premature senescence affects photosynthetic decline of wheat flag leaves during growth in elevated carbon dioxide. Int. J. Plant Sci. 159, 798 – 804.
Smart, C.M., 1994. Gene expression during leaf senescence. New Phytol. 126, 419 – 448.
Stitt, M., Krapp, A., 1999. The interaction between elevated carbon dioxide and nitrogen nutrition: the physiological and molecular background. Plant Cell Environ. 22, 583 – 621. Theobald, J.C., Mitchell, R.A.C., Parry, M.A.J., Lawlor, D.W.,
1998. Estimating the excess investment in ribulose-1,5-bis-phosphate carboxylase/oxygenase in leaves of spring wheat grown under elevated CO2. Plant Physiol. 118, 945 – 955.
Thompson, J.E., Froese, C.D., Madey, E., Smith, M.D., Hong, Y.W., 1998. Lipid metabolism during plant senescence. Prog. Lipid Res. 37, 119 – 141.
Tottman, D.R., Broad, H., 1987. The decimal code for the growth stages of cereals, with illustrations. Ann. Appl. Biol. 110, 441 – 454.
Van Kraalingen, D.W.G., 1990. Effects of CO2enrichment on
nutrient-deficient plants. In: Goudriaan, J., Van Keulen, H., Van Laar, H.H. (Eds.), The Greenhouse Effect and Primary Productivity in European Agroecosystems. Pudoc, Wa-geningen, pp. 42 – 45.
Vermehren, B., Fangmeier, A., Ja¨ger, H.-J., 1998. Influnce of elevated CO2on nitrogen economy of wheat. In: Peter, D.,
Maracchi, G., Ghazi, A. (Eds.), Climate Change Impact on Agriculture and Forestry. European Commission, Brussels, pp. 497 – 505.
Webber, A.N., Nie, G.Y., Long, S.P., 1994. Acclimation of photosynthetic proteins to rising atmospheric CO2.
Photo-synth. Res. 39, 413 – 425.
Weigel, H.J., Manderscheid, R., Ja¨ger, H.-J., Mejer, G.J., 1994. Effects of season-long CO2enrichment on cereals. I. Growth
performance and yield. Agric. Ecosyst. Environ. 48, 231 – 240.
Wilson, J.B., 1997. An evolutionary perspective on the ‘death hormone’ hypothesis in plants. Physiol. Plant. 99, 511 – 516. Yen, C.H., Yang, C.H., 1998. Evidence for programmed cell death during leaf senescence in plants. Plant Cell Physiol. 39, 922 – 927.