B
RIEF
R
EPORTS
CSF Oxytocin and Vasopressin Levels after Recovery
from Bulimia Nervosa and Anorexia Nervosa,
Bulimic Subtype
Guido K. Frank, Walter H. Kaye, Margaret Altemus, and Catherine G. Greeno
Background: When ill, people with eating disorders have
disturbances of the neuropeptides vasopressin and
oxytocin.
Methods: To avoid the confounding effects of the ill state,
we studied women who were recovered (more than 1 year,
normal weight, and regular menstrual cycles, no bingeing
or purging) from bulimia nervosa (rBN) or binge eating/
purging–type anorexia nervosa (rAN-BN), and matched
healthy control women.
Results: Vasopressin was elevated in rAN-BN and showed
a trend towards elevation in rBN. In rBN, elevated
cerebrospinal fluid vasopressin may be related to having a
lifetime history of major depression. In comparison,
cere-brospinal fluid oxytocin was normal in recovered subjects,
but elevated levels in some rBN might be related to birth
control pill use.
Conclusions: These data confirm and extend the
possibil-ity that elevated cerebrospinal fluid vasopressin may be
related to the pathophysiology of eating disorders, and/or
a lifetime history of major depression. Biol Psychiatry
2000;48:315–318 © 2000 Society of Biological Psychiatry
Key Words: Anorexia nervosa, bulimia nervosa,
oxyto-cin, vasopressin, major depression, obsessive– compulsive
disorder
Introduction
B
ulimia nervosa (BN), a disorder of unknown etiology
that has its onset in young women, is characterized by
binge eating followed by compensatory methods to
pre-vent weight gain (American Psychiatric Association
1994). Most commonly, women with BN maintain a
normal weight. Bulimic symptoms also occur less
com-monly in people with the binge eating/purging type of
anorexia nervosa (AN-BN). People who are ill with BN
and AN-BN have a similar range of comorbid symptoms
(Garfinkel et al 1995) and a wide variety of
neuroendo-crine disturbances (Kaye et al 1998a), which could be a
consequence of central nervous system neuropeptide
dys-regulation. For example, altered concentrations of
oxyto-cin (OXT) and vasopressin (AVP) have been found in
people with eating disorders (EDs) (Demitrack et al 1990,
1992; Gold et al 1983a).
OXT and AVP are produced in the hypothalamic
supraoptic and paraventricular nucleus and are released
peripherally via pituitary projections to the
neurohypoph-ysis, and also centrally directed into the brain (Swaab et al
1993). Central and peripheral OXT and AVP are
indepen-dently regulated (Geiner et al 1988), with the peripheral
activities of these peptides best known. AVP controls
free-water clearance of the kidney, whereas OXT
pro-motes uterine contraction and milk let-down. In the brain,
these peptides are long-acting neuromodulators and exert
complex behavioral effects. OXT administration to rats
disrupts memory consolidation and retrieval, whereas
AVP administration enhances memory function. In animal
models, OXT has anxiolytic actions, whereas AVP has
anxiogenic effects (Liebsch et al 1996; McCarthy et al
1995). Thus, in several ways the effects of OXT appear to
be reciprocal to the effects of AVP. In terms of EDs, these
neuropeptides are of interest because they influence
feed-ing behavior (Burlet et al 1990; Olson et al 1991) and have
also been implicated in obsessional behaviors (Leckman et
al 1994) and depression (Gjerris et al 1985).
It is not known whether alterations in OXT or AVP are
a consequence of pathologic eating or malnutrition, or if
they are premorbid traits that contribute to a vulnerability
to develop EDs. Determining whether abnormalities are a
consequence or a potential antecedent of pathological
feeding behavior is a major question in the study of EDs.
It is impractical to study people with EDs prospectively,
because of the young age of onset and difficulty in
premorbid identification of people who will develop this
illness. Therefore, we studied women who were recovered
for more than a year from BN or AN-BN. Any persistent
psychobiological abnormalities might be trait-related and
might potentially contribute to the pathogenesis of the
disorders.
From the Department of Psychiatry, University of Pittsburgh, School of Medicine, Western Psychiatric Institute and Clinic, Pittsburgh, Pennsylvania (GKF, WHK, CGG) and the Department of Psychiatry, Cornell University Medical College, New York, New York (MA).
Address reprint requests to Walter H. Kaye, M.D., University of Pittsburgh, Western Psychiatric Institute and Clinic, 3811 O’Hara Street, E-724, Pittsburgh PA 15213.
Received June 10, 1999; revised December 28, 1999; accepted January 3, 2000.
Methods and Materials
Thirty-three women who had previously met DSM-IV criteria for an ED were recruited: 23 women recovered from BN (rBN), and 10 subjects recovered from binge eating/purging–type AN (rAN-BN). The rBN women had never had AN. Recovered ED subjects were compared to 17 female normal control subjects (NC), who were recruited through advertisements in local news-papers and who were without lifetime psychiatric or major medical illnesses. In the year before the study, all participating subjects had to maintain a body weight of.90% average body weight (%ABW) (Metropolitan Life Insurance Company 1959), have regular menstrual cycles; have not binged, purged, or engaged in restrictive eating patterns; and have not met criteria for substance-related disorders. None of the subjects had a current major depressive episode. Current depressive symptoms were assessed with the Hamilton Depression Rating Scale (HDRS; Hamilton 1967) and current obsessive and compulsive symptoms with the Yale-Brown Obsessive Compulsive Scale (Y-BOCS; Goodman et al 1989). All subjects completed a 7-day diary at home, in which they listed individual food items consumed, portion sizes, and exercise frequency. This informa-tion was used to confirm the presence of relatively normal dietary intake. Subjects were free of medication for 6 weeks prior to the study and gave informed consent.
A lumbar puncture (LP) for obtaining cerebrospinal fluid (CSF) samples without preservative was performed during the early follicular phase by methods previously described (Kaye et al 1998b). CSF OXT and AVP assays were performed by radioimmunoassay also as previously described (Altemus et al 1994). A modified version of the Schedule for Affective Disor-ders and Schizophrenia-Lifetime Version (SADS-L) was used for assessment of lifetime axis I diagnoses (Kaye et al 1998b). Three NC, two rAN-BN, and nine rBN women were taking birth control pill (BCP) at the time of the study.
One-way analysis of variance (ANOVA) and Tukey’s post hoc test were used to investigate three group comparisons. Analysis of covariance (ANCOVA) was applied for confounding vari-ables. Two-way ANOVA investigated possibly interacting inde-pendent within-group factors when all cells were filled; other-wise stepother-wise regression was applied. Correlations were assessed
by Spearman correlation coefficients. For statistical analyses, the SPSS (Chicago) software package was used (Barcikowski 1984).
Results
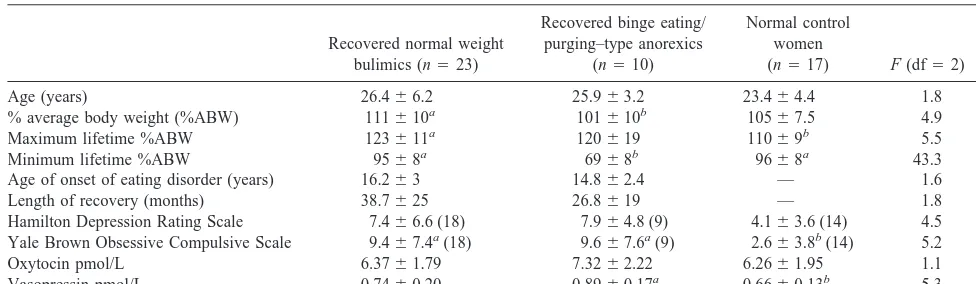
The three subject groups (Table 1) were of similar ages.
Current %ABW was lower in rAN-BN compared to rBN
women, and as expected, there were significant
differ-ences in high and low lifetime %ABW. ED subjects had a
similar age of onset of their ED.
The three subject groups had similar CSF OXT values;
however, CSF AVP concentrations were significantly
elevated in the rAN-BN compared to NC, and tended to be
higher compared to rBN women (p
5
.06, Tukey’s post
hoc test). When the three groups were combined, age was
significantly associated with CSF AVP (r
5
.43, p
,
.05).
Separate ANCOVAs with age as covariate showed that
CSF AVP in the rAN-BN was significantly higher than for
NC subjects [F(1)
5
12.1, p
,
.002], or rBN [F(1)
5
6.2,
p
,
.02].
A series of exploratory analyses was done in rBN
subjects to determine whether depression or obsessive–
compulsive disorder (OCD) or other factors were related
to neuropeptide levels. The rAN-BN group was not
analyzed because of their small cell sizes. rBN with a
lifetime history of OCD had higher CSF OXT
concentra-tions (n
5
5, 7.74
6
2.0 pmol/L) than did rBN subjects
without a lifetime OCD diagnosis (n
5
16, 5.89
6
1.6
pmol/L, p
5
.05). In addition, rBN with a lifetime history
of major depressive disorder (MDD) had higher CSF AVP
concentrations (n
5
17, 6.50
6
2.0 pmol/l) than did rBN
without a lifetime MDD diagnosis (n
5
4, 5.63
6
1.2
pmol/L, p
5
.03). Neither CSF AVP or OXT was related
to current depression or OCD symptoms in rBN. To
determine whether BCP use contributed to these findings,
a stepwise regression analysis was done. This showed that
elevated CSF OXT in rBN was mainly related to BCP
Table 1. Comparison of Study Subjects on Demographic Variables and Cerebrospinal Fluid Oxytocin and Vasopressin
Recovered normal weight
% average body weight (%ABW) 111610a 101
610b 105
67.5 4.9 .01
Maximum lifetime %ABW 123611a 120
619 11069b 5.5 .00
Minimum lifetime %ABW 9568a 69
68b 96
68a 43.3 .00
Age of onset of eating disorder (years) 16.263 14.862.4 — 1.6 .22
Length of recovery (months) 38.7625 26.8619 — 1.8 .19
Hamilton Depression Rating Scale 7.466.6 (18) 7.964.8 (9) 4.163.6 (14) 4.5 .15
Yale Brown Obsessive Compulsive Scale 9.467.4a(18) 9.6
67.6a(9) 2.6
63.8b(14) 5.2 .01
Oxytocin pmol/L 6.3761.79 7.3262.22 6.2661.95 1.1 .35
Vasopressin pmol/L 0.7460.20 0.8960.17a 0.66
60.13b 5.3 .01
Values are mean6SD.
p,.05 foravs.b(Tukey’s post hoc test).
316 BIOL PSYCHIATRY G.K. Frank et al
intake (t
5
3.2, p
5
.005) but not to a lifetime history of
lifetime OCD (t
5
1.1, p
5
.3); however, birth control use
had no effect on CSF AVP (t
5
0.87, p
5
.40) as the
relationship of CSF AVP and a history of MDD (t
5
2.28,
p
5
.03) remained significant.
Discussion
These data confirm and extend the possibility that long-term,
recovered eating disorder subjects with bulimic-type
symp-toms have increased CSF AVP. Previous studies (Demitrack
et al 1990; Gold et al 1983a) found elevated CSF AVP levels
in ill and recovered subjects with eating disorders but
subgrouping by types of eating behavior was not done.
This study raises the possibility that elevated CSF AVP
is particularly associated with depressive symptoms. Such
questions were not asked in previous studies of people with
eating disorders. The small size of subject groups after
segregating by ED type and Axis I diagnosis limits our power
to investigate the interactions of these diagnoses and AVP.
Other studies have found elevated CSF AVP in people with
lifetime MDD who were not currently depressed (Sorensen et
al 1985), whereas other studies suggest reduced CSF AVP
levels in depression (Gold et al 1983b). We also found that
CSF AVP was associated with age. We are not aware of a
study in humans investigating age as a covariate for this
peptide; however, age was related to the circadian pattern of
CSF AVP in an animal study (Jolkkonen et al 1986).
As a group, recovered subjects had normal CSF OXT
levels. These data, however, raise a question of whether
CSF OXT is related to a lifetime history of OCD or BCP
use. CSF OXT has not been previously measured in
recov-ered ED subjects. People with OCD have been reported to
have both normal (Altemus et al 1999) and elevated levels of
CSF OXT (Leckman et al 1994). Studies in animals suggests
that peripheral administration of gonadal steroids can alter
brain OXT mRNA; however, changes in CSF OXT or AVP
have not been found (Amico et al 1990; Insel et al 1997;
Parry et al 1991). In terms of limitations, the small size of the
recovered ED groups makes segregation by diagnosis or BCP
problematic. Thus, these preliminary data raise interesting
questions, but replication is needed in a larger sample.
Supported in part by grants from the National Institute of Mental Health (No. 2 R01 MH 42984-04, “The Neurobiology of Feeding Behavior in Bulimia”), the Children’s Hospital Clinical Research Center, Pittsburgh, Pennsylvania (No. 5M01RR00084), and the Christina Barz-Foundation, Du¨sseldorf, Germany.
References
Altemus M, Jacobson KR, Debellis M, Kling M, Pigott T, Murphy DL, et al (1999): Normal CSF oxytocin and NPY in OCD. Biol Psychiatry 45:931–933.
Altemus M, Swedo SE, Leonard HL, Richter D, Rubinow D, Potter W, et al (1994): Changes in cerebrospinal fluid neurochemistry of obsessive-compulsive disorder with clomi-pramine. Arch Gen Psychiatry 51:794 – 803.
American Psychiatric Association (1994): Diagnostic and
Sta-tistical Manual of Mental Disorders, 4th ed. Washington, DC:
American Psychiatric Press.
Amico JA, Janosky JE, Cameron JL (1990): Effect of estradiol and progesterone administration upon the circadian rhythm of oxytocin in the cerebrospinal fluid of rhesus monkeys.
Neu-roendocrinology. 51:543–551.
Barcikowski, Robert S, editors (1984): Computer Packages and
Research Design, vol. 3: SPSS & SPSSX. Lanham, MD:
University Press of America.
Burlet A, Desor D, Max JP, Nicolas JP, Krafft B, Burlet C (1990): Ingestive behaviors of the rat deficient in vasopressin synthesis (Brattleboro strain). Effect of chronic treatment by dDAVP. Physiol Behav 48:813– 819.
Demitrack MA, Kalogeras KT, Altemus M, Pigott TA, Listwak SJ, Gold PW (1992): Plasma and cerebrospinal fluid mea-sures of arginine vasopressin secretion in patients with bulimia nervosa and in healthy subjects. J Clin Endocrinol
Metab 74:1277–1283.
Demitrack MA, Lesem MD, Listwak SJ, Brandt HA, Jimerson DC, Gold PW (1990): CSF oxytocin in anorexia nervosa and bulimia nervosa: Clinical and pathophysiological consider-ations. Am J Psychiatry 147:882– 886.
Garfinkel PE, Kennedy SH, Kaplan AS (1995): Views on classification and diagnosis of eating disorders. Can J
Psy-chiatry 40:445– 456.
Geiner H, Altstein M, Whitnall WS (1988): The biosynthesis and secretion of oxytocin and vasopressin. In: Knobil E, Neill J, editors. The Physiology of Reproduction. New York: Raven, 2265–2281.
Gjerris A, Hammer M, Vendsborg P, Christensen NJ, Rafaelsen OJ (1985): Cerebrospinal fluid vasopressin changes in de-pression. Br J Psychiatry 147:696 –701.
Gold PW, Kaye WH, Robertson GL, Ebert MH (1983a): Abnor-malities in plasma and cerebrospinal-fluid arginine vasopressin in patients with anorexia nervosa. N Engl J Med 308:1117–1123. Gold PW, Robertson GL, Ballenger JC, Rubinow DR, Kellner CR, Post RM, et al (1983b): Neurohypophyseal function in affective illness. Psychopharmacol Bull 19:426 – 431. Goodman WK, Price LH, Rasmussen SA, Mazure C,
Fleisch-mann RL, Hill CL, et al (1989): The Yale-Brown Obsessive Compulsive Scale (YBOCS): I. Development, use and reli-ability. Arch Gen Psychiatry 46:1006 –1011.
Hamilton M (1967): Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol 6:278 –296. Insel TR, Young L, Wang Z (1997): Central oxytocin and
reproductive behaviors. Rev Reprod 2:28 –37.
Jolkkonen J, Tuomisto L, van Wimersma Greidanus TB, Laara E, Riekkinen PJ (1986): Vasopressin levels in the cerebro-spinal fluid in rats of different age and sex.
Neuroendocri-nology 44:163–167.
Vasopressin, Oxytocin, Bulimia, and Anorexia BIOL PSYCHIATRY 317
Kaye WH, Gendall K, Kye C (1998a): The role of the central nervous system in the psychoneuroendocrine disturbances of anorexia and bulimia nervosa. Psychiatr Clin North Am 21:381–395.
Kaye WH, Greeno CG, Moss H, Fernstrom J, Fernstrom M, Lilenfeld LR, et al (1998b): Alterations in serotonin activity and psychiatric symptoms after recovery from bulimia ner-vosa. Arch Gen Psychiatry 55:927–935.
Leckman JF, Goodman WK, North WG, Chappell PB, Price LH, Pauls DL, et al (1994): Elevated cerebrospinal fluid levels of oxytocin in obsessive-compulsive disorder. Arch Gen
Psychi-atry 51:782–792.
Liebsch G, Wotjak CT, Landgraf R, Engelmann M (1996): Septal vasopressin modulates anxiety related behavior in rats.
Neurosci Lett 217:101–104.
McCarthy MM, McDonald CH, Brooks PJ, Goldman D (1995): An anxiolytic action of oxytocin is enhanced by estrogen in the mouse. Physiol Behav 60:1209 –1215.
Metropolitan Life Insurance Company (1959): New weight standards for men and women. Stat Bull Metropolitan Insur
Company 40:1–11.
Olson BR, Drutarosky MD, Chow M-S, Hruby VJ, Stricker EM, Verbalis JG (1991): Oxytocin and oxytocin agonist adminis-tered centrally decrease food intake in rats. Peptides 12:113– 118.
Parry BL, Gerner RH, Wilkins JN, Halaris AE, Carlson HE, Hershman JM, et al (1991): CSF and endocrine studies of premenstrual syndrome. Neuropsychopharmacology 5:127– 137.
Sorensen PS, Gjerris A, Hammer M (1985): Cerebrospinal fluid vasopressin in neurological and psychiatric disorders. J
Neu-rol Psychiatry 48:50 –57.
Swaab DF, Hofman MA, Lucassen PJ, Purba JS, Raadsheer FC, Van de Nes JAP (1993): Functional neuroanatomy and neuropathology of the human hypothalamus. Anat
Em-bryol187:317–330.
318 BIOL PSYCHIATRY G.K. Frank et al