Abstrak
an laten virus varisela-Toster
(VW)
menyebablan terjadinya herpes zoster. Dalam keadaan laten, dapaldide
oRF-29, oRF-62, dan oRF-63. sistem imunberperanpenting dalampengendalianreaktivasivZV. variselabanya
eszoster
fur dan variselautPaI
fidakme
Hasil beberapapeneli
beri respAbstract
Reactivation
oster.Threelatentinfection.Th
incontrollinzos
er in childhood, the latterPai
conditions; both are tife ththe
Keywords: Latency, Immune respons, pathogenesis, Herpes zoster, Treatment Vol 6, No 3, JuIy - September I99V
Reactivation of l-,atent Varicella
Zosten
Virus
M.
Cholis
The varicella-zoster virus
(VZV) is
amember
of
the herpesvirus family
andresponsible
for
both
varicella
andherpes zoster. The
important
cytologic
al
charac-teristics
of
YZY
arethe
affinity for
tissuesof
ectoder_mal
origin
and theability
to
causelatent
infection.
Latency
is
required
for
the persistenceof
VZV
in
thehost
andthis virus
hasevolved elaborate
mechanisrnto
control the latent
state.
Reactivation
of
the
latent
virus,
usuall
zoster.
The
i
virus involve
This paper discuss some
aspects
of
reactivation
of
VZV
including
latency, mechanism,
clinical
implica_
tion,
and methods
of
limiting
the
consequencesof
reactivation.
VIRAL
FACTORS
IIWOLVED
IN LATENCY
Latent
viral infections
aredefined
as persistent infec_tions
in
which
the
viral
genome
is
piesent,
but
genehool of Medicine, Hospital
Varicella Tnster Virus
t49
expression
is
limited
and
infectious
virus
is
not
produced.
erp
lytic
pathogens,
the
nfect.
However,
of
ction,
some viruses express latent
functions which restrict
the expressionof viral
genes and enablethe
cell
harbour_ing
thevirus
to
survive.
The
VZV
genome consists
of
a linear molecule
of
It
::
It
contains
openreading frame
(ORD
corresponding to
approximately 70
genes,many
of
which
have
DNA
150 Cholis
In
contrast
to
HSV,
where
only
latency-associatedtranscripts
(LATs)
aretranscribed,
severalregions
of
theYZY
genome are expressedduring
latentinfection,
although
no
viral-encoded antigens
have
been
detected.
During latent infection, three VZV
genes,ORF-29, ORF-62
andORF-63
have been detectedby
in
situ
hybridization.
ORF-29
encodes theVZV
major
DNA-binding
protein. The
exactrole of this protein
isstill
unknown.
But the
DNA-
binding property
indi-catesthat
it
hasregulatory activities on
gene expres-sion.ORF-62
encodes themajor immediate early
genetransactivator, and ORF-63
down-regulates
transac-tivation
by ORF-
62
and
may therefore
repress
theproduction
of infectious virus.l
THE HOST FACTORS
INVOLVED IN LATEN.
CY
'Wood
(1993)
statesthat intact immune
responsesto
VZV
especially
cell
mediated
immunity
(CMI),
arerequired
in
maintainingYZY
in
the
latent
state. Theimmune
system has
animportant role in limiting
theeffect
of
primary
VZV infection
andin
constraining
the
infection
to
alatent
state.Although
hostimmunity
doesnot prevent reactivation,
it
is likely
that
the host responseplays
animportant role in
rapidly controlling
this
events. Therefore immunocompromised
patientsare much
more likely to have frequent and
severereactivation of
latent
VZV
infection.
The loss
of
cel-lular
immune
function
allows the latent
VZV
toreac-tivate
and causes herpes zoster.3The
primary
aims
of
thehost immune
response areto
promote host survival,
rescueinfected cells
andestab-lish
lifelong immunity
toreinfection.
In
achieving this
aims the
uii'.r,
-uy
becontained or
eliminated.3'{
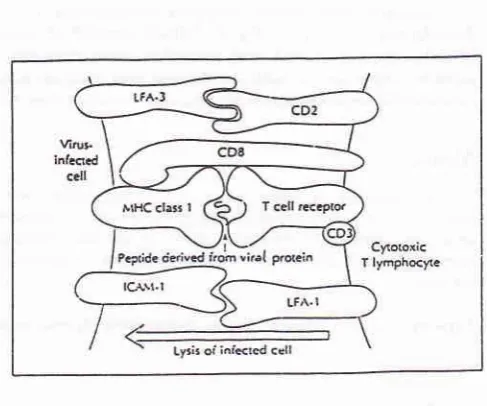
Antibody-dependent cell-rnediated
cytotoxicity
(ADCC)
is
an important
component
of
the
host
response
to
infection. Virally-encoded proteins
are processedinto
peptides and
presentedby major
his-tocompatibility complex (MHC)
classI
molecules to
the
T-cell
receptors
of
CD 8+
cytotoxic
T-lym-phocytes,
causes subsequentlysis of
the infected
cell
(Figure
1). Theclassic
antiviral
response,which
cause thelysis of infected
cellsby
cytotoxic
T- lymphocytes,
is
essentialwhen
thehost
needsto destroy
aninfected
cell.2's
In
general, herpesviruses
and the human host arevery
well
adaptedto
one another
andit
may
not
be
in
the interestof
the host to eradicate latency. In theory,down
regulation
of
the
MHC
class
I
molecules
which
arevital for immune recognition, would
be
a
valuable
Med J Indones
means
of
evading the immune
response. Numerous
cytokines
are
secretedby cellular immune cells,
e.g.y-interferron, which
can affect the
transcriptional
en-vironment
of
neighbouring
cells.
In
this
way, lytic
infection
may be prevented andcellular function
[image:2.595.300.544.183.386.2]main-tained
in
adelicate
balancewith
thelatent
state.2'4Figure 1. Interaction between the immune system and a
viral
infected ceII. CD8+ cytotoxic T-Iymphocyte (CTL) recognition of a virus-infected cell: a short peptide fragment deived from a viral protein is presented to the T cell receptor in associa-tion with a parrtcular MHC class I molecule; the CTL-nrBet cell interaction is stabilized by CD8 binding to the MHC class I molecule and by the specific binding of various adhesion molecules (only theLFA-I-ICAM-|
and LFA3-CD2 interac-lions are shown).'REACTIVATION OF LATENT
VZV
INFEC.
TION
During
the course
of
varicella,
VZV
passes
from
lesions
in
the
skin
andmucosal
surfacesinto
thecon-tiguous
endings
of
sensorynerves
andis
transported
centripetally
up the
sensory
fibers
to
the
sensoryganglia. In the ganglia
alatent
infection
is
established and thevirus
then persistssilently
andharmlessly. The
mechanisms
involved
in
the
activation of
latent
VZV
are unclear,
but
a number
of
conditions have
been associetedwith
theoccurence
andlocalization of
her-pes zoster. These
include immunosuppression
in
HIV
infection, Hodgkin's
disease andother malignancies;
administration
of
immunosuppressive drugs
andVol 6, No 3, July - September 1997
The
level
of
antibody is
considered
to
be the
critical
determinant
of
the Àost's capacity
to
contain
yZy
reversions. There is evidence
for
subclinical
reactiva-tions
of
YZY
in
immunocompromised patients
andin
the
elderly,
and
it
hasbeen
suggestedthat
a
similar
phenomenon may
periodically
occur
in
immunocom-petent individuals^
to
boost specific immunity
andmaintain
latency.T'8In
latent
VZV
infection,
theminute
doseof
infectious
virus that results
is
immediately neutralized
by
cir-culating antibody
or
destroyed
by
cellular
immune
responsesbefore
it can
infect
other
cells
andmultiply
enough
to
causeperceptible
damage.However,
in
theactivation of
latentVZV
infection, virus
multiplies
andspreads
within
theganglion,
causing neuronal necrosis andintense
inflammation; this
processis usually
ac-companied
by
severeneuralgia.
Infectious
VZV then
spreadsantidromically
down the
sensory nerve,caus-ing
intense
neuritis,
andis
releasedfrom
the
sensorynerve
endings
to
the
surrounding
skin
where
it
produces the
characteristic cluster
of
zoster vesicles.The incidence
of
herpes
zosteris
estimated to
bebe-IMPLICATION
OF
REACTIVATION
With
increasing
numberof elderly
peoplein
developedcountries,
the
number
of herpes
z-ostér caseswill
in_ crease and hencethe
patientssuffering
from
the
most
frequent
ion of
herpes
zoster:
chronic
zoster-assocring
preg-varicella
a serious
Varicella Zoster Virus
t5l
METHODS OF
LIMITING
THE
CONSEQUEN.
CES
OF
RBACTIVATION
Vaccination
While CMI
appears to beimportant
for
theprevention
of
reactivation, the
persistence
of
humoral
immune
responsesespecially
has beencorrelated
with
protec-tion from reinfecprotec-tion, which
may occur
in individuals
who
developlow titers of antibody
afterimmunization.
It
is therefore important
thatboth
armsof
theimmune
response aremaintained after vaccination.
Trials
with
the Oka strain
of VZV
showed
that
persistence
of
cellular
andhumoral immune
responses occuredin
themajority
of
those vaccinated, and were consistentwith
those achieved
after
natural
infection with
yZy
at
6 years afterimmunization.
The Oka vaccine has been used
successfully in
children
in
Japan
to
prevent varicella
and
has recently
been approvedfor
usein
theUSA.
However,
thevirus
usedin
the vaccine is
just
asneurotropic
asthe wild-type
and
it
is
possible
that
widespread vaccination
of
children could
resultin
ashorter-lasting
immunity,
andconsequently, there will be more cases
of
herpes zosterfollowing
avaccination
compared tonatural
infection.
This
hasnot
been the caseto
date,but
researchfor
thedevelopment of
anon-neurotropic vaccine
shouldcon-tinue.23
It
is not
yet
known
what impact
cination
of
adultswill
have onth
zoster. Studiesin
leukemic
child
higher risk
ofdeve
zoster(incidence
rateof
10-20
Vo)comp
y
children
(incidence
rate
of
0.05-0.074
that the incidence
of
herpes zoster was
actually
lower in
vaccine recipients
than
among thosewho
acquired
varicella
nuturuily.2,gVaricella
immunization
in
elderly
boosts
VZV
_specific
CMI
and may therefore decrease theincidence
andseverity
of
herpes zoster.
Currently, no
data
areavailable to
confirm
whether
immunization
attenuatesor prevents
herpein
theUSA, invol
60
years
of
agevaccination.
Antiviral
therapy
r52
CholisTo
impact
onchronic
zoster-associatedpain,
antiviral
therapy should
subsequent
nerve
damage.
A
7-day courseof aciclovir
(800
mgfive
times
daily)
has beenshown
to confer significant benefit to
the patient
for
the
management
of
zoster-associatedpain. Aciclovir
also
acceleratesthe
events
of
cutaneoushealing
and shortenstire duration
of virus
shedding.ll'12
The
daily
doseregimen
of aciclovir for
herpes zostermentioned above
is
recommended also
by
the
In-donesian
llerpes
Study Group (1995).13
Th"
"o--bined
datafrom all
the multicenter
trials
indicate that
oral
aciclovir
results
in simificant
benefit
for
patientswith
herpes
zo"ter.t3'14'lf
The
advent
of
new antiviral
agents
will
expand
therange
of
antiviral
therapies
available
to
the
prac-titioner. A7
-day courseof
valaciclovir
(1000 mg threetimes
daily)
resolves zoster-associated pain aboutone-third
faster than
aciclovir.
Other
published
datashowed
that famciclovir (250 mg three times daily
(UK)
or 500 mg
three
times daily (USA))
produced
similar
results
to aciclovir in
reducing
theduration
of
zoster-associated
pain.
In
the
immunocompromised
individual
with
herpes zoster,
intravenous aciclovir
therapy
may
b-e_r_equiredif
there
is
arisk
of
visceral
dissemination.2' l)
CONCLUSION
1.
Latency
of YZY
is
not a resting, but a
dynamic
state.
It
involves a complex interrelationship
be-tween
thevirus, host
cell
and theimmune
system.2.
Reactivation
of
thelatent
YZY
can betriggered by
risk
factors
andresult
in
herpes zoster.3.
In
elderly,
but
otherwise
immunocompetent
patients,
themost common complication
of
herpes zoster ischronic
zoster- associated pain orpost-her-petic neuralgia.
4. The
useof Oka-live vaccine for
theprevention
of
varicella
in
healthy children
is
controversial,
thoughit
wasproven beneficial in reducing
therisk
of
varicella
in
immuno-compromised
individuals.
5.
Antiviral
therapy
should beoffered
routinely
to
all
patients
over the
ageof
50
years presenting
with
herpes zoster
within
72
hours
of
rash
onset.
Valaciclovir
appears to be the mosteffective
choiceof antiviral
therapy.
Med J Indones
REFERENCES
l.
Otmann MN, Alani R.Varicella
and Herpes Zoster. In: Fitzpahick TB, Eizen AZ, WolfK,
FreedbergIM,
AustenKT, eds.
Dermatolology In General Medicine.vol 2.4ûH.
New York; Mc GrawHill,
1993:2543-67.2. \Mhitely RJ, Sandstrom E, editors. The Clinical Implications
of
llerpes virus Latency. Proceedingof
the 3td Annual Meeting of The Intemational Herpes Management Forum on The Clinical Implicationsof
Herpes virus Latency; 1995Nov
17-20; Istambul.Istambul: lnternational
Herpes ManagementForum, 1995.3. Wood
M,
editor.flow
can the burden of zoster-associated pain be reduced? Proceeding of The International Herpes Management Forurn Strategies Workshop on How can the burden of zoster-associated pain be reduced? ; 1993 May 1 5 ;Washington
DC.
WashingtonDC:
Intemational llerpes ManagementForum, 1993.4. Paul WE. Infectious Diseases And The Immune System. Scientific American 1993; September:S1 -63.
5. Joklik
WK,
rWillet HP, Amos DB,Wilfert
CM. Immune responses in infection. In: Joklik WK, Willet HP, Amos DB, Wilfert CM, editors. Znsser Microbiology. 20s Ed. Con-necticut; Prentice Hall, 1992:352-7 .6.
Arvin AM.
Cell
Mediated Immunityto
Varicella-zoster Virus. J Infect Dis 1992;166 Suppl l:35-41.7. Masaoka T, Hirauka A, Teshima H, Tominaga N. Varicella-zoster Virus Infection In Immunocompromised Patients. J Med
Virol
1993;1 Suppl l:82-4.8. Teroda
K,
Kawano S, YoshihitoK.
Varicella-zoster virus reactivation is related to the low response of YZY
specificimmunity
after chickenpox infancy.J
Infect
Dis
1994; 169:650-2.9. Hardy I, Cherson AA, Steinberg SP. The Incidence of zoster after immunization
with
the live-attenuated varicella vac-cine,A
Studyin
childrenwith
leukemia.N
Engl J Med1 9 9 I :3 25 (22) : 1545 - 5 0.
10. Lumintang
H.
EpidemiologiVirus
Herpes Hominis di RSUD Dr. Soetomo Surabaya. Airlangga Period Dermatol-Venereol 1993;6(l):l-8.11. Moorthy
TT.
HerpesVirus
Infection, The Enigma and Chalengers. Airlangga Period Dermatol-Venereol 1993; 6(t):13-21.I 2. B alfour HH. Current Management of Varicella Zoster Virus
Infection. J Med
Virol
1993;l Suppl 1:74-80.13. Daili SF, Budiardja SA, Zuber F, Makes WIB, eds. Infeksi
Herpes,
Pedoman PengobatanAsiklovir
untuk Dokter. Jakarta; Kelompok Studi Herpes Indonesia, 1995.14. Fiddian AP,
Mills
J. Therapeutic OptionsFor
Zoster. J Intemational Herpes Management Forum 1995; 2(3):77 -80. 15. Balfour HH. Clinical Aspectsof
Chicken Pox and Herpes