Abstrak
Status kardiovaskular dan peran dobutamin terhadapnya belum pernah diteliti pade pasien dengan penyakit membran hialin (PMH) ringan. Tujuan penelitian ini adalah untuk mengetahui pengaruh pemberian dobutamin pada pasien PMH ringan (PMH yang tidak memerlukan pemberian ventilasi mekanik). Pasien PMH ringan dilakukan randomisasi, untuk dibertkan dobutarnin atau plasebo, selain pengobatan suporrtf stundar yang berlaku
di Subbagian
Peinatologi, Bagian llmu Kesehatan Anak FKUI-RSCM, Jalarta. Pengobatan diberikan sampai sertap pasien berusia 96 jam atau bila terjadi kegagalan (memerlukan ventilasi mekanik atau mengalamiperburukan
yang
mengancam jiwa). Empat puluh satu pasien disertakan dalam penelitian; 21 dibeikan plasebo, 20 mendapat dobutamin 10 1tg/kg/menit. Selama perlakuan tidak terdapat perbedaan yang bermakna dalam laju jantung dan laju napas antara kedua kelompok, namun telanan sistolik, tekanan diastolik, dan tekanan arteri rerata secara bermalçta lebih tinggi pada kelompok ding pada kelompok plasebo. Terdapat 8 pasien yang mengalami kegagalan pada kelompok plasebo, dan 7 pada min, termasukI
pasien yang mengalami peningkatan tekanan darah sistolik sampai 85 mmHg. Uji xz tidak memper-lihatkan perbedaan kegagalan antara kedua kelompoh namun analisis kesintasan (survival analysis) dengan menggunalcan metode Kaplan-Meier danuji
Breslow menunjukkan bahwa pasien kelompok dobutarnin mempunyai masa bebas ventilasi mekanik dan perburukanklinis (rerata78 jam, intervalkepercayaan95Vo antaraT0 sampaiS4 jam) lebihlamadibandingkandenganpasienkelompok plasebo (rerata kesintasan 61 jam, interval kepercayaan 95Vo antara5I sampai
71 jam). Disimpulkan bahwa penambahan dobutamin pada terapi suportif standar memperlambat perburukan klinis NKB dengan PMH ringan, sehingga dianjurkan pemberian dobutamin1O 1s.g/kg/minute secara dini pada pasien PMH ringan.
Abstract
In contrast to the situationin severe hyaline membrane disease (HMD), cardiovascular status and effect of inotropic agentinmild HMD have not beeninvestigated. This study aimed to sndy the effect of dobutamine on the clinical course of mild HMD. Preterminfants withmildHMD (thosewho didnot requiremechanicalventilation)were randomly assignedto eitherreceive dobutamine 10;tglkg/minute or placèbo in addition to standard supportive teatment. AII patients were managed accord.ing to standard treatment at the Division
of
Perinatology, Department of Child Health, Medical School, (lniversity of Indonesia.'The teatment was continued until each patient reached the age of 96 hours, or needed mechanical ventilation, or experiencedIift
threatening clinical deterioration. Fofi-one infants were enrolled in the study; 20 were treated with dobutamine and2l
with placebo. Clinical observation disclosed that there was nodffirence between heart rate and respiratory rate between the 2 groups, but systolic, diastolic, and mean arterial bbod pressures were significantly higher in dobutamine-treatedthaninplacebo-teated group. Therewere Sfailures inplacebo group andTfailures (including
l
with systolic hypertension) in the dobutamine group. There was no significant dffirence of the occurrence of treatmentfailure between the tvvo groups. However, on survival analysis using Kaplan-Meier and Breslow tusrtng it was observed that dobutamine-treated patients had a significantly longer mean mechanical-ventilation-free survival (mean: 78 hours; 95Vo conf,dence interyals: 70; 84 hours) than placebo- treated patients (mean: 61 hours; 95Vo confidence intervals 5 I;7I
hours). Administration of dobutamine to standard treatment delays the deterioration of preterm infants with mild HMD, so that use of dobutarnine 10 1tg/kg/minute early in the course of the disease is recommended.Keywords: Dobutamine, preterm infants, lryaline membrane disease, mechanical ventilation free suwival Vol 7, No 4, October - December 1998
Effect of
dobutamine
on
the
hyaline membrane
disease
Sudigdo
SastroasmoroHyaline
membrane
disease(HMD)
is
oneof
themost
challenging problems
in neonatal period
both
in
Departrnent of Child Health, Faculty of Medicine, University of Indonesia/Dr. Cipto Mangunkusumo Hospital, Jakarta, Indonesia
Role of dobutamine on the clinical course of mild
HMD
227clinical
course
of preterm
infants with mild
syn-228 Sastroasmoro
thesized
by
type
II
pneumocytes, starting
at22
weeks
gestational age and reaching
its
optimal amount
atterm."-'The clinical
courseof
HMD
varieswidely. In
almost
all
instances
the clinical
manifestations
of
respiratory
distress syndrome
have
appearedby
theage of 4
hours.*
A
proportion of
patients present them-selveswith
breathing
difficulty
atbirth,
and becomemore
severe
in
the
subsequenthours
and days.
Theusual
clinical
course, however,
is that the
symptomsand signs become
worse
in
the
first 48
hours,
thentend
to
beplateau
for
the next 24 hours,
followed
by
gradual improvement
in
l-2
days.
While
patients
present themselveswith
severemanifestations usually
persistin
severecondition,
thosewho
initially
presentwith mild respiratory
distress may become
severe sothat
mechanical
ventilation
isrequired.
Proper management
of
severeHMD
requiressophisti-cated
technology, i.e.,
use
of
mechanical
ventilation
with
orwithouisurfactant
replacement therapy.5'6 Thetwo
modalities
arenot
available
for
most patients
be-causeof
problems
with facility
andcost. One
of
thecomplications
of HMD is
its effects
on thecardiovas-cular
system.In
severeHMD
the cardiacfunctions
aredisturbed;7'8
however,
in mild form of HMD this
hasnot
been elucidated. Since cardiac
functions
are
dis-turbed
in
severeHMD,
it
is logical to
assumethat
in
mild HMD
thecardiac
functions
are alsodisturbed
in
a
lesser degree.
Furthermore,
dobutamine, a
titrable
inotropic
agentwhich
is known to improve
cardiovas-cular function
in
severeform of HMD,
hasnot
beenstudied
in
patients
with mild HMD.
Consequently,
therole
ofinotropic
agent on theclinical
courseofpatients
is
not
known.
The purpose
of this
study
wasto
deter-mine the clinical
course
of
patients
with mild HMD
following
continuous
infusion of
dobutamine.
METHODS
This
was adouble
blind,
randomizedplacebo-control-led clinical
trial to
determine
the clinical
course
of
preterm
infants
initially
diagnosed
asmild HMD
fol-lowing
dobutamine infusion.
The study was conductedat the
Departments
of Child
Health
andObstetrics
andGynecology,
Dr. Cipto
Mangunkusumo Hospital,
Jakarta,from
November
1997through
May
1998. The diagnosis ofHMD
was based onclinical
andradiologi-cal criteria.a Preterm
infant with
HMD
wasjudged to
have
mild
disease
when
he
or
she
did
not
require
mechanical
ventilation support
(see
below).
To
beeligible for
thestudy,
apatient
with mild HMD
had tobe less than 37
completed
weeksof
gestation,weighed
morethan
1250 grams, and aged less than 48hours
atthe
time of
enrollment.
Infants who
met
theinclusion
Med J Indones
criteria
wereexcluded
from
thestudy
if
there rù/as anyof
the
followings: twins,
had
structural
heart
diseaseexceptpersistent
ductus arteriosus, had anovert
severecongenital
disease,or
if
the
parentsdisagreed to
join
the
study.This
study
was
apart
of
alarger study
to
determine
physiological
andepidemiological
aspectsof
preterm
infants
with HMD.
Patientswho met
the studycriteria
were
randomized
by
using
computer-generated
ran-domization
scheme,
to
either receive
dobutamine
(Dobutamine,
GiuliniR,
10pglkg/minute) or
placebo.Clinical
andlaboratory characteristics were examined
before
the
study,
including
complete physical
ex-amination, blood
gasesanalysis, and
electrolyte
ex-amination.
All
infants were
treated
in
the
incubator
with
the ambient temperatureof
35oC andhumidity
of
90o.Oxygen were given
2-4llminute by
using
a nasalcatheter
or
a
head
box. Prophylactic antibiotic
(in-travenousamoxycillin
50mg/kg/day)
wasgiven.
Theclinical condition
wascontinuously
monitored, while
acid-base andelectrolyte
measurements were repeatedevery
\2
hours,
andif
the
clinical condition
dictated.
Headultrasound to
detect evidenceof
periventricular-intraventricular
hemorrhage(PIVH)
was done on day3 or 4 by
apediatric radiologist. The
study was
ter-minated when the
infant
had reached
the
age
of
96hours,
or
if
heor
she neededmechanical
ventilation,
or
if
life
threatening
deterioration
of
any
cause
oc-curred. Thecriteria
for
mechanical
ventilation
support
were: PaCO2>
65mmHg
for
babies>
1500 gramsor
>
50mmHg
for
babies<
1500 grams,with pH <7.2.e
All
patients received
standard managementaccording
to
theprocedure
used at theDivision of
Perinatology,
Department
of Child
Health,
Medical
School,
Univer-sity of
Indonesia.
They were
managed
by
pediatric
residents underthe supervision
ofperinatologists.
All
clinical
observations, examinations, medications
andprocedures were recorded. The
investigator
observedbut did not interfere
with
thepatient
care and manage-ment.The sample size was estimated
by the use of
QuickBASIC 4.0 (Microsoft
Inc.)
Emulates
Vol 7, No 4, October - December 1998
failures in both
groups.
Second,analysis considering
thetime of event (time
dependentanalysis) of
patientswas
performed,
sincewhat
wasimportant
was notonly
whether
the failure
had occurred,
but
also
how
long
after
thebeginning
of
treatment
it
occurred. For this
purpose, survivals
of
patients
in
dobutamine
andplacebo groups were
constructed
by
using
Kaplan-Meier
method,
andBreslow
test wasapplied
for
com-paring the survivals
between thetwo
groups.parental
informed
consent was obtainedfrom
every patient. Thestudy
protocol has been
approved
by
the
ResearchEthical
Committee
of
theMedical
School,
University
of
Indonesia.
RESULTS
There were 41 patients met the study
criteria,
consisted of 23 males and 18 females. The gestational age rangedfrom 27
to
35
(mean 32.3,
SD 2.7)
weeks
while
thebirth weight
ranged
from 1250
to
2230 (mean
1689,SD 294)
grams.Twenty
patients received dobutamine
10
pg/kg/minute while
rheother
2l
received
placebo,each
of
them was given
by
an infusion pump.
Theclinical
and
laboratory
characteristics
of both
groupsare depicted
in
Table
l.
No
apparent
difference
wasfound
in
any
of
thecharacteristics.
RoIe of dobutamine on the clinical course of rnild
HMD
229Comparison
of clinical
andlaboratory
characteristics
between
placebo
and dobutamine
groups during
thestudy was
performed by presenting
the values at hours0,24,48,
and 60 after thebeginning of
theintervention
(Figure
I
and Table 2). Since some of the patientswere
excluded
from the study
and the
number
of
patients
was
getting
smaller, comparison was
only
done
until
60 hours
after
thebeginning
of
theintervention.
Vital
signs
The patient clinical condition, especially heart
rate,respiratory
rate,systolic
anddiastolic blood
pressures,and mean
arterial
pressure
were
examined
every
12hours.
Figure
1 shows the comparison
of
mean
vital
sign values
of dobutamine
and placebo groups at hours0,
24,
48, and60. Since
somepatients were excluded
from
the study due toclinical
deterioration,
thenumber
of
patients
in
each
time
of
evaluation was different
(decreased). This figure shows that there were
nosignificant
differences
in
mean
heart rate
and
respiratory rate
in
both groups. On the other
hand,mean
arterial
pressure
in
the
dobutamine
group
washigher than that
in
the placebo group;
this
difference
wasclinically significant
and wasstatistically
closeto
thesignificance value (p= 0.056).
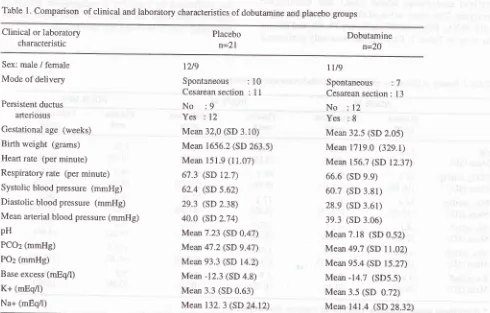
Table 1. Comparison of clinical and laboratory characteristics of dobutamine and placebo groups
Clinical or laboratory
characteristic Placebon=21
Dobutamine n=20
Sex: male / female Mode of delivery
Persistent ductus arteriosus
Gestational age (weeks) Birth weight (grams) Heart rate (per minute) Respiratory rate (per minute) Systolic blood pressure (mmHg) Diastolic blood pressure (mmHg) Mean arterial blood pressure (mmHg) pH
PCO2 (mmHg)
PO2 (mmHg)
Base excess (rnEqn) K+ (mEqÂ) Na+ (mEq/l)
t2/9
Spontaneous
:l0
Cesareansection :ll
No
:9
Yes
: 12Mean 32,0 (SD 3.10) Mean 1656.2 (SD 263.5) Mean 151.9 (11.07)
67.3 (SD 12.7) 62.4 (SD 5.62)
29.3 (SD 2.38)
40.0 (sD 2.74) Mean7.23 (SD 0.47) Mean 47.2 (SD 9.47) Mean 93.3 (SD 14.2)
Mean -12.3 (SD 4.8) Mean 3.3 (SD 0.63) Mean 132. 3 (SD 24.12)
tU9
Spontaneous
'.7Cesarean section:
l3
No
:12
Yes :8
Mean 32.5 (SD 2.05) Mean 1719.0 (329.1) Mean 156.7 (SD 12.37) 66.6 (SD e.9)
60.7 (SD 3.81)
28.9 (SD 3.61)
39.3 (SD 3.06) Mean 7.18 (SD 0.52) Mean49.7 (SD 11.02)
[image:3.595.43.533.414.727.2]2-j0 Sastroasmoro
Figure 1. Dingram shows
yital
signs at the beginning of the study and 24, 28, and 60 hours later. MAP = mean arterial pressure; SEM = standard error ofthe mean.Laboratory results
Besides physical examination, every 12 hours
eachpatient
underwent
blood
gases and
electrolyte
analyses.
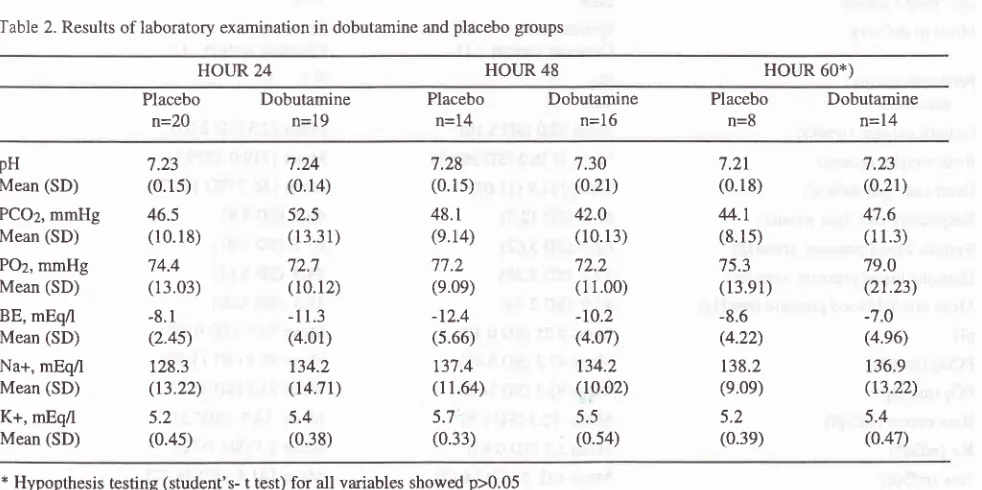
The
meanvalue of the blood
gases analysis(pH,
PCO2, POz, andBE)
athours 24,48,
and 60 canbe
seenin Table 2.
Comparison was
only performed
Med J Indones
until
60 hours afterdobutamine
or placeboadministra-tion, since
athour T2there was only
1patient of
the placebo group remainedin
thestudy.
Hypothesis
test-ing
performed
by
t-test
for 2
independent
groups
athour 60
disclosed
no
significant difference
of
thelaboratory
results between thetwo
groups.Table 2. Results of laboratory examination in dobutamine and placebo groups
p=0,106
p=0,234
p=0,056
HOURS
24 48
E
= dobutaminegroup
A
= placebo group[
=t""n
+/'1,sG sEMHOUR 24 HOI.JR 48 HOUR 60*)
Placebo
Dobutaminen=20
n=19Placebo
n=14
Dobutamine n=16
Placebo Dobutamine
n=8
n=14pH
Mean (SD)
PCO2, mmHg Mean (SD)
POz, mmHg Mean (SD) BE, mEq/l Mean (SD) Na+, mEq/l Mean (SD) K+, mEq/l Mean (SD)
7.23
(0.15)
46.5 (r 0.1 8)
74.4 (13.03)
-8.1
(2.4s)
128.3
(13.22)
5.2
(0.4s)
7.24 (0.14)
52.5 (r 3.31)
72.7 (10.12) -11.3 (4.01)
134.2
(r4.7t)
5.4 (0.38)
7.28
(0.15)
48.1
(e.14) 77.2 (e.0e) -12.4 (5.66) r37.4 (r 1.64)
5.7
(0.33)
7.30 (0.2r) 42.0
(10. r3)
72.2 (11.00)
-10.2
(4.07)
134.2
(10.02)
5.5
(0.s4)
7.21
(0.18) 44.r
(8. l 5)
75.3
(13.e1) -8.6 (4.22)
138.2
(e.0e)
5.2
(0.39)
7.23 (0.21) 47.6
(1 r.3) 79.0 (21.23) -7.0 (4.e6)
136.9 (t3.22)
5.4 (0.47)
[image:4.595.50.541.481.726.2]Role of dobutamine on the clinical course of mild HMD 231 Vol 7, No 4, October - December
l99g
Treatment
failure
placebo groups.
SeeTable
3.Table 3. Results
of
treatmentin
dobutamine and placebo groupsFailure
Success
TotalRelative
risk:
I .10 (Cl 95Vo: 0.48; 2.44); p = 0.837Survival analysis
As
mentioned
earlier,
sincethe
time of
the occurenceof
failure
was asimportant
as theincidence
of failure,
survival
analyses were performed.
First,
survival
analysis
of
all
patients
was
perfo
d
by
comparison
of
survivals
betw
and
dobutamine
groups. The event ofin
n thepatient
neededmechanical
ventilation,
or when
alife
threatening
condition occurred.
Basedon
thenatural
history
of
HMD,
rhatthe active
processof
the diseaseusually
subsidesafter
the
ageof
72
to
96hours,
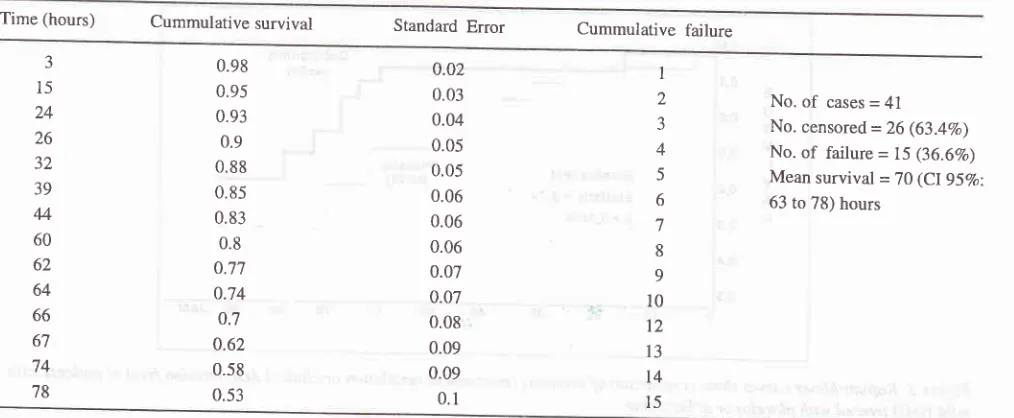
sur_Table
4.
Life table (Kaplan-Meier) in41
patientswith
mild HMDvival
analysis was doneuntil
theinfant
reached the ageof
96 hours.Analysis
was doneusing
the SpSSprogram for
Win_
dows
Release
6.0,
with
Kaplan-Meiei method
andTable
4
is
the survival table
of
the
4l
patients;
15patients
failed
and
26
patients
*"."
""nrored.
Themean
survival
was 70hours,
with
the 95Voconfidence
intervals
from 63 to 78
hours. Figure
2
shows
thesurvival
curve
of
all
patients
(41)
according
to
Table
4.
From
both
the table
andfigure
it
is
seenthat
therewere
only
2 eventsoccurred during
thefirst
24 hours;the rest
of
the events were more
or
less
equally
dis_tributed
within
the second andthird
24houiperiod.
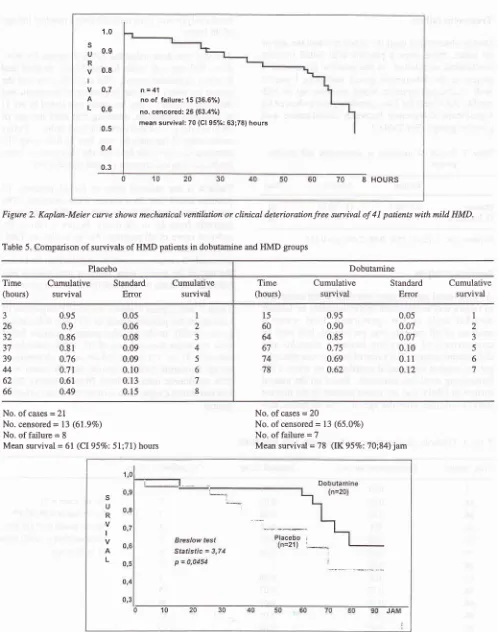
Table 5
and
Figure 3
shows
survival comparison
of
patients
of
theplacebo group
(n=21)
anddàbutamine
group (n=20).
In
the placebo
group g
pa
dwith
the mean
survival
rate
of
61
ejEo
eintervals 51
to
71) hours,
while
in
the
egroup 7
patients
failed (survival rate 7g hours with
95Vo confidence
intervals from
70to
g4hours).
Bres_low
test shows asignificant difference
between thetwo
groups.
Placebo
Dobutamine
8 (38Vo) 7 (35Vo)
13 (62Vo)
t3 (65Vo)
21 20
Time (hours) Cummulative survival Standard Error
Cummulative failure
J 15
24
26 32 39
44
60 62 64 66 67
74
78
0.98 0.95
0.93 0.9
0.88 0.85
0.83 0.8
0.77 0.74
0.7
0.62 0.58
0.53
0.02
0.03
0.04 0.05 0.05 0.06 0.06 0.06 0.07 0.07 0.08 0.09 0.09 0.1
I
2
J
4
5
6 7
8
9 10 12
t3 I4
l5
No.
of
cases = 41No. censored
=26
(63.4Vo)No.
of
failure = 75 (36.6Vo) [image:5.595.45.552.526.735.2]232 Sastroasmoro Med J Indones
s
U R
v
I
v A L
1.0
0.9
0.8
0.7
0.6
0.5
o.4
0.3
n=41
no of failure: 15 (36.6%) no. cencored: 26 (63.4Yo1
mean suwival:70 (Cl 95%: 63;78) hours
[image:6.595.47.545.68.700.2]8
HOURSFigure 2. Kaplan-Meier curve shows mechnnical ventilntion or clinical deterioration free sun ival of
4I
patients with mild HMD.Table 5. Comparison of survivals of HMD patients in dobutamine and HMD groups
Placebo Dobutamine
Time (hours)
Cumulative survival
Standard
Error
Cumulative survival
I
2 34
5 6 7 8
Time (hours)
Cumulative survival
Standard
Error
Cumulative suwival
1
2 J 5 6 7 J
26 32 37 39
44
62 66
0.95 0.9 0.86
0.81
0.76 0.7r
0.61
0.49
0.05
0.06 0.08 0.09 0.09 0.10
0.13
0.15
15 60
64
67
74
78
0.95 0.90 0.85 0.75 0.69 0.62
0.05 0.07 0.07 0.10
0.l l
0.12
No. ofcases =
2l
No. censored
=
13 (6l.9Vo) No. of failure = 8Mean survival =
6l
(CI 95%: 5l;71) hoursNo. of cases = 20
No. of censored = 13 (65.0Vo)
No. of failure = 7
Mean survival =
78
(IK 95Vo:70;84) jamFigure 3. Kaplan-Meier cuwes show comparison of survivals (mechanical ventilation or clinical detertoration free) of patients with mild HMD treated with placebo or dobutamine.
s
U R
v
IVol7, No 4, October - December 1998
fntracranial
hemorrhage
Examination
for
thepossibility of
intracranial
hemor-rhage was done
with
a headultrasonography,
on day-3or
4
after
birth.
Data collected
from
27patients,
showthat 7 had intracranial
hemorrhage,
i.e.,
4 out
of
15patients
of
the
dobutamine group and
3
out
of
12patients
of
the placebogroup.
Side effects
During the observation the
only
side
effect was
in-crease
of
systolic
blood
pressure
(up
to
85
mmHg)
found
in
a
patient
of
the
dobutamine
group
ap-proximately 15 hours after the start
of
dobutamine
administration. This patient
was
then
excluded
from
the study
andconsidered failed.
DISCUSSION
Of
the
41
patients
studied,
20
patients received
dobutamine
and2l
received placebo; the differences
of all
clinical
andlaboratory
parameters of both groupswere
not clinically
important.
The
study was
ter-minated
as theinfant
reached the ageof
96 hours.The
main
reason, as
put forward.earlier,
becauseof
thenatural
history of
the
disease.4Furthermore, many
of
preterm infants
with
HMD
who
survive
from
their
critical
period,
notinfrequently
suffer from
manycom-plications, e.g. hyperbilirubinemia, diarrhea,
sepsis,meningitis,
etc.which
may leadto
death.At
the ageof
96 hours, on the
one hand
theactive
processof
HMD
is
over, and on the other
hand
the
presence
of
anycomplication, especially
nosocomial
infection which
is
the main threàt
in
preterm infant
care,lOhas
not
appeared
yet.
Analysis
of
clinical
parameters,
i.e.,
vital
signs after
dobutamine
or placebo administration, was
done by
comparing values at hours
24,48,
and60,
as seenin
Figure
1.It
showsthat the heart
andrespiratory
rateswere
not significantly different, while
mean arterial
pressure
of
the
two
groups
wasdifferent.
Thesefind-ings showed that dobutamine administered
for
a
re\a-tively long time
(3
days)
did not
cause
significant
changes
on heart
and
respiratory
rates,
but
it
could
maintain
its
effect
on
increasing
the mean
arterial
pressure.
It
should
be noted that since
some
of
theinfants died
or wereexcluded
from
the study and some datacould not
beobtained,
thenumber
of
infants
was decreasingwith
time.
On laboratory results comparison (Table 4-15)
it
is
obvious
thatin
all parameters there were nosignificant
RoIe of dobutamine on the clinical course of mild
HMD
233differences between the
two
groups(hypothesis testing
was
doneonly on
the data athour
60). The question
is,
why
did
clinical
(especially
vital
signs, excepts theblood
pressure) andlaboratory
characteristics betweenthe
two
groups not
differ
despite
difference
in
sur-vivals? This
canbe explained
by
the
fact that
almost
all
ofthe
patients entered the studyin a
relatively
stablecondition.
During
hospitalization
some
of
them
developed
clinical
and/orlaboratory deterioration that
made them excluded
from
the study. Patientswho
wereexcluded
from the
study
must
have had
clinical
andlaboratory deterioration, but they were
not
analyzed.
Treatment
failure
The
proportions
offailure in both
groupswere similar,
which
were
7
out
of
20
subjects
in
the
dobutamine
group
and
8 out
of
21
subjects
in
the
placebo group
(Table 5).
Based
on this
finding
it
can be concluded
that dobutamine does not reduce the number of
patients
with mild
HMD
who
needmechanical
ventilation
in
the
first
4
days.
This
comparison was done
with
the numberof
subjects less than what wasactually
needed(recalculation
showed that thepower
of the
study
wasnotSÙVo; instead
it
has decreased to67Vo).ltshows that
the
incidence difference
between
thetwo
groups
wasclinically not
important. However, there was
onepatient
in
the
dobutamine
group who was
excluded
from
the study
dueto
increasedblood
pressure.Had
this
patient
not
beenconsidered
failed,
then the
con-clusion
should have been
that
dobutamine
decreasesthe need
for
mechanical
ventilation more
than2OVo, afigure that should be
considered
if
it
is
applied
on hundreds of thousandHMD
patientsin
thecommunity.
To find
out
whether there
was any
clinical
andlaboratory difference
(at
start
of
therapy)
between
patients
who failed
and
survived,
a
comparison
was done (datanot shown).
It
wasfound
that there were nosignificant clinical
nor laboratory differences
betweenthe
two
groups.
In
other words,
the
clinical
and/
or
laboratory
condition
at thebeginning
of
the courseof
the
illness is not
a goodpredictor for
thelater
needof
mechanical
ventilation.
This
is
in
accord
with
thenatural
history
of
HMD,
thatmany patients who
are atfirst
diagnosed ashaving
amild
illness
tendto worsen
in 48
hours, then
relatively
stable, and
gradually
im-prove after 72
hours.
At
the time
of
deterioration,
some patients
might
have
respiratory failure so
that
they
needed
mechanical ventilation.
These patients
could not
bepredicted
basedon
clinical or
laboratory
234 Sastroasmoro
Survival analysis
In daily
medical practice, both
from
thedoctor
and thepatient
points
of
view,
the
time of
the occurrence
of
an
event is
just
asimportant
asthe
occurenceof
the eventitself.
This fact is often overlooked
in
analyzing
astudy result,
which
may affect
its
application
in
themanagement
of
patients.
To
avoid this, an
analysisconsidering
the time
of
occurrence (time-dependent
analysis) ortime
to event data,r I shouldbe
performed.
Usually,
this
analysis
is mainly
doneto
evaluate
theresults
of
treatment
of
malignancy,
i.e.
cancer
or
leukemia.
Ideally
this
analysis
should
be
done in
various diseases, since even
ifboth
groups havesimilar
incidence
rate,but
if the
time of
the occurrenceof
theevent
differs, this
may
give
abenefit
in
managing
the subsequentpatients.
For
that reason,survival
analysis hadbeen designed since the
beginning of this
study.There
are severalways
to
construct
survival
analysis,the
two most popular ways
arethe
actuarial method
(Cutler-Ederer)
ind
theKâplan-Meier method.l2 Both
methods
usually
give
similar
curves,
eventhough
sometimesthey
don't.
There
are
also numeroustech-niques
for
testing the hypothesis
in
survival
analysis.In
this study
Kaplan-Meier
method
was usedfor
sum-marizing survival
data,
with
Breslow method for
hypothesis
testing.
The
results show
that
the two
groups
differed significantly
in how long
apatient
was freefrom
mechanical ventilation
orfatal complication.
With
the
limited number
of
subjects,
the
data
sup-ported
thehypothesis that
inotropic
agentadministra-tion
delays the needfor
mechanical
ventilation,
orput
in
other way,
dobutamine delays the progress
of
the disease.A
delay
of
the needfor
mechanical
ventilation
can beexplained
with 2
definite
reasons and
2
probable
reasons.The
2 definite
reasonsare:
(1)
improvement
of diastolic
and systolic
functions
dueto
dobutamine
administration
and increaseof
meanarterial
pressurewill
decrease the degreeof circulus vitiosus.
Improve-ment
of
ventricular functions
and increase
of
meanarterial
pressurein
dobutamine
group
result
a more stablecardiovascular
system comparedto
the placebogroup;
and(2)
dobutamine
haspositive
effects
on the secretion anddistribution ofsurfactant.
Other possiblepositive effects
of
dobutamine are:
(1)
dobutamine
decreases
pulmonary
vascular pressure;
(2)
dobutamine may
increasesurfactant
production.'
Side effects
On
clinical
andlaboratory
observations nodobutamine
sideeffect
wasfound,
except onepatient who
showedMed J Indones
an increased systolic blood pressure
on
day
I
of
dobutamine administration. This patient
wasdelivered
through
a cesareansection
with
gestational
ageof 32
weeks,
with
the
first
minute
Apgar
score
of
4.
Ten
hours after
dobutamine
infusion the blood
pressure increasedfrom62136 mmHg
to76142
mmHg,
and
15hours after
dobutamine administration it
became 85/58mmHg. Although
clinical or laboratory
deterioration
was
not
observed,it
wasfinally
decidedto terminate
thetrial,
and thepatient
was treated as usual.Role
of
patent ductus arteriosus
Studies on preterm
infants
alwaysinvolve
the presenceof
patent ductus arteriosus
(PDA).
The prevalence
of
PDA
isinversely
related togestational
age; the less the gestational age the larger theproportion
ofinfants
with
PDA.
In
full-term infants, the incidence
of
PDA
is
about 1 per thousandlivebirths,13
while in
infants
with
32
weeks gestational
agethe significant
incidence is
about
25Vo,and
in
28
weeks
or
less the
incidence
becamemore
than 50%.In
infants
andchildren,
moderate andlarge
degrees ofPDA
causes aleft
to
right
shuntthat may
cause heartfailure which would afféct
therespiratorysystem.14
In
full-term
and preterminfants without
HMD,
in thefirst
2-3
daysafter
birth, usually PDA
hasnot
manifested
itself.
Theinfants
donot suffer
from
distress, and evenon physical
examination
aheart
murmur
may
not
befound, or only systolic murmur
is
heard.
This is
be-causein
the
first
daysof life
the
pulmonary
vascular
resistanceis still high,
so thereis no significant
pres-sure
difference between
the
aorta
and_pulmonary
artery,
and
no significant
shunt
occurr.'t
If
the
pul-monary vascular resistance decreases then a
significant
left
toright
shuntwill
occur.
The
samething
happenin
preterm
infants with HMD
or
PDA;
in
thefirst
2or
3 daysof
theHMD
course, thepulmonary
vascular
resistanceis
still
high
sothat
nosignificant left to right
shunt is
present.As
the
HMD
improves,
thepulmonary
vascular resistance decreasesand
causesa
significant
pressuredifference
betweenthe
aorta andpulmonary
artery.
In
result
asignificant
left to right
shunt
is
present.
In
preterm
infants with
HMD
and
large PDA large
left to
right
shunt
may
follow
causing reappearanceof
dyspnea as amanifes-tation
of
heartfailure.
In this
series,of
41HMD
casesthere
were2l
patientsVoI7, No 4, October - December 1998
had
continuous murmur.
Onhigh
parastemal short axisview
aduct could be detected on the
left
side
of
thepulmonary
artery branch.
By using the color Doppler
acontinuous
flow
could be
seenin
the duct and
themain pulmonary
artery,l6-18usually with
alow
dias-tolic
component.
An
M-mode examination showed
anormal
left
atrium dimension meaning that the duct
washemodynamically not significant.
Some authors study the blood
flow
patterns
of the
arterial duct
in
preterm
infants,lT'19-20including
theeffect
of
PDA to the ventricular functions
and organblood
flow. In preterm infants,
alarge PDA may give
a significant effect in infants without
HMD
or
in
in-fants
who have recovered
from
HMD.
In
such cases,not
only
theinfant
is prone to heartfailure but
a.changeof
the cerebral
blood
flow
may also happen.'l Ther"
infants are good
candidates
for
indomethacin or
other anti-prostaglandin
drugsto
closethe
dùcl3'22-2swhich
are
believed
to
decrease
the
incidence
of
periventricular-intraventricular
hemorrhage.26'27Since
all the subjects
in
this
seriesdid
not show
anysignificant
PDA,
the presence
of
PDA was not
in-cluded in
the analysis.To sum
up, this study shows that
dobutamine could
alter the clinical course of patients
who were initally
diagnosed
as having
mild HMD.
Definite
practical
implication
of
this result
needsfurther confirmation,
as medical
knowledge
isnotbuilt only from
experienceof
some patients
but
as a result
of
accumulation
of
experiencewith
hundreds or thousandsofpatients. For
the time
being, however, the author
think that suffice
it
to
suggest
to
administer dobutamine
l0
pglkg/minute
to all preterm infants with HMD who
donot
needmechanical
ventilation
besides standardsup-portive
treatment.
For rural
hospitals
or those which
are not equipped
with
neonatalintensive
carefacilities,
a
delay of
the needfor mechanical
ventilation gives
achance
to the doctor
in
chargeto modify
treatment or
to refer
thepatient to
abetter equipped hospital.
ACKNOWLEDGMENTS
The author wishes to thank Professor Hans E
Monintja,
MD,
Professor Sofyan Ismael,
MD,
and
ProfessorB ambang
Madiyono,
MD
for continuously supervising
the study and
reviewing
thearticle,
toAsril Aminullah,
MD
and thePerinatology Staff
for supervising patient
management,to Kemas
Firman,
MD,
for performing
ultrasonographic examination,
and
to
Dyani
Kusumowardhani,
MD
for
helping
with
manuscript
preparation.
This
study was
partly
funded
by
'Tim
Role of dobutamine on the clinical course of mild
HMD
235Manajemen
Program Doktor'
(Department
of
Educa-tion
andCulture, Republic
of
Indonesia), South-East
Regional
Office
(SEARO)
-
V/HO,
Manila,
The
Management
of
'Riset
Bimbingan
llmu
Pengetahuan
dan Teknologi Kedokteran' (National
ResearchCoun-cil),
PT Solvay
Pharmaceutical Indonesia,
pT
Dexa-Medica,
and
PT Kimia
Farma. Dobutamine
andplacebo were prepared and supplied
by Solvay
phar-maceutical, Hannover, Germany.
REFERENCES
1. Hjalmarson
O.
Epidemiology and classificationof
acuteneonatal respiratory disorders. Acta Paediatr Scand 1981;
70:773-8.
2. Martin RJ, Fanaroff AA. The respiratory distress syndrome and
its
management.In:
FanaroffAA, Martin
RJ, eds.Neonatal-perinatal medicine. Diseases of the fetus and
in-fant. 6th ed. St Louis: Mosby; 1997. p. l0l8-21-.
3. Jobe AH. Lung development. In: Fanaroff
AA,
Martin RJ,eds. Neonatal-perinatal medicine. Diseases of the fetus and
infant. 6th ed. St Louis: Mosby; 1997. p.991-1008. 4. Walsh MC, Carlo W, Miller MJ. Respiratory diseases of the
newborn.
In: Carlo WA,
ChatburnRL,
eds. Neonatal respiratory care. 2nd ed. Chicago:year Book;
1988. p.260-88.
5. Gregory GA. Treatment of the idiopathic respiratory distress
syndrome with continuous positive airway pressure. N Engl
J Med 1971; 24:1333-40.
6. Fujiwara
T,
KonishiM,
Chida S. Surfactant therapy in neonatal respiratory distress syndrome.In: yeh
TF, eds.Neonatal therapeutics. 2nd ed,. St Louis: Mosby; 1991. p.
70-86
7. Tambunan T, Monintja HE, Karyomanggolo WT, Tamaela
LA. Gambaran radiologis paru pada bayi baru lahir dengan ' respiratory distress' . Maj Kedokt Indones I 978 ; 28 : i 09- I 6.
8.
Monintja
HE.
Perinatologi dan hubungannya dengan kualitas manusia. Pidato Pengukuhan Guru Besar FKUI.Jakarta: Balai Penerbit FKUI, 1989.
9. Brady JP, Gregory GA. Assisted ventilation. In: Klauss MH, Fanaroff
AA,
eds. Careof high-risk
neonate.3rd
ed.Philadelphia: Saunders; 1986. p. 202-lB.
10. Monintja HE. Infeksi sistemik pada neonatus. In:
yu
VyH,
Monitja HE. Beberapa masalah perawatan intensif neonatus.
Jakarta: FKUI; 1997. p.217-30.
I
l.
Altman DG, Bland JM. Time to event (survival) data.Brit
Med J 1998; 317:468-9.
12. Sastroasmoro S, Firmansyah A, Said M, Akib Ap. Analisis
kesintasan. In: Sastroasmoro S, Ismael S, eds. Dasar-dasar
metodologi penelitian
klinis.
Jakarta: Binarupa Aksara;1995. p. 143-55.
13.
Yu VHY,
Monintja HE. Duktus arteriosus persisten padaneonatus.
In: Yu VYH,
MonitjaHE.
Beberapa masalahperawatan intensif neonatus. Jakarta: FKUI ; I 997. p. 57 -7 6.
14. DiCarlo JV, Steven JM. Respiratory failure
in
congenitalheart disease. Pediatr Clin North Am 1994; 4l,,525-42. 15. Drayton
MR,
SkidmoreR.
Duchrs arteriosus blood flow236 Sastroasmoro
16. Shimade S. Effect
of
PDA on left ventricular output and organ blood flow in preterm infants with respiratory distresssyndrome treated with surfactant. J Pediatr 1994., 125.2'70-j. 17. Hirsimaki
H,
Kero P, Wanne O. Doppler ultrasound andclinical evaluation in detection and grading of patent ductus
arteriosus in neonates. Crit Care Med 1990; l8:490-3. 18. Su B-H, Watanabe T, Shimizu M, Yanagisawa M.
Echocar-diographic assessment ofpatent ductus arteriosus shunt flow
pattem in premature infants. Arch Dis Child Fetal Neonatal 1997;77:F36-40.
19. Macdonald FD,
Yu VYH.
Simultaneous measurement ofpreductal and
postductal oxygen saturationby
pulseoximetry
in
hyaline membrane disease.Arch Dis
Child 1992:67:1166-8.20. Clyman RI. Ductus arteriosus. In: Gluckman PD, Heymann
MA, eds. Pediatrics and perinatology - the scientific basis.
Edisi ke-2. London: Amold; 1996.p.755-61.
21. Mahony
L,
Caldwell RL, GirodDA,
Hurwitz RA, Jansen RD, Lennons JA, et a1. Indomethacin therapy on the first dayof life in infants with very low birthweight. J pediatr 1985;
106:801-5.
Med J Indones
22. Kueger E, Mellander M, Bratton D, Cotton R. Prevention
of
symptomatic patent ductus arteriosus with a single dose of
indomethacin. J Pediatr 1987 ; 1 1 1 :7 49 - 54.
23. Rennie JM, Doyle J, Cooke RV/Y. Early administration
of
indomethacin
to
preterm infants.Arch Dis Child
1986;6l:233-8.
24.Yeh TF, Luken JA, Thalji
A,
Raval D, CarrI,
Pildes RS. Intravenous indomethacin therapy in premature infants withpersistent ductus arteriosus: a double-blind controlled study.
J Pediarr
l98l;
98:137-45.25. Ment LR, Duncan CC, Ehrenkranz RA, Kleinman CS, Pitt BR, Taylor KfW, et al. Randomized indomethacin trial for prevention
of
intraventricular hemorrhagein
very
lowbirthweight infants. J Pediatr 1985; lO7:937-43.
26. Bada HS, Green RS, Pourcyrous
M,
Leffler CW, Korones SB,Magill HL,
et al. Indomethacin reduces the risksof
severe intraventricular hemorrhage. J Pediatr I 989; I I 5 : 63 I
-7.
27. Hanigan WC, Kennedy G, Roemisch F, Anderson R, Kusack
T,
PowersW.
Administrationof
indomethacinfor
the prevention of periventricular-intraventricular hemorrhage in