Vol6, No I, January - March 1997 Effects of PEM on Sperm Quality and Spermatogenesis
The Effects
of Protein
Energy
Malnutrition
(PEM)
on Sperm
euality
and
Spermatogenesis of
Male
Rats
Injected
by Testosterone Enanthaie+)
Eliza*, N. Moeloek**, N. Suhana**, S. Sri Sukmaniah***
Abstrak
Telah diketahui, bahwa penyuntikan testosteron enantat (TE) kepada
i
aenyebabiun aloopermia; namun sampai sekarang belum diketahui apakah akibat penyuntikanTE terseùutdip
^4 onon. Ooro
iùld ià'ùl|"rà
nya berbeda terhadap penyuntikanTE (pada orang Kaukasia hanya ia menyebabkan 90Vo azoospermia), Diduga perbedaan tersebut untuk mengetahui pengaruh diet rendah enersi protein terhadap kualins sperma dan spermatogenesis tikus inggu kepada tikus pEM akan menyebabkan penurunan kualitas dalampenelitian ini adalah: konsentrasi sperma, morfologi normal ormal maupun pada kelompok diet rendah kaloi protein; namun matosonium-A dan jumtah spermatosit pakhiten jugamenurun
::itHr::;;"!o"ii;i"::;:;
enersi protein, tetapi pada kelompok diet rendah enersi proteinmpok diet normal.
Abstract
The relationship of tesbsrerone enanthate (TE) with azoosDe is also influenced by nutritionalfactor. Data of WHd andWaitisî
ns no difference between the two groups. Testis weighr, diameter of the the number ofA-spermatogonium, and the number ofpachyiene spermatocyte were decreased er TE injection; but there was a dffirence betvveen normal diet animals and pEM animals,
Keywords: Protein energy malnutrion, Testosterone Enanrharc, Sperm euality, Spermatogenesrs, Àars
Concern over the rapidly growing world's population has stimulared andrologist to
find
effeciivè, safe, and reversible contraceptive substancesfor
men.+) S_upported by The Project of Research Team URGE (University Research For G raduate Educatio n) No. : 02 3/HTp p/U RGA I gg5. Department of Education & Culture, Directorate General of Higher Education.
* Focutty tf Medicine, University rf An_ nt of Biomedical post Graduate program, +* D.epartment of Bioktgy, Facutty of Medicine, University of In_
donesia, Jukurta.
Eliza et al. Med J Indones
so that spermatogenesis can be induced' However, protein dàficiency reduces the hypothalamus activity, ihat in turn affecting the production of ABP'
This research is conducted to investigate the effect of TE injection and malnutrition (protein energy
mal-nutrifion
=
PEM)
to
spermquality
and
sper-matogenesis of albino male rats. It is hypothesized that PEM male rats injected with 1 mg TE once a week forevaluated.
MATERIALS AND METHOD
Animals used
in
this research were male albino rat strainLMR
(Lembaga Makanan Rakyat) (V/istar derived).The
number of experimental animals were 36, which were divided into two groups:1.
Group one, consistsof
l8
animals and was given food àn ad libitum base for up to three months old, the animal weight of approximately between150-200g(Normal=N)
2.
Group two, consists of 18 animals and was given a diet food to induce protein energy malnutrition; therefore, their weights were lower, around6O-7OVo of normal animals (correspond to protein energy malnutrion = PEIvD.Each group was divided into 3 subgroups:
-
C (untreated control) animals (n = 6)-
TC (treated control = placebo control) animals (n = 6) injected with 0.2 ml solvent (wijen oil) once a week begining at 12th week after birth.-
T (treated) animals (n = 6) injected with 1 mg TE in 0.2 ml solvent (wijen oil) once a week begining at 72th week after birth.weeks afterbirth
normal food (group l)/diet food (group 2) injected with TE or solvent
mone secreted from hypothalamus, stimulates the syn-thesis and release
of
LH
(luteinizing hormone) and(androgen binding protein) by the Sertoli cell of the testis. Both testosterone and ABP have very important role
in
the processof
spermatogenesis' But the tes-tosterone in high concentration decreases the produc-tion of LH and FSH directlY.On the other hand, at the hyphophysis level hormone with a direct influence to the process is more needed'Of
various hormone contained in contraceptive pills, com-bination
of
TE (testosterone enanthate) and DMPA een shown a or oligo-hormonesof
sperm,normal
tilitY' Inaddi-tion,
th
gritY of sPerTmembr
is hamPered'"zoosPermia has hether its effect .DataofWHO5 and Mongolian men response differently to TE. It has been suggested that the different
in
their eating pattern might causen
to TE (907o (onlY 50% af-Bronson/, and ciencY in male rats can reduce their hyphofisis activities. Consequent-ly, the production of hypophysis controlled hormone, zuchai
FSH andLH,
which are importantin
the spermatogenesis, will bedisturbed-Another research showed that lower concentration of
Timetable
0 I 2 3 45 6 7 8 9l01ll2l3t4l5
161718192021:=l=l==-:i::===:=-:
Vol 6, No I, January - March 1997
Animals were housed individually
in 26x2lxll
cm cagesand
they were floored by saw-dust to absorb urine. Testosterone enanthate(TE)
andits
solvent (wijen oil) was made by PT Schering Indonesia. The food was prepared in the Diponegoro unit of Nutritionof
the Ministry of HealthOffice.
At the end of the treatment(21
weeks afterbirth)all
animals were sacrificed; both vas deferenses were excised and placein
petri dish containing 0.25 m1 0.85Vo NaCl. Both testis were also excised, weighted and then fixed inBouin's
solution then processedfor
histological preparation and evaluation.The parameters evaluated of sperm collected from yas
pachytene-spermatocyte.
A
Randomized Block Design was used with two factors (Normal and pEM animal) and three steps (injected by TE, solvent oil andcontrol). Data were normalized and homogenized before statistical techniques were applied. Factorial tests two by three were used to compare six different
treatments.
mal,then
non-pa
is ofvariance
(A
cantTest)
were
ele-ments.
RESULTS
The results of parameters evaluated are as follows: Sperm concentration (vas deferens)
lion/ml) of
(PEM)rats is depicted
80
60
C
TCTN
CTCT
PEùI
lsr
tr
2ndEffects of PEM on Sperm Quality and Spermatogenesis
[image:3.595.176.539.66.826.2]crcT
cTcc
Figure 2.
C = untreated control; lÇ = placebo/treated control;
T = treated, injected with TE
Sperm viability
Mean and SD of sperm viability (Vo) of normal and
protein energy malnutrition (pEM) rats injected with TE, is depicted in Figure 3.
Figure 3.
C = untreated control; TC -- placebo/treated control; T = treated, injected with TE.
Testis weight
Mean and SD
of
testis weight (mg)of
normal andprotein energy malnutrition (pEM) rats injected with TE, is depicted in Figure 4.
Sperm morphology (Normal head sperm)
Mean and SD of normal head sperm (%) of normal and protein energy malnutrition (pEM) rats injected with TE, is depicted in Figure 2.
Figure L
Figure 4.
C = untreated control; lÇ = plncebo/treatedcontrol; T = treated, injectedwithTE.
Testis diameter
Mean and SD
of
testis diameter (cm) of normal andprotein energy malnutrition (PEM) rats injected with TE, is depicted in Figure 5.
CTCT
CTCT
N
PEI\,IFigure 5.
C = untreated control; TC -- placebo/treated control;
T = treated, injectedwithTE.
Seminiferous tubule diameter
Mean and SD of seminiferous tubule diameter (pm) of
normal and protein energy malnutrition (PEM) rats
injected with TE, is depicted in Figure 6'
Med J Indones
250
C
TC
T
C
TCT
Figure 6.
C = untreated control; lÇ = plncebo/treated control; T = treated, injectedwithTE.
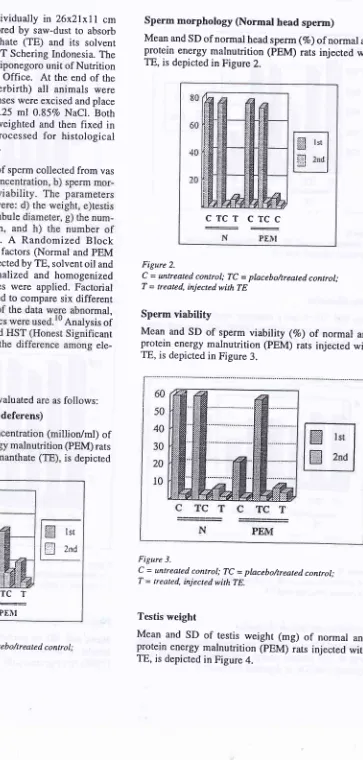
A-Spermatogonium
Mean and SD of A-spermatogonium number/tubule of
normal and protein energy malnutrition (PEM) rats
injected with TE, is depicted in Figure 7.
C TCT C
TCT
N
PEMFigure 7.
C = untreated conlrol; TC
-
placebo/treated control; T = treated, injected with TE.Pachytene spermatocYte
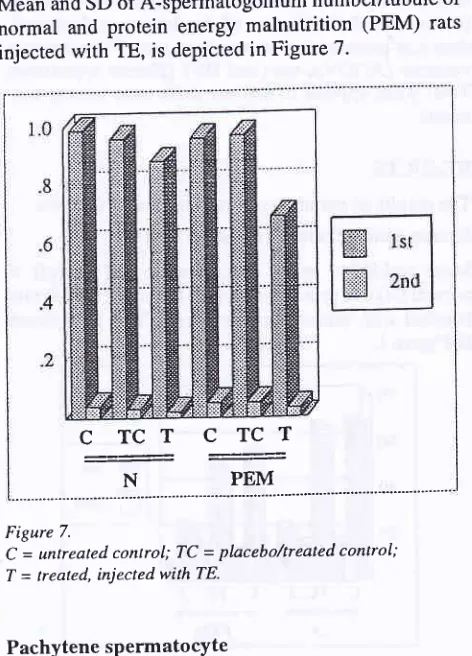
Mean and SD
of
pachytene spermatocyte number/tubule
of
normal and protein energy malnutrition (PEM) rats injected with TE is depicted in Figure 8' Eliza et al.1200
1000
800
600
400
200
2W
150
100
50
I
Vol 6, No 1, January - March 1997 Effects of PEM on
Sperm euatity and Spermatogenesis
C TCT C TCT
N
-E-'
the sperm concentration (p > 0.05) between TE in_ jected PEM and TE injected normal animals.
Sperm viability
ropped al con-as well
Testis weight
ANOVA tesr for testis weight showed
that
both inTestis diameter
S eminiferous tubule diamete
r
[image:5.595.61.293.70.351.2]A-spermatogonium Figure 8.
C = untreated control; TC = placebo/treated conlrol; T = treated, injected with TE.
The
Statistical AnalysisDistribution and Homogeinity of data
Before analysis of variance (ANOVA) test was carried
Sperm concentration
VoI6, No I, tanuary - March 1997 H uman R ec omb inan t Ery throp oi
e tin 43 't6
14
12
10
I
64
2
0
2468101214
[image:6.595.57.557.61.749.2]Weeks
Figure 4. Case no.4, male, 62 years old, Muttiple Myeloma
Ht 30
25
20
15
10
5
o
Hb
12
10
I
6
4
2
0
14
12
10
I
l;Hbl
.
l*t,
I 42
0 Ht
2468101214
16Weeks
Figure 5. Case no. 5, male, 6g years old, Multiple Myeloma
a a
4
6
''= I
10
12Weeks
Figure 6. Mean Hb, Ht,from 5 cases
Hb
40
30
20
P achyt ene s P e rmut o cY t e
ANOVA
test
showedthat
both in normal (N) andp-Èin
"n"tgy malnutrition (PEM) animals TE injec-iion declineà- the number of pachytene spermatocyteGii'*'rry
G < 0.01) compaied to tn"*ti,""ri$J.]
in
TEin-al animin-als.
DISCUSSION
The declining of sperm concentration after TE injec-tion either on the normal (N) and protein energy
mal-"",iiii".
(PEM) animals is very likely' This is due to"
t"g",it"
t"edÉack of exogenous testosterone directly io-inïrtvpophysis which*pt"tt"t
the production ofirr
unaîsÉ
or may be through the hypothalamus so ,n"ftoat",ion
ofdH-lH
decieases and eventually thef.oâo"ti*
of
LH
and FSHwill
also drop'In
bothcases,
finally
the process of spermatogenesiswill
be suppressed' Since there is no difference of spermcon-centration between normal
(N)
and protein energy malnutrition (PEM) animals, there is no interactionb",*""n
TE injection and protein energy-malnutritionon spermatogènesis, so the declining
of
spermcon-cent;tion
in this experiment is solely due to the TE injection.The same explanation is hold true also for sperm mor-fnorogy, ,o
ih"
decreasing number-of. normal sperm-orpnôtogy
was
dueto
the
TE
injection
thathampered
IÉ" pro""tt of
organel cell .development'Since there was no difference the number of normal
,f".-
*otpttology between TE injected in both normal(fr)
ana pfur4 animats, soit
was very likely that the à"ér"uring number of sperm morphology was due to the TE injection.Thç
same results and very likely also the mechanism were also found on the viability of the sperm in thesense that the number of viable sperm also declined
after TE injection in both normal and PEM animals;
this declining was also due to exogenous testosterone'
because there was no tnteraction between TE injection and PEM.
The most interesting phenomenon is that the viability
of
sperm in the untiéated control PEM animals alsodeclined,
although
in
the treated controlof
PEManimals did
not.
This is probably due to the calorydeficiency; this deficience might be disrupt the cell
membrane with the end result affected the viability' In
the treated control of PEM, this deficience has been
Med J Indones
than in normal (N) grouP.
VoI6, No 1, January - March 1997
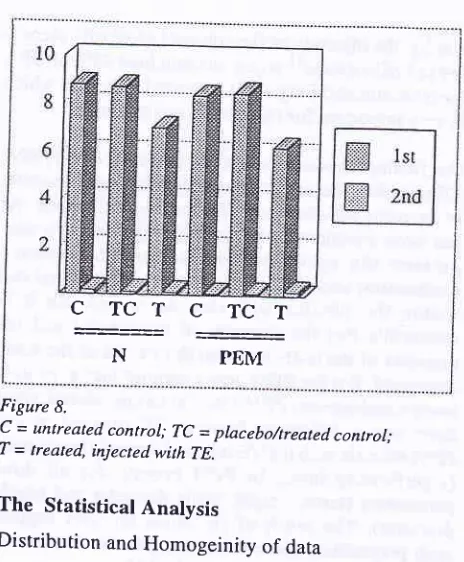
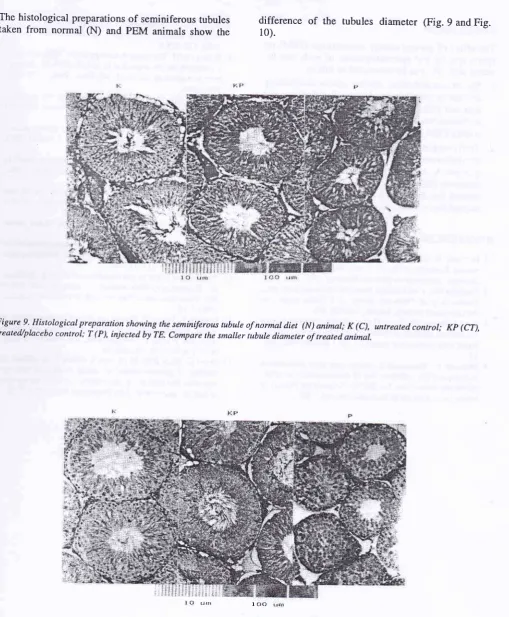
The histological preparations of seminiferous tubules
taken from normal
(N)
and pEM animals show theEffects of PEM on Sperm Quality and Spermatogenesis
difference
of
the
rubules diameter (Fig. 9 and Fig.10).
Figure 9' Histological preparation showing the seminiferous tubule of normal diet (N) animal; K (C), untreated control; Kp (CT),
treated,/placebo control; T (P), injected by TE. Compare the smaller tubule diameter of treated animal.
[image:8.595.29.513.44.399.2] [image:8.595.19.529.67.684.2]tn tr{n I CIO
Figure I0' Histological preparation showing the seminiferous rubule of protein energy malnutrition (pEM) animal; K (C),
t0 Eliza et aL
CONCLUSION
The effect of protein energy malnutrition (PEM) on spenn quality and spermatogenesis
of male rats
in-jected
with
TE, can be concluded as follow:l.
Sperm concentration, normal sperm morphologyand sperm viability was decreased, in both normal
diet and PEM animal; nevertheless there was no
difference between treated normal diet animals and
treated PEM animals.
2.
Testis weight, diameter of the testis, diameter of theseminiferous tubule, the number
of
spermato-gonium-A, and the numberof
pachytenesper-matocyte decreased in both normal
diet and
PEManimal; but there was a difference between treated
normal diet animals and treated PEM animals.
REFERENCES
l. Moeloek N. Kontrasepsi pria masa kini dan masa akan
datang. Presented at The VI Yearly Scientific Meeting,
In-donesian Andrological Society, Bandung, 1987.
2. Tadjudin MK. Cara keluarga berencana hormonal pada pria.
Proceeding
of National
KongresI,
IndonesianEn-d ocri nologi cal S oci ety, Jakarta, I 9 86;22-229.
3. Frick J, Danner Ch, Kunit G et al. Spermatogenesis in men treated with injection of mendroxyprogesterone acetate com-bined with testosterone enanthate. Int J Androl
1982;246-52.
4. Moeloek
N.
Penurunan kesuburan pria pada penyuntikantestosteron (TE) + DMPA dan l9 Nortestosteron heksilok-sifenil propionat ( I 9-NT) + DMPA. Dissertation, Faculty of
Medicine University of Indonesia, Jakarta, 1991.
Med J Indones
5. Word Health Organization. Contraceptive efficasy
of
tes-tosterone induced azoospermia
in
normal men. Lancet,1990; 336: 955-9.
6. Waites GMH. The research strategy of the WHO task force
on methods for the regulation of male fertility. In:
Perspec-tives in Andrology (Serio, M, ed), Raven Press, New York. 989;3-516.
7. Hamilton GD, Bronson FH. Food restriction and reproduc-tive development male and female mice and male rats.Am J
Physiol 1986; 250: 37 0-6.
8. Vawda AL, Mandlevana JG. The effect of dietary protein
deficiency on rat testicular function. Int J Androl 1980;
22:575-83.
9. French FS, Ritzen EM. Androgen binding protein (ABP) in efferent duct fluid of rat testis. J Reprod Ferfil., 1973;
32:479-83.
10. Nainggolan M. Experimental design (perencanaan dan
pengerjaan percobaan). GIS Research Institute, Medan Branch, Medan, Sumatra, 1965.
I 1. Sediaoetama AD. Faktor gizi. Bharata Karya Aksara,
Jakar-ta,1985.
12. CIermontY. Quantitative analysis of spermatogenesis of the
rat : A Revised Model for the Renewal of Spermatogonia. Am J Anat 1972:'lll: 111-29.
13. Franchimont P. Hormonal requalition of testicular function.
In : Regulation of Male Fertility. Cuningham GR, Sehell WB, Hafez ESE (eds). Martinus Nifhoff Publisher, London.
1980;5-14.
14. Lermite V, Tergui M. Plasma sex steroid binding protein in mature Heifers: Effect of the productive status, nutritional level, and porcine growth hormone, and tradiol-l78 treat-ment. Bio Rep 1991; 44:864-70.
15. Street C, Herry RJS, Al-Othman S, Chard T. Inhibition of
binding of gonadal steroid to serum binding protein by
P achyt en e s P ermat o cY t e
DISCUSSION
injection.
the TE injection.
this declining was also due to exogenous testosterone' because there was no interaction between TE injection and PEM.
Med J Indones
lvent (wijen oil). Acord-il contained 437o of
WA
essential fattY acid which mbrane integritY'Our finding showed that both in normal (N) and PEM'
fe
ini""tià'n caused decrease of weight and diameteroitt
"
t"ttit
and also seminiferous tubule diameter' As has been mentioned earlier,TE
injection can sup-Dressedthe
spermatogenesisthrough
feedbackIt""ft*it*t
acËordingly the number of germinal cell*irttin
the
tubules*itt
utto
decreased' Soit
isr"uto*tf"
that the diameterof
the tubules and the àiurn"t", of the testis and also the weight of the testisà""i"ut"a. But for PEM group another factor' protein ""r-r1y
^tl"utrion
(PEMI should be considered'since therJ was a difference
between{E,iliiïîï"iffiï,:
grouP),
for all
three s diameter and tubule tatistical analYsis suPPort such preposition, since there was positive interaction betwËenlacbr TE injection and PEM'According to T ermite and Tergui 14 reported that nutri-tional status seem to be an additional factor regulating
ir*-tt"roU
binding protein (SBP) level which may alter the percentageof
SBP available for positive ori"g",i*
ieedback; the decline of SBP' results in the inJreasine concentrationof
free androgenin
serum' 'Stree*eiat,l5
found that nonesterifiedfatty
acids modify binding affinitiesof
sex steroid hormone to SBPt;
vitro' Since in this experiment PEM group is undernourish, itis
predicted that supply of fatty acidand
protein
is
very limited;
it
follows
by
theavaitaUitity
of
SBPin
the blood, more overif
the affinity to testosteroneis
alsolow,
then the blood free téstosterone increases sharply upon the introduc-tion of exogenous testosterone; followed by stronger feedback michanism in PEM group compared tonor-mal
(N)
group. Thus,this
conditionwill
induce,t
ong"i
ùppàssionof
spermatogenesisin
PEM than in normal (N) grouP.The same explanation is hold true also for the number of A-spermatogonim and pachytene spermatocyte' so the decreasing number of both the germinal cells were due to the TE injection that hampered the proliferation during spermatogenests. Since there was a difference
U"t*J"tt fg injected in normal (N) and PEM animals concerning to the number of germinal cells' it was very
I ikely thaithe decreasin g number of A-sperm atogoni a
and iachytene spermatotyte in PEM animals was due