www.elsevier.com/locate/eja
E
ff
ects of water deficit and plant interaction on morphological
growth parameters and yield of white clover (
Trifolium
repens
L.) and ryegrass (
Lolium perenne
L.) mixtures
D.W. Lucero, P. Grieu *, A. Guckert
Unite´ associe´e INRA-Agronomie et Environnement, ENSAIA, 2 Avenue de la Foreˆt de Haye, F-54500 Vandœuvre-le`s-Nancy, France
Accepted 29 March 1999
Abstract
The effects of soil water deficit and interspecific plant interaction were studied on the dry matter (DM ) yield of white clover and ryegrass and on the morphogenesis of white clover. Plants were grown either: (1) individually (no interaction); or in a mixture of equal plant numbers with either (2) just shoot interaction, or (3) both shoot+root interaction. Plants were subjected to soil water deficits corresponding to no (0 MPa), moderate (−0.5 MPa) or severe (−1.0 MPa) water deficit. Ryegrass had higher and below-ground DM yields than white clover. The above-and below-ground DM yield for ryegrass, above-and the above-ground DM yield, stolon growth above-and relative growth rate (RGR), and leaf appearance rate (LAR) for white clover decreased as soil water deficit increased. Inshoot+root interactionat no and moderate soil water deficit levels, white clover had the highest proportion of above-ground growth in the leaf form (69%) and had, respectively, 11%and 32%more above-ground DM yield than when grown in justshoot interaction; ryegrass had, respectively, 20%and 25%more above-ground DM yield than ryegrass grown in justshoot interaction. In shoot+root interaction at severe soil water deficit, ryegrass had twice as much above-ground DM yield as white clover (3.50 g per plant versus 1.59 g per plant, respectively) and white clover had 60% less above-ground DM yield than when grown in justshoot interaction. In a soil column of restricted depth (30 cm) at no and moderate soil water deficit levels, remarkable increases in shoot biomass yield were observed for both white clover and ryegrass grown inshoot+root interaction. The increased shoot biomass yield of ryegrass can be attributed to benefits from white clover’s N fixing ability, whereas the causes for increased white clover biomass yield need to be studied further. However, at severe soil water deficit, ryegrass had a competitive advantage over white clover when grown in shoot+root interaction. This was due to the larger root system of ryegrass and its ability to control transpirational losses more efficiently, thus prolonging its growth period compared with white clover. © 1999 Elsevier Science B.V. All rights reserved.
Keywords:Biomass yield; Grass–legume mixtures;Lolium perenne; Morphology; Plant interaction; Ryegrass;Trifolium repens; Water deficit; White clover
* Corresponding author. Tel.:+33-03-8359-5856; fax:+33-03-8359-5799.
E-mail address:[email protected] (P. Grieu)
1. Introduction Preference) at the Ecole Nationale Superieure
d’Agronomie et des Industries Alimentaires
White clover (Trifolium repens L.) is commonly (ENSAIA, Nancy, France). In March of 1995
used as a perennial forage legume in the temperate seedlings were grown in specially constructed PVC
zones of both hemispheres. Usually, white clover is tubes and boxes (described below). Three
composi-grown with one or more grass companions, the most tions of white clover and ryegrass plant interaction
common being perennial ryegrass (Lolium perenne were combined with three levels of soil water
L.) (Hill and Michaelson-Yates, 1987). Grown deficit in a randomized complete block experiment
together, white clover and ryegrass are major
contrib-with four replications. White clover was inoculated utors to the productivity and quality of pastures.
with Rhizobium triflii SB116 on day 10. From In mixture, grasses and legumes compete for
week four to nine a complete nutrient fertilizer water, light and nutrients and may also compete for
solution (Robin et al., 1992) was applied once per O
2, CO2and space (Haynes, 1980). Mixture studies week. This nutrient solution was slightly modified
have often looked at the effects of cutting and
in order to supply both white clover and ryegrass grazing ( Wilman and Asiegbu, 1982; Evans and
with N, P, and K at rates equivalent to Williams, 1987; Woledge et al., 1992a), inorganic N
165 kg ha−1, 60 kg ha−1 and 60 kg ha−1, respec-and temperature (Davidson respec-and Robson, 1986;
tively. Average daily maximum and minimum air Ledgard, 1991; Nesheim and Boller, 1991) and light
temperatures were 26±4°C and 13±3°C,
and photosynthesis (Dennis and Woledge, 1985;
respectively. Thompson and Harper, 1988; Woledge et al., 1992b),
as well as competition (Martin and Field, 1984;
2.2. Plant interaction treatments Menchaca and Connolly, 1990; Lu¨scher et al., 1992).
The dynamics of water availability and use are
White clover and ryegrass were grown as fol-essential factors differentiating plant survival in a
lows: (1) individually (no interaction); (2) shoot pasture. Little, however, is known concerning
only interaction (shoot interaction); and (3) both white clover and ryegrass response to water deficit
root and shoot interaction (shoot+root
inter-in a mixed pasture. Reduced white clover growth
action). White clover and ryegrass grown in no in pasture mixtures under drought has been
attrib-uted to its less extensive rooting system ( Thomas, interaction were seeded into plastic PVC tubes
1984; Guobin and Kemp, 1992). Yet the capacity 5 cm in diameter and 30 cm in depth filled with
of white clover to extract water from deep soil soil made of a quartz sand (filtered to pass a 2 cm
reserves has also been reported (Guckert et al., sieve) added to a potting mix at a weight
propor-1993). Therefore, the results concerning white tion of 70:30 and packed to simulate a bulk density
clover are conflicting. This greenhouse study was of 1.25±0.10 g cm−3. Individual PVC tubes were
conducted to determine the effects of soil water spaced at intervals of 20 cm. Forshoot interaction,
deficit and interspecific plant interaction on the 20 PVC tubes (10 white clover and 10 ryegrass
dry matter (DM ) yield of white clover and ryegrass plants), identical in nature to those containingno
and on the morphogenesis of white clover. White interactionplants, were placed in a 5×4 grid in an
clover and ryegrass were grown in a shallow soil, alternative pattern in specially constructed crates
either individually (no interaction) or in mixtures (30 cm×24 cm×30 cm). The 14 plants on the
of equal numbers with just shoot interaction or outer edge of the 5×4 grid were defined as border
with bothshoot+root interaction. plants and were not included in experimental
meas-urements. Each PVC tube was placed in a drilled hole of diameter slightly larger than the PVC tube
2. Methods and materials itself. Holes were drilled in a sheet of 5 mm
ply-wood. The plywood sheet was supported to the 2.1. Experimental conditions
same height as the lip of each PVC tube. In this manner the 5 mm plywood sheet became the sup-A greenhouse experiment was conducted with
as the artificial soil level. Seeds of white clover and ber (Scho¨lander et al., 1965) at three dates in order to verify plant water stress levels. Soil water deficit ryegrass were sown in the center area of each tube,
hence the spacing between plants was 6 cm. Thus, levels were obtained at day 66 after sowing. Tubes
and boxes containing plants were thereafter while the above-ground canopy was allowed to
grow together, the PVC tubes kept the below- weighed every 2 days and at that time the water
lost to transpiration was added in order to bring ground plant parts completely separated. In the
shoot+root interactiontreatment, white clover and tube and box weights back to soil water deficit levels (due to plant cover soil evaporation was ryegrass were seeded not into plastic PVC tubes
but into plastic boxes (of the same dimension as negligible). For bothno interactionandshoot
inter-actiontreatments, the tubes containing white clover the shoot interaction crates, i.e. 30 cm×24 cm×
30 cm) in an identical number (10 white clover or ryegrass were weighed individually. For plants
grown inshoot interactionthis required the careful and 10 ryegrass plants) and in an identical pattern
to that of plants in theshoot interactiontreatment lifting up (by approximately 1 cm) of the tube
containing the plant in such a manner as to
(5×4 grid, 6 cm spacing). Hollow plastic PVC
tubes identical to those in whichno interactionand minimize disturbance of the developed
above-ground canopy. Once weighed, the tube containing shoot interactionplants were grown were sealed at
both ends and placed alternatively between the the plant was gently re-lowered into the
above-ground undisturbed canopy. For the shoot+root
seeded white clover and ryegrass plants in the
plastic boxes of the shoot+root interaction treat- interaction treatment, boxes were weighed in
entirety. Once soil water deficit levels were ment. These hollow PVC tubes were thus placed
in order to both simulate similar rooting conditions obtained, plants were subjected to soil water
defi-cits for a 37 day period before harvest. and to assure that the soil volume per plant was
identical for each of the three plant interaction
treatments. Plants in the shoot+root interaction 2.4. Dry matter yield
treatment were allowed to develop with both
above- and below-ground plant parts in complete Two replications (108 total plants) were
har-vested on day 102 and the other two on day 103 interaction.
after sowing. All plants were separated into above-and below-ground material. Above-ground white 2.3. Soil water deficit levels
clover plant parts were further separated into leaf lamina, petioles, stolons and flowers. Roots of Soil water deficit levels were allowed to develop
gradually by the progressive withdrawal of water both white clover and ryegrass were carefully
washed with a jet of tap water to remove soil. In at day 50, at which time the leaf area index (LAI )
for theshoot interactionand theshoot+root inter- order to minimize root loss during washing the
plant roots were individually rinsed in wooden actiontreatments was approximately 3.
Pre-experi-mental tests using identical PVC plastic tubes, boxes with a base made of a 2 mm mesh screen
and the root material separated during the washing boxes and soil had determined the soil weight
(70% and 55% of field capacity, respectively) per process was carefully gathered and included for
plant root DM measurements. Harvested plant tube and box necessary to obtain the desired plant
water stress levels. Plant water stress levels were parts were immediately dipped in liquid nitrogen,
stored at−30°C until freeze drying and weighed.
based on pre-dawn leaf water potentials of white
clover and ryegrass, i.e. no (−0.0 MPa), moderate The harvest of the shoot interaction and the
shoot+root interactiontreatments necessitated the
(−0.5 MPa) and severe (−1.0 MPa). Soil water
deficits corresponding to plant stress below careful separation of the above-ground canopy.
Measurements were carried out on the six plants
−1.0 MPa drastically reduce growth and can lead
to plant death (Guckert et al., 1993). During the in the inner 12×18 cm2experimental area. For the
shoot+root interaction treatment, root parts also experiment, pre-dawn leaf water potentials were
clover and 12 ryegrass plants were harvested for each interaction treatment and soil water deficit level.
2.5. Morphological measurements
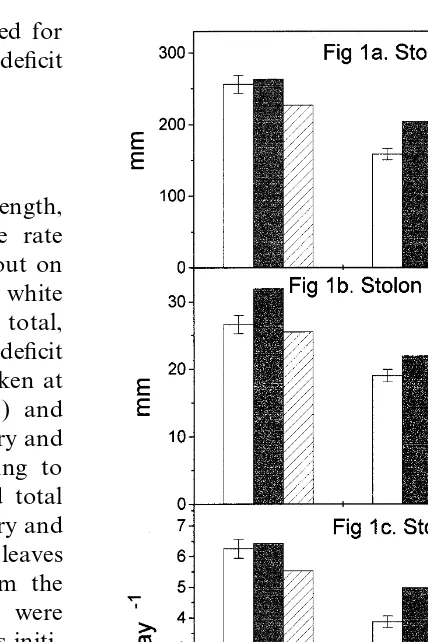
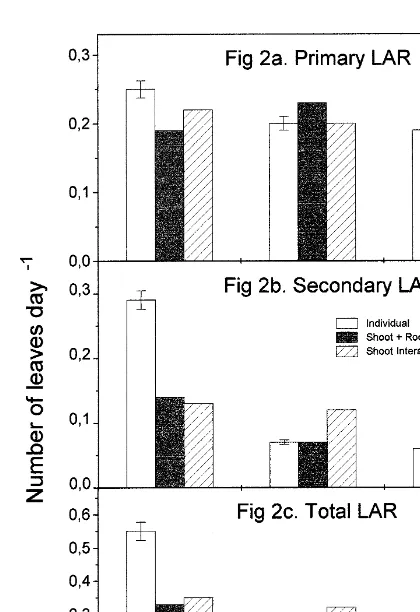
Non-destructive measurements [stolon length, internode length, primary leaf appearance rate (LAR) and secondary LAR] were carried out on two randomly selected stolons from two white clover plants per replication (72 plants in total, eight plants per interaction and soil water deficit level ). Measurements for time zero were taken at the establishment of water stress (day 66) and thereafter on days 77, 88, 96 and 102. Primary and secondary LARs were determined according to criteria established by Carlson (1966), and total leaf index was a combination of both primary and secondary LARs [see Fig. 2(a)–(c)]. Primary leaves were defined as those leaves growing from the primary stolon axis and secondary leaves were defined as those leaves developing on stolons initi-ated from the main axis of selected stolons. The relative growth rate (RGR) of stolons and leaves was a function of growth divided by time [Figs. 1(c), 2(a)–(c)]. At each measurement, stolon length, from one internode to another, was recorded and average internodal length was a
Fig. 1. Stolon length, stolon internodal length and stolon
rela-function of the lengths of all internodes summed
tive growth rate (RGR) of white clover plants grown
individu-and divided by the number of internodes. ally, in shoot+root interaction, or justshoot interaction with
ryegrass and subjected to three levels of water deficit. Error bars on observed data indicate±1 standard deviation.
2.6. Statistical analysis
The effect of treatments was obtained from highest root DM yield, and plants grown in
shoot+root interactionthe lowest root DM yield. analyses of variance. Mean separations were
per-formed by an LSD procedure where theFvalues For ryegrass grown in no interaction, an increase
in soil water deficit from no to moderate reduced were significant at the 0.05 probability level (SAS
Institute, 1990). root DM yield by over 50% and an additional
increase to severe soil water deficit further reduced
root DM yield by 36%.
3. Results
3.2. Above-ground dry matter yield 3.1. Below-ground dry matter yield
Shoot DM yield decreased as soil water deficit increased ( Table 1). Plants grown inno interaction Root DM yield tended to decrease as soil water
deficit increased (Table 1). Ryegrass produced at no soil water deficit had the highest shoot DM
yields. At no and moderate soil water deficit, more root DM than white clover at each soil water
ilar. Ryegrass grown in shoot+root interaction
had, respectively, 19% and 25% higher DM yields
at no and moderate soil water deficit than ryegrass grown in justshoot interaction. An increase in soil water deficit from no to moderate level decreased
white clover shoot DM yield by on average 35%;
however, for white clover grown in shoot+root
interaction this decrease was less (26% compared
with 40% and 38%, respectively for white clover
plants grown in no interaction or in shoot
inter-action). On the other hand, an increase in soil water deficit from no to moderate level reduced
ryegrass shoot DM yield by on average only 24%
(range 20 to 29%). As soil water deficit level
increased, the differences in DM yield observed
between the different interaction treatments tended to decrease. However, this relationship was not
true for plants grown in shoot+root interaction,
where an increase in soil water deficit from moder-ate to severe level significantly increased the reduc-tion in white clover shoot DM yield. Though shoot DM yield was higher for ryegrass grown in shoot+root interactioncompared withshoot inter-action, an increase in soil water deficit from moder-ate to severe level decreased ryegrass shoot DM
yield by 32% in shoot+root interaction but only
20%inshoot interaction.
Fig. 2. Primary, secondary and total leaf appearance rate (LAR) of white clover plants grownindividually, inshoot+root
3.3. White clover leaf and stolon growth
interaction, or justshoot interactionwith ryegrass and subjected to three levels of water deficit. Error bars on observed data
indicate±1 standard deviation. At no and moderate soil water deficit white
clover grown in shoot+root interaction had
sig-nificantly higher leaf DM yield than white clover
shoot DM yield than plants grown inshoot
inter-action. At moderate soil water deficit ryegrass had grown inshoot interaction( Table 2). This increase in total leaf DM yield was in the form of petioles higher shoot DM yield than white clover, whereas
at severe soil water deficit ryegrass had higher as leaf lamina DM yield was similar for white
clover grown in shoot+root interaction or shoot
shoot DM yield than white clover only when
grown in shoot+root interaction. At severe soil interaction. Both leaf and stolon DM yields
decreased as soil water deficit increased. At no and
water deficit the white clover grown in no
inter-action and in shoot interaction had, respectively, moderate soil water deficit, white clover grown in shoot+root interactionhad a higher percentage of
107% and 57% more shoot DM yield than
white clover grown in shoot+root interaction. shoot DM in the form of leaf tissue than did white
clover grown in shoot interaction (69 and 68.5%
Conversely, at no and moderate soil water deficit,
white clover grown inshoot+root interactionhad, compared with 62 and 63%, respectively). At
mod-erate soil water deficit, white clover grown in
respectively, 11% and 32% more shoot DM yield
than white clover grown in shoot interaction and shoot+root interaction had 20% and 40% more
leaf DM yield than white clover grown in either
the shoot DM yields of white clover grown inno
Table 1
Dry matter yield and root/shoot ratio of white clover and ryegrass plants grown eitherindividually, withshoot interactionor with bothshoot+root interactionin the greenhouse and subjected to three levels of soil water deficit
Treatment Dry matter yielda(g plant−1) (Assoc./water stress) level )
Below-ground Above-ground Total Root/shoot ratio
WC RG WC RG WC RG WC RG
Individual/0.0 MPa 0.91 cdeb 3.52 a 6.84 ab 7.42 a 7.75 b 10.94 a 0.13 cd 0.48 e Individual/−0.5 MPa 0.71 efgh 1.66 b 4.07 e 5.27 c 4.78 fgh 6.93 bcd 0.17 de 0.31 de Individual/−1.0 MPa 0.57 ghi 1.05 cd 3.30 fg 3.75 ef 3.87 ij 4.80 fgh 0.17 de 0.28 d Shoot/0.0 MPa 0.60 fghi 1.12 c 5.17 cd 5.39 cd 5.67 def 6.51 cde 0.12 bc 0.21 cd Shoot/−0.5 MPa 0.64 fghi 1.02 cd 3.19 fg 4.09 e 3.83 hij 5.11 efg 0.20 e 0.25 cd Shoot/−1.0 MPa 0.45 hij 0.81 defg 2.50 g 3.28 fg 2.95 jk 4.09 ghij 0.18 e 0.25 d Shoot+root/0.0 MPa 0.44 ij 0.86 def 5.75 b 6.40 ab 6.19 cde 7.26 bc 0.08 a 0.15 a Shoot+root/−0.5 MPa 0.45 hij 0.84 def 4.21 e 5.11 cd 4.66 ghi 5.95 def 0.11 b 0.16 b Shoot+root/−1.0 MPa 0.29 j 0.65 fghi 1.59 h 3.50 efg 1.88 k 4.15 ghij 0.18 e 0.19 bc
aBoth WC and RG treatments compared within below-, above-ground, total and root/shoot ratio columns. bLSD followed by different letters are significantly different (P≤0.05).
Table 2
White clover leaf, stolon and flower dry matter and percentage of above-ground yield from plants grown in the greenhouseindividually, withshoot interactionor with bothshoot+root interactionwith ryegrass and subjected to three levels of soil water deficit
Treatment Final harvest dry matter yield (Assoc./water stress)
Petiolea(g) Leaf (g) Leaf (g) Leaf (%) Stolon (g) Stolon (%) Total leaf Flower
Individual/0.0 MPa 2.05 ab 2.18 a 4.33 a 63.3 2.20 a 34.1 97.4 2.6 Individual/−0.5 MPa 1.08 cd 1.32 c 2.40 c 59.0 1.43 cd 35.1 94.1 5.9 Individual/−1.0 MPa 0.83 de 1.00 de 1.83 d 55.4 1.17 de 35.5 90.9 9.1
Shoot/0.0 MPa 1.56 b 1.64 b 3.20 b 61.9 1.82 b 35.2 97.1 2.9
Shoot/−0.5 MPa 0.98 cd 1.04 de 2.01 d 63.0 1.11 de 34.8 97.8 2.2 Shoot/−1.0 MPa 0.72 de 0.83 ef 1.55 de 62.0 0.89 e 35.6 97.5 2.4 Shoot+root/0.0 MPa 2.18 a 1.79 b 3.97 a 69.0 1.61 bc 28.0 97.0 3.0 Shoot+root/−0.5 MPa 1.66 b 1.22 cd 2.88 c 68.5 1.30 cde 31.0 99.5 0.5 Shoot+root/−1.0 MPa 0.39 e 0.58 f 0.97 f 61.0 0.61 f 38.4 99.4 0.6
aTreatments compared only within columns.
bLSD column means followed by different letters are significantly different (P≤0.05).
Conversely, at severe soil water deficit, white clover leaf decreased and flower increased (as a
percen-tage of shoot DM yield ) as soil water deficit
grown inno interactionor inshoot interactionhad
90% and 45% higher leaf DM yields, respectively increased.
than white clover grown inshoot+root interaction.
Stolon as a percentage of shoot DM yield increased 3.4. Root and shoot ratios
as soil water deficit increased for white clover
grown inshoot+root interaction, and at severe soil Root/shoot ratios were highly variable for all
treatments ( Table 1). For white clover, roots
water deficit made up almost 40%of above-ground
DM yield (compared with 28%and 31%at no and tended to increase in proportion to shoots as soil
water deficit level increased. This effect was most moderate soil water deficit, respectively). For white
inter-action, but was due more to a decrease in shoot Increasing soil water deficit had a much greater
effect on secondary LAR than on primary LAR
growth than to an increase in root growth, as soil
volume in this experiment was restricted. [Fig. 2(b)]. White clover subjected to no soil water
deficit and grown in no interaction had twice as
high a secondary LAR as the other two interaction 3.5. Stolon growth and relative growth rate
treatments. Total LAR was highest for white clover
grown in no interaction at no soil water deficit,
Average stolon and stolon internodal length
decreased as soil water deficit increased [Fig. 1(a) almost twice as much as the other interaction
treatments and soil water deficits [Fig. 2(c)]. At and (b)]. At no and moderate soil water deficit,
average stolon and stolon internodal length was moderate soil water deficit a similar total LAR
was observed for each interaction treatment; how-highest in white clover grown inshoot+root
inter-action, followed by plants grown inno interaction ever, the total LAR of white clover grown in shoot+root interaction was the most severely
and then by plants grown inshoot interaction. At
severe soil water deficit, white clover grown in reduced when soil water deficit was severe.
shoot+root interaction had shorter stolons than
white clover grown in either no interaction or in
shoot interaction. For average stolon internodal 4. Discussion length this same relationship was true for white
clover grown at each soil water deficit level. Van Loo (1992) observed that root growth of
ryegrass plants grown individually was reduced Stolon RGR decreased as soil water deficit
increased [Fig. 1(c)]. The RGR of stolons was more than shoot growth at similar soil water deficit
levels to those used in this study. In our study, substantially higher at no and moderate soil water
deficit than at severe soil water deficit for white increased soil water deficit decreased root DM
yield for ryegrass grown in no interaction. Plant
clover grown in shoot+root interaction. Stolon
RGR for white clover grown inshoot+root inter- interaction (shoot or shoot+root) lessened the
effects of reduced ryegrass root DM yield that was actionat moderate soil water deficit was as high as
that observed for white clover grown in shoot observed in ryegrass grown inno interaction. That
white clover root DM yield was only slightly interaction at no soil water deficit. At no and
moderate soil water deficit, the order of white clover affected by increased soil water deficit could be
attributed to the smaller root system of white
stolon RGR was: shoot+root interaction>no
interaction>shoot interaction; however, at severe clover compared with grasses ( Thomas, 1984). In a restricted soil area, such as that used in this soil water deficit this trend was: no interaction>
shoot interaction>shoot+root interaction. study, the degree to which white clover root sys-tems could have been reduced by soil water deficit was thus less than for ryegrass.
3.6. Leaf appearance rate
Wilson (1988) reported that root competition had a greater effect than shoot competition in 70% Highest primary LAR was observed for white
clover grown in no interaction at no soil water of 47 cases found in the literature and concluded
that root competition is usually more important deficit [Fig. 2(a)]. White clover grown inno
inter-actionhad higher primary LAR at each soil water than shoot competition in determining competitive balance, the intensity of competition, and resource
deficit level than white clover grown in shoot
interaction. The highest primary LAR observed at use. Root DM yield of ryegrass was double that of white clover in this experiment ( Table 1). Jupp moderate soil water deficit level was for white
clover grown in shoot+root interaction. White and Newman (1987) observed that water-stressed
ryegrass plants had three to five times more lateral
clover grown inshoot+root interactionat
moder-ate soil wmoder-ater deficit had as high a primary LAR root initiation and growth than controls at severe
soil water deficit. The ability of one plant to extract
as white clover grown in either no interaction or
enhanced by greater root densities. In this study, shootorshoot+root, offset the propensity of white clover to flower under water stress ( Table 2). ryegrass grown inshoot+root interactionat severe
soil water deficit was more competitive for soil Generally, white clover spreads vegetatively by the
growth of stolons and, therefore, is continually water than white clover. Burch and Johns (1978)
have also observed that white clover grown in establishing new, adventitious root systems from
stolon nodes. The long-term sustainability of white
competition with Festuca had poorer control of
leaf transpiration, which resulted in low leaf water clover in mixed swards subjected to soil water
deficits could, however, still be compromised by potentials and increased leaf senescence. Grasses,
on the other hand, have better stomatal control the reduction in inflorescence caused by plant
interaction observed in this study. and higher leaf water potential, which increased
and prolonged grass growth in drought conditions The increased DM yield observed at no and
moderate soil water deficits for ryegrass grown in (Johns and Lazenby, 1973; Thomas, 1984). This
may further explain why, in this study, reductions shoot+root interactioncompared withshoot
inter-action with white clover could be attributed to in the shoot DM yield of white clover subjected
to severe soil water deficit were considerably increases in N availability through N fixed by
white clover, though this was not measured in greater than for ryegrass, and especially when the
two plants were grown inshoot+root interaction. our study. The transfer of N fixed
atmospheri-cally from white clover to companion grasses is Stolon growth and RGR and LAR of white
clover were markedly reduced by increased soil well documented and can amount to about
70 kg ha−1 per year (Ledgard, 1991); this may
water deficit ( Turner, 1991; Belaygue et al., 1996).
In our study,shoot+root interaction lessened the explain why ryegrass grown in shoot+root
inter-actionwith white clover produced more shoot DM reduction in stolon growth due to soil water deficit
that was observed for white clover grown inshoot than when grown in just shoot interaction with
white clover. Shamsun-Noor et al. (1989) studied interaction. Beinhart (1963) has shown that the
most marked effect of shading on white clover was the effects of drought on white clover and reported
that N fixation rates were maximum at soil water the reduction in the formation of stolons from
auxiliary buds. In this study, stolon length and deficits of between 60 and 70%, a level quite similar
to that used in this study to define moderate soil
RGR were markedly reduced byshoot interaction
(compared with plants grown in no interaction), water deficit. The optimization of N fixation by
white clover at this level of soil water deficit has yet at no and moderate soil water deficit levels
stolon length, internodal length and RGR were also been observed for white clover grown in sandy
soil by Laperrie`re (1984) and Holter (1978) and highest for plants grown inshoot+root interaction
[Fig. 1(a)–(c)]. by Wahab and Zahran (1983) for red clover and
alfalfa. In our experiment, the removal of soil
For white clover grown in no interaction or in
shoot interaction, increased soil water deficit did inorganic N by the roots of ryegrass grown in interaction with white clover would have led the not change the percentage of stolon as part of
shoot DM yield ( Table 2). However, increased soil companion white clover plant to increase N
fixa-tion in order to obtain sufficient N for growth.
water deficit did change leaf as a percentage of
shoot DM yield in white clover plants grown in Inorganic N can inhibit many phases of the N
fixing process (Postgate, 1982). However, Morris no interaction or in shoot+root interaction. For
plants grown inno interaction, flowers as a percen- and Weaver (1987) have observed that N fertilizer
rates did not significantly reduce the amount of N tage of shoot DM doubled at each increase in soil
water deficit — this increase was at the expense of fixed by clover in clover and ryegrass mixtures due
to grass removal of soil inorganic N. leaf DM yield. It has been observed that soil water
deficit can affect the balance between vegetative Additionally, at no and moderate soil water
deficit, the shoot DM yield of white clover grown and reproductive growth in white clover plants
( Turner, 1991) and that long-term soil water deficit in shoot+root interaction was higher than when
grown in just shoot interaction ( Table 1). This
promotes the production of inflorescences ( Turner,
especially petiole. Also, white clover grown in soil water deficit (Burch and Johns, 1978). Burch shoot+root interaction at no and moderate soil and Johns (1978) have also shown that, owing to
water deficit had the highest percentage of shoot white clover poor stomatal control during a severe
growth in the form of leaf (69 and 68.5%) and the drought, leaf senescence is a mechanism used by
lowest in the form of stolon (28 and 31%) white clover to balance rate of transpiration rate
( Table 2). Though stolon, as a percentage of and water uptake. In our experiment, white clover
above-ground DM, was the lowest of all treat- drought avoidance in response to severe soil water
ments, it must be noted that stolon length, inter- deficit can explain the low shoot DM yield
nodal length and RGR were still the highest of all (Table 1) and more importantly the low leaf DM
treatments [Fig. 1(a)–(c)]. As mentioned pre- yield as a percentage of shoot DM yield observed
viously, the moderate soil water deficit level used for white clover grown in shoot+root interaction
in this experiment has been shown to optimize (Table 3). The more efficient control of stomatal
white clover N fixation (Laperrie`re, 1984; closure and ability to reduce transpiration by leaf
Shamsun-Noor et al., 1989), which would have rolling in response to severe soil water deficit
favored white clover DM yield. Additionally, allowed ryegrass to prolong its growth period and
Tomm et al. (1994), in a short-term N-transfer furthered ryegrass competitiveness for limited soil
experiment, showed that N is transferred from water when grown in shoot+root interaction in
both the N-fixing legume to an associated non- our experiment.
legume and also from the non-legume to the Though no N fixation parameters were
mea-N-fixing legume. In this manner, where there was sured in our experiment, the reduction in shoot
shoot+root interactionin our experiment between DM yield observed for white clover grown in ryegrass and white clover, increased N from white
shoot+root interactionat severe soil water deficit clover fixation that benefited ryegrass DM yield
level might have been due to a reduction in atmo-could also have benefited white clover growth. The
spherically fixed N by white clover nodules. Wilson
question of ryegrass shoot+root interaction
(1931) first reported that drought adversely increasing the soil mineral uptake by white clover
affected legume root nodules. Engin and Sprent
must also be asked. Whatever the reason, the
(1973) attributed the initial effect of drought on
response of white clover grown in shoot+root
interaction to produce more petiole leaf material
would have favored white clover photosynthetic Table 3
Variance ratios for plant interaction (PI ), soil water deficit
activity by positioning a maximum amount of leaf
( WD) and plant interaction by soil water deficit (PI×WD) for
lamina to receive sunlight. A higher efficiency of
ryegrass and white clover plant part dry matter yield
white clover to intercept light has been observed
in mixed swards (Dennis and Woledge, 1985; Dry matter F association F water F interaction
Woledge, 1988). Increased light interception leads yield deficit
to increased plant photosynthetic activity, which
Ryegrass
would affect C assimilation as well as other plant
Below ground 77.50*** 37.90*** 24.25***
metabolic processes such as water use, mineral
Above ground 7.99*** 23.02*** 2.16
uptake and N fixation. Total 21.71*** 35.03*** 6.61***
The severe reduction in DM yield, especially
White clover
leaf growth, observed for white clover grown in
Below ground 31.00*** 12.24*** 2.00
shoot+root interaction treatment at severe soil Above ground 1.19 71.14*** 4.19**
water deficit compared withshoot interactiontreat- Total 2.20 69.52*** 4.13**
Petriole 8.49*** 72.48*** 3.09**
ment ( Table 1) may be due to several factors.
Leaf lamina 1.03 50.09*** 4.02**
First, at severe soil water deficit, white clover is
Leaf total 1.55 16.87*** 3.28**
unable to reduce either relative canopy
conduc-Stolon 6.78** 45.45*** 1.70
tance or rate of leaf transpiration as efficiently as
ryegrass and, therefore, is not as well adapted as **Significant atP=0.01.
***Significant atP=0.001.
white clover to reduction in the N fixing capacity in shoot+root interaction. Ryegrass increased
of nodules and noted that the severity of nodule shoot biomass yield can be attributed to benefits
response depended on the length and degree of the from white clover’s N fixing ability, whereas the
water stress. In our study, the 37 day period of causes for increased white clover biomass yield
water stress at the plant level would have been of need to be studied further. However, at severe soil
sufficient length to have led to the reduction in the water deficit, ryegrass had a competitive advantage
N fixing capacity of white clover nodules. Butler over white clover when grown inshoot+root
inter-and Ladd (1985) found a direct decrease in action. This was due to the larger root system of
MedicagoN fixation due to ryegrass competition ryegrass and its ability to control transpirational
and indicated that N fixation was directly related losses more efficiently, thus prolonging its growth
to legume weight. In our study, the DM yield of period compared with white clover.
white clover grown inshoot+root interactionwith To explain these findings more fully, studies are
ryegrass at severe soil water deficit was 60% of needed that look at the influence of white
that when grown inshoot interactionwith ryegrass. clover/ryegrass root interaction on (1) plant soil
In addition, the increased DM yields observed in carbon allocation, and (2) the plant use of soil
ryegrass grown in shoot+root interaction with resources other than water.
white clover at no and moderate soil water deficit levels was not observed at severe soil water deficit,
Acknowledgements which further indicates that N fixation by white
clover was most probably reduced by severe soil
The first author was supported as an American water deficit. Butler and Ladd (1985) additionally
Fulbright Scholar in France by the Franco– noted that during plant competition the observed
American Commission for Educational Exchange, increases in the DM yield of ryegrass were directly
to whom thanks are due. Additional appreciation related to an equal percentage decrease in the
is extended for funding received from the Integrated amounts of fixed N by the companion clover. The
Program GALILEE, a Franco–Italian collabora-reduced DM yield of white clover observed in our
tion partially funded by the Ministe`re Franc¸ais des experiment at severe soil water deficit would not
Affaires Etrange`res. The authors are very grateful only have decreased the capacity of white clover
for the tremendous help given by all Ph.D. students to fix N, but further allowed ryegrass to become
and technical staff of the Laboratoire Agronomie
much more aggressive for limited available soil
et Environnement, ENSAIA, Nancy, France, as water. In our experiment at severe soil water
well as innumerable others who helped in the
deficit, white clover made up only 31% of the
measurements and harvests. shoot canopy DM yield at harvest when grown in
shoot+root interaction with ryegrass, whereas
when grown in no interaction or in just shoot
References interaction these percentages were 49% and 43%,
respectively. Thus, as soil water deficit increased, Beinhart, C., 1963. Effects of environment on meristematic
the RGR of white clover decreased compared with development, leaf area and growth of white clover. Crop
Sci. 3, 209–213.
ryegrass. This has not been observed under
non-Belaygue, C., Wery, J., Cowan, A.A., Tardieu, F., 1996.
Contri-water limiting conditions for white clover and
bution of leaf expansion, rate of leaf appearance, and stolon
ryegrass subjected to various cutting regimes
branching to growth of plant leaf area under water deficit
( Woledge, 1988; Woledge et al., 1992a,b). in white clover. Crop Sci. 36, 1240–1246.
Burch, G.J., Johns, G.G., 1978. Root absorption of water and physiological responses to water deficits byFestuca arundi-naceaScreb. andTrifolium repensL.. Aust. J. Plant Phys.
5. Conclusion
5, 859–871.
Butler, J.H.A., Ladd, J.N., 1985. Growth and nitrogen fixation
At moderate soil water deficit in shallow soil, byMedicago littoralisin pot experiments: effect of plant
remarkable increases in shoot biomass yield were density and competition fromLolium multiforum. Soil Biol.
Biochem. 17, 355–361.
Carlson, G.E., 1966. Growth of clover leaves developmental arrowleaf clover–gulf ryegrass mixtures. Soil Sci. Soc. Am. J. 51, 115–119.
morphology and parameters at ten stages. Crop Sci. 6,
Nesheim, L., Boller, B.C., 1991. Nitrogen fixation by white 293–294.
clover when competing with grasses at moderately low tem-Davidson, I.A., Robson, M.J., 1986. Effect of temperature and
peratures. Plant Soil 133, 47–56. nitrogen supply on the growth of perennial ryegrass and
Postgate, J.R., 1982. The Fundamentals of Nitrogen Fixation. white clover 2. A comparison of monocultures and mixed
Cambridge University Press, New York. swards. Ann. Bot. 57, 709–719.
Robin, C., Varlet-Grancher, C., Gastel, F., Flenet, F., Guckert, Dennis, W.D., Woledge, J., 1985. The effect of nitrogenous
fer-A., 1992. Photomorphogenesis of white clover (Trifolium
tilizer on photosynthesis of white clover/perennial ryegrass
repensL.): phytochrome mediated effects on14C-assimilated sward. Ann. Bot. 55, 171–178.
partitioning. Eur. J. Agron. 1, 235–240. Engin, M., Sprent, J.I., 1973. Effects of water stress on growth
1990. SAS Users’ Guide: Statistics, Version 6. SAS Institute, and nitrogen-fixing activity ofTrifolium repens. New Phytol.
Cary, NC.
72, 117–126. Shamsun-Noor, L., Robin, C., Schontz, D., Heulin, T., Guck-Evans, D.R., Williams, T.A., 1987. The effect of cutting and ert, A., 1989. E
ffet d’un de´ficit hydrique sur le tre`fle blanc grazing managements on dry matter yield of white clover (Trifolium repensL.) I. Importance du cultivar. Agronomie varieties (Trifolium repens) when grown with S23 perennial 9, 251–257.
ryegrass. Grass Forage Sci. 42, 153–159. Scho¨lander, P.F., Hammel, H.T., Bradstreet, E.D., 1965. Sap Guckert, A., Robin, C., Grieu, Ph., Gras, F., 1993. Influence pressure in vascular plants. Science 148, 339–346.
du de´ficit hydrique sur le tre`fle blanc. Fourrages 135, Thomas, H., 1984. Effects of drought on growth and competi-369–381. tive ability of perennial ryegrass and white clover. J. Appl.
Ecol. 21, 591–602. Guobin, L., Kemp, D.R., 1992. Water stress affects the
produc-Thompson, L., Harper, J.L., 1988. The effect of grasses on the tivity, growth components, competitiveness and water
rela-quality of transmitted radiation and its influence on the tions ofPhalarisand white clover growing in mixed pasture.
growth of white clover Trifolium repens. Oecologia 75, Aust. J. Agric. Res. 43, 659–672.
343–347. Haynes, R.J., 1980. Competitive aspects of the grass–legume
Tomm, G.O., van Kessel, C., Slinkard, A.E., 1994. Bi-association. Adv. Agron. 33, 227–261.
directional transfer of nitrogen between alfalfa and brome-Hill, J., Michaelson-Yates, T.P.T., 1987. Effects of competition
grass: short and long term evidence. Plant Soil 164, 77–86. upon the productivity of white clover–perennial ryegrass
Turner, L.B., 1991. The effect of water stress on the vegetative mixtures: analysis of and interrelations between characters.
growth of white clover (Trifolium repensL.): comparison of Plant Breeding 98, 161–170.
long-term water deficit and a short-term developing water Holter, V., 1978. Nitrogen fixation of four legumes in relation stress. J. Exp. Bot. 42, 311–316.
to above-ground biomass, root biomass, nodule number and Turner, L.B., 1993. The e
ffect of water stress on floral charac-water content of the soil. Oikos 31, 230–235. ters, pollination and seed set in white clover (Trifolium repens Johns, G.G., Lazenby, A., 1973. Defoliation, leaf area index L.). J. Exp. Bot. 44, 1155–1160.
and the water use of four temperate pasture species under Van Loo, E.N., 1992. Tillering, leaf expansion and growth of irrigated and dryland conditions. Aust. J. Agric. Res. 24, plants of two cultivars of perennial ryegrass grown using 783–795. hydroponics at two water potentials. Ann. Bot. 70, 511–518. Jupp, A.P., Newman, E.I., 1987. Morphological and anatomical Wahab, A.M.A., Zahran, H.H., 1983. The effect of water stress
on N
2(C2H2) fixation and growth ofMedicago sativaL. Acta effects of severe drought on the roots ofLolium perenneL.
Agron. Acad. Sci. Hung. 32, 114–118. New Phytol. 105, 393–402.
Wilman, D., Asiegbu, J.E., 1982. The effects of clover variety, Laperrie`re, C., 1984. Etude de la fixation d’azote par le Trefle
interval and nitrogen application on herbage yields, propor-blanc. Aspects biologiques et agronomiques, Ph.D.
Disserta-tions and heights in perennial ryegrass–white clover swards. tion, INPL-ENSAIA, Nancy, France.
Grass Forage Sci. 37, 1–13. Ledgard, S.F., 1991. Transfer of fixed nitrogen from white
Wilson, J.K., 1931. The shedding of nodules by beans. J. Am. clover to associated grass in swards grazed by dairy cows
Soc. Agron. 23, 670 using15N methods. Plant Soil 131, 215–223.
Wilson, J.B., 1988. Shoot competition and root competition. Lu¨scher, A., Connolly, J., Jacquard, P., 1992. Neighbor
speci-J. Appl. Ecol. 25, 279–296. ficity betweenLolium perenneandTrifolium repensfrom a
Woledge, J., 1988. Competition between grass and clover in natural pasture. Oecologia 91, 404–409. spring as a
ffected by nitrogen fertilizer. Ann. Appl. Biol. Martin, M.P.L.D., Field, R.J., 1984. The nature of competition 112, 170–175.
between perennial ryegrass and white clover. Grass Forage Woledge, J., Reyneri, A., Tewson, V., Parsons, A.J., 1992a. The Sci. 36, 247–253. effect of cutting on the proportions of perennial ryegrass Menchaca, L., Connolly, J., 1990. Species interference in white and white clover mixtures. Grass Forage Sci. 47, 169–179.