Penanggung Jawab:
Kapuslit Metalurgi – LIPI
Dewan Redaksi :
Ketua Merangkap Anggota:
Ir. Bambang Sriyono Dipl.Ing.
Anggota:
Dr. Ir. Rudi Subagja Dr. Ir. F. Firdiyono Dr. Agung Imadudin Dr. Efendi Mabruri
Ir. Adil Jamali, M.Sc (UPT BPM – LIPI) Prof. Riset. Dr. Ir. Pramusanto (Puslitbang TEKMIRA)
Prof. Dr. Ir. Johny Wahyuadi, DEA (UI) Dr. Ir. Sunara, M.Sc (ITB)
Sekretariat Redaksi:
Dedi Irawan, ST
Daniel Panghihutan Malau, ST Arif Nurhakim, S.Sos
Penerbit:
Pusat Penelitian Metalurgi – LIPI
Kawasan PUSPIPTEK, Serpong, Gedung 470
Telp: (021) 7560911, Fax: (021) 7560553
Alamat Sekretariat:
Pusat Penelitian Metalurgi – LIPI
Kawasan PUSPIPTEK, Serpong, Gedung 470
Telp: (021) 7560911, Fax: (021) 7560553 E-mail: Majalah ilmu dan teknologi terbit berkala setiap tahun, satu volume terdiri atas 3 nomor.
VOLUME 25 NOMOR 1, APRIL 2010 ISSN 0126 – 3188
AKREDITASI : SK 187/AU1/P2MBI/08/2009
Pengantar Redaksi ………. iii Application of Mechanochemistry in Mineral Processing
Abstrak …..……….. v
Urgency to Develop Biocompatible Materials for Medical Implant Applications in Indonesia
Solihin……….. 1
Proses Pemanasan Temperatur 700°C Mineral Magnesit dari Padamarang
Andika Widya Pramono…..……….…… 7
Eko Sulistiyono dan Bintang Adjiantoro…13 Pembuatan Baja Lapis Titanium dengan Metoda Cladding
Analysis of The Mg-Ti-Fe Alloy Prepared by High Energy Ball Milling and its Hydrogen Capacity Sri Mulyaningsih dan Budi Priyono …..….19
Prospek Paduan Magnesium untuk Aplikasi Biomedis
Hadi Suwarno..………..………...………25
Pembuatan Material Komposit Matriks Paduan Al-4,5%Cu-4%Mg/Sic(P) dengan Proses Tempa (1)
Yusuf………...33
Recovery TiO2 dari Larutan TiO(SO4) Hasil Ekstraksi Bijih Ilmenite Bangka Menggunakan Proses Sol Gel
Bintang Adjiantoro dan Bambang Sriyono.41
Penghalusan Butir Titanium Murni untuk Aplikasi Biomedis dengan Teknik Equal Channel Angular Pressing (ECAP)
F. Firdiyono, dkk.…………..………...………49
Pengantar Redaksi
| iii
PENGANTAR REDAKSI
Syukur Alhamdullilah, terbitan Majalah Metalurgi pada edisi kali ini lebih awal,
direncanakan majalah ini akan terbit tiga kali dalam setahun
Majalah Metalurgi Volume 25 Nomor 1, April 2010 kali ini menampilkan sembilan
buah tulisan, terdiri atas enam buah tulisan hasil penelitian dan tiga buah studi. Tulisan hasil
penelitian disampaikan oleh Eko Sulistyo dan Bintang Ajiantoro tentang “Proses Pemanasan
Temperatur 700ºC Mineral Magnesit dari Padamarang”. Selanjutnya Sri Mulyaningsih dan
Budi Priyono menyajikan tulisan tentang “Pembuatan Baja Lapis Titanium dengan Metoda
Cladding”. Berikutnya Hadi Suwarno menulis tentang “Analysis of The Mg-Ti-Fe Alloy
Prepared by High Energy Ball Milling and its Hydrogen Capacity”; berikutnya dan Bambang
Sriyono menulis tentang “Pembuatan Material Komposit Matriks Paduan
Al-4,5%Cu-4%Mg/SiC(p) dengan Proses Tempa”. F. Firdiyono dan Kawan-Kawan menyajikan tulisan
tentang “Recovery TiO
2dari Larutan TiO(SO
4Pada bagian berikutnya ada tiga buah hasil studi yaitu “Application of
Mechanochemistry in Mineral Processing” yang disampaikan oleh Solihin dan “Urgency to
Develop Biocompatible Materials for Medical Implant Applications in Indonesia ” yang
ditulis oleh Andika Widya Pramono. Terakhir disajikan “Prospek Paduan Magnesium untuk
Aplikasi Biomedis” yang dipaparkan oleh Yusuf.
) Hasil Ekstraksi Bijih Ilmenit Bangka
Menggunakan Proses Sol Gel”. Tulisan berikutnya disajikan oleh Efendi dengan tajuk
“Penghalusan Butir Titanium Murni untuk Aplikasi Biomedis dengan Teknik Equal Channel
Angular Pressing (ECAP)”.
Semoga penerbitan Majalah Metalurgi volume ini dapat bermanfaat bagi perkembangan
dunia penelitian di Indonesia
.
Abstrak
| v
METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 660.2
Solihin (Research Center for Metallurgy, Indonesian Institute of Science)
Application of Mechanochemistry in Mineral Processing
Fenomena mekanokimia telah lama dikenal mampu menginisiasi transformasi struktural atau reaksi kimia pada temperature kamar. Selain itu, fenomena mekanokimia juga membuat kinetika reaksi dapat diakselerasi sehingga dapat terjadi pada temperature kamar. Transformasi struktural dengan memanfaatkan fenomena mekanokimia ini dapat diarahkan untuk meningkatkan unjuk kerja pemrosesan mineral atau ekstraksi logam berharga. Sulfidisasi mineral oksida, pembentukan senyawa yang mampu larut dalam air dan meningkatkan luas permukaan spesifik merupakan contoh-contoh reaksi mekanokimia atau transformasi structural yang dapat meningkatkan unjuk kerja pemrosesan mineral.
Metalurgi, Volume 25 No.1 April 2010
Kata kunci : Mekanokimia, Pemrosesan mineral, Reaksi antar padatan, Kinetik, Milling
Mechanochemical phenomenon has been known to be able to conduct structural transformation or chemical reaction at room temperature. The kinetics of the reaction can also be accelerated at room temperature through mechanochemical reaction. This transformation through mechanochemical reaction can be used to enhance mineral processing or metal extraction. Sulphidation of oxides minerals, formation of water soluble compound, and increasing specific surface area of minerals are among the mechanochemical reaction or structural transformation capable to enhance the mineral processing of certain minerals.
vi |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 619.600
Andika Widya Pramono (Research Centre for Metallurgy, Indonesian Institute of Sciences)
Urgency to Develop Biocompatible Materials for Medical Implant Applications in Indonesia
Makalah ini memberi gambaran tentang arti pentingnya pengembangan material biokompatibel untuk aplikasi implan medis di Indonesia. Berbagai latar belakang permasalahan dan perkembangan yang terjadi di dunia dibahas dimulai dari: peningkatan prosentase manula, tingkat kecelakaan dalam berkendara di Indonesia, millennium development goals, kemajuan riset dan pengembangan material biokompatibel di dunia dan Indonesia, penggunaan nanoteknologi sebagai sarana terobosan inovatif dan peningkatan nilai tambah, sampai dengan perlunya mengedepankan keunggulan kompetitif di atas keunggulan komparatif bagi Indonesia. Di bagian akhir makalah dikemukakan tentang upaya ke depan dalam pengembangan komponen implan biokompatibel yang murah dan berkualitas melalui kolaborasi internasional, termasuk dengan Amerika Serikat. Aspek manfaat bagi semua pihak yang berkolaborasi ditekankan baik dari segi kemanusiaan maupun tekno-ekonomi.
Metalurgi, Volume 25 No.1 April 2010
Kata kunci : Biokompatibel, Implan, Millennium development goals, Keunggulan komparatif, Keunggulan kompetitif, Nanoteknologi, Tekno-ekonomi
This paper underlines the significance of developing biocompatible materials for medical implant applications in Indonesia. Various problems and development worldwide concerning implant materials are discussed including: the increase in percentage of elderly people, the extent of accidents during vehicle driving in Indonesia, the millennium development goals, the advanced research and development of biocompatible materials worldwide and in Indonesia, the utilization of nanotechnology as the means for innovative breakthrough and added values, as well as the importance of bringing forward competitive advantages over the comparative advantages for Indonesia. The final part of paper discusses the possible future attempts to develop affordable biocompatible implant materials through the international collaboration including with the USA. The mutual benefits for all parties are emphasized from the aspects of humanity and techno-economy.
Abstrak
| vii
METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 660
Eko Sulistiyono dan Bintang Adjiantoro (Pusat Penelitian Metalurgi-LIPI)
Proses Pemanasan Temperatur 700 ºC Mineral Magnesit dari Padamarang
Telah dilakukan kegiatan proses pemanasan pada temperatur tinggi terhadap mineral magnesit dari Padamarang untuk melihat pengaruh pemanasan. Dari hasil percobaan dengan pemanasan pada temperatur 700 °C dengan berbagai variabel ukuran partikel menunjukkan bahwa ukuran butiran tidak berpengaruh padsa reaksi. Secara keseluruhan pada tempatur 700 °C telah menunjukkan adanya pembentukan MgO yang cukup tinggi yaitu sekitar 95 % pada waktu proses diatas 6 jam. Hasil dari proses pemanasan ini selanjutnya dilakukan analisis SEM, memperlihatkan bentuk kristal yang berupa lembaran-lembaran yang mengelompok dalam bentuk kluster-kluster.
Metalurgi, Volume 25 No.1 April 2010
Kata kunci : Magnesit, MgO, Padamarang
Activity has been carried out at high temperature heating process of the mineral magnesite from Padamarang to see the influence of heating. From the results of experiments with heating at a temperature of 700 °C with a variety of variable particle size showed that particle size has no effect on the reaction. Overall at 700 °C tempature have shown the formation of MgO is high enough, it was 95% in processing time by more than 6 hours. The result of this heating process is then performed SEM analysis, showing crystal shape in the form of sheets are clustered in the form of clusters
viii |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 620
Sri Mulyaningsih, Budi Priyono ( Pusat Penelitian Metalurgi – LIPI )
Pembuatan Baja Lapis Titanium dengan Metoda Cladding
Telah dilakukan penelitian tentang baja lapis titanium dengan metoda mechanical cladding untuk meningkatkan ketahanan korosinya. Proses cladding dilakukan dengan menyusun secara berturut-turut pelat baja, tembaga dan titanium setelah sebelumnya dibersihkan permukaannya, kemudian diikat dan dipanaskan pada temperatur diatas temperatur austenit. Percobaan dilakukan dengan memvariasikan temperatur pemanasan yaitu; 750, 800 dan 900°C dan ditahan selama 1 jam. Proses cladding dilakukan menggunakan metoda mekanik yaitu di roll dalam keadaan panas. Dari hasil percobaan diperoleh bahwa hasil proses cladding terbaik adalah pemanasan pada temperatur 900 °C yaitu hasil lapisan yang melekat merata pada semua sampel.
Metalurgi, Volume 25 No.1 April 2010
Kata kunci : Cladding, Intermetalik, Pengerollan panas
There has been done research on steel and titanium cladding mechanical cladding method by mean hot rolled cladding for increasing its corrosion behavior. The cladding process was done by put the titanium, cuprum and steel layer by layer to united, than heat treated over the austenite temperature. The heat treatment temperature was varies from 750, 800 and 900 °C, holding time at 1 hour. Continue to the cladding process with allow the unite hot plate into the roll machine. The best result from the experiment is heat treating the sample at 900°C which is show the cladding process inherent on the samples.
Abstrak
| ix
METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 669.7
Hadi Suwarno ( Center for Technology of Nuclear Fuel, National Nuclear Energy Agency )
Analysis of the Mg-Ti-Fe Alloy Prepared by High Energy Ball Milling and Its Hydrogen Capacity
Hidrogen diprediksi akan menjadi sumber energy penting untuk masa depan. Menyimpan hidrogen dalam bentuk metal hidrid merupakan metoda yang cukup menarik untuk menyimpan hidrogen dalam bentuk padat. Logam paduan Mg-Ti-Fe berukuran nano partikel dibuat dengan menggunakan mesin high energy ball milling untuk maksud menyimpan hidrogen. Analisa menggunakan mesin sinar-X atas spesimen yang di-milling selama 30 jam menunjukkan bahwa paduan sintetis senyawa Fe2Ti dan FeTi dapat dibentuk, sementara tak teramati adanya senyawa Mg-Fe maupun Mg-Ti. Adanya Mg di dalam spesimen berfungsi sebagai katalis yaitu menyediakan ruang bebas untuk hidrogen agar berinteraksi dengan fasa Fe-Ti dan Ti membentuk senyawa metal hidrid. Kapasitas hidrogen sebesar 5,7 % berat pada suhu kamar dan sebesar 1,2 % berat pada suhu 70 °C memenuhi persyaratan suhu operasi untuk fuel cell tipe polymer electrolyte membrane (PEMFC). Dari hasil penelitian disimpulkan bahwa paduan Mg-Ti-Fe dapat dipromosikan sebagai bahan penyimpan hidrogen dalam bentuk senyawa metal hidrid.
Metalurgi, Volume 25 No.1 April 2010
Kata kunci: Ball milling energi tinggi, Paduan sintesis, Material penyimpan hidrogen
Hydrogen will become a very important energy source in the near future. Storing hydrogen in the form of metal hydride presents a challenging method for solid hydrogen storage. The Mg-Ti-Fe alloy in the nanosize particles is prepared to develop a solid hydrogen storage material using a high energy ball milling. X-ray diffraction analyses of the specimen after 30 h of milling shows that the synthetic alloying of Fe2
Keywords : High energy ball milling, Synthetic alloying, Hydrogen storage material
x |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
UDC (OXDCF) 620.18
Yusuf ( Pusat Penelitien Metalurgi – LIPI )
Prospek Paduan Magnesium untuk Aplikasi Biomedis
Paduan magnesium memiliki prospek yang sangat baik sebagai material untuk aplikasi biomedis. Sifatnya yang ringan, kuat, kaku dan mudah dikerjakan sangat menarik untuk aplikasi apapun. Sedangkan sifatnya yang ramah dan bersahabat dengan cairan dan organ tubuh menjadi unggulan untuk aplikasi biomedis. Hasil interaksi antara logam magnesium dengan cairan tubuh menghasilkan magnesium khlorida tidak meracuni tubuh dan dengan mudah dikeluarkan dari tubuh lewat air seni. Sifatnya yang reaktif dan mudah terkorosi mendapat tempat sebagai material implan yang biodegradabel. Sifat ini sangat cocok untuk kebutuhan implan yang bersifat sementara, seperti pen atau baut penahan tulang yang patah. Keberadaan pen atau baut itu bersifat sementara dan harus diambil sesudah patah tulangnya berhasil dipulihkan. Pengambilan implan ini harus dilakukan dengan tindakan operasi yang agak merepotkan. Paduan magnesium yang dirancang larut pada saat patah tulang pulih, akan menghindari pengambilan pen atau baut tadi. Untuk aplikasi biomedis dalam bentuk implan yang permanen, paduan magnesium memerlukan perlakuan khusus. Paduannya sendiri bisa ditambah dengan unsur untuk menambah ketahanan korosi seperti zirkon atau kalsium. Untuk lebih meningkatkan ketahanan korosinya, paduan magnesium dapat diberi berbagai macam lapis lindung. Mulai dari lapis oksida, lapis logam, lapis polimerhingga lapis keramik. Metode pelapisannyapun bisa sederhana semacam konversi kimia, elektrolisa anodisasi, semprot dingin, lapis plasma, hingga pelapisan canggih berskala nano semacam self assembled monolayer (SAM).
Metalurgi, Volume 25 No.1 April 2010
Kata kunci : Magnesium, Paduan, Biomedis, Implan, Korosi, Lapis lindung
Magnesium alloys have a good prospect as materials for biomedical aplications. Their character as light, strong, stiff and good workability materials looks very interesting for many applications. On top of these characters, their compatibility with body liquids and human organs will become advantages in their biomedical applications. Reaction products between a magnesium metal and body liquids will produce a magnesium chloride solution which is not harmful to human body and will be secreted out from the human body through the urine solution. Their character as reactive and corrosive materials is finding its role as biodegradable temporaly implants, like temporary pin and scrscrews to connect broken bones. The pin and screws are only needed as a temporary tools before the bones are growth and connected again. The pin and screws should be removed from human body, usually by surgery. With a certain magnesium alloy as a biodegradable material the pin and screws can be left and degrade in the human body. quirFor the biomedical application as permanent implants, the alloys require a rather special treatment to improve its corrosion resistance through alloying and protective coatings. Addition of zircon or calcium are known to improve the corrosion resistance. The protective coating might be one of the following materials: oxide,metal, polymer or ceramic. The coating method also varied from a simple chemical conversion or anodizing up to a sophisticated nano scale technology such as a self assembled monolayer (SAM) method.
Abstrak
| xi
METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
Bintang Adjiantoro dan Bambang Sriyono ( UDC (OXDCF) 620.19
Pembuatan Material Komposit Matriks Paduan Al-4,5%Cu-4%Mg/SiC(p) dengan Proses Tempa (1) Pusat Penelitian Metalurgi-LIPI)
Penelitian pembuatan material komposit matriks logam telah dilakukan dengan menggunakan metoda stirrcasting pada matriks paduan Al-4,5%Cu-4%Mg dengan penguat partikel SiC. Percobaan dilakukan dengan memvariasikan persen fraksi volume partikel (5% dan 7,5%) dan ukuran partikel (147
Metalurgi, Volume 25 No.1, April 2010
µm dan 74µ
Kata kunci : Komposit matriks logam, Paduan terner AlCuMg, Senyawa karbida SiC
m). Dari hasil percobaan menunjukkan bahwa persen fraksi volume partikel sangat berpengaruh terhadap sifat mekanik dan struktur mikro dari material komposit matriks paduan Al-4,5%Cu-4%Mg/SiC(p). Hal ini ditunjukkan dengan meningkatnya kekuatan tarik, kekerasan dan memperbaiki tingkat keausan namun material komposit matriks paduan Al-4,5%Cu-4%Mg/SiC(p) cenderung memiliki sifat lebih getas.
Research the manufacture of metal matrix composite materials has been carried out by using the method stirrcasting the matrix alloy Al-4.5% Cu-4% Mg with SiC particle reinforcement. Experiments carried out by varying the particle volume fraction percent (5% and 7.5%) and particle size (147 µ m and 74 µ m). Experimental results show that the percent volume fraction of particles affect the mechanical properties and microstructure of the alloy matrix composite material Al-4.5% Cu-4% Mg / SiC (p). This is indicated by the increased tensile strength, hardness and improve wear but the alloy matrix composite material Al-4.5% Cu-4% Mg / SiC (p) tend to have more brittle nature.
xii |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
F. Firdiyono, Rudi Subagja, Latifa Hanum i, Iwan Setiawan, Nurhayati UDC (OXDCF) 669.7
Recovery TiO
( Pusat Penelitian Metalurgi – LIPI )
2 dari Larutan TiO(SO4) Hasil Ekstraksi Bijih Ilmenite Bangka Menggunakan Proses Sol Gel
Penggunaan TiO
Metalurgi, Volume 25 No.1 April 2010
2 setiap tahunnya terus meningkat antara 10 sampai 15 % di pasaran (US Department of
Commerce June, 2001). Hal ini karena TiO2 merupakan material yang banyak digunakan sebagai pigmen,
sunscreens, cat, kosmetik dan bahan baku industri kimia. Adanya manfaat dan keunggulan yang begitu banyak tersebut mendorong iklim penelitian terkait dengan pembuatan TiO2 dari berbagai prekursor. Pemanfaatan
mineral ilmenit (FeTiO3) Bangka Indonesia untuk membentuk TiO2 dengan kemurnian tinggi merupakan
potensi yang bagus dalam upaya menaikkan nilai ekonominya. Tujuan penelitian adalah melakukan recovery TiO2 dari larutan TiO(SO4) hasil ekstraksi bijih ilmenite Bangka menggunakan proses sol gel. Proses yang
dilakukan dalam penelitian adalah hidrolisis dengan pelarut H2O dalam reaktor berpengaduk dan reflux dalam
berbagai rasio volume pelarut (v/v) H2O/TiOSO4 (0, 1, 3, 5, 8, 10, 15, 19), pH (0, 1, 3, 4, 5,dan 6), pengaruh
pengadukan, dan pencucian dengan asam. Tahapan penelitian adalah larutan TiOSO4 direaksikan dengan H2O
dalam berbagai kondisi sesuai variabel pada suhu 90 °C selama 2 jam. Proses ini menghasilkan gel TiO2. Gel
TiO2 yang terbentuk kemudian dipisahkan dari filtratnya dan dicuci sampai pH netral. Proses pengeringan pada
suhu 100°C menggunakan oven dilakukan untuk mendapatkan bubuk TiO2. Bubuk titanium dioksida yang
dihasilkan kemudian dianalisa AAS, SEM, dan SEM. Hasil percobaan menunjukkan bubuk TiO2 hasil sintesis
rasio volume (v/v) H2O/TiOSO4 yang lebih kecil mempunyai ukuran partikel lebih besar dengan kadar pengotor
besi lebih kecil. Proses hidrolisis ini mampu menyisihkan pengotor Fe cukup significan. Sedangkan bubuk TiO2
hasil hidrolisis pada pH semakin kecil mempunyai ukuran partikel titanium dioksida lebih kecil dengan morfologi partikel yang seragam dan kadar pengotor besi lebih kecil. Fraksi kristalin semakin meningkat pada produk titanium dioksida yang dihasilkan pada hidrolisis pH rendah. Morfologi titanium dioksida mempunyai partikel yang seragam pada proses hidrolisis dalam reaktor berpengaduk. Proses pencucian menggunakan asam H2SO4 pada gel TiO2 dapat menurunkan kadar pengotor Fe dalam bubuk TiO2. Penelitian yang dilakukan ini
diharapkan menjadi masukkan dalam sintesis titanium dioksida dari mineral ilmenit. Keberhasilan recovery TiO2
Kata kunci : Ilmenit, Titanium sulfat, Titanium dioksida, SEM
dengan kadar pengotor besi yang rendah diharapkan dapat diaplikasikan sebagai pigmen atau bahan baku industri kimia.
The use of TiO2 each year continues to increase between 10 to 15% on the market (U.S. Department of
Commerce June, 2001). This is because TiO2 is material which is widely used as pigments, sunscreens, paints,
cosmetics and industrial raw materials chemistry. The existence of the benefits and advantages that so many of the climate to encourage research related to the production of TiO2 from various precursors. Utilization of
mineral ilmenite (FeTiO3) Bangka Indonesia to form TiO2 with high purity is a great potential in an effort to
increase its economic value. The purpose of this research is to perform recovery of TiO2 from a solution of TiO
(SO4) Bangka ilmenite ore extracted using sol gel process. The process is carried out in research is hydrolysis
with solvent H2O in a strirred reactor and reflux in various solvents volume ratio (v/v) H2O/TiOSO4 (0, 1, 3, 5,
8, 10, 15, 19), pH (0, 1, 3, 4, 5 and 6), the influence of stirring, and washing with acid. Stages of the research is TiOSO4 solution reacted with H2O in a variety of conditions as variable at 90 °C for 2 hours. This process
produces TiO2 gel. TiO2 gel was then separated from the filtrate and washes until neutral pH. The process of
drying at a temperature of 100 °C using the oven do to get the TiO2 powder. The resulting titanium dioxide
powder is then analyzed AAS, SEM, and SEM. The results showed a synthesis of TiO2 powder volume ratio
(v/v) H2O/TiOSO4 smaller particles have a size larger with smaller levels of iron impurities. This hydrolysis
process capable of removing Fe impuritiesis significant. While the results of hydrolysis of TiO2 powder at pH
less titanium dioksida have a smaller particle size with uniform particle morphology and lower levels of iron impurities. Increasing crystalline fraction in the titanium dioxide product produced at low pH hydrolysis. The morphology of titanium dioxide particles have a uniform in the process of hydrolysis in a stirred reactor. The washing process using H2SO4 acid on TiO2 gel can reduce levels of impurity Fe in TiO2 powder. This research is expected to be entered in the synthesis of titanium dioxide from the mineral ilmenite. The successful recovery of TiO2 with low levels of iron impurities is expected to be applied as a pigment or chemical industrial raw material.
Abstrak
| xiii
METALURGI (Metallurgy)
ISSN 0126 – 3188 Vol 25 No. 1 April 2010 Kata Kunci bersumber dari artikel. Lembar abstrak ini boleh diperbanyak tanpa izin dan biaya.
Efendi Mabruri, Bambang Sriyono, Sri Mulyaningsih, Solihin UDC (OXDCF) 620.19
(Pusat Penelitian Metalurgi-LIPI
Penghalusan Butir Titanium Murni untuk Aplikasi Biomedis dengan Teknik Equal Channel Angular Pressing (ECAP)
)
Tulisan ini memaparkan penghalusan butir titanium murni (Commercial Purity Titanium/CP-Ti) untuk aplikasi biomedis dengan teknik equal channel angular pressing (ECAP). Die ECAP yang dibuat untuk percobaan memiliki sudut rongga Φ=120°dan Ψ= 7°
Metalurgi, Volume 25 No.1 April 2010
yang menghasilkan regangan geser 0,65 untuk individual pass. Rute deformasi ECAP (A dan Bc) dan jumlah pass dievaluasi terhadap perubahan struktur mikro CP-Ti. Hasil percobaan menunjukkan bahwabahwa sampel CP-Ti setelah ECAP(Φ=120°, Ψ= 7°) pada masing-masing rute deformasi menunjukkan penghalusan butir yang signifikan dibandingkan dengan struktur mikro CP-Ti awal. Rute deformasi Bc menghasilkan ukuran butir yang lebih halus dibandingkan dengan yang dihasilkan oleh rute A pada jumlah pass yang sama. Penambahan jumlah pass pada masing-masing rute deformasi semakin menghaluskan ukuran butir CP-Ti.
Kata kunci : Penghalusan butir, CP-Ti, Aplikasi biomedis, Equal channel angular pressing, Rute deformasi
This paper reports the grain refinement of pure titanium (Commercial Purity Titanium/CP-Ti) for biomedical application by using equal channel angular pressing (ECAP). The ECAP dies used in the experiment have the die angle of Φ=120° and Ψ= 7° giving the shear strain of 0.65 for individual pass. The deformation routes (A and Bc) and the number of passes were evaluated with respect to microstructure evolution of CP-Ti. The experimental results showed that the grain size of CP-Ti significantly decreased after extrusion through the ECAP(Φ=120°, Ψ= 7°) dies for both deformation routes A and Bc. The ECAP route Bc resulted in the finer grain sizes compared to those were resulted by route A for the same pass number applied. Furthermore, the grain sizes of CP-Ti decreased with increasing the number of passes of both ECAP routes.
APPLICATION OF MECHANOCHEMISTRY IN MINERAL
PROCESSING
Solihin
Research Center for Metallurgy, Indonesian Institute of Science
Kawasan Puspiptek Serpong Tangerang Selatan, Banten Provence, Indonesia
Intisari
Fenomena mekanokimia telah lama dikenal mampu menginisiasi transformasi struktural atau reaksi kimia pada temperature kamar. Selain itu, fenomena mekanokimia juga membuat kinetika reaksi dapat diakselerasi sehingga dapat terjadi pada temperature kamar. Transformasi struktural dengan memanfaatkan fenomena mekanokimia ini dapat diarahkan untuk meningkatkan unjuk kerja pemrosesan mineral atau ekstraksi logam berharga. Sulfidisasi mineral oksida, pembentukan senyawa yang mampu larut dalam air dan meningkatkan luas permukaan spesifik merupakan contoh-contoh reaksi mekanokimia atau transformasi structural yang dapat meningkatkan unjuk kerja pemrosesan mineral.
Kata kunci : Mekanokimia, Pemrosesan mineral, Reaksi antar padatan, Kinetik, Milling
Abstract
Mechanochemical phenomenon has been known to be able to conduct structural transformation or chemical reaction at room temperature. The kinetics of the reaction can also be accelerated at room temperature through mechanochemical reaction. This transformation through mechanochemical reaction can be used to enhance mineral processing or metal extraction. Sulphidation of oxides minerals, formation of water soluble compound, and increasing specific surface area of minerals are among the mechanochemical reaction or structural transformation capable to enhance the mineral processing of certain minerals.
Keywords : Mechanochemistry, Mineral processing, Solid-state reaction, Kinetics, Milling
INTRODUCTION
Mechanochemistry is
defined as a
branch of chemistry, which concern with
chemical and physicochemical
transformations of substances in all states
of aggregation produced by the effect of
mechanical energy
[1]The spontaneity of any chemical and
psychochemical transformations depends
on its standard free energy change.
Thermodynamically speaking, the reaction
takes place spontaneously when the free
energy changes become negative. It can be
found in any textbook elsewhere that
standard free energy change is a variable
that depend on temperature and
atmospheric pressure of matters
.
[2-4]
The mechanochemical reaction breaks
the bonding energy between atoms in
different way; it uses mechanical energy
instead of temperature and atmospheric
pressure change. The continuous
mechanical energy, transmitted to the
powder through collision and impact,
makes the particles deform plastically,
flattened, cold-welded, fractured and
re-welded. The continuation of the
mechanical deformation results in
progressive particles size reduction leading
. The
changes in temperature and pressure of
2 |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188/ hal 1-6to the increasing of surface energy, as well
as changes in chemical, physicochemical
and structural properties. This is
manifested by the presence of a variety of
crystal defects such as increasing of grain
boundaries, dislocations, vacancies,
stacking faults and deformed and ruptured
chemical bonds
[5]. Soon after the chemical
bonds are ruptured, the atoms or molecular
ions will be in the active state, and thus
further chemical reaction with other active
atoms or ions can possibly takes place. The
chemical reaction can be a displacement
reaction
[6-7], or the combination
reaction
[8-9]In some cases, transmission of
mechanical energy through milling only
change the crystal structure of the
substance rather than promoting solid-state
chemical reaction. The changes can be
polymorphic transformation,
amorphisation, or re-crystallization. For
example, the structure of CaCO
.
3
can be
transformed from calcite to aragonite; for
that of TiO
2from anatase to rutile; and for
that of PbO from massicot to litharge.
The polymorphic transformation
proceeds in two stages
[10]1.
Preliminary stage, in which the crystal
size decreases with increasing lattice
distortion, the critical stress for fracture
is being larger than for distortion.
:
2.
Polymorphic transformation stage, in
which the stored energy is released with
a small (or zero) decrease in crystallite
size.
The direction of polymorphic
transformation depends on temperature and
pressure applied to the materials. The
direction of transformation can be
predicted by using the
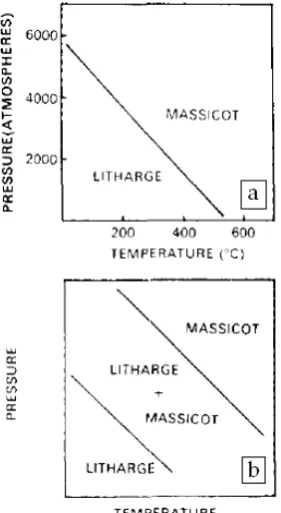
pressure-temperature transformation diagram
(Figure 1 (a)).
In the case of mechanochemistry, in
which shears are very much applied, the
line of phase transformation shifts toward
lower temperature and pressure, along with
the existance of the area containing both
phases (Figure 1 (b))
[11]Amorphisation often happens to the
particles treated by milling. The
mechanical energy is believed to distort the
crystal lattice resulting irregular
orientations of crystal. The broadening or
disappearance of the peak of intensity in
XRD pattern is the indication that
amorphisation has occurred. For example,
the product reaction of milled LiOH.H
. The shifting of
this line illustrates that the crystal structure
transformation can take place at relatively
low temperature with the aid of rapid
shears.
2
O-MnO
2and Mg-Ni become amorphous after
being milled with the broadening intensity
peak as indication of amorphisation
[12]; and
Al(OH)
3and LaBr
3becomes amorphous
after being milled with Y
2O
3and La
2O
3respectively with the disappearance of
their intensity peak as indication
amorphisation
[13-14]The mechanism of amorphisation
during milling is initiated by the formation
of structural defect such as vacancies and
dislocations that drives to grain boundaries
sliding. The deformed grains consist of
many dislocations with disorder
orientation, as well as those with ordered
orientation. The disorder orientation
dislocation is believed to drift, driven by
tendency of shear stress relaxation, which
leads to the formation of local amorphous
region. The drifting of disorder orientation
dislocation that takes place rapidly leads to
grain boundary sliding, which results in
amorphisation of entire crystal structure
.
[15]
Application Of Mechanochemistry …../ Solihin
| 3
Figure 1. Pressure-Temperature transformationDiagram [10] (a) Hydrostatic pressure (b) Hydrostatic pressure and shears
EFFECT OF MECHANOCHEMICAL
PHENOMENON TO KINETICS OF
SOLID-STATE REACTION
The mechanical energy transmitted to
particles accelerates the kinetics of
reaction. An ordinary chemical reaction
involving solid-state matter normally takes
place through diffusion mechanism of
reactants followed by chemical reaction
among reactants. The kinetic of reaction is
influenced by both the rate of diffusion and
chemical reaction. When diffusion rate is
slower than chemical reaction rate, then
the total reaction depends a lot to the rate
of diffusion
[16]. Solid-state reaction is
usually followed by the formation of
product phases on the interfaces of the
reactants. Further growth of these product
phases will in turn cover the surface of
reactants. This coverage makes the atomic
diffusion, which is very necessary for the
continuing of that solid-state reaction is
retarded or at least slowed down. At this
moment, the rate of entire reaction is
controlled by diffusion rate. The flux of
atoms that diffuse through the product
layer
follows the Fick’s diffusion
Equation
[17].
where
D
= diffusion coefficient
∆
c and
∆
x
= gradient of the reactant
concen- tration and the
diffusion path
According to equation above, one way
to increase the atomic flux is by changing
the value of diffusion coefficient (D) so
that the atoms can diffuse easily through
the layer of reaction products. The
diffusion coefficient is a temperature
dependence variable with exponential
relation
[18]Mechanochemical method increases the
reaction kinetics through different path.
Instead of inputting heat, the product layer
that retard diffusion is broken by repetitive
impact, shears and cold welding. The
repetitive impact and shear decreases the
particle size, therefore provides an
increasing of contact area and a continuous
supply of fresh surfaces. The cold-welding
of particles during milling increases the
kinetics of reaction through dissolution of
solid materials. The kinetics of reaction is
also improved by the presence of crystal
defects. The defects enhance the diffusivity
of atoms within crystal, which finally
results in quite homogen atomic
distribution
.
By increasing temperature,
the value of diffusion coefficient increases
exponentially, and therefore the reactant
molecule can diffuse rapidly through the
layer of reaction product.
[5]
.
APPLICATION OF
MECHANOCHEMICAL REACTION
IN MINERAL PROCESSING
4 |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188/ hal 1-6different fields have creatively proposed a
new route of process based on
mechanochemical reaction. In this
sub-chapter the application of
mechanochemical reaction to aid the
extraction of metals from their mineral is
discussed.
Solihin, from Indonesia, has proposed
the soft process for palladium recycling
from waste through mechanochemical
reaction
[19]P. Balaz, a Slovakian scientist in the
field of mineral dressing, has proposed the
sulphidation of certain oxide mineral to aid
the separation of those mineral through
flotation. Inspired by Theophrastus of
Efesus (371-286 BC) a disciple of Aristotle
who has extracted mercury from cinnabar,
Balaz also has proposed a
mechanochemical reaction to extract
mercury from cinnabar using brass pestle
in the presence of vinegar
combined with water-leaching.
A waste containing palladium is
mechanochemically reacted to produce
water-soluble compound of palladium
which can be leached by only water. By
this new process, around 80% palladium
can be extracted after leaching by using
only pure water.
[20]
Another well-known scientist in the
field of metallurgy and mineral processing
who published many textbook in the field
of metallurgy, Fathi Habashi, has
introduced mechanochemical reaction to
activate chalcopyrite in a unit operation of
copper and iron extraction
.
[21]
. It is found
that the kinetic of extraction reaction can
be increased by increasing specific surface
area, which is a parameter engineered
through milling
[22]. Figure 2 shows the
dependence of kinetic constant to specific
surface area. The mechanism of reaction
rate dependence to specific surface area is
explained briefly by E. Gock
[23]•
The first step is the dissolution of
surface layers formed
by
mechanochemical surface reaction,
:
•
The active sites, which are characterized
by the presence of
β
-phase are leached
out.
Sphalerite also has been activated
before acid leaching. The similar behavior
is found. The activation of sphalerite leads
to the increasing of recovery of zinc, as
shown in Figure 3
[28].
Figure 2. Structural sensitivity of reaction for mechanically activated chalcopyrite. ko=initial
rate constant, S=specific surface area, X=crystallinity degree[22]
Figure 3. The influence of leaching time on zinc recovery for sphalerite[28]
(1)
Mechanically activated ZnS for 60
min,
(2)
Mechanically activated ZnS for 60 min
and annealed,
Application Of Mechanochemistry …../ Solihin
| 5
SUMMARY
Transmission of mechanical energy into
minerals can induce mechanochemical
reaction that leads to chemical reaction or
structural transformation. Both of them can
be applied in many fields to create a new
route of process or to accelerate the rate of
a process.
In the field of mineral
processing, the structural change of
materials can be used to activate the
minerals to enhance leaching. The specific
surface area of minerals can be increased
through mechanochemical reaction which
leads to the increasing of metal recovery.
REFERENCES
[1]
G. Heinicke, Tribochemistry.
Akademie - Verlag, Berlin (1984).
[2]
R.W. Missen, C.A. Mims, B.A.
Saville, Introduction to chemical
reaction Engineering and kinetics,
John Wiley and Son, New York
(1999).
[3]
JJ Moore, Chemical metallurgy,
Buttenworth and Co, England (1981)
[4]
S. Stolen, T. Grande, Chemical
thermodynamics of materials, John
Wiley and Son, England (2004).
[5]
C. Suryanarayana, Mechanical
alloying and milling, Progress in
Materials Science Vol. 46 pp. 1-184
(2001).
[6]
P. Matteazzi, G.. Le Caër,
Mechanically activated room
temperature reduction of sulphides,
Mater Sci and Eng
Vol. A156
pp.229-37 (1992).
[7]
J. Kano, E. Kobayashi, W. Tongamp,
F. Saito, Preparation of GaN powder
by mechanochemical reaction between
Ga
2O
3and Li
3[8]
Q. Zhang, J. Lu, F. Saito,
Mechanochemical synthesis of LaCrO
by grinding constituent oxides,
Powder Technology
Vol.122 pp.145–
149 (2002).
N,
Journal of Alloys
and Compounds
Vol.464 No.1-2
pp.337-339 (2008).
[9]
Q. Zhang, F. Saito, Mechanochemical
synthesis of LaMnO from LaO and
MnO powders,
Journal of Alloys and
Compounds
Vol.297 pp. 99–103
(2000).
[10]
I.J. Lin, S. Nadiv, Review of the Phase
Transformation and Synthesis of
Inorganic Solids Obtained by
Mechanical Treatment
(Mechanochemical Reactions),
Materials Science and Engineering
Vol.39 pp.193 – 209 (1979).
[11]
Y. Tanaka, Q. Zhang, F. Saito,
Synthesis of spinel Li
4Mn
5O
12[12]
D. Guzm´ana, S. Ordo˜neza, D.
Serafinib, P. Rojasc, O. Bustos, Effect
of the milling energy on the
production and thermal stability of
amorphous Mg
with
an aid of mechanochemical treatment,
Powder Technology
Vol.132 pp.74–
80 (2003).
50
Ni
50[13]
Q. Zhang, F. Saito, Mechanochemical
solid reaction of yttrium oxide with
alumina leading to the synthesis of
yttrium aluminum garnet,
Powder
Technology
Vol.129 pp.86– 91 (2003).
,
Journal of
Alloys and Compounds
Vol. 456
No.1-2 (No.1-2008).
[14]
J. Lee, Q. Zhang, F.
Saito,
Mechanochemical Synthesis of LaOX
(X=Cl,Br) and Their Solid State
Solutions,
Journal of Solid State
Chemistry
Vol.160 pp.469-473 (2001).
[15]
I.A. Ovid’ko, Defects and
amorphisation process in plastically
deformed metals,
J.Phys.,
D.Appl.Phys
. Vol.23 pp.365-367
(1990).
[16]
F. Habashi, Principles of Extractive
Metallurgy, Volume 1. General
Principles, Gordon & Breach, New
York (1980).
[17]
H.D. Baehr, K. Stephan, Heat and
mass transfer, Springe-Verlag, Verlin
(2006).
[18]
R.C. Ropp,
Solid-state chemistry
,
Elsevier Amsterdan (2003).
6 |
Majalah Metalurgi, V 25.1.2010, ISSN 0126-3188/ hal 1-6Method, P
roceeding of Metal and
Mineral Institute of Japan Tohoku
Branch,
Autumn Conference 2006.
[20]
P. Balaz, Mechanical activation in
hydrometallurgy,
International Jornal
of Mineral Processing
72 (2003)
341-354.
[21]
F. Habashi, Chalcopyrite, its
Chemistry and Metallurgy.
McGraw-Hill, New York (1978).
[22]
P. Balaz, Intensification of oxidative
leaching of chalcopyrite.
PhD thesis
,
Mining Institute of Slovak Academy
of Sciences, Kosˇice, (1981).
[23]
E. Gock, The influence of solid state
reaction in vibratory mill on
leachability of sulphidic raw materials,
Habilitations-schrift
, TU Berlin,
German.
[24]
D. Maurice, D. Hawk, Ferric chloride
leaching of mechanically activated
chalcopyrite.
Hydrometallurgy
49
(1998), 103–124.
[25]
D. Maurice, J.A. Hawk, Simultaneous
autogeneous grinding and ferric
chloride leaching of chalcopyrite.
Hydrometallurgy
51 (1999), 371– 377.
[26]
M. Kobayashi, , J.E. Dutrizac, , JM
Toguri, A critical review
of the
ferric chloride leaching of galena.
Canadian Metals Quarterly
29 (1990),
201–211.
[27]
P. Balaz, Influence of solid state
properties on ferric chloride leaching
of mechanically activated galena.
Hydrometallurgy
40 (1996), 359– 368.
[28]
X.M. Li, C. Jiayong, R. Kammel, F.
Pawlek, Non-oxidative dissolution of
mechanically activated sphalerite.
International Journal of Mechanical
Alloying 1 (1994), 166– 171.
CURRICULUM VITAE
Solihin,
lahir di Karawang, Jawa Barat,
Menyelelesaikan Pendidikan di Teknik
Pertambangan ITB pada tahun 1995
kemudian menyelesaikan Pendidikan S2
pada tahun 2006 dan S3 pada tahun 2009
di
Tohoku University
Jepang. Telah
Indeks
![Figure 3. The influence of leaching time on zinc recovery for sphalerite [28]](https://thumb-ap.123doks.com/thumbv2/123dok/2674008.1304794/18.595.314.518.158.391/figure-influence-leaching-time-zinc-recovery-sphalerite.webp)