Comparative assessment of published atrial
fi
brillation stroke risk strati
fi
cation
schemes for predicting stroke, in a non-atrial
fi
brillation population: The Chin-Shan
Community Cohort Study
Gregory Y.H. Lip
a,⁎
, Hung-Ju Lin
b, Kuo-Liong Chien
a,b,c,⁎⁎
, Hsiu-Ching Hsu
c, Ta-Chen Su
c,
Ming-Fong Chen
c, Yuan-Teh Lee
c,da
University of Birmingham Centre for Cardiovascular Sciences, City Hospital, Birmingham, B18 7QH, United Kingdom b
Department of Internal Medicine, National Taiwan University Hospital, Taipei, Taiwan c
Institute of Epidemiology & Preventive Medicine, College of Public Health, National Taiwan University, Taipei, Taiwan dInstitute of Clinical Medical Science, Chinese Medical University, Taichung, Taiwan
a b s t r a c t
a r t i c l e
i n f o
Article history:
Received 2 August 2012
Received in revised form 15 September 2012 Accepted 22 September 2012
Available online 13 October 2012
Keywords:
Stroke risk stratification Atrialfibrillation CHADS2
Background:In patients at high risk of stroke, such as atrialfibrillation (AF), there has been great interest in developing stroke risk prediction schemes for identifying those at high risk of stroke. Stroke risk prediction schemes have also been developed in non-AF populations, but are limited by lack of simplicity, which is more evident in schemes used in AF populations. We hypothesized that contemporary stroke risk stratifi ca-tion schemes used in assessing AF patients could predict stroke and thromboembolism in a non-AF commu-nity population, comparably to that seen in AF populations.
Methods:We tested the CHADS2and CHA2DS2-VASc schemes, as well as the AF stroke risk stratification
schemes from the Framingham study, Rietbrock et al., 2006 ACC/AHA/ESC guidelines, the 8th American Col-lege of Cardiology (ACCP) guidelines and NICE, for predicting stroke in a large community cohort of non-AF subjects, the Chin-Shan Community Cohort Study.
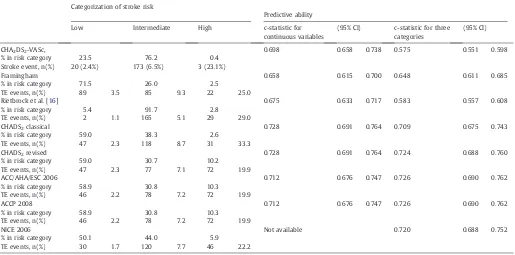
Results:The tested schemes had variable classification into low, moderate and high risk strata, with the pro-portion classified as low risk ranging from 5.4% (Rietbrock et al. to 59.0% (CHADS2classical). Rates of stroke
also varied in those classified as‘low risk’ranging from 1.1% (Rietbrock et al. to 3.5% (Framingham). All com-mon risk schemes had broadly similar c-statistics, ranging from 0.658 (Framingham) to 0.728 (CHADS2
clas-sical) when assessed as a continuous risk variable for predicting stroke in this population, with clear overlap between the 95% CIs. In an exploratory analysis amongst AF subjects in our population, the c-statistics were broadly similar to those seen in non-AF subjects.
Conclusion:Contemporary stroke risk stratification schema used for AF can also be applied to non-AF populations with a similar (modest) predictive value. Given their simplicity (e.g. CHADS2score), these scores
could potentially be used for a‘quick’evaluation of stroke risk in non-AF populations, in a similar manner to AF populations.
© 2012 Elsevier Ireland Ltd. All rights reserved.
1. Introduction
In patients at high risk of stroke, such as atrial
fi
brillation (AF),
there has been great interest in developing stroke risk prediction
schemes for identifying those at high risk of stroke. Stroke risk
predic-tion schemes have also been developed in non-AF populapredic-tions, but
are limited by lack of simplicity
[1]
, which is more evident in schemes
used in AF populations, such as the CHADS
2and CHA
2DS
2-VASc
schemes
[2,3]
.
The CHADS
2scheme is an amalgamation of stroke risk factors
identi
fi
ed from 2 trial-based stroke risk strati
fi
cation schemes, the
AF Investigators and the SPAF-1 schemes
[2]
. However, the CHADS
2scheme has many limitations, and does not include many stroke risk
factors
[4,5]
. To complement the CHADS
2scheme, the CHA
2DS
2-VASc
has been developed
[3]
, by being more inclusive (rather than
exclu-sive) of stroke risk factors. The CHA
2DS
2-VASc scheme has been
shown to be as good as (and possibly better) than the CHADS
2scheme in predicting high risk patients with AF who develop stroke
and thromboembolism (TE)
[6,7]
, but performs particularly well in
identifying those patients with AF who are
‘
truly low risk
’
of
throm-boembolism, who do not need any antithrombotic therapy
[6
–
10]
.
–
⁎ Corresponding author. Tel.: +44 121 5075080; fax: +44 121 5544083.
⁎⁎ Corresponding author at: Department of Internal Medicine, National Taiwan University Hospital, Taipei, Taiwan. Tel.: +886 2 23123456x62830; fax: +886 2 23511955.
E-mail addresses:[email protected](G.Y.H. Lip),[email protected]
(K.-L. Chien).
0167-5273/$–see front matter © 2012 Elsevier Ireland Ltd. All rights reserved.
http://dx.doi.org/10.1016/j.ijcard.2012.09.148
Contents lists available at
ScienceDirect
International Journal of Cardiology
The CHA
2DS
2-VASc scheme is used in the 2010 European Society of
Cardiology guidelines on AF management
[11]
.
The CHADS
2score has been applied to small cohorts of non-AF
populations, and has been reported to have modest predictive value
for predicting stroke and thromboembolism
[2,12]
. One recent
analy-sis found the CHADS
2score to have predictive value for adverse
car-diovascular events in patients admitted with stable coronary artery
disease
[13]
. In the REACH registry, the CHADS
2score was also related
to cardiovascular events in a large population of patients with
atherothrombosis
[14]
. However, we are unaware of any
comprehen-sive analysis of contemporary AF stroke risk strati
fi
cation schemes in
a large prospective community cohort of non-AF subjects, let alone in
a Far Eastern population.
We therefore hypothesized that contemporary stroke risk strati
fi
ca-tion schemes could predict stroke and thromboembolism, comparably
to that seen in AF populations. To test this hypothesis we applied the
CHADS
2and CHA
2DS
2-VASc schemes, as well as the AF stroke risk
strat-i
fi
cation schemes from the Framingham study
[15]
, Rietbrock et al.
[16]
,
2006 ACC/AHA/ESC guidelines
[17]
, the 8th American College of
Cardiol-ogy (ACCP) guidelines
[18]
and NICE
[19]
, to a large community cohort
of non-AF subjects, the Chin-Shan Community Cohort Study.
2. Methods
2.1. Study design and study participants
Details of this cohort study have been published previously[20]. In brief, the Chin-Shan Community Cohort (CCCC) Study began in 1990 by recruiting 1703 men and 1899 women of Chinese ethnicity aged >35 years from the town of Chin-Shan, 30 km north of metropolitan Taipei, Taiwan. Information about lifestyle and medical conditions and anthropometric measures was assessed by interview questionnaires and physical examinations in 2-year cycles for the initial 6 years; the validity and reli-ability of the collected data and measurements have been reported in details elsewhere
[20,21]. The cohort was followed up from 1990 to the end of 2007 (a total of 49 281 person-years, median 15.9 years, interquartile range: 12.8 to 16.9 years)[20].
2.2. Description of stroke risk stratification schema
The various stroke risk schema compared and/or validated in this‘real world’cohort are summarized inTable 1. The Framingham, CHADS2and CHA2DS2-VASc schemes are
point-based scores, with the Framingham one based on a mathematical formula[15]
and the CHADS based on 1 point for CHAD (congestive heart failure, hypertension, age>75 and diabetes) and 2 points for stroke/TIA[2]. The CHA2DS2-VASc score is based
on 2 points for stroke/TIA and age≥75, and 1 point for CHAD, age 65–75, vascular disease and female gender[3].
In order to compare their predictive ability with other schema for distinguishing low, intermediate and high risk strata, we categorized also the scores into three groups. We defined the CHADS2score in two ways: (i)classical, whereby scores of 0 = low, 1–2 =
intermediate, >2=high risk; or (ii)revised, whereby scores of 0=low, 1=intermediate,
≥2=high risk. The CHA2DS2-VASc score was categorized as 0=low, 1=intermediate
and≥1 as high risk. We categorized the Framingham score in a similar manner to that pro-posed by Fang et al.[22], as follows: score 0–7=low, 8–15=intermediate, 16–31=high risk. In addition to these categorized definitions (commonly used in clinical practice), the Framingham, CHADS2and CHA2DS2-VASc scores were also tested as continuous variables.
2.3. Follow-up strategy and outcome ascertainment
Procedures for our documentation of incident stroke have been previously described and validated[23–25].
Incident stroke cases were ascertained according to the following standard criteria: a sudden neurological symptom of vascular origin that lasted >24 h with supporting evi-dence from brain imaging studies. Fatal stroke cases were included. Deaths were
identi-fied from official certificate documents and verified by house-to-house visits. The cases were confirmed by cardiologists and neurologists. Transient ischemic attacks were not included in this study, especially since this is a‘soft’endpoint. The National Taiwan University Hospital Committee Review Board approved the study protocol.
2.4. Statistical analysis
We used descriptive analyses with proportions and means (±standard deviation) to describe the validation cohort, categorization of the three risk groups per schema and the event rates per risk group. We calculated the 95% confidence interval of event rates using the binomial approximation. We performed logistic regression with each schema, containing three risk groups, as independent variable and TE during 1 year as dependent variable. We calculated the area under the curve for the receiver-operating characteristic (ROC) which represents the ability of a schema to correctly classify risk for TE events, which is also referred to as the c-statistic (Harrell's c).
The cohort largely consisted of non-AF subjects (n = 3524), but as a sensitivity
exploratoryanalysis, we calculated the c-statistics in a small separate cohort of AF cases (n = 38) within our cohort.
All statistical tests were 2-sided with a Type I error of 0.05, and probability values of
b0.05 were considered statistically significant. Analyses were performed with SAS Version 9.1 (SAS Institute, Cary, NC), Stata Version 9.1 (Stata Corporation, College Station, Texas), and R Version 2.9.0 (The R Foundation for Statistical Computing).
3. Results
Our study population and associated risk factors are shown in
Table 2
. As compared to AF subjects, non-AF subjects tended to be
younger, and to have less proportion with use of cigarettes,
hyperten-sion, type 2 diabetes mellitus, heart failure and coronary artery
disease (p
b
0.05). As to prior stroke, there was no difference between
those with or without presence of baseline AF.
Risk strati
fi
cation, incidence of stroke, and predictive ability for
risk stratum amongst the 3524 CCCC study participants without
baseline AF are shown in
Table 3
.
The schemes had variable classi
fi
cation into low, moderate and
high risk strata, with the proportion classi
fi
ed as low risk ranging
from 5.4% [
16
] to 59.0% (CHADS
2classical). Rates of stroke also varied
Table 1
Risk stratification schemes used to predict thromboembolism in atrialfibrillation.
Risk scheme Ref Low risk Intermediate risk High risk
CHADS2(2001)—classical [2] Score 0 Score 1–2 Score 3–6
CHADS2—revised [3] Score 0 Score 1 Score 2–6
Framingham (2003) [24] Score 0–7 Score 8–15 Score 16–31 Rietbrock et al (2008) [16] Score 0 Score 1–5 Score 6–14 NICE guidelines (2006) [19] Ageb65 years with
no moderate/high risk factors
Age≥65 years with no high risk factors Ageb75 years with hypertension, diabetes or vascular diseasea
Previous stroke/TIA or thromboembolic event
Age≥75 years with hypertension, diabetes or vascular disease Clinical evidence of valve disease or heart failure, or impaired left ventricular function
ACC/AHA/ESC guidelines (2006) [17] No risk factors Age≥75 years, or hypertension, or heart failure, or LVEF≤35%, or diabetes
Previous stroke, TIA or embolism, or≥2 moderate risk factors of (age≥75 years, hypertension, heart failure, LVEF≤35%, diabetes)
8th ACCP guidelines (2008) [18] No risk factors Age > 75y, or hypertension, or moderately or severely impaired LVEF and/or heart failure, or diabetes
Previous stroke, TIA or embolism, or≥2 moderate risk factors of (age≥75 years, hypertension, moderately or severely impaired LVEF and/or heart failure, diabetes) CHA2DS2-VASc (2009) [3] No risk factors One‘clinically relevant nonmajor’risk factor
(heart failure/LVEF≤40, hypertension, diabetes, vascular diseasea, female gender,
age 65–74)
Previous stroke, TIA or embolism, or age≥75 years, or≥2
‘clinically
relevant nonmajor’risk factors (heart failure/LVEF≤40, hypertension, diabetes, vascular diseasea, female gender,
age 65–74)
a
in those classi
fi
ed as
‘
low risk
’
ranging from 1.1% [
16
] to 3.5%
(Framingham).
All common risk schemes had broadly similar c-statistics, ranging
from 0.658 (Framingham) to 0.728 (CHADS
2classical) when assessed
as a continuous risk variable for predicting stroke in this population,
with clear overlap between the 95% CIs (
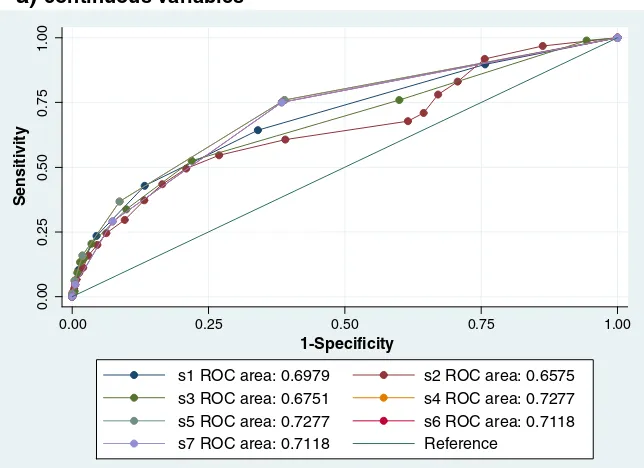
Fig. 1, Table 3
). When analysed
as 3 categories, the c-statistics for modi
fi
ed CHADS
2[
16
] and CHA
2DS
2-VASc schemes were less impressive compared to other schemes.
In an exploratory analysis amongst the small cohort of AF subjects
in our population (who had 12 stroke events), the point estimates of
the c-statistics were broadly similar for CHA
2DS
2-VASc (0.623),
Fra-mingham (0.643), Rietbrock et al. (0.660), CHADS
2classical (0.595),
CHADS
2revised (0.595), ACC/AHA/ESC and ACCP (both 0.593), with
much overlap in 95% CIs given the small numbers in this cohort
(full data not shown).
4. Discussion
In this analysis, we show that many contemporary stroke risk
strati
fi
cation schema used for AF can also be applied to non-AF
populations with a similar (modest) predictive value, as re
fl
ected by
the c-statistic. This would have advantages, since schemes such as
CHADS
2are simple and easily remembered, whilst other stroke risk
scores are usually based on weighted formulae derived from
multi-variate analyses. Of note, the c-statistics in our community study
were broadly similar for the schemes whether applied to non-AF
and AF populations.
The present analysis shows that the CHADS
2scheme allows a
sim-ple and rapid assessment of stroke risk, even in a non-AF population.
This would enhance rapid clinical assessment of patients who may be
at risk of stroke. Other stroke risk scoring systems have been evident
for many years, and these prediction models for the risk of stroke
have been helpful to guide screening and interventions and to predict
stroke events, but derivation of some prediction models were based
on hospital-based patients with various co-morbid conditions. One
older stroke risk assessment model based on a community cohort,
the Framingham risk score included variables such as age, systolic
blood pressure, antihypertensive therapy, diabetes mellitus, smoking,
history of CVD, AF and LVH
[1]
.
Based on the present Chin-Shan Community Cohort Study, we
re-cently published a model for predicting the 15-year incidence of
stroke in a community-based Chinese adult population, based on
age (8 points), gender (1 point), systolic blood pressure (3 points),
di-astolic blood pressure (2 points), family history of stroke (1 point),
atrial
fi
brillation (3 points), and diabetes (1 point), where the
c-statistic was 0.772 (95% CI, 0.744 to 0.799)
[20,21]
. This model
in-cludes some variables of the CHADS
2score, but is weighted (and
thus, more complex compared to (say) the CHADS
2score), includes
family history of stroke and AF as additional variables, and is designed
for
‘
general
’
population assessments.
Nonetheless, the CHADS
2score has been shown to predict ischemic
stroke in the absence of AF amongst subjects with stable coronary heart
disease
[13]
. Indeed, Welles et al.
[13]
studied 916 non-anticoagulated
Table 2Baseline characteristics of the study cohort by absence or presence of atrialfibrillation (AF)a
Age, years, mean (SD) 54.8 (12.3) 67.0 (10.2) 54.9 (12.3) Age 65–75, % 15.9 36.8 16.1
Age >=75, % 6.4 23.7 6.5
Female, % 53.0 36.8 52.8
Systolic blood pressure, mm Hg, mean(SD) 125 (21) 138 (29) 126 (21) Diastolic blood pressure, mm Hg, mean(SD) 77 (11) 81 (16) 77 (11) Body mass index, kg/m2
, mean (SD) 23.5 (3.4) 24.0 (3.6) 23.5 (3.4) LV ejection fraction,%, mean (SD) 68.4 (10.4) 62.1 (16.1) 68.3 (10.4)
Baseline medical conditions
Alcohol use, % 29.7 39.5 29.9 Current smoker, % 36.1 52.6 36.4 Hypertension, % 29.8 52.6 30.0
Diabetes, % 13.0 31.6 13.3
Heart failure, % 1.4 7.4 1.4 Coronary artery disease, % 3.4 2.6 3.4
Prior stroke, % 2.5 2.6 2.5
a
Only 2621 cases with echocardiography data were available.
Table 3
Risk stratification, incidence of stroke, and predictive ability for risk stratum amongst the CCCC study participants without baseline AF status (n = 3524).
Categorization of stroke risk
CHA2DS2-VASc, 0.698 0.658 0.738 0.575 0.551 0.598
% in risk category 23.5 76.2 0.4 Stroke event, n(%) 20 (2.4%) 173 (6.5%) 3 (23.1%)
Framingham 0.658 0.615 0.700 0.648 0.611 0.685
% in risk category 71.5 26.0 2.5 TE events, n(%) 89 3.5 85 9.3 22 25.0
Rietbrock et al. [16] 0.675 0.633 0.717 0.583 0.557 0.608
% in risk category 5.4 91.7 2.8 TE events, n(%) 2 1.1 165 5.1 29 29.0
CHADS2classical 0.728 0.691 0.764 0.709 0.675 0.743
% in risk category 59.0 38.3 2.6 TE events, n(%) 47 2.3 118 8.7 31 33.3
CHADS2revised 0.728 0.691 0.764 0.724 0.688 0.760
% in risk category 59.0 30.7 10.2 TE events, n(%) 47 2.3 77 7.1 72 19.9
ACC/AHA/ESC 2006 0.712 0.676 0.747 0.726 0.690 0.762
% in risk category 58.9 30.8 10.3 TE events, n(%) 46 2.2 78 7.2 72 19.9
ACCP 2008 0.712 0.676 0.747 0.726 0.690 0.762
% in risk category 58.9 30.8 10.3 TE events, n(%) 46 2.2 78 7.2 72 19.9
NICE 2006 Not available 0.720 0.688 0.752
outpatients with stable coronary heart disease and after 5821
person-years of follow up, the ischemic stroke/TIA was 0.69/100 person-person-years,
and the c-statistic was 0.65. When compared to low risk (CHADS
20
–
1)
subjects, the risk of stroke in intermediate risk patients was increased
2.4-fold, and for high risk patients, 4.0-fold. In the present analysis, we
have extended our previous work
[20]
and that of Welles et al.
[13]
to
show that all the stroke risk scores used in AF can also be applied to
non-AF populations with a similar (modest) predictive value to AF
co-horts, even in a Chinese community cohort.
Other risk assessment schemes have concentrated on prediction of
overall cardiovascular risk, with an endpoint that includes
(myocardi-al infarction, coronary heart disease, stroke, and transient ischaemic
attack), rather than the prediction of stroke per se. In a recent analysis
from the United Kingdom, the QRISK cardiovascular disease risk
equa-tion offered an improvement over the Framingham score in identifying
a high risk population for cardiovascular disease in
[26,27]
. The QRISK
score did underestimate the 10 year cardiovascular disease risk, but
the magnitude of underprediction was smaller than the overprediction
with Framingham score. Other models for cardiovascular disease risk
prediction have been described, including ASSIGN
[28]
. Of note, even a
cardiovascular risk prediction score has prognostic implications in
post-stroke patients
[29]
, although some debate over the applicability
of various scores to different ethnic groups is evident
[30]
.
Notwithstanding how the different scores are presented in
Table 1
,
the arti
fi
cial categorization into low, moderate and high risk strata is
perhaps less relevant in the non-AF population, as stroke risk is a
a)
continuous variables
0.00
0.25
0.50
0.75
1.00
Sensitivity
0.00 0.25 0.50 0.75 1.00
1-Specificity
s1 ROC area: 0.6979
s2 ROC area: 0.6575
s3 ROC area: 0.6751
s4 ROC area: 0.7277
s5 ROC area: 0.7277
s6 ROC area: 0.7118
s7 ROC area: 0.7118
Reference
b)
categorical (ie. low/moderate/high) variables
r1 ROC area: 0.5747
r2 ROC area: 0.648
r3 ROC area: 0.5825
r4 ROC area: 0.7092
r5 ROC area: 0.724
r6 ROC area: 0.726
r7 ROC area: 0.726
r8 ROC area: 0.7199
Reference
0.00 0.25 0.50 0.75 1.00
1-Specificity
0.00
0.25
0.50
0.75
1.00
Sensitivity
continuum
—
in AF, the identi
fi
cation of the
‘
high risk
’
stratum was such
that such patients could be targeted for the
‘
inconvenient
’
oral
anticoagulation available, which was warfarin. This necessity is less
ap-parently with the availability of new oral anticoagulants that overcome
the dis-utility of warfarin, and may also be relatively safer. Indeed, a
Markov decision analysis model recently suggested that anticoagulation
with one of these new
‘
safer
’
agents should even be considered at a
lowered stroke threshold of 0.9%/year amongst AF populations
[31]
.
The c-statistics in the present study for the various schemes were
broadly comparable to the c-statistics derived for the different
schemes in AF patients from the EuroHeart survey
[3]
. Of note, the
c-statistics in our cohort were also broadly comparable to the
explor-atory analysis in the small number of patients with AF in our wider
non-AF study cohort. Nonetheless, it would be dif
fi
cult to make
com-parisons between c-statistics tested in one validation cohort, with
those derived from another one.
4.1. Limitations
This study is limited by its registry-based design, but its strength is
the prospective follow up
[20,21]
. Unfortunately, we only had small
numbers of subjects with AF at baseline, but our limited exploratory
analysis suggests that the risk schemes had broadly similar predictive
value both AF and non-AF subjects. Our AF diagnosis was based on
documented AF, and more intense/prolonged ECG monitoring may
have pick up AF in what was presumed to be non-AF subjects
[32]
.
In-deed, continuous monitoring may identify AF in 30% of patients with
stroke risk factors, but without previous known AF or stroke/TIA over
a mean followup of 1.1 years
[33]
. The presence analysis would
sup-port the possible use of the AF stroke risk strati
fi
cation schema in
non-AF populations. Also, we included all types of stroke in this
study and did not specify ischemic and haemorrhage subtypes. Also,
our cohort would relate to
‘
all stroke
’
as not all patients had detailed
cerebral imaging, but stroke would be a
‘
hard
’
endpoint, in contrast to
TIAs (which were not included) which are a
‘
soft
’
endpoint. Of note,
the healthcare system for stroke in this community-based cohort
was consistent over time, and we ascertained the stroke cases
according to careful medical history and hospitalization records.
Fur-ther validation studies of these scores in the general population, as
well as other non-AF populations should be performed, that include
both Asian and non-Asian cohorts.
In conclusion, contemporary stroke risk strati
fi
cation schema used
for AF can also be applied to non-AF populations with a similar
(mod-est) predictive value. Given their simplicity (e.g. CHADS
2score) and
pending further validation studies, these scores could possibly be
used for a
‘
quick
’
evaluation of stroke risk in non-AF populations, in
a similar manner to AF populations.
Competing interests
Prof Lip has served as a consultant for Bayer, Astellas, Merck, Sano
fi
,
BMS/P
fi
zer, Daiichi-Sankyo, Biotronik, Portola and Boehringer Ingelheim
and has been on the speakers' bureau for Bayer, BMS/P
fi
zer, Boehringer
Ingelheim, and Sano
fi
Aventis.
Other authors
—
none declared, as relevant to this mauscript.
Acknowledgements
The authors of this manuscript have veri
fi
ed that they comply
with the principles of ethical publishing in the International Journal
of Cardiology.
References
[1] Wolf PA, D'Agostino RB, Belanger AJ, Kannel WB. Probability of stroke: a risk
pro-file from the Framingham Study. Stroke 1991;22:312-8.
[2] Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Valida-tion of clinical classification schemes for predicting stroke: results from the Na-tional Registry of Atrial Fibrillation. JAMA 2001;285:2864-70.
[3] Lip GY, Nieuwlaat R, Pisters R, Lane DA, Crijns HJ. Refining clinical risk stratifi ca-tion for predicting stroke and thromboembolism in atrialfibrillation using a novel risk factor-based approach: the euro heart survey on atrialfibrillation. Chest 2010;137:263-72.
[4] Karthikeyan G, Eikelboom JW. The CHADS2score for stroke risk stratification in
atrialfibrillation—friend or foe? Thromb Haemost 2010;104:45-8.
[5] Keogh C, Wallace E, Dillon C, Dimitrov BD, Fahey T. Validation of the CHADS2
clin-ical prediction rule to predict ischaemic stroke. A systematic review and meta-analysis. Thromb Haemost 2011;106:528-38.
[6] Olesen J, Lip GYH, Hansen ML, et al. Validation of risk stratification schemes for predicting stroke and thromboembolism in patients with atrialfibrillation: a nation-wide cohort study. BMJ 2011 Jan 31;342:d124.
[7] Boriani G, Botto G, Padeletti L, et al. Improving stroke risk stratification using the CHADS2and CHA2DS2-VASc risk scores in paroxysmal atrialfibrillation patients
by continuous arrhythmia burden monitoring. Stroke 2011;42:1768-70. [8] Van Staa TP, Setakis E, Di Tanna GL, Lane DA, Lip GY. A comparison of risk
strat-ification schema for stroke in 79884 atrialfibrillation patients in general prac-tice. J Thromb Haemost 2011;9:39-48.
[9] Abu-Assi E, Otero-Raviña F, Allut Vidal G, et al. Comparison of the reliability and validity of four contemporary risk stratification schemes to predict thromboem-bolism in non-anticoagulated patients with atrial fibrillation. Int J Cardiol 2013;166:205-9.
[10] Chao TF, Lin YJ, Tsao HM, et al. CHADS(2) and CHA(2)DS(2)-VASc scores in the prediction of clinical outcomes in patients with atrialfibrillation after catheter ab-lation. J Am Coll Cardiol 2011;58:2380-5.
[11] Camm AJ, Kirchhof P, Lip GY, et al. Guidelines for the management of atrialfibrillation: the task force for the management of atrialfibrillation of the European Society of Car-diology (ESC). Eur Heart J 2010;31:2369-429.
[12] Lip GY. Stroke in atrialfibrillation: epidemiology and thromboprophylaxis. J Thromb Haemost 2011;9(Suppl. 1):344-51.
[13] Welles CC, Whooley MA, Na B, Ganz P, Schiller NB, Turakhia MP. The CHADS2score
predicts ischemic stroke in the absence of atrialfibrillation among subjects with cor-onary heart disease: data from the Heart and Soul Study. Am Heart J 2011;162: 555-61.
[14] Goto S, Bhatt DL, Röther J, et al. Prevalence, clinical profile, and cardiovascular out-comes of atrialfibrillation patients with atherothrombosis. Am Heart J 2008;156: 855-63 [863.e2].
[15] Wang TJ, Massaro JM, Levy D, et al. A risk score for predicting stroke or death in individuals with new-onset atrialfibrillation in the community: the Framingham Heart Study. JAMA 2003;290:1049-56.
[16] Rietbrock S, Heeley E, Plumb J, van Staa T. Chronic atrialfibrillation: incidence, prevalence, and prediction of stroke using the Congestive heart failure, Hyperten-sion, Age >75, Diabetes mellitus, and prior Stroke or transient ischemic attack (CHADS2) risk stratification scheme. Am Heart J 2008;156:57-64.
[17] Fuster V, Ryden LE, Cannom DS, et al. ACC/AHA/ESC 2006 guidelines for the man-agement of patients with atrialfibrillation—executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Prac-tice Guidelines and the European Society of Cardiology Committee for PracPrac-tice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Manage-ment of Patients with Atrial Fibrillation). Eur Heart J 2006;27:1979-2030. [18] Singer DE, Albers GW, Dalen JE, et al. Antithrombotic therapy in atrialfibrillation:
American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th edition). Chest 2008;133(6 Suppl.):546S-92S.
[19] National Collaborating Centre for Chronic Conditions. Atrialfibrillation: national clinical guideline for management in primary and secondary care. London: Royal College of Physicians; 2006.
[20] Chien KL, Su TC, Hsu HC, et al. Constructing the prediction model for the risk of stroke in a Chinese population: report from a cohort study in Taiwan. Stroke 2010;41:1858-64.
[21] Lee YT, Lin RS, Sung FC, et al. Chin-Shan Community Cardiovascular Cohort in Taiwan: baseline data andfive-year follow-up morbidity and mortality. J Clin Epidemiol 2000;53:836-46.
[22] Fang MC, Go AS, Chang Y, et al. Comparison of risk stratification schemes to pre-dict thromboembolism in people with non-valvularatrialfibrillation. J Am Coll Cardiol 2008;51:810-5.
[23] Chien KL, Su TC, Jeng JS, et al. Carotid artery intima-media thickness, carotid plaque and coronary heart disease and stroke in Chinese. PLoS One 2008;3: e3435.
[24] Chien KL, Sung FC, Hsu HC, Su TC, Lin RS, Lee YT. Apolipoprotein A1 & B, and stroke events in a community-based cohort in Taiwan: report of Chin-Shan Community Cardiovascular Study. Stroke 2002;33:39-44.
[25] Chen PC, Chien KL, Hsu HC, Su TC, Sung FC, Lee YT. Metabolic syndrome and C-reactive protein in stroke prediction: a prospective study in Taiwan. Metabo-lism 2009;58:772-8.
[26] Collins GS, Altman DG. An independent external validation and evaluation of QRISK cardiovascular risk prediction: a prospective open cohort study. BMJ 2009;339:b2584.
[27] Hippisley-Cox J, Coupland C, Vinogradova Y, et al. Predicting cardiovascular risk in England and Wales: prospective derivation and validation of QRISK2. BMJ 2008;336: 1475-82.
[29] Ovbiagele B, Liebeskind DS, Kim D, Ali LK, Pineda S, Saver JL. Prognostic value of Framingham Cardiovascular Risk Score in hospitalized stroke patients. J Stroke Cerebrovasc Dis 2011;20:222-6.
[30] Bhopal R, Fischbacher C, Vartiainen E, Unwin N, White M, Alberti G. Predicted and observed cardiovascular disease in South Asians: application of FINRISK, Framing-ham and SCORE models to Newcastle Heart Project data. J Public Health (Oxf) 2005;27:93–100.
[31] Eckman MH, Singer DE, Rosand J, Greenberg SM. Moving the tipping point: the decision to anticoagulate patients with atrialfibrillation. Circ Cardiovasc Qual Outcomes 2011;4:14-21.
[32] Liao J, Khalid Z, Scallan C, Morillo C, O'Donnell M. Noninvasive cardiac monitoring for detecting paroxysmal atrialfibrillation orflutter after acute ischemic stroke: a systematic review. Stroke 2007;38:2935-40.