Respiration Rate and Respiratory Quotient of Harvested Oyster Mushrooms at Different Storage Temperatures
By:
Gede Arda1, B. Rahardjo2and Nursigit Bintoro2
1
Department Agricultural Engineering
Faculty of Agricultural Technology, Udayana University Bali, Indonesia
2
Department of Food and Agricultural Engineering
Faculty of Agricultural Technology, University of Gadjah Mada, Yogyakarta, Indonesia
Abstract
Temperature management on postharvest handling of fruits and vegetables has an important role to preserve the product to prolong their sales appeal period or shelf life. Besides, availability of gases around the product influences the product behavior also. The important aspects of the related-physiological behaviors are respiration rate and respiratory quotient. Those provide the initial information about the changes of gas composition in inner space of storage jar as well as the sign of the shift of aerobic to anaerobic respiration occurred. Based on that issue, the aim of this work is to study the physiological behaviors of Oyster mushroom on various temperature storage and gas concentration. The object of study was Oyster mushrooms (Pleurotus ostreatus)
produced by the local farmers. Respiration rate was determined by closed or static system until gas composition shows 1-2% of O2 and 20-21 % of CO2 under 5
0
C, 15 0C and ambient storage temperature. The result showed that respiration rate of Oyster mushroom was influenced by storage temperature with activation energies of 2.33 kJ/mol and 2.08 kJ/mol for O2 consumption and CO2 evolution respectively. Respiration rates resulted from this measurement were 55.45 mlO2/kg.h, 135.97 mlO2/kg.h, 318.97 mlO2/kg.h and 61.59 mlCO2/kg h, 165.34 mlCO2/kg h, 335.76 mlCO2/kg h for 5
0
C, 150C and ambient storage temperatures respectively. In this work also was found that Oyster mushrooms were not CO2 sensitive (tolerant of high CO2 concentration). The value of respiratory quotients were not much different although the concentrations of CO2 reach more than 15 %. Respiratory quotient of mushroom under 5 0C, 15 0C and ambient storage temperatures were 1.11, 1.22 and 1.05 respectively.
1. Introduction
Some agricultural produces are perishable that characterized by their short shelf life. Physical, chemical or biological damages on product accelerate the deterioration of products. The improper management of storage conditions, i.e., too warm or too cold for a certain product, is another factor that increases physiological deterioration. The shelf life of certain products could be extended by applying the proper packaging system also. Temperature and humidity were essential factors for fresh products shelf-life-extension-efforts. Respiration rate which indicates many metabolic activities of the tissues as well as the rate of quality degradation of stored products could be suppressed through lowering the storage temperature to slightly above the critical temperature of certain product (Saltveit, 2004). The second factor that has great influence on product quality is humidity. Weight loss can be controlled by reducing the gradient of water vapor pressure (WVP) between product's surface and surrounding atmosphere (Perkins et al, 2008). Losing 1-2 % of water of produce can reduce sales appeal of produce of fresh produce (Kader, 2002). Therefore, the packaging system that can maintain the oxygen and carbon dioxide at certain concentration as well as humidity is needed. The characteristics of packaging system have strong relationship with characteristics of packed product. It has been proved that every product has their own unique properties; therefore, designing of packaging system should be based on those properties. The initial knowledge that is important on designing packaging system is respiration rate on different temperature storage. Respiration rate is main factor that changes the composition of atmosphere dynamically on jar’s head space. The gases concentration inside jar’s head space is always changing dynamically until the equilibrium condition is obtained. This
equilibrium condition reached when the respiration rate, transfer of gas oxygen and carbon dioxide were equal. Oxygen that consumed by packed products was compensated by oxygen permeating from the surrounding atmosphere. In the same way to oxygen, the carbon dioxide which diffuses to surrounding atmosphere is replaced by diffusion from product’s tissues. On one hand, oxygen must be available for respiration to avoid anaerobic respiration during storage. On the other hand, over supply of oxygen makes the packaging has no benefit for prolonging shelf life. The critical threshold of gas concentration is considered to design the magnitude of gas transfer of packaging film (Beaudry et al., 1992).
The water vapor that produced from respiration and transpiration continuously adds the water vapor contained in jar’s head space. Water vapor diffuses to ambient environment with a certain magnitude. Most of them condense on product’s or packaging’s surface when saturated water vapor pressure at certain temperature is exceeded. This liquid water encourages the microbial development and reduces the diffusivity of packaging film.
Mushroom is one of the vegetables that have short shelf life, only 3 days on ambient temperature (Czapski and Szudyga, 2000). Their dermal tissues have no cuticle to protect them from physical damages or microbial attack or water loss (Martine et al., 2000). High moisture content in their fruit body’s makes mushrooms respire and transpire with high rate. Mushroom need low temperature storage (0 - 20C), low O2 concentration
and relatively high CO2 concentration as
well as high humidity (90-98%) (Thompsonet al, 2002).
of Oyster mushroom, that were still rare, to choice the right kind of bag plastic or to design the proper packaging system. Therefore, the aim of this work is to study the physiological behaviors of Oyster mushroom on several variations of temperature storage and gas concentration.
2. Theoretical Approach
2.1 Respiration
Respiration is the oxidative breakdown of complex substrate normally present in plant cells -such as starch, sugars, and organic acids- to simpler molecules such as CO2 and H2O.
Concomitant with this catabolic reaction is the production of energy and intermediate molecules that are required to sustain the myriad of anabolic reactions essential for the maintenance of cellular organization and membrane integrity of living cells (Kader and Saltveit, 2003). Oxygen which is needed for oxidation must be available in the course of respiration process. Respiration on fresh produce located on free atmosphere can be considered that availability of oxygen is unlimited, therefore availability of oxygen does not inhibits the respiration. Otherwise, limitation of oxygen availability on surrounding atmosphere suppresses the rate of respiration (Kader and Saltveit, 2003). Depletion of oxygen and accumulation of carbon dioxide inhibit the reaction on internal tissue. Respiration is enzymatic reaction that has common characteristic such as depend on substrate concentration, pH, and temperature and of course the concentration of enzyme that catalyses the reaction (Metzler, 2002). Enzyme needs pH range 5.5 to 7.5 to perform optimum activity on reaction. Besides, enzyme needs optimum reaction temperature also (Kader and Saltveit, 2003). Enzyme can be inactivated if the reaction temperature very low otherwise enzyme will be denaturalized on too high reaction temperature (Belitz, Grosch,
Schiberle, 2009). So, three factors that influence the activity of enzyme are substrates concentration, pH and reaction temperature. Heat is released during respiration, 686 kcal or 2870 kJ heat is released per 1 mole of glucose. Besides, 0.8-1 fraction of heat released is used to heat the surrounding atmosphere or to increase temperature of product (Ooraikul
and Stiles, 1991).
2.2 Respiratory quotient
Respiratory quotient is defined as ratio of production rate of CO2 to consumption rate of O2. According to respiration equation in which mole O2 consumed is equal to mole of CO2 produced indicated by coefficient of every species that involve in reaction.
+ 6 6 + 6 +
(Ooraikul and Stiles, 1991).
From the definition it can be concluded that the respiratory quotient is preferable to evaluate the respiration. Increasing on magnitude of respiratory quotient indicates respiration is approaching to anaerobic respiration or production rate of CO2 is higher than consumption rate of O2. Kader (2002) suggested that respiratory quotient on aerobic respiration range between 0.8 to 1.3 and the exact value is depending on the intrinsic properties of product and the storage condition.
3. Method
3.1 Sample preparation
which covers the mushroom’s surface. Mushrooms were weighted before packed into box. Mushroom then transported to Operation Unit Laboratory of Agricultural Technology Faculty of Gadjah Mada University. The last step was storing the mushrooms at 5 0C or 15 0C for 1 hour to equilibrate the mushroom’s temperature according to treatment temperature.
Three replications were prepared to investigate the respiration rate on different storage temperatures. The plastic jars 9 liters were used on this work. Two holes were made on the lid of plastic jars to facilitate the measurement of gas concentration.
3.2 Respiration rate measurement
Mushrooms were weighted range from 300 to 500 grams according to storage temperature and then were placed in 9 liters plastic jars. Paraffin wax was used to cover the gap between the lid and the body of the jar as well as at the hole at its lid. The mass of mushroom used were ±500 grams, ± 400 grams and ±300 grams for 5oC, 15oC and room temperature respectively. Those masses differences were intentionally used for avoiding small gas concentration change on measurement-time-range in which was difficult to measure due to lack of accuracy of instruments. Gas compositions were measured by Cosmotector XP-314 type (Sensorex Oy, Finland) for Oxygen concentration and Cosmotector XP-318 type (Sensorex Oy, Finland) for CO2
concentration. The instruments have an accuracy of 0.1%. Gas composition measurements were taken every 4 hours, 3 hours and 2 hours for 50C, 150C and room temperature respectively, until 1-2% oxygen level was obtained.
4. Results and Discussion
4.1 Respiration rate
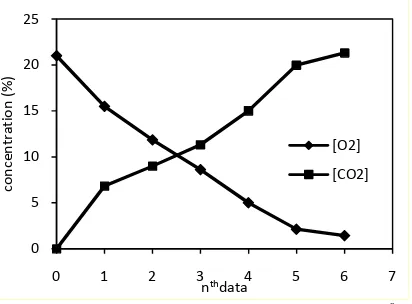
Respiration rate measured in this work was not daily respiration development of harvested mushrooms but respiration rate at different gas composition. The gas composition change occurred when oxygen was consumed and carbon dioxide was produced by product. The mushroom stored without interruption and measurements were periodically conducted during storage. Each data point in the figures below was taken to be initial gas composition for the next measurement.
Amount of measurement data indicated the rate of gas composition change inside the jar’s head space. Amount of measurement data time by period of measurement showed the spending time for the head space to obtain 1% oxygen or 20% carbon dioxide level. The lowest storage temperature, 5 0C, spent 52 hours to obtain those levels. This period was longer than period that mushrooms spent at 15 0C with 18 hours and 14 hours at room temperature. Crossing point between O2and CO2curves
occur at near 10% level. This indicates that mushrooms spend equal time to obtain the half level of head space concentration and also indicates that the ratio between O2
consumption rate and CO2 production rate
approaches 1.
Figure 1. Gas concentration development at 50C (data are taken every 4 hours)
0 5 10 15 20 25
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14
co
n
ce
n
tr
a
ti
o
n
(%
v/
v)
nthdata
[O2]
Figure 2. Gas concentration development at 150C (data are taken every 3 hours)
Figure 3. Gas concentration development at room temperature (data are taken every 2 hours)
Figure 1-3 show that rapid respiration occurs at initial storage of mushrooms. Abundant oxygen on head space and field heat that still remain accelerate the respiration. Respiration continuously decreases when oxygen is getting thinner and carbon dioxide getting thicker as well as product temperature getting colder. Oyster mushroom respires rapidly faster than 60 ml O2/hr.kg, therefore mushroom
Oyster is grouped in vegetables with very high respiration rate (Kader, 2002).
4.2 Respiration and temperature
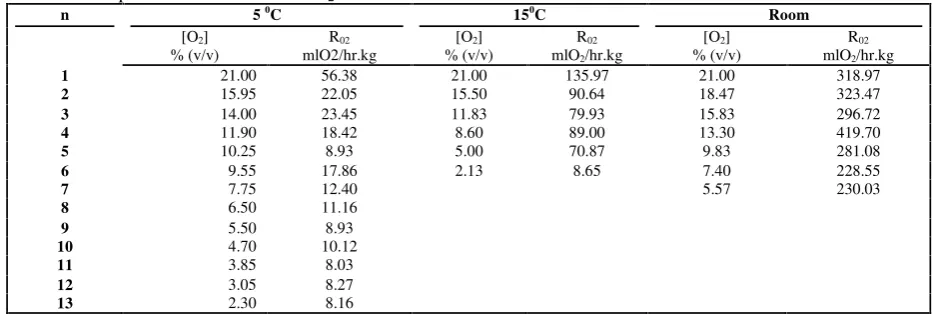
Data demonstrate that temperature had major influence on Oyster mushroom respiration rate. Air temperature is the most important variable because it tends to control flesh temperature of perishable commodities (Thompson, 2002). Lowering temperature to 150C and 50C could suppress the consumption rate of O276.4%
and 95%, respectively. Slight different effect occurred to production rate of CO2.
Lowering temperature to 150C and 50C suppressed the production rate of CO2
50.7% and 81.7%, respectively. On the other hand, gas composition has minor effects on Oyster mushroom respiration i.e, at room temperature respiration rate decreases to 27.9%. All of this value was calculated from the data taken at first measurement of gas concentration. First measurement was considered indicate the influence of temperature alone. It was different to next measurement which was considered indicate the influence of temperature and gas concentration. This value is as great as respiration of broccoli. Relative to held in the ambient air, the reduction in respiration rate is about 28% for broccoli heads in 2% O2 (Kader and
Saltveit, 2003).
Table 1.Respiration rate on 50C, 150C and room temperature storage
replication Consumption rate of O2 Production rate of CO2 Respiratory Quotient (RQ)
Ea (kJ/mol)
(ml/hr.kg) (ml/hr.kg) O2 CO2
50C 150C Room 50C 150C Room 50C 150C Room
2.33 2.09
1 55.82 135.97 302.18 62.52 173.05 319.38 1.12 1.27 1.06
2 56.94 135.97 302.18 64.20 162.68 319.38 1.13 1.20 1.06
3 53.59 135.97 352.55 58.05 160.29 368.51 1.08 1.18 1.05
average 55.45 135.97 318.97 61.59 165.34 335.76 1.11 1.22 1.05
0 5 10 15 20 25
0 1 2 3 4 5 6 7
co
n
ce
n
tr
a
ti
o
n
(
%
)
nthdata
[O2]
[CO2]
0 5 10 15 20 25
0 1 2 3 4 5 6 7 8
co
n
ce
n
tr
a
ti
o
n
(
%
v
/v
)
nthdata
Table. 2 Respiration rate at different O2concentration
n 50C 150C Room
[O2] R02 [O2] R02 [O2] R02
% (v/v) mlO2/hr.kg % (v/v) mlO2/hr.kg % (v/v) mlO2/hr.kg
1 21.00 56.38 21.00 135.97 21.00 318.97
2 15.95 22.05 15.50 90.64 18.47 323.47
3 14.00 23.45 11.83 79.93 15.83 296.72
4 11.90 18.42 8.60 89.00 13.30 419.70
5 10.25 8.93 5.00 70.87 9.83 281.08
6 9.55 17.86 2.13 8.65 7.40 228.55
7 7.75 12.40 5.57 230.03
8 6.50 11.16
9 5.50 8.93
10 4.70 10.12
11 3.85 8.03
12 3.05 8.27
13 2.30 8.16
Columns of Table.2 showed the influence of O2 concentration changes. But,
combination of temperature and gas concentration changes gives the great influence on mushroom respiration.
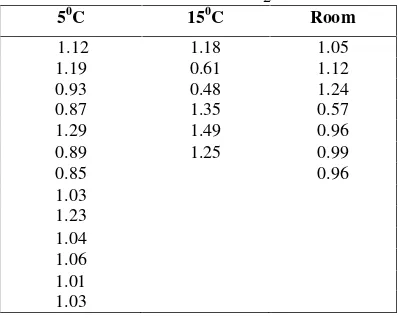
4.3 Respiratory quotient (RQ)
Respiratory quotient could be define as the rate ratio of CO2 production to O2 consumption; CO2 and O2 could be measured in moles or volumes depending on the substrate being oxidized. RQ values for fresh commodities range from 0.7 to 1.3 for aerobic respiration. When carbohydrates are being aerobically oxidized, the RQ is near 1, while it is < 1 for lipids, and > 1 for organic acids. Very high RQ values usually indicate anaerobic respiration in those tissues that produce ethanol. In such tissues, a rapid change in the RQ could be used as an indication of the shift from aerobic to anaerobic respiration (Saltveit, 2004). The value of the RQ presented in Table 1. Although has the various value at different temperature, but the deviations is small. Those values were calculated from initial respiration where the surrounding atmosphere was still rich in O2. Kader (2002) and Saltveit
(2004) suggest that aerobically respiration on fresh produces stored at physiological temperature range from 0.7 – 1.3. Therefore, those values indicate that respiration was aerobic. This conditions continuously changes according to O2
changes, but was still in the aerobically respiration range (Table 3). Give more attentions on value on 50C and 150C, they show that Oyster mushrooms was not sensitive to O2low level or CO2high level. Kader (2002) suggest that mushrooms are one of vegetables that respire highly but withstand to high level of CO2. Evaluation on mushroom appearance that are stored at 50C and packed with plastic jar for 3 days give the prove that they can preserve with low temperature and low O2concentration.
The excessive accumulation of CO2 (> 12%) inside the package can cause physiological injuries to the produce, which in the case of mushrooms, results in severe browning (Nichols and Hammond, 1973; Lopez-Briones et al., 1992) is not proven in this case. But authors suggest that this fact need to be investigated further, because this research is not intended to prove or to refuse that fact. Especially, the similar influences occur on different species and different storage conditions.
4.4 Activation energy of respiration
The gradient of each linear equations come from linearization represents the activation energy and universal gas constant ratio (Ea/R).
Table 3. Respiratory quotient on different temperature and O2composition*
50C 150C Room
1.12 1.18 1.05
1.19 0.61 1.12
0.93 0.48 1.24
0.87 1.35 0.57
1.29 1.49 0.96
0.89 1.25 0.99
0.85 0.96
1.03 1.23 1.04 1.06 1.01 1.03
*TheO2concentrations refers to Table 2
Applying the Arrhenius’ law, respiration rate of mushrooms along physiological temperature can be counted.
= 0.28 ...(a)
= 0.25 ...(b),
Where subscript on parameter RR and T represent value of respiration rate on storage temperature T at condition 1 (reference) and storage temperature T will be found. Equations (a) and (b) are the predictive equation for O2 consumption
and CO2 production rate, respectively.
With the activation energy (Ea) are 2.33 kJ/mol and 2.08 kJ/mol for O2
consumption and CO2 evolution
respectively, prediction of respiration rate of Mushroom Oyster at 50C to 30 0C is easy to count. This Arrhenius equation assists the engineer to design of packaging system.
5. Conclusions
The important physiological aspects of postharvest handling of fruits and vegetables are physiological behaviors including respiration rate and respiratory
quotient. Oyster mushrooms show the significant influence of temperature on its respiration but less dependent on atmospherically gas composition. Combination of low temperature and low oxygen give the great effects to mushroom respiration and less effect on respiratory changes. Predictions of the respiration rate of mushrooms can be established satisfyingly by applying the Arrhenius equations.
References
Beaudry, R.M., Cameron, A.C., Shirazi, A. and Lange, D.D. 1992. Modified atmosphere packaging of blueberry fruit: Effect of temperature on package oxygen and carbon dioxide. J. Amer. Soc. Hortic. Sci. 117: 436-441.
Belitz, H.-D, W. Grosch and P. Schieberle. 2009. Food Chemistry, 4th revised and extended edition. Springer-Verlag Berlin Heidelberg. German Czapski, J., & Szudyga, K. 2000. Frozen
mushroom quality as affected by strain, flush, treatment before
freezing and time of storage.Journal of Food Science, 65(4), 722–725 Kader, A.A and Michael E. Saltveit.
Respiration and Gas Exchange. In Postharvest Physiology and Pathology of Vegetables, 2nd edition. Marcel Dekker. Inc. New York. Kader, A.A. 2002.Postharvest Technology
of Horticultural Crops. Third Edition. Regent of University of California, Divison of Agriculture and Natural Resources. Publication 3311 USA
Lopez-Briones, G., Varoquaux, P., C. Yves, B. J., Bureau. G. and Pascat, B. 1992. Storage of common mushroom under controlled atmospheres. Int. J. Food Sci.Technol.27: 493-505.
hydrogen peroxide to extend the shelf life of fresh sliced mushrooms, Lebensmittel-Wissenschaft und-Technologie33(2000), pp. 285–289. Mikal E. Saltveit. 2004. Respiratory Metabolism. In The Commercial Storage of Fruits, vegetables, florist
and Nursery Stocks.
http://www.ba.ars.usda.gov
Nichols, R. and Hammond, J.B.W. 1973. Storage of mushrooms in pre-packs: the effect of changes in carbon dioxide and oxygen on quality. J. Food Sci. Agric. 24 : 1371-1381. Ooraikul, B. and M.E. Stiles. 1991.
Modified Atmosphere Packaging of Food. Ellis Harwood. Great Britain Penelope Perkins-Veazie, Julie K. Collins ,
Luke Howard. 2008. Blueberry fruit response to postharvest application of ultraviolet radiation. Postharvest Biology and Technology 47 (2008) 280–285