Brain Research 887 (2000) 482–483
www.elsevier.com / locate / bres
Short communication
The effects of neocortical ectopias on Lashley III water maze learning
in New Zealand Black mice
a ,1 c a a,b ,
*
Lynn A. Hyde
, Gordon F. Sherman , Amy Jo Stavnezer, , Victor H. Denenberg
a
Biobehavioral Sciences Graduate Degree Program, University of Connecticut, Storrs, CT 06269-4154, USA b
Department of Psychology, University of Connecticut, Storrs, CT 06269-4154, USA c
Dyslexia Research Laboratory, Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA 02215, USA
Accepted 3 October 2000
Keywords: New Zealand Black mouse; Neocortical ectopia; Maze learning; Lashley maze
Approximately 50% of New Zealand Black (NZB) mice ectopic NZB mice demonstrated learning impairments in have neocortical ectopias, which are misplaced clusters of the Lashley maze when compared to non-ectopic controls. neurons located in layer I of cortex [17,18]. Previous We used a water version of the Lashley III maze that experiments have found that ectopias in NZB mice were can be learned by the use of extra-maze cues or by related to differences in learning [5,15]. This has been memorizing the chain of correct left and right turns [7]. A shown to be the case for BXSB / MpJ mice as well [1– total of 38 NZB / BINJ (21 male and 17 female) mice were 4,6,9–11,19]. Specifically, left-pawed ectopic NZB mice given six trials per day (three trials in the morning and learned a simple water escape task better than left- and three in the afternoon, separated by at least 4 h) for 5 days right-pawed non-ectopics, that, in turn, learned better than [9]. The inter-trial interval for the three trials was|10 min.
right-pawed ectopics [5]. In addition, NZB ectopics were A learning index (number of correct entries divided by less active in a small activity chamber, escaped faster in a total number of entries) was calculated and the number of non-spatial water T-maze, and were poorer in several forward, forward T-choice, forward cul-de-sac entry, and measures of spatial Morris maze learning [15]. backward errors were counted. Mice were transcardially Another learning task in which NZB mice have been perfused with physiological saline and 10% formalin, and tested is the Lashley III water maze [7]. This is a relatively their brains were examined for the presence of neocortical complex maze, containing cul-de-sacs that the animal has ectopias.
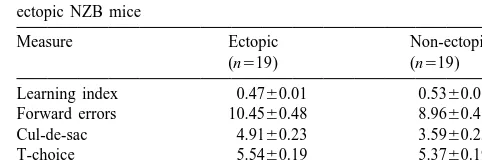
to learn to avoid, and T-choices where the animal has to Of the 38 mice, 19 (50%) were non-ectopic and 19 learn to make the correct left or right turn. The mice are (50%) were non-ectopic. Table 1 shows the Lashley maze given two widely spaced trials per day over several days. learning measures for the two groups. Ectopics had lower However, NZB mice, regardless of ectopia status, were learning index scores, made more forward errors, made reported to be poor learners in the Lashley maze, even with more forward cul-de-sac entries, and tended to make more extra training trials [14], making learning comparisons backward errors, than non-ectopics (F ’s(1,36)55.82, 4.87, between ectopic and non-ectopic mice meaningless.
Re-Table 1
cently, however, we have been able to obtain proficient
Lashley III water maze behavioral means6S.E. for ectopic and
non-Lashley maze learning in NZB mice by giving the mice ectopic NZB mice extra training trials spaced more closely in time [9]. This
Measure Ectopic Non-ectopic
enabled us to compare NZB ectopics to non-ectopics in
(n519) (n519)
Lashley maze learning. In the present study, we found that
Learning index 0.4760.01 0.5360.01*
3
Forward errors 10.4560.48 8.9660.48*
Cul-de-sac 4.9160.23 3.5960.23*
*Corresponding author. Present address: Biobehavioral Sciences T-choice 5.54 3 60.19 5.3760.19
Graduate Degree Program, 3107 Horsebarn Hill Road, Storrs, CT 06269- [
Backward errors 12.0860.57 10.0760.57 4154, USA. Tel.:11-860-486-3826; fax:11-860-486-3827.
E-mail address: [email protected] (V.H. Denenberg). * Main effect of ectopia, P,0.05. 1Present address: Department of Pediatrics and Psychiatry, University 3Ectopia
3linear trend for trial blocks, P,0.05. [
of Colorado School of Medicine, Denver, CO 80262, USA Main effect of ectopia, P,0.08.
L.A. Hyde et al. / Brain Research 887 (2000) 482 –483 483
[2] G.W. Boehm, G.F. Sherman, B.J. Hoplight, L.A. Hyde, N.S. Waters,
8.30, and 3.37, P’s,0.03, 0.04, 0.007, and 0.08,
respec-D.M. Bradway, A.M. Galaburda, S.A. Ahmed, V.H. Denenberg,
tively). In addition, ectopics had a flatter rate of decline of
Learning in year-old females autoimmune BXSB mice, Physiol.
the number of forward and forward T-choice errors over Behav. 64 (1998) 75–82.
trial blocks as compared to non-ectopics (ectopia3linear [3] G.W. Boehm, G.F. Sherman, B.J. Hoplight, L.A. Hyde, N.S. Waters,
trend for trial blocks: F ’s(1,504)57.01 and 5.50, P’s, D.M. Bradway, A.M. Galaburda, V.H. Denenberg, Learning and memory in the autoimmune BXSB mouse: effects of neocortical
0.01 and 0.025).
ectopias and environmental enrichment, Brain Res. 726 (1996)
Therefore, ectopic NZB mice were impaired in learning
11–22.
the Lashley maze compared to their non-ectopic controls. [4] G.W. Boehm, G.F. Sherman, G.D. Rosen, A.M. Galaburda, V.H. We have evidence that NZB mice were using extra maze Denenberg, Neocortical ectopias in BXSB mice: effects upon
cues to a greater extent than memorizing the left / right reference and working memory systems, Cereb. Cortex 6 (1996) 696–700.
turns to learn the maze, suggesting that the Lashley maze
[5] V.H. Denenberg, G.F. Sherman, L.M. Schrott, G.D. Rosen, A.M.
is a spatial task [9]. The results of this study are in
Galaburda, Spatial learning, discrimination learning, paw preference
agreement with another study which demonstrated that and neocortical ectopias in two autoimmune strains of mice, Brain ectopic NZB mice were inferior to non-ectopics in learning Res. 562 (1991) 98–104.
the spatial Morris maze [15]. [6] V.H. Denenberg, G.F. Sherman, L.M. Schrott, N.S. Waters, G.W. Boehm, A.M. Galaburda, L.E. Mobraaten, Effects of embryo
BXSB / MpJ (BXSB) mice also have neocortical
ec-transfer and cortical ectopias upon the behavior of BXSB-Yaa and
topias [17,18]. Both BXSB and NZB ectopics were inferior
BXSB-Yaa1mice, Dev. Brain Res. 93 (1996) 100–108.
to non-ectopics in learning the Lashley maze [7,8] (see [7] V.H. Denenberg, N. Talgo, D.A. Carroll, S. Freter, R. Deni, A also this study). However, BXSB ectopics were superior to computer-aided procedure for measuring Lashley III maze
per-non-ectopics in learning the Morris maze [3,6], while NZB formance, Physiol. Behav. 50 (1991) 857–861.
[8] G.R. Fink, K. Zilles, A. Schleicher, Postnatal development of
ectopics were inferior [15]. It is not known why ectopias
forebrain regions in the autoimmune NZB mouse, Anat. Embryol.
have the same effect in both strains on Lashley maze
183 (1991) 579–588.
learning, but the opposite effect on Morris maze learning. [9] L.A. Hyde, Spatial and non-spatial working and reference memory This discrepancy may be related to the cortical location of in BXSB mice and massed and spaced learning in NZB mice,
the ectopia(s), since ectopias are usually located in somato- Doctoral dissertation, The University of Connecticut, 1998. [10] L.A. Hyde, G.F. Sherman, V.H. Denenberg, Radial-arm maze
sensory cortex in NZB mice [17,18], but in prefrontal /
learning in BXSB mice: effects of neocortical ectopias, Soc.
motor cortex in BXSB mice [17]. Another explanation
Neurosci. Abstr. 22 (1996) 485.
may be that NZB mice have other neuroanatomical abnor- [11] L.A. Hyde, G.F. Sherman, V.H. Denenberg, Non-spatial radial-arm malities that are absent in BXSB mice. For example, in maze learning in BXSB mice with neocortical ectopias, Soc.
addition to ectopias, NZB mice have a 100% incidence of Neurosci. Abstr. 24 (1998) 439.
[12] R.S. Nowakowski, Abnormalities in neuronal migration in the
ectopic hippocampal granule cells and pyramidal cells
hippocampal formation, Soc. Neurosci. Abstr. 12 (1986) 317.
[8,12,13] and small hippocampal intra- and infra-pyramidal
[13] R.S. Nowakowski, Development of the hippocampal formation in
mossy fiber projections [16]. Therefore, the combination of mutant mice, Drug Dev. Res. 15 (1988) 315–336.
having ectopia(s) and these additional hippocampal abnor- [14] L.M. Schrott, V.H. Denenberg, G.F. Sherman, G.D. Rosen, A.M.
malities may affect learning in ectopic NZB mice differ- Galaburda, Lashley maze learning deficits in NZB mice, Physiol. Behav. 52 (1992) 1085–1089.
ently than ectopic BXSB mice.
[15] L.M. Schrott, V.H. Denenberg, G.F. Sherman, N.S. Waters, G.D. Rosen, A.M. Galaburda, Environmental enrichment, neocortical ectopias, and behavior in the autoimmune NZB mouse, Dev. Brain
Acknowledgements Res. 67 (1992) 85–93.
[16] H. Schwegler, H.-P. Lipp, W.E. Crusio, NZB mouse: hippocampal mossy fiber patterns and behavioral profiles of young and older
The authors wish to thank Wei Wei, Nancy Talgo, and
animals, Drug Dev. Res. 15 (1988) 297–305.
Antis Zalkalns for technical assistance. This work was
[17] G.F. Sherman, A.M. Galaburda, P.O. Behan, G.D. Rosen,
Neuro-funded by NIH Grant 20806. anatomical anomalies in autoimmune mice, Acta Neuropathol.
(Berl.) 74 (1987) 239–242.
[18] G.F. Sherman, A.M. Galaburda, N. Geschwind, Cortical anomalies in brains of New Zealand mice: a neuropathologic model of References
dyslexia?, Proc. Natl. Acad. Sci. USA 82 (1985) 8072–8074. [19] N.S. Waters, G.F. Sherman, A.M. Galaburda, V.H. Denenberg, [1] S.A. Balogh, G.F. Sherman, L.A. Hyde, V.H. Denenberg, Effects of Effects of cortical ectopias on spatial delayed-matching-to-sample neocortical ectopias upon the acquisition and retention of a non- performance in BXSB mice, Behav. Brain Res. 84 (1997) 23–29. spatial reference memory task in BXSB mice, Dev. Brain Res. 111