Brain Research 888 (2001) 158–162
Institute of Biomedical Engineering, Kansai Denryoku Hospital, Imaichi 2-7-14, Asahi-ku, Osaka 535-0011, Japan
b
Department of Tissue Regeneration, Field of Clinical Application, Institute for Frontier Medical Science, Kyoto University, Kyoto, Japan
Accepted 19 September 2000
Abstract
This paper describes the regeneration of severed peripheral nerve axons along collagen filaments without a tube. Two thousand collagen filaments were grafted to bridge 20 mm defects of rat sciatic nerve. The number of myelinated axons was approximately 4800 in the distal end of the nerve autograft at 8 weeks postoperatively; while in the collagen-filaments nerve guide it was 5500. The results suggested the collagen filaments guided regenerating axons effectively. 2001 Elsevier Science B.V. All rights reserved.
Theme: Development and regeneration
Topic: Regeneration
Keywords: Nerve; Peripheral; Regeneration; Axon; Collagen; Filament
Axonal regeneration has been studied using a tubular polyethylene glycol diglycidyl ether as the cross-linking
prosthesis for a long time. Entubulation repair has a reagent does not elicit the macrophage response. The
lengthy history and several experimental and clinical collagen filament was further stabilized using ultraviolet
studies have explored the effectiveness of many biodegra- radiation (250 nm, 30 W, 30 min). Two-thousand collagen
dable and non-biodegradable materials with or without filaments were used to make a 22-mm-long nerve guide
some protein additives and cells [1,2,4–10,14,15,17– (Fig. 1). Twenty-two millimeter long collagen tube
(inter-22,24,25,27]. It has been reported that increased per- nal diameter 1.8 mm, wall thickness 50mm) was made and
meability—extent of exposure to surrounding tissue—im- used as a control.
proves axonal regeneration [11–13,16]. We developed a Thirty-nine 6-month-old male Wistar rats weighing
nerve guide made of collagen filaments, instead of a tube, about 250 g were used for the study. Adequate measures
to improve resorbability and permeability of the material were taken to minimize pain or discomfort. Experiments
employed and assessed its effect in peripheral nerve were carried out in accordance with the European
Com-regeneration. The nerve guides made of collagen filaments munities Council Directive of 24 November 1986. Under
were provided by Koken Co. Ltd., (Tokyo, Japan). The deep pentobarbital anesthesia, the right sciatic nerve was
collagen filament, 20mm in diameter, was made of highly exposed from the sciatic notch to the popliteal region, and
purified type I collagen. Bovine skin was subjected to a 20-mm segment of the tibial division of the nerve was
enzymatic and chemical treatments to remove non-col- removed. The proximal and distal nerve stumps were
lagenous components. The collagen filament was stabilized sutured to the 20-mm long sciatic nerve autograft with two
using polyethylene glycol diglycidyl ether as the cross- sutures using 10-0 monofilament nylon epineurial sutures
linking reagent, which cross-linked ´-NH2 groups of to bridge the nerve defect in Autograft Group. The
collagen molecules. The cross-linked collagen is resorbed proximal and distal nerve stumps were sutured to the
more slowly compared to the non cross-linked collagen collagen-filament nerve guide with two sutures using 10-0
and resists bacterial collagenase digestion. The use of monofilament nylon epineurial sutures to bridge the nerve
defect in the Fiber Group. The proximal and distal nerve stumps were inserted 1 mm into the collagen tubes and
*Corresponding author. Tel.: 181-6-6956-2729; fax: 1
81-6-6458-were held in place with two epineurial sutures in the Tube
6994.
E-mail address: [email protected] (S. Yoshii). Group. Fifteen rats received a sciatic nerve autograft
Fig. 1. Collagen-filaments nerve guide. Two-thousand collagen filaments were used to make a 22-mm long nerve guide.
(Autograft Group) and another group of 15 rats underwent nerves including the nerve guides or autografts were
implantation of nerve guides made of collagen filaments dissected out, fixed in 2.5% glutaraldehyde and postfixed
(Fiber Group). Nine rats underwent implantation of col- in 2% osmium tetroxide. Each nerve was embedded in
lagen tubes (Tube Group). In the Fiber Group, three of the 100% Epon. One micrometer thick transverse sections
removed sciatic nerves were examined histologically as the were made from the nerve to obtain sections at successive
normal control. two-millimeter intervals. Each section was stained with
Motor function was assessed for 12 weeks at 4-week toluidine blue and examined under a light microscope.
intervals using the sciatic functional index (SFI). Three Ultrathin sections of selected areas of the nerves were
rats in each group underwent the assessment. Hind feet examined under an electron microscope (Nihon Denshi
were wiped with India ink and the rat walked down a JEM 200cx, Tokyo, Japan) at 100 kV.
walking track. Pawprints were measured for both the The axonal count and fiber diameter were used to
operated and unoperated sides. The values were submitted evaluate axonal regeneration. The selected sections were
to a sciatic functional index as described by Bain et al. [3]. photographed under a light microscope (original
magnifica-Axonal regeneration across the repair site was electro- tion 3400, enlarged to 31000). Montages of whole
physiologically evaluated at 4, 8 and 12 weeks postopera- section were constructed. All myelinated axons were
tively using nine rats in each group. Before removal of the counted and the fiber diameter was measured. The number
nerve, bipolar stimulating electrodes were inserted into the of axons in the tibial division of the sciatic nerve of normal
planter surface of the foot in the receptive area of the rats was approximately 5200 (52306360), and the
diam-sciatic nerve. The previously operated diam-sciatic nerve was eter was 6.9mm. The dissected nerve guides in the Fiber
exposed and recording electrodes were placed on the Group were brown-colored at 4 weeks postoperatively and
sciatic nerve 3 mm proximal and 3 mm distal to the repair their mean diameter was 2.4 mm, which was larger than
site. The latency was measured and the nerve conduction that of the grafted tibial division of the sciatic nerve (mean
velocity was calculated (Electronic stimulator SEN-3201, diameter 1.1 mm). The dissected collagen tubes were
Dual-beam memory oscilloscope VC-10, Add scope brown-colored and semitransparent in Tube Group. In all
ATAC-210, Nihon Kohden, Japan). In addition to conduc- collagen tubes, proximal stumps had small outgrowth
tion velocity, the sciatic nerve was stimulated proximal extending less than 6 mm. Neuromas were found at both
and distal to the repair site using two pairs of electrodes, ends of the all six autografts in the Autograft Group at 4
and a needle electromyogram (EMG) was recorded from weeks postoperatively. No neuroma was found at the
the short flexor muscles of the foot. suture in the Fiber Group. Regenerated unmyelinated
At 4 and 8 weeks postoperatively, six rats in the axons were found, but no regenerated myelinated axon was
Autograft and Fiber Groups, and three rats in the Tube found at the distal end of the graft—1 mm proximal to the
160 S. Yoshii, M. Oka / Brain Research 888 (2001) 158 –162
Fiber Group at 4 weeks postoperatively. The distal ends of EMG was recorded from the short muscles of the foot in
the regenerated myelinated axons were found at 10–14 all groups at 4 and 8 weeks postoperatively. Interpretable
mm distal to the proximal suture threads in the Autograft compound action potentials were recorded for two out of
Group at 4 weeks postoperatively. The distal ends of the three rats in the Autograft Group and two out of three rats
regenerated myelinated axons were found at 12–16 mm in the Fiber Group at 12 weeks postoperatively. The mean
distal to the proximal suture threads in the Fiber Group. nerve conduction velocity was 51.7 m / s for the Autograft
The distal ends of the regenerated myelinated axons were Group and 40.0 m / s for the Fiber Group. In all six rats
found at 2–4 mm distal to the proximal suture threads in from the Autograft and Fiber Groups, good EMG was
the Tube Group. Under the electron microscope, many recorded from the short flexor muscles of the foot when
macrophages were found around the collagen material to the nerve was stimulated proximal or distal to the repair
phagocytose it in the Fiber Group and the Tube Group at 4 site. No interpretable compound action potential or no
weeks postoperatively. EMG from the short muscles of the foot was recorded in
The dissected nerve guides in the Fiber Group were the Tube Group at 12 weeks postoperatively.
white-colored at 8 weeks postoperatively and their mean Myelinated axons of the rat sciatic nerve had
regener-diameter was 1.8 mm, which was larger than that of the ated 20 mm along collagen-filaments without a tube or
grafted tibial division of the sciatic nerve (1.1 mm) in the neurotrophic additives by 8 weeks postoperatively in this
Autograft Group. No neuroma was found at the suture in study. It has been reported that increased permeability
the Autograft and Fiber Groups at 8 weeks postoperatively. improves axonal regeneration [11–13,16]. Yoshii et al.
The collagen tubes were not found in all three rats and a reported that they had obtained good regeneration of axons
very thin white structure linked the nerve stumps in the using a nerve guide which was made of laminin-coated
Tube Group. Regenerated myelinated and unmyelinated filaments, instead of a tube [26]. A nerve guide made of
axons were found at the distal end of the graft—1 mm filaments without a tube has high permeability. In our
proximal to the distal suture threads—in both the Autograft study, the number of myelinated axons of the tibial
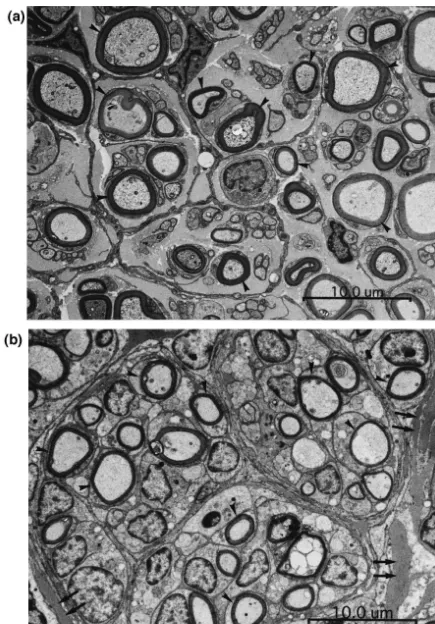
Group and the Fiber Group at 8 weeks postoperatively division of the normal sciatic nerve was smaller than the
(Fig. 2a,b). The mean number of myelinated axons was previously reported one [23]. This may be due to
differ-approximately 4800 (48376604) in the distal end of the ences in staining and counting methods. The number of
nerve autograft (Autograft Group, six rats) at 8 weeks regenerated myelinated axons in the collagen-filaments
postoperatively; while in the distal end of the collagen- nerve guide was not smaller than the number of
regener-filaments nerve guide (Fiber Group, six rats) it was ated myelinated axons in the nerve autograft. The mean
approximately 5500 (54916617). There were no statisti- fiber diameter and mean nerve conduction velocity were
cally significant differences between groups (Wilcoxon’s significantly less in the collagen-filaments nerve guide
tests, P50.05). The mean fiber diameter was 3.3mm in the group than those in the Autograft Group at 8 weeks
distal end of the nerve autograft at 8 weeks postoperative- postoperatively. A study with an extended period should
ly; while in the distal end of the nerve guide it was 2.3 be made to investigate these findings. The collagen
fila-mm. The mean fiber diameter was significantly larger in ments were considered easy to resorb because only small
the Autograft Group (Student’s t-test, P50.05). No myeli- residues of collagen filaments were found among
regener-nated and unmyeliregener-nated axon was found in the thin white ated axons at 8 weeks postoperatively (Fig. 2b).
structure in the Tube Group. Small residues of collagen filaments were found among regenerated axons in the Fiber Group (Fig. 2b). Few macrophages were found around the
References
collagen filaments.
The SFI values decreased after the nerve transection and
[1] A.D. Ansselin, T. Fink, D.F. Davey, Peripheral nerve regeneration
repair. The mean SFI values for the rats in the Autograft
through nerve guides seeded with adult Schwann cells, Neuropathol.
Group was2106.4 at 4 weeks postoperatively. The value Appl. Neurobiol. 23 (1997) 387–398.
was2121.5 in the Fiber Group. The value was 2117.2 in [2] S.J. Archibald, C. Krarup, J. Shefner, S.T. Li, R. Madison, A collagen-based nerve guide conduit for peripheral nerve repair: an
the Tube Group. The values did not increase in all groups
electrophysiological study of nerve regeneration in rodents and
at 8 weeks postoperatively. The values increased slightly
nonhuman primates, J. Comp. Neurol. 307 (1991) 1–12.
in all groups at 12 weeks postoperatively. The mean SFI
[3] J.R. Bain, S.E. Mackinnon, D.A. Hunter, Functional evaluation of
values for the rats in the Autograft Group was271.0 at 12 complete sciatic, peroneal and posterior tibial nerve lesion in the rat, weeks postoperatively. The value was 284.2 in the Fiber Plast. Reconstr. Surg. 83 (1989) 129–138.
Group. The value was 2105.8 in the Tube Group. The [4] R.M. Braun, Experimental peripheral nerve repair tubulation, Surg. Forum 15 (1964) 452–454.
three groups were considered to be not different practically
[5] D.T.W. Chiu, I. Janecka, T.J. Krizek, M. Wolff, R.E. Lovelace,
in SFI values. Toe contracture was not found in all groups
Autogenous vein graft as a conduit for nerve regeneration, Surgery
through the experimental period. 91 (1982) 226–233.
No interpretable compound action potential was re- [6] W. Colin, R.B. Donoff, Nerve regeneration through collagen tubes,
162 S. Yoshii, M. Oka / Brain Research 888 (2001) 158 –162
[7] N. Danielsen, L.B. Dahlin, Y.F. Lee, G. Lundborg, Axonal growth in [18] G. Lundborg, L.B. Dahlin, N.P. Danielsen, R.H. Gelberman, F.M. mesothelial chambers: the role of the distal nerve segment, Scand. J. Longo, H.C. Powell, S. Varon, Nerve regeneration in silicone Plast. Reconstr. Surg. 17 (1983) 119–125. chambers: influence of gap length and presence of distal stump [8] A.L. Dellon, S.E. Mackinnon, An alternative to the classical nerve components, Exp. Neurol. 76 (1982) 361–375.
graft for the management of the short nerve gap, Plast. Reconst. [19] R.D. Madison, C.F. Da Silva, P. Dikkes, Entubulation repair with Surg. 82 (1988) 849–856. protein additives increases the maximum nerve gap distance suc-[9] T. Gluck, Ueber Neuroplastik auf dem Wege der Transplantation, cessfully bridged with tubular prostheses, Brain Res. 447 (1988)
Arch. Klin. Chir. 25 (1880) 606–616. 325–334.
[10] E.W. Henry, T.-H. Chiu, E. Nyilas, T.M. Brushart, P. Dikkes, R.L. [20] C.R. Noback, J. Husby, J.M. Giorado, C.A.L. Bassett, J.B. Camp-Sidman, Nerve regeneration through biodegradable polyester tubes, bell, Neural regeneration across long gaps in mammalian peripheral Exp. Neurol. 90 (1985) 652–656. nerves: early morphological findings, Anat. Rec. 131 (1958) 633– [11] C-B. Jenq, R.E. Coggeshall, Nerve regeneration through holey 647.
silicone tubes, Brain Res. 361 (1985) 233–241. [21] E. Nyilas, T.H. Chiu, R.L. Sidman, E.W. Henry, T.M. Brushart, P. [12] C-B. Jenq, R.E. Coggeshall, Permeable tubes increase the length of Dikkes, R. Madison, Peripheral nerve repair with bioresorbable the gap that regenerating axons can span, Brain Res. 408 (1987) prosthesis, Trans. Am. Soc. Artif. Intern. Organs 29 (1983) 307–
239–242. 313.
[13] D.H. Kim, S.E. Connolly, S. Zhao, R.W. Beuerman, R.M. Voorhies, [22] R.L. Reid, D.E. Cutright, J.S. Garrison, Biodegradable cuff an D.G. Kline, Comparison of macropore, semipermeable, and non- adjunct to peripheral nerve repair: a study in dogs, Hand 10 (1978) permeable collagen conduits in nerve repair, J. Reconst. Microsurg. 259–266.
9 (1993) 415–420. [23] H. Schmalbruch, Fiber composition of the rat sciatic nerve, Anat. [14] D.G. Kline, G.J. Hayes, The use of a resorbable wrapper for Rec. 215 (1986) 71–81.
peripheral nerve repair, J. Neurosurg. 21 (1964) 737–750. [24] B.R. Seckel, T.H. Chiu, E. Nyilas, R.L. Sidman, Nerve regeneration [15] R.A.W. Lehman, G.J. Hayes, Degeneration and regeneration in through synthetic biodegradable nerve guides: regulation by the
peripheral nerve, Brain 90 (1967) 285–296. target organ, Plast. Reconst. Surg. 74 (1984) 173–181.
[16] F.M. Longo, M. Manthorpe, S.D. Skapper, G. Lundborg, S. Varon, [25] P. Weiss, A.C. Taylor, Guides for nerve regeneration across gaps, J. Neurotrophic activities accumulating in vivo within silicone nerve Neurosurg. 3 (1946) 375–389.
regeneration chambers, Brain Res. 261 (1983) 109–116. [26] S. Yoshii, T. Yamamuro, S. Ito, M. Hayashi, In vivo guidance of [17] G. Lundborg, L.B. Dahlin, N.P. Danielsen, H.A. Hansson, K. regenerating nerve by laminin-coated filaments, Exp. Neurol. 96
Larsson, Reorganization and orientation of regenerating nerve fibers, (1987) 469–473.
perineurium, and epineurium in preformed mesothelial tubes – an [27] B.L. Young, P. Begovac, D.G. Stuart, G.E. Goslow Jr, An effective experimental study on the sciatic nerve of rats, J. Neurosci. Res. 6 sleeving technique in nerve repair, J. Neurosci. Methods 10 (1984)