volatile flavours ofBrassica rapaseeds. Bull. Fac. Pharm. Cairo Oleszek, W. 1987. Allelopathic effects of volatiles from some cruci-Univ. 33:91–93. ferae species on lettuce, barnyard grass and wheat growth. Plant Fenwick, G.R., R.K. Heaney, and W.J. Mullin. 1983. Glucosinolates Soil 102:271–273.
and their breakdown products in food and food plants. Crit. Rev. Parthipan, B., and A. Mahadevan. 1994. Effects of methyl isothiocya-Food Sci. Nutr. 18:123–301. nate on soil microflora and the biochemical activity of soils. Envi-Krishnan, G., D.L. Holshauser, and S.J. Nissen. 1998. Weed control ron. Pollut. 87:283–287.
in soybean (Glycine max) with green manure crops. Weed Tech- Petersen, J. 1999. Untersuchungen zur Unkrautbekaempfung mit Bo-nol. 12:97–102. dendeckern in herbizidresistentem Mais. Cuvillier Verlag, Goet-Kruskal, W.H., and W.A. Wallis. 1953. Use of ranks in one-criterion tingen.
variance analysis. J. Am. Stat. Assoc. 48:907–911. Seefeldt, S.S., J.E. Jensen, and P.E. Fuerst. 1995. Log-logistic analysis Michel, A., J. Petersen, M.N. Dogan, and V. Ernst. 1999. Anleitung of herbicide dose response relationship. Weed Technol. 9:218–227.
zur Anlage und Auswertung von Versuchen zur Erstellung quanti- Streibig, J.C. 1988. Herbicide bioassay. Weed Res. 28:479–484. tativer Dosis-Wirkungsbeziehungen am Beispiel der Wirksamkeit Tang, C.S. 1971. Benzyl isothiocyanate of papaya fruit. Phytochemis-von Herbiziden. Gesunde Pflanz. 1:10–19. try 10:117–121.
Mithen, R. 1992. Leaf glucosinolate profiles and their relationship to pest and disease resistance in oilseed rape. Euphytica 63:71–83.
Laboratory Bioassay for Phytotoxicity: An Example from Wheat Straw
Bader Al Hamdi, Inderjit,* Maria Olofsdotter, and Jens C. Streibig
ABSTRACT nism of plant interference. The observed growth pattern
is better explained by a synergistic action of several
Allelopathy involves complex plant3plant chemical interactions.
mechanisms of interference (Inderjit and Del Moral,
Although a large number of laboratory bioassays have proposed to
1997). It is almost impossible to demonstrate allelopathy
demonstrate allelopathy, most of them have little or no relevance in
terms of explaining behavior in the field. In this paper, we discuss by following the above six criteria. We, therefore, will
the phytotoxicity of wheat (Triticum aestivumL.) straw leachate to restrict our discussion to phytotoxicity. We argue that the seedling growth of perennial ryegrass (Lolium perenneL.). The laboratory bioassays can generate some meaningful objective of this study was to discuss the significance of (i) soil, (ii) data, provided that attention is paid to following points: leachate concentrations in bioassays of plant debris and soil, (iii) the
(i) soil, (ii) several concentrations of phytotoxic
mate-role of N fertilizer in overcoming plant growth inhibition, (iv) organic
rial, (iii) elimination of possible inhibition by N
defi-molecules in plant inhibition, and (v) actual assay species. The results
ciency due to added organic material, (iv) involvement
show the phytotoxic nature of wheat straw leachate (WSL) and the
of organic molecules in plant inhibition, and (v) assay
possible involvement of organic molecules in the growth inhibition of
species. A study with a wheat straw–perennial ryegrass
perennial ryegrass. However, laboratory studies can not demonstrate
system is designed to address the above criteria.
Acti-allelopathy as the sole factor responsible for the observed growth
inhi-bition. vated charcoal was added to the system, as suggested,
to isolate the interference by organic molecules (Mahall and Callaway, 1992; Inderjit and Foy, 1999).
Many ecologists often argue that the addition of plant
C
oncerns are often raisedabout the relevance ofdebris, leachate, or both into the soil results in enhanced laboratory bioassays for allelopathy (Connell,
microbial activity, which causes N depletion. Any 1990; Inderjit and Olofsdotter, 1998; Inderjit and
Dak-growth suppression, they argue, is due to N depletion, shini, 1995, 1999). Willis (1985, p. 72) listed a six-point
rather than organic molecules (Harper, 1977). To ad-protocol necessary to demonstrate allelopathy in natural
dress this concern and invoke the probable involvement systems: “(i) a pattern of inhibition of one species or
of organic molecules in growth suppression, a series of plant by another must be shown, (ii) the putative
aggres-experiments was conducted. These aggres-experiments investi-sive plant must produce a toxin, (iii) there must be a
gated the effect of soil amended with WSL on the seed-mode of toxin release from the plant into the
environ-ling growth of perennial ryegrass and whether the inter-ment, (iv) there must be toxin transport and/or
accumu-ference due to wheat straw is modified after the addition lation in the environment, (v) the afflicted plant must
of activated charcoal and different amounts of N fertiliz-have some means of toxin uptake, and (vi) the observed
ers. The objective of this paper is to demonstrate that pattern of inhibition cannot be explained solely by
phys-laboratory bioassays for phytotoxicity can generate ical factors or other biotic factors, especially competition
some meaningful data, provided that experiments are and herbivory.” Blum et al. (1999) recently concluded
conducted under realistic conditions. that no study has ever demonstrated all of these criteria.
Nature is too dynamic to be solely explained by a
mecha-Why Wheat Straw?
Wheat straw has been reported to possess allelopathic B. Al Hamdi, M. Olofsdotter, and J.C. Streibig, Dep. of Agric. Sci.
activities (Guenzi and McCalla, 1962; Guenzi et al., (Weed Sci.), The Royal Veterinary and Agric. Univ., Thorvaldsensvej
40, DK-1871 Frederiksberg C, Copenhagen, Denmark; Inderjit, Dep. 1967). Guenzi and McCalla (1966) found phytotoxicity of Botany, Panjab Univ., Chandigarh 160014, India. Received 20 Nov. of phenolic acids, particularly p-coumaric acid, from 1999. *Corresponding author ([email protected]).
sity Experimental Station in Høbakkega˚rd, Denmark (558409 residues of wheat and other cereals. However, the
bio-N, 128309E). The soil was allowed to dry at room temperature
assays conducted by these authors to demonstrate
phy-and was sieved (1.8 mm sieve) phy-and stored in paper bags.
totoxicity had several shortcomings: (i) organic solvent
Wheat straw (50 g) was obtained from Højbakkega˚rd and
was used to prepare the phenolic acid solution, (ii)
au-soaked in 900 mL of distilled water (hereafter referred to as
thentic phenolic acids were taken, (iii) soil was not in- water) for 72 h and then filtered. The filtrate was described volved in the bioassays, and (iv) no consideration was as full-strength (FS) WSL. Appropriate amounts of water were paid to the significance of mixtures of chemicals (Einhel- added to the FS WSL to obtain 50, 25, and 12.5% (v/v, WSL/ lig, 1999). Another major problem with studies on phe- water) WSL. In addition, 50 g of wheat straw was burned,
nolic acids is the lack of relevance of the tested con- and WSL of different strengths (FS, 50, 25, and 12.5%) were prepared by soaking the burned wheat straw in water as
de-centration in field settings. The three common
scribed above.
concentrations used by Guenzi and McCalla (1966) in their bioassays were 1250, 2500, and 5000 ppm. With
these concentrations, each petri plate received 7.5, 15, Soil Amendments
and 30 mg of phenolic acid for the respective
concentra-The soil (90 g) was amended with 40 mL of FS, 50, 25, and
tion, with 10 wheat seeds sown. This is an unrealistic
12.5% WSL of unburned and burned wheat straw. Soil that
amount for wheat seeds to experience in a natural setting.
was amended with 40 mL water served as the control.
Alam (1990) studied the effect of wheat straw extracts Soil (90 g) was amended with 0.25, 0.50, and 1 g of activated on the germination and seedling growth of wheat. How- charcoal (Sigma, USA) and 40 mL FS, 50, 25, and 12.5% WSL. ever, he ground the wheat straw to make the extract. Soil that was amended with 0.25, 0.50, and 1 g of activated Grinding may lead to the release of certain enzymes, charcoal and 40 mL of water served as control.
amino acids, and other organic compounds that would Appropriate amounts of ammonium nitrate (NH4NO3) have never been released from wheat straw in nature were dissolved in water to obtain final concentrations of 0.25,
(Chou and Muller, 1972; Inderjit and Dakshini, 1995). 0.50, 0.75, and 1 mM of N fertilizer. The soil (90 g) was amended with 20 mL of 0.50, 1, 1.5, and 2 mMof fertilizer
Another problem with this study was the absence of soils.
and 20 mL of FS, 50, 25, and 12.5% WSL. Soil that was
Soil is important because abiotic and biotic soil factors
amended with 0.50, 1, 1.5, and 2 mMof fertilizer and 20 mL
significantly influence the quantitative and qualitative
of water served as the control for the respective treatments.
levels of allelochemicals (Cheng, 1995; Inderjit et al., 1999). Steinsiek et al. (1982) reported that allelopathic
interference of wheat to selected weed species was depen- Growth Experiments
dent on the extract, species, and temperature. They
re-Perennial ryegrass was selected as an assay species because
ported that ivyleaf morning-glory (Ipomoea hederacea
(i) it is often found in wheat fields, and (ii) it has been shown
Jacq.) was most affected and barnyardgrass [Echinochloa that wheat has the potential to suppress the root growth of crus-galli(L.) Beauv.] was least affected. However, con- perennial ryegrass (unpublished, 1999). Soil (90 g) as described clusive evidence for allelopathy is still lacking. Growth above (soil 1, 2, and 3), was placed in 9-cm petri dishes, and response due to soaked, agitated, or leached extracts at 15 perennial ryegrass seeds were sown on the soil surface.
different temperatures cannot be attributed to the occur- The experiment had four replications and was repeated once
rence of allelopathic compounds. No soil was included (identified as Exp. 1 and 2). Data on the root and shoot length of wheat and perennial ryegrass were recorded after 10 d, and
in the bioassay of Steinsiek et al., and it is difficult to
the longest root and shoot of each seedling was measured.
argue for allelopathy in the absence of soil. Using an
The average environmental conditions were light regimes of
extract for bioassay, an investigator can only
demon-12.3mmol photons m22s21and a temperature of 228C. strate the potential phytotoxicity of the extract. More
realistic experimentation is needed to determine
whe-ther the observed phytotoxicity of the extract is ex- Data Analyses
pressed in a natural environment. Hicks et al. (1989)
Data were subjected to analyses of variance and linear
reported allelopathic effects of wheat straw on the ger- regression. The analyses of variance and lack-of-fit tests for mination, emergence, and yield of cotton (Gossypium the linear regressions were used to check whether the linear hirsutumL.). They found that the maximum inhibition regressions were adequate to describe the variation in data. in cotton germination and emergence occurred when (Weisberg, 1985).
wheat straw was mixed throughout the soil. Later, Opoku et al. (1997) implicated phenolics in the
allelo-RESULTS AND DISCUSSION
pathic interference of wheat straw to corn (Zea mays
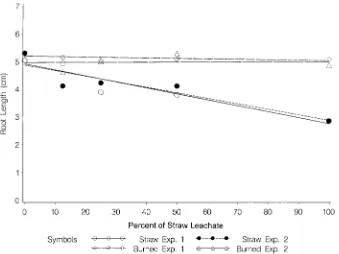
L.). They reported that the total phenolic levels of soil In general, soil that was amended with different in surface-placed straw were higher compared with soil amounts of WSL suppressed the root growth of peren-alone. However, the phenolic content of soil mixed with nial ryegrass (Fig. 1). The shoot growth of perennial straw was not different from that of soil alone. There- ryegrass, however, was not affected by soil amendment. fore, their results were not conclusive. These results show the inhibitory effects of WSL on the root growth of perennial ryegrass. Further, the fact that MATERIALS AND METHODS
straw leachate was prepared in an aqueous medium
General Procedures explains the root suppression of perennial ryegrass in
the field where natural precipitation and irrigation are
Soil (sandy loam) was collected from a wheat-free field
to germinate due to adequate soil moisture, their further leachate could be due to the absence of organic com-pounds, which were present in the leachates prepared establishment would be restricted because of phytotoxic
interference by the WSL. from unburned wheat straw. To further confirm the
involvement of organic compounds in the root growth The paracelsus (Streibig et al., 1993) axiom is very
much applied to the concentration-dependent dose re- inhibition of perennial ryegrass by WSL, we used acti-vated charcoal, which adsorbs organic molecules, to sep-sponses. The English translation of the German axiom
is that all chemicals are poisonous—it is a question of arate the phytotoxic effects from other interference (Wardle and Nilsson, 1997; Inderjit and Foy, 1999). In dose. It is important to select more than three levels of
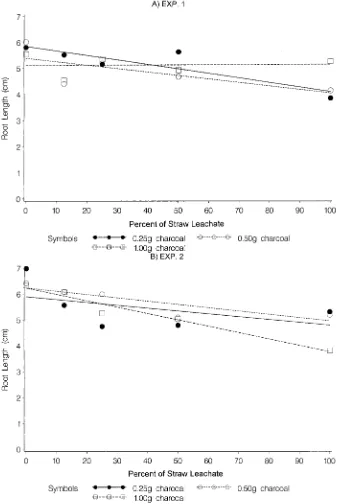
concentrations of phytotoxic material to know whether general, the addition of different amounts of charcoal to soil amended with WSL eliminated the leachate ef-the dose response relationship is properly explained by
a linear regression. Any conclusion drawn with one or fects on the root growth of perennial ryegrass (Fig. 2A, 2B). The significant affect on the root growth of peren-two concentrations of phytotoxic material is not enough
in a biological system. In the present study, we have nial ryegrass was observed in soil amended with FS WSL and lower amounts of charcoal (Fig. 2A, 2B). This can selected different levels of straw leachates to study the
growth responses. Furthermore, it is important to select be explained due to the concentration-dependent nature of phytotoxins. This means that every interaction be-a rbe-ange of concentrbe-ations thbe-at be-are close to the nbe-aturbe-al
agroecosystem. The amount of wheat straw used in the tween charcoal and leachate and the test for lack of fit was significant. It is likely that amount of organic present study is relevant in terms of incorporating wheat
straw in Danish arable fields. However, it is difficult to compounds that were contributed by FS WSL was not completely sorbed by the amount of added charcoal. predict what concentration of leachate will be added in
the field setting. This is because of an uneven distribu- In the present study, different levels of N fertilizer were added in soil amended with WSL. In general, the tion of straw in the field as well as a varied site specific
climatic and edaphic conditions. Phytotoxic material root growth of perennial ryegrass was still suppressed even though the amended soils were supplemented with (e.g., wheat straw) is often distributed unevenly in
na-ture, and there are pockets with varying amounts of lower levels of N fertilizers, e.g., 0.25 and 0.50 mM (Fig. 3A, 3B). There was a highly significant interaction phytotoxic material in the same field (Liebl and
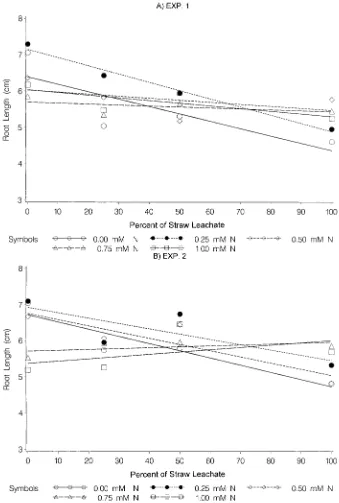
Wors-ham, 1983). between leachate and N, and a test of the lack of fit of the
linear regression was significant because of nonlinearity. Compared with the control, soil amended with
differ-ent amounts of burned straw leachate had no effect on Less influence on the root growth of perennial ryegrass was recorded when the amended soils were supple-either the root (Fig. 1) or shoot growth of perennial
ryegrass. Most of the organic compounds present in mented with higher amounts of 0.50 and 1 mM of N fertilizer (Fig. 3A, 3B). While a significant promotion wheat straw are likely to be destroyed after burning.
The observed elimination of the root growth inhibition in the shoot growth of perennial ryegrass was observed in the amended soils that were supplemented with N of perennial ryegrass in soil amended with burned straw
Fig. 2. Effect of soil amended with four concentrations 12.5, 25, 50, and 100% (full strength) of wheat straw leachate (WSL) and three concentrations (0.25, 0.50, and 1 g) of charcoal on root growth of perennial ryegrass for (A) Experiment 1 and (B) Experiment 2. Soil amended only with charcoal served as the control. Experiments were replicated four times and repeated once.
fertilizer, no affect on the shoot growth was recorded of organic compounds, which could have resulted in a significant influence on the seedling growth.
when the experiment was repeated. Any conclusion on the promotion or lack of promotion in the shoot growth of perennial ryegrass in amended soils that were
supple-CONCLUSION
mented with N fertilizer can not be argued because two
experiments were carried out and gave different results. The present study indicates the phytotoxic nature of WSL and the possible involvement of organic molecules We did not measure the microbial activity, but higher
microbial activity due to N fertilizer is likely (Novak in the growth inhibition of perennial ryegrass. However, no laboratory study can predict the possibility of allelo-et al., 1995). The higher microbial activity might have
Fig. 3. Effect of soil amended with four concentrations 12.5, 25, 50, and 100% (full strength) of wheat straw leachate (WSL) and five concentrations (0, 0.25, 0.50, 0.75, and 1 mM) of N fertilizer on root growth of perennial ryegrass for (A) Experiment 1 and (B) Experiment 2. Soil amended with 0 mMof N fertilizer served as the control. Experiments were replicated four times and repeated once.
some controlled experiments under simulated field con- 1997). What we can interpret from the present study is the phytotoxic action of wheat straw to the root growth ditions, which can help in deciding whether further
ex-perimentation or field study on phytotoxicity is justified. of perennial ryegrass under given conditions.
We feel that experiments carried out under controlled It is important how researchers interpret their data. For
example, in the present study, we can not argue the conditions can never confirm allelopathy to be opera-tional in nature (Blum, 1999). We have avoided using phytotoxic potential of wheat straw in nature because
there may be several interference mechanisms (resource the term allelopathy in the discussion and used the term phytotoxicity instead because the terms are often mis-competition, allelopathy, nutrient immobilization,
Inderjit, H.H. Cheng, and H. Nishimura. 1999. Plant phenolics and pathy should not be used until data are available on (i)
terpenoids: Transformation, degradation, and potential for allelo-the natural release of compounds from allelo-the aggressive pathic interactions. p. 255–266.In Inderjit et al. (ed.) Principles plant, (ii) the concentration and persistence of these and practices in plant ecology: Allelochemicals interactions. CRC compounds in the environment, and (iii) the direct Press, Boca Raton, FL.
Inderjit, and K.M.M. Dakshini. 1995. On laboratory bioassays in allel-involvement of these compounds with the inhibition of
opathy. Bot. Rev. 61:28–44.
the target plant, which can be demonstrated by uptake Inderjit, and K.M.M. Dakshini. 1999. Bioassays for allelopathy: Inter-studies. However, the term phytotoxicity can be used. actions of soil organic and inorganic constituents. p. 35–44.In Inderjit et al. (ed.) Principles and practices in plant ecology: Allelo-chemicals interactions. CRC Press, Boca Raton, FL.
REFERENCES Inderjit, and R. Del Moral. 1997. Is separating allelopathy from re-source competition realistic? Bot. Rev. 63:221–230.
Alam, S.M. 1990. Effect of wheat straw extract on the germination and
Inderjit, and C.L. Foy. 1999. Nature of the interference potential of seedlings growth of wheat (cv. Pavon). Wheat Inf. Serv. 71:16–18.
mugwort (Artemisia vulgaris). Weed Technol. 13:176–182. Blum, U. 1999. Designing laboratory plant debris-soil bioassays: Some
Inderjit, and M. Olofsdotter. 1998. Bioassays for rice allelopathy: reflections. p. 17–23.InInderjit et al. (ed.) Principles and practices
Some concerns. p. 45–55.InM. Olofsdotter (ed.) Allelopathy in in plant ecology: Allelochemical interactions. CRC Press, Boca
rice. Int. Rice Res. Inst. Press, Manila, Philippines.
Raton, FL. Liebl, R.A., and A.D. Worsham. 1983. Inhibition of morning-glory
Blum, U., S.R. Shafer, and M.E. Lehman. 1999. Evidence for
inhibi-(Ipomoea lacunosaL.) and certain other weed species by phytotoxic tory allelopathic interactions involving phenolic acids in field soils:
components of wheat (Triticum aestivum L.) straw. J. Chem. Concepts vs. experimental model. Crit. Rev. Plant Sci. 18:673–693. Ecol. 9:1027–1043.
Cheng, H.H. 1995. Characterization of the mechanisms of allelopathy: Mahall, B.E., and R.M. Callaway. 1992. Root communication mecha-Modeling and experimental approaches. p. 132–141.InInderjit et nisms and intracommunity distributions of two Mojave desert al. (ed.) Allelopathy: Organisms, processes, and applications. Am. shrubs. Ecology 73:2145–2151.
Chem. Soc., Washington, DC. Novak, J.M., K. Jaychandra, T.B. Moorman, and J.B. Weber. 1995. Chou, C.H., and C.H. Muller. 1972. Allelopathic mechanisms ofArc- Sorption and binding of organic compounds in soil and their rela-tostaphylos glandulosavar.zacaensis. Am. Midl. Nat. 88:324–347. tion to bioavailability. p. 13–31.InH.D. Skipper and R.F. Turco Connell, J.H. 1990. Apparent versus “real” competition in plants. p. (ed.) Bioremediation: Science and applications. SSSA Spec. Publ.
9–25.In J.B. Grace and D. Tilman (ed.) Perspectives on plant 43. SSSA, Madison, WI.
competition. Academic Press, San Diego, CA. Opoku, G., T.J. Vyn, and R.P. Voroney. 1997. Wheat straw placement Einhellig, F.A. 1999. An integrated view of allelochemicals amid multi- effects on total phenolic compounds in soil and corn seedling
ple stresses. p. 479–494.InInderjit et al. (ed.) Principles and prac- growth. Can. J. Plant Sci. 77:301–305.
tices in plant ecology: Allelochemical interactions. CRC Press, Romeo, J.T., and J.D. Weidenhamer. 1998. Bioassays for allelopathy Boca Raton, FL. in terrestrial plants. p. 179–211.InK.F. Haynes and J.G. Miller (ed.) Guenzi, W.D., and T.M. McCalla. 1962. Inhibition of germination Methods in chemical ecology. Vol. 2. Bioassay methods. Kluwer
and seedling development by crop residues. Soil Sci. Soc. Am. Academic Publ., Norvell, MA.
Proc. 26:456–458. Steinsiek, J.W., L.R. Oliver, and F.C. Collins. 1982. Allelopathic po-Guenzi, W.D., and T.M. McCalla. 1966. Phenolic acids in oats, wheat, tential of wheat (Triticum aestivum) straw on selected weed species.
sorghum, and corn residues and their phytotoxicity. Agron. J. Weed Sci. 30:495–497.
58:303–304. Streibig, J.C., M. Rudemo, and J.E. Jensen. 1993. Dose-response
Guenzi, W.D., T.M. McCalla, and F.A. Norstadt. 1967. Presence and curves and statistical models. p. 29–55.InJ.C. Streibig and P. Kudsk persistence of phytotoxic substances in wheat, oat, corn, and sor- (ed.) Herbicide bioassays. CRC Press, Boca Raton, FL. ghum residues. Agron. J. 59:163–165. Wardle, D.A., and M.-C. Nilsson. 1997. Microbe-plant competition, Harper, J.L. 1977. Population biology of plants. Academic Press, allelopathy, and arctic plants. Oecologia 109:291–293.
London. Weisburg, S. 1985. Applied linear regression. 2nd ed. John Wiley &
Hicks, S.K., C.W. Wendt, J.R. Gannaway, and R.B. Baker. 1989. Sons, New York.
Allelopathic effects of wheat straw on cotton germination, emer- Willis, R.J. 1985. The historical bases of the concept of allelopathy. J. Hist. Biol. 18:71–102.