www.elsevier.nlrlocateraqua-online
The nutritional value of algae grown under different
culture conditions for Mytilus edulis L. larvae
Nikos Leonardos
1, Ian A.N. Lucas
)School of Ocean Sciences, Menai Bridge, North Wales LL59 5EY, UK
Received 4 January 1999; received in revised form 19 July 1999; accepted 19 July 1999
Abstract
Continuous cultures of Skeletonema costatum, Chaetoceros muelleri, Rhinomonas reticulata and PaÕloÕa lutheri were subjected to phosphorus or nitrogen limitation at two light intensities in order to manipulate their biochemical composition. They were then fed to Mytilus edulis larvae over a 2-week period, and the larval growth and mortality were assessed. All monospecific diets supported growth, sometimes equal to or better than a control diet containing a mixture of species ŽR. reticulata and P. lutheri . Survival was not affected by the diets but significantly better larval. growth was obtained with low-light, nitrogen-limited or non-nutrient-limited cells of S. costatum and high-light, nitrogen-limited or low-light cells of C. muelleri grown under no limitation or nitrogen limitation. High-light phosphorus-limited or low-light non-nutrient-limited R. reticulata cells and high- light phosphorus-limited cells of P. lutheri were also superior in relation to their counterparts. A novel computer-aided image analysis technique was used for measuring the length of the larvae. A multidimensional model was used in an effort to correlate algal biochemical components with larval growth. Some fatty acids were found to be significant in determining the algal nutritional value, with protein and carbohydrate playing a secondary ‘‘modifying’’ role. In
Ž .
P. lutheri, the 16:0 and saturated fatty acids SAFA were significantly positively correlated with
larval growth while the contrary was found for dietary ny3 fatty acids, suggesting their strong negative effect on larval growth. Similar results were found in R. reticulata, although in this case, both protein and carbohydrate content were found to determine the algal nutritional value concurrently with some fatty acids. In the other two diatoms, S. costatum and C. muelleri, no consistent relationship could be established, thus suggesting that either there is a species-specific
)Corresponding author. Telefax:q44-1248-382871; E-mail: [email protected]
1
E-mail: [email protected].
0044-8486r00r$ - see front matterq2000 Elsevier Science B.V. All rights reserved.
Ž .
relationship or that other components are important in determining the algal nutritional value.
q2000 Elsevier Science B.V. All rights reserved.
Keywords: Chaetoceros muelleri; Fatty acids; Light; Larvae; Mytilus edulis; Nutrients; PaÕloÕa lutheri; Rhinomonas reticulata; Skeletonema costatum
1. Introduction
Many marine animals rely on phytoplankton as a food source especially in their early stages of development. For a number of bivalve molluscs which are of commercial interest, live phytoplankton cultures are routinely used as food for the larval and adult stages. From the plethora of phytoplankton species found in nature, only a small number
Ž .
have been used as food sources De Pauw et al., 1984 . In early research, the nutritional value of phytoplankton cultures in terms of biochemical composition was not considered
ŽWalne, 1963 . Instead, palatability and digestibility were suggested as being more.
significant. However, later studies, benefitting from advances in analytical methods, emphasized the indisputable role of the algal biochemical composition in determining its
Ž .
nutritional value Wikfors et al., 1984; Enright et al., 1986b . Factors influencing algal
Ž .
biochemical composition have been described by Leonardos 1998 .
There is only general agreement on which biochemical constituents of phytoplankton cells are most significant in determining their food value for planktonic herbivores. Numerous reports have examined biochemical classes of components, primarily protein, carbohydrate and lipid and have reached different conclusions about the significance of
Ž
gross biochemical composition Epifanio, 1979; Webb and Chu, 1983; Enright et al.,
.
1986a,b; Wikfors et al., 1992; Thompson et al., 1994 . However, since the observation
Ž .
that certain long-chain polyunsaturated fatty acids PUFA are essential to many marine organisms, considerable research has focused on the availability and the role of PUFA in
Ž . Ž
hatcheries Watanabe et al., 1983 and in the natural environment Sargent and Whittle,
.
1981 . But there is remarkably little experimental evidence that diets containing more PUFA are superior for herbivores and there is now growing evidence that the
impor-Ž
tance of dietary PUFA may have been overgeneralized Dickey-Collas and Geffen,
.
1992; Thompson et al., 1993, 1994 . This confusion may reflect true differences in the nutritional requirements of various bivalve species. However, even minor changes in the culturing conditions employed in different laboratories may lead to different
morpholog-Ž
ical, physiological andror biochemical properties of the algae in question Gallager and
.
Mann, 1981; Thompson et al., 1993 . In most hatcheries and laboratory feeding trials concerning the nutritional value of algae which essentially use the larvae as bioassay organisms, the algal biochemical composition is not constant over the whole time range of the feeding trials. Thus, direct comparisons of results from different laboratories may
Ž
be misleading. Although this conflict is commonly recognized by phycologists Baars,
.
1981 , its potential importance to mariculture, in general, has received comparatively little attention.
biochemical composition, thus rendering the experiments internally comparable; how-ever, in practice, some variation in the algal biochemical composition is to be expected. Using monospecific diets, the growth performance of the larvae can be correlated with algal biochemical properties, without introducing differences in digestibility, cell size and palatability which may have compromised previous studies using multispecies diets. Special emphasis was placed on algal fatty acids due to ever increasing evidence suggesting the important role of these compounds as diet components of particular
Ž .
nutritional value Thompson et al., 1996 .
2. Materials and methods
2.1. Algal cultures, chemical and biochemical analyses
The algal species diet used as food for the larvae of Mytilus edulis were PaÕloÕa Ž
lutheri obtained from Culture Centre of Algae and Protozoa, Oban, Scotland, CCAP
. Ž . Ž .
931r1 , Chaetoceros muelleri CCAP 1010r3 , Rhinomonas reticulata CCAP 995r2
and Skeletonema costatum, the strain of which was isolated by the first author, from a
Ž .
sample taken from Syros island Greece . The algae were grown using a continuous culture regime under a light–dark cycle of 14–10 h, at 208"18C. Two light intensities
Ž y1 y2
were employed high-light, HLs890–950 kphotons s m and low-light, LLs
y1 y2.
275–337 kphotons s m , and three nutrient conditions were used in each light
Ž . Ž . Ž .
intensity: no nutrient limitation fr2 , phosphorus P and nitrogen N limitation. The
phosphorus-limited medium and nitrogen-limited medium were prepared by adding a quarter of the amount of the respective salt. The density of the cultures was determined twice daily and adjustments to the flow rate were made on a daily basis to keep the cell density constant. Typically, the cultures were maintained in the selected cell density
"10% throughout the feeding trials. The collector flasks were emptied regularly and algae, which were to be used either as food or for samples for cell density determination and for the subsequent biochemical analyses, were left to stand for no more than 3 h in that vessel. The algae used in the control diet were cultured in 20 l glass round flasks at a light intensity of 715–827 kphotons sy1 my2 using a semi-continuous batch culturing system and Conway medium under constant illumination. All cultures were maintained in uni-algal condition. Chemical and biochemical methods and techniques are described
Ž .
in Leonardos and Lucas 2000a and the biochemical results are expressed in a per cell basis since this was the unit of standardization for the feeding trials. Three samples underwent protein and carbohydrate analyses while the fatty acid profile was determined in two samples.
2.2. Mussels spawning and feeding trials
Ripe mussels, M. edulis, collected from the Conwy estuary, N. Wales, in the spring
Ž .
mm in length were selected and epifauna and byssals threads were removed. Spawning
Ž .
was induced as in Loosanoff and Davis 1963 . Fertilized eggs were incubated, unfed at 148C for 4 days and at the D-stage larval pool, the percentage of normality and
Ž Ž .
abnormality according to the developmental stages described by Bayne 1965 as well as the total number of larvae were calculated. A small sub-sample of larvae was taken
Ž
from this group and photographed or videotaped see later part of the section for image
.
analysis under a microscope for later calculation of the initial mean length of the larvae.
Ž .
All batches of larvae used had a small -10% percentage of abnormal larvae.
An appropriate volume was taken from the larval D-stage group and diluted to a concentration of 30,000 larvae ly1. These were transferred into sufficient plastic 1-l
beakers to give three replicates for each treatment. To each beaker, the respective species and quantities of algae were added and then filled to 1 l and left at 148"18C. Each diet was tested in triplicates. Feeding densities of 50 cellsmly1 were the same for
all diets; the control diet was a mixture of P. lutheri and R. reticulata in a 4:1 ratio. A
change of the 0.2 mm filtered water and feeding of the larvae was carried out every
second day with the respective algal food added to a final concentration of 50 cells mly1
. For the high-light and low-light experiments of the two diatoms, S. costatum and C. muelleri, two different batches of larvae were used for each light intensity; hence,
Ž .
results are compared against two sets of controls control diet and unfed larvae . The high- and low-light experiments of R. reticulata and P. lutheri were carried out with the same batch of larvae; therefore, only one set of controls was needed for each trial. After 2 weeks, samples were taken from the larval cultures to calculate the total
Ž .
number of live larvae as well as percentage of normal individuals Bayne, 1965 ; the samples were stored in a glass vial and fixed with few drops of Lugol’s iodine solution. Each sample was, at a later stage, decanted into a sedimentation chamber and counted using an inverted microscope.
Growth was estimated on the basis of length increase of the larvae. To measure length, two image analysis techniques were used. The larvae were measured at the
Ž .
beginning of every experiment while at D-stage and at the end, after a 2-week period. The first technique was based on measuring photographic negatives of the animals and was used for the S. costatum and C. muelleri feeding trials, while the second was based on processing videotaped information of the larvae and was employed in the P. lutheri and R. reticulata experiments. The first steps for both techniques were the same: the larvae from each container were concentrated by pouring its contents into a 45-mm nylon sieve. A small sub-sample was taken with a pipette and placed onto a slide.
In the photographic technique, the mean length of the larvae was estimated from
Ž .
negatives taken with a Nikon FM2 camera; 60 normal Bayne, 1965 , clearly focused, larvae were measured from the negative using the same microscope fitted with an eyepiece scale. The length of the larvae was calculated using a magnification factor computed from an image of a standard graticule.
In the videotape technique, the slide with the larvae was placed on a Nikon inverted
Ž
microscope with a microscope video camera Hitachi MOS Colour Video Camera,
.
VK-C150ED attached. The resulting picture was recorded with a video recorder
ŽPanasonic, NV-J35HQ . The recorded images were later transferred to an IBM-compat-.
Ž .
focus were grabbed with the aid of Microsoft’s Media Manager Video capture 32 and the resulting still images were saved as bitmap images on the computer’s hard disk.
Ž .
Then an image analysis software package Jandel Scientific Sigma Scan was used to measure the length of 60 larvae. This technique was compared with the photographic technique by measuring the same slide of larvae. The mean values obtained by the two techniques did not differ by more than 5%.
2.3. Statistical analysis
Before applying any parametric test each data set was tested for normality and homogeneity of variances, with a normal probability plot and with Barlett’s test, respectively. Significance was tested at the 95% level.
Logarithmic transformation of data resulted in more data sets having a normal distribution; however, in no instance were all the distributions of all data sets normal. Even when one of the assumptions necessary for applying parametric tests was violated
Ž .
in a small part of the data set typically one or two sets of larval lengths , parametric tests were used. The experimental design was such that nested ANOVA was used in all experiments to assess whether significant differences existed between diets. This test also reveals whether there is significant variation between the replicates of the same diet.
Ž
In order to identify the groups that demonstrated significant differences 95% confidence
. X
Ž
limit , the multiple comparisons between means of T -method was used Sokal and
.
Rohlf, 1987 . All tests were carried out with the aid of the statistical software package,
Minitabw
, while preliminary calculations and graphs were made in Quattrow
Pro. To identify any relationship between algal biochemical component and larval growth, correlation analysis was used. This type of analysis was used instead of ordinary regression analysis because the assumptions of model I regression analysis do not hold
Ž .
true with this type of data Sokal and Rohlf, 1987 .
3. Results
The gross biochemical composition of the algal species grown under various nutrient
Ž .
light and nutrient conditions is taken from Leonardos and Lucas 2000a and reproduced here in Table 1 for their protein, carbohydrate and proportions of some of the major fatty acids. The percentage of change in per cell protein content in S. costatum, C. muelleri
Ž
and P. lutheri as a result of the various culture conditions employed was smaller
one-.
to twofold change than in R. reticulata where protein content varied from 4.17 pg
y1 y1Ž .
cell to 18.27 pg cell fourfold change . Protein levels of the two diatoms and of R.
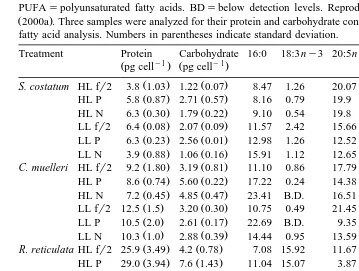
Table 1
Responses of S. costatum, C. muelleri, R. reticulata and P. lutheri to various growth conditions
Ž .
HLshigh light; LLslow light; fr2sfr2 medium no nutrient limitation ; Psphosphorus-limited medium, Nsnitrogen-limited medium. Fatty acids are expressed as percentages of TIFA. SAFAssaturated fatty acids; PUFAspolyunsaturated fatty acids. BDsbelow detection levels. Reproduced from Leonardos and Lucas
Ž2000a . Three samples were analyzed for their protein and carbohydrate content while two samples underwent.
fatty acid analysis. Numbers in parentheses indicate standard deviation.
Treatment Protein Carbohydrate 16:0 18:3ny3 20:5ny3 SAFA PUFA ny3 FAs
y1 y1
LL P 6.3 0.23 2.56 0.01 12.98 1.26 12.52 29.91 34.42 17.60
Ž . Ž .
LL N 3.9 0.88 1.06 0.16 15.91 1.12 12.65 30.00 37.79 18.31
Ž . Ž .
C. muelleri HL fr2 9.2 1.80 3.19 0.81 11.10 0.86 17.79 24.33 50.99 22.44
Ž . Ž .
HL P 8.6 0.74 5.60 0.22 17.22 0.24 14.38 29.93 42.08 17.57
Ž . Ž .
HL N 7.2 0.45 4.85 0.47 23.41 B.D. 16.51 32.20 37.11 19.85
Ž . Ž .
LL fr2 12.5 1.5 3.20 0.30 10.75 0.49 21.45 23.22 52.02 25.11
Ž . Ž .
LL P 10.5 2.0 2.61 0.17 22.69 B.D. 9.35 42.71 29.53 13.11
Ž . Ž .
LL N 10.3 1.0 2.88 0.39 14.44 0.95 13.59 27.99 37.87 17.28
Ž . Ž .
R. reticulata HL fr2 25.9 3.49 4.2 0.78 7.08 15.92 11.67 17.04 69.71 59.77
Ž . Ž .
HL P 29.0 3.94 7.6 1.43 11.04 15.07 3.87 24.12 53.36 34.33
Ž . Ž .
HL N 36.2 2.12 18.3 0.8 25.09 13.43 2.08 49.64 33.21 27.32
Ž . Ž .
LL fr2 25.4 1.68 8.0 2.31 6.10 12.31 9.95 12.93 78.09 52.44
Ž . Ž .
LL P 28.2 2.89 4.7 0.04 8.37 17.44 8.63 12.33 75.39 59.01
Ž . Ž .
LL N 32.8 3.66 6.3 0.25 13.03 12.36 8.10 23.16 66.24 42.64
Ž . Ž .
P. lutheri HL fr2 3.7 1.25 0.51 0.06 18.50 0.99 27.88 27.71 55.71 43.61
Ž . Ž .
HL P 6.6 0.51 0.94 0.09 25.20 1.48 16.51 34.55 40.84 28.18
Ž . Ž .
HL N 4.1 0.2 0.45 0.17 17.62 1.88 17.66 27.11 57.40 39.69
Ž . Ž .
LL fr2 4.5 0.54 0.55 0.02 14.15 1.31 22.18 22.97 64.07 49.00
Ž . Ž .
LL P 5.7 1.93 0.54 0.15 17.75 2.44 25.63 26.01 59.84 42.81
Ž . Ž .
LL N 7.1 0.74 0.74 0.08 16.29 1.53 27.45 24.07 62.53 47.24
In all four algal species used, carbohydrate content per cell was not greatly affected by the culture conditions showing one- to twofold changes. Carbohydrate content of S. costatum was generally increased with decreased irradiance except for the nitrogen-limited cultures where carbohydrate content remained practically unchanged at both irradiance levels. R. reticulata, on the other hand, showed a different response with conservative changes in carbohydrate content; at the higher-light intensity, carbohydrate content was increased only under phosphorus limitation and at the lower-light under nitrogen limitation. The carbohydrate content of C. muelleri was affected more by nutrient limitation at the higher-light intensity while at low-light, there was little change. In contrast, P. lutheri showed little change in its carbohydrate content under the conditions employed.
With regard to the fatty acid content of the algal species used, it appeared that
Ž .
saturated fatty acids SAFA were increased as a proportion of the total identifiable fatty
Ž .
Fig. 1. Length of D-stage larvae as well as final length of M. edulis larvae after the 14-day feeding trial using the Control, the S. costatum and P. lutheri as food cultured under various light and nutrient conditions. Results shown are average of three replicate cultures and vertical lines indicate "standard deviation.
Ž .
fr2sfr2 medium no nutrient limitation ; Psphosphorus-limited medium, Nsnitrogen-limited medium.
Fig. 2. Length of D-stage larvae as well as final length of M. edulis larvae after the 14-day feeding trial using the Control, the C. muelleri and the R. reticulata diets as food cultured under various light and nutrient conditions. Results shown are average of three replicate cultures and vertical lines indicate "standard
Ž .
Ž .
picture emerged for the PUFA and the ny3 series of fatty acids ny3 . In S.
costatum, PUFA and ny3 decreased with nutrient limitation under the higher-light intensity, but under low-light, PUFA proportions increased in the phosphorus-limited cells and decreased in the nitrogen-limited cells. C. muelleri increased its SAFA proportion under nutrient stress at both light intensities, while ny3 FA decreased under the same conditions. A similar decrease occurred with PUFA, except with phosphorus limitation at low light, where its proportions were approximately the same as with the
Ž .
non-limiting nutrient fr2 conditions. The proportion of PUFA and ny3 in R.
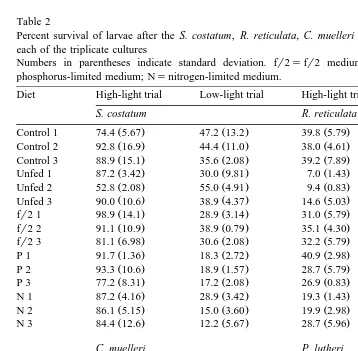
Table 2
Percent survival of larvae after the S. costatum, R. reticulata, C. muelleri and P. lutheri feeding trials, for each of the triplicate cultures
Ž .
Numbers in parentheses indicate standard deviation. fr2sfr2 medium no nutrient limitation ; Ps phosphorus-limited medium; Nsnitrogen-limited medium.
Diet High-light trial Low-light trial High-light trial Low-light trial
S. costatum R. reticulata
Ž . Ž . Ž . Ž .
Control 1 74.4 5.67 47.2 13.2 39.8 5.79 39.8 5.79
Ž . Ž . Ž . Ž .
Control 2 92.8 16.9 44.4 11.0 38.0 4.61 38.0 4.61
Ž . Ž . Ž . Ž .
Control 3 88.9 15.1 35.6 2.08 39.2 7.89 39.2 7.89
Ž . Ž . Ž . Ž .
Unfed 1 87.2 3.42 30.0 9.81 7.0 1.43 7.0 1.43
Ž . Ž . Ž . Ž .
Unfed 2 52.8 2.08 55.0 4.91 9.4 0.83 9.4 0.83
Ž . Ž . Ž . Ž .
Unfed 3 90.0 10.6 38.9 4.37 14.6 5.03 14.6 5.03
Ž . Ž . Ž . Ž .
Control 1 74.4 5.67 47.2 13.2 35.0 2.36 35.0 2.36
Ž . Ž . Ž . Ž .
Control 2 92.8 16.9 44.4 11.0 40.0 7.20 40.0 7.20
Ž . Ž . Ž . Ž .
Control 3 88.9 15.1 35.6 2.1 43.3 3.60 43.3 3.60
reticulata showed smaller changes under low irradiance, whereas at the high light, there was a peak of PUFA when nutrients were not limited. It was also noticeable that there was a marked increase in the SAFA proportion in R. reticulata grown under nitrogen limitation at the higher-light conditions. For P. lutheri, there was little change in overall SAFA components with the various treatments. The exception occurred under high-light phosphorus limitation where the SAFA proportion was considerably increased and there
was a corresponding reduction in the PUFA and ny3 proportions.
The final length of M. edulis larvae achieved after the 14-day feeding trial, as well as the corresponding length of the control unfed and at the initial D-stage, is illustrated in Fig. 1, when the larvae were fed either on S. costatum or P. lutheri and in Fig. 2, when fed on C. muelleri or R. reticulata. With the aid of nested ANOVA and analysis of means, the S. costatum diets were ranked, in decreasing order of exhibited larval
Ž .
growth, with ‘‘)’’ indicating significant difference 95% , as follows: LL NsLL
fr2)Control)HL fr2sHL PsHL N)LL P. The C. muelleri diets were ranked,
in decreasing quality, in the high-light experiment as: HL N)Control)HL fr2sHL
P, and for the low-light experiment, LL fr2sLL N)ControlsLL P. The R.
reticulata diets were ranked as: HL NsLL fr2sControl)LL N)HL fr2sHL PsLL P. Finally, the P. lutheri diets were ranked, again in decreasing quality order as:
HL P)HL fr2sHL N)LL NsLL P)LL fr2sControl.
Ž .
It becomes clear that survival varied between experiments Table 2 , which were carried out using different batches of larvae, while in the same experiment, survival could not be correlated with the algal diet. This argument is supported by the variation
Ž
in survival between triplicates of the same diet showing significant differences P
-.
0.05 .
Ž .
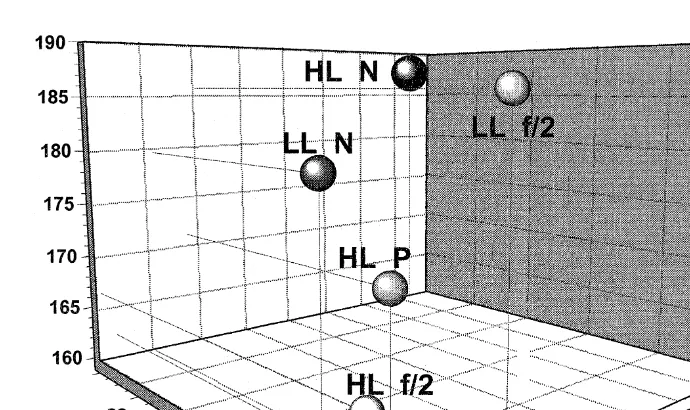
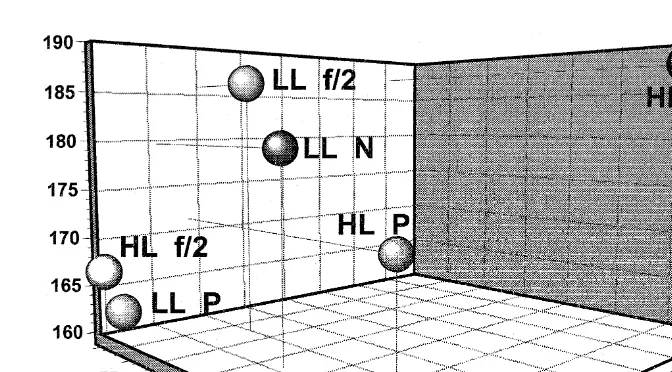
Fig. 4. Relationship between 16:0 proportion as percentage of total identifiable fatty acids, TIFA , protein and carbohydrate content of R. reticulata and larval length achieved after feeding for 2 weeks on this species. Protein content is shown colour-coded.
There were few occasions that a coherent relationship between algal biochemical components and larval size could be established. In P. lutheri, there was very clear
Ž .
Ž .
Fig. 6. Relationship between total ny3 FA proportion as percentage of total identifiable fatty acids, TIFA , protein and carbohydrate content of R. reticulata and larval length achieved after feeding for 2 weeks on this species. Protein content is shown colour-coded.
positive correlation between the 16:0 and SAFA, with growth and negative correlations
Ž .
existing between PUFA and ny3 FA with larval size Fig. 3 , with correlation values
of 0.97, 0.98, y0.98 and y0.97, respectively. Protein and carbohydrate content were
not significantly correlated with larval growth, with correlation values of 0.19 and 0.57, respectively. However, protein, carbohydrate and some fatty acids of R. reticulata were correlated with larval growth. Generally, lower correlation values were calculated with
0.73 and 0.48 for protein and carbohydrate content and 0.55, y0.91, 0.56, y0.6 for
16:0, 18:3ny3, SAFA and ny3 FA, respectively. Four-dimensional plots of larval
length, algal protein, carbohydrate and one of these algal fatty acids components are
shown in Figs. 4–6 for the 16:0, 18:3ny3 and ny3 FA, respectively. Collectively,
these graphs, together with the correlation analysis, indicate that there is a positive
correlation between algal 16:0 and SAFA with larval growth while algal 18:3ny3 and
ny3 FA were negatively correlated with larval growth. Despite careful examination of
the data for C. muelleri and S. costatum, no consistent relationship could be established.
4. Discussion
Culturing live algae for food in hatcheries is essential, especially given the limited
Ž .
success, up to now, of artificial feeds Laing, 1987 ; hence, optimizing the nutritional value of any alga for a specific animal should be of critical importance. Environmental
Ž
conditions have been shown to influence algal biochemical composition Leonardos and
.
S. costatum is a species used extensively in hatcheries mainly because of its ability to thrive in a variety of environments while also supporting good growth of animals
ŽO’Connor et al., 1992 . It was found to be the second highest-ranked diatom for Ostrea.
Ž .
edulis growth by Enright et al. 1986a among the species tested and one of the best
Ž .
diets for the hard clam, Mercenaria mercenaria Wikfors et al., 1992 . C. muelleri has
Ž
been shown to have good potential in a subtropical greenhouse bivalve hatchery Nelson
.
et al., 1992 . A good nutritional value record is also reported for this diatom for the
Ž .
Sydney rock oyster, Saccostrea commercialis by O’Connor et al. 1992 and its high nutritional value is further confirmed here with M. edulis larvae. It appears that this Chaetoceros species is no exception from the generally good nutritional value record
Ž
that this genus has been shown to demonstrate Chu, 1989; O’Connor and Heasman,
. Ž .
1997 . Of all the species tested here, R. reticulata previously Rhodomonas baltica is the least studied, although other Cryptomonads, described as Rhodomonas species, have
Ž .
been found to be good diets for O. edulis Enright et al., 1986a . On the basis of the very high nutritional value found here, the apparent lack of extensive usage of this species in mariculture is difficult to substantiate. P. lutheri has an ambiguous record of nutritional value in which it appears to be a very good food for larvae of several
Ž
bivalves, e.g., doughboy scallops, Mimachlamys asperrima O’Connor and Heasman,
. Ž
1997 , American and European oysters, Crassostrea Õirginica and O. edulis Walne,
.
1963; Chu and Dupuy, 1980 , but mediocre and poor for some others like the Japanese
Ž .
scallop, Patinopecten yessoensis Thompson et al., 1994 and the Pacific oyster, C.
Ž .
gigas Langdon and Waldock, 1981 . The present work classifies this species as an excellent food for M. edulis larvae since in almost all of its biochemical compositions, it performed better than the control diet. This control two-species diet was expected to be of superior value, because it was considered to contain the greater diversity of biochemical constituents to satisfy most nutritional requirements for growth than any
Ž .
one monospecific diet Widdows, 1991 .
Ž .
Thompson et al. 1996 correlated positively the proportion of dietary 16:0 and
negatively the dietary 20:5ny3 of P. lutheri with larval mortality in C. gigas; both
these results were unequivocally confirmed here with the use of correlation analysis. It
appears that in PUFA and ny3 FA, in general, rather than specifically, the 20:5ny3
was negatively correlated with larval growth. Hence, it is not clear whether the positive and negative correlations of 16:0 and 20:5ny3, respectively, are due to their specific
role or is a reflection of the positive and negative correlations of SAFA and ny3 FA,
generally.
The use of a multidimensional model enables a more comprehensive inclusion of dietary components that have been shown in the past to be important in determining the algal nutritional value, overcoming the problematic usage of multilinear regression
Ž .
Again, the decrease of nutritional value was found when higher proportions of 18:3ny3 and ny3 fatty acids, in general, were observed. The descriptive nature of this approach does not provide an exact measure by which the contributions to the nutritional value of any algal component can be determined, but it provides a means of demonstrating how
Ž
dietary components interact to determine the overall dietary value Enright et al.,
. Ž .
1986a,b . However, the comments of Dickey-Collas and Geffen 1992 and Thompson
Ž .
et al. 1996 about the positive effects of increased 16:0 and SAFA and the negative
effects of increased PUFA and more specifically, the ny3 fatty acids are generally
confirmed here. It would seem that in the cases where such a clear-cut relationship is found, with dietary fatty acid being the main factor in determining nutritional value, the other biochemical components are of minor importance. In other cases, there is a positive effect of increased protein and carbohydrate content, with fatty acids also contributing to the nutritional value.
Ž
These results are apparently in conflict with the widely publicized held view e.g., Su
.
et al., 1988 that increased dietary PUFA enhance the dietary value of an algal diet, not least because larval PUFA have been shown to be a positive growth index for M. edulis
ŽLeonardos and Lucas, 2000b . Langdon and Waldock 1981 demonstrated the impor-. Ž .
tance of PUFA, especially the 20:5ny3, to C. gigas. Many researchers in the
aquacultural field, largely because of this strong correlation of the larval 20:5ny3 fatty acid and PUFA, in general, with growth, use the occurrence and quantity of these
Ž .
compounds as ‘‘a rule of thumb’’ guide to rank algal diets Su et al., 1988 . Other workers have indirectly further substantiated this relationship by trying to explain the nutritional deficiency of particular algal species in terms of the lack of some essential
Ž .
fatty acids, such as some of the ny3 groups De Pauw et al., 1984 . From the current
findings, this is a misleading overgeneralization to make. A clear distinction should be made between the nutritional elements in the diet and the way that they influence larval growth and development on one hand and the biochemical composition of the larvae themselves. A low level or lack of one dietary component, e.g., a particular fatty acid, will not necessarily be reflected by a deficiency in the same component in the larvae, since the animals may transform almost all of the components that they digest, through their metabolic pathways; a deficiency in the input element will translate into a deficiency of the end product inside the animal cells. Therefore, it is not entirely correct to substantiate the correlation of a larval fatty acid with larval growth by observing the effect that it has if it is excluded or inadequately present in the diet, or vice versa. The sole significance is related to the nutritional quality of the diet for that given organism and not as to the importance as a growth index for the animals themselves.
which environmental conditions determine the nutritional value of an alga, but the range of species and clones investigated must also be widened. Further research should aim at enlightening the interactive way in which discrete algal biochemical components deter-mine the nutritional value of the species, with emphasis given to their fatty acid profile.
Acknowledgements
The first author would like to thank Andy Beaumont for his helpful comments throughout this work and J. East for his invaluable help with the fatty acid analyses. The present work is part of a PhD research thesis funded by the Greek State Scholarship
Ž .
Foundation I.K.Y. whose assistance is acknowledged.
References
Baars, J.W.M., 1981. Autecological investigations on marine diatoms: II. Generation time of 50 species. Hydrobiol. Bull. 15, 137–151.
Bayne, B.L., 1965. Growth and the delay of metamorphosis of the larvae of Mytilus edulis L. Ophelia 2, 1–47.
Chu, K.H., 1989. Chaetoceros gracilis as the exclusive feed for the larvae and postlarvae of the shrimp
Metapenaeus ensis. Aquaculture 83, 281–287.
Chu, F.L.E., Dupuy, J.L., 1980. The fatty acid composition of three unicellular algal species used as food
Ž .
sources for larvae of the American oyster CrassostreaÕirginica . Lipids 15, 356–364.
De Pauw, N., Morales, J., Persoone, G., 1984. Mass cultures of microalgae in aquaculture systems: progress and constraints. Hydrobiologia 116r117, 121–134.
Dickey-Collas, M., Geffen, A.J., 1992. Importance of the fatty acids 20:5v3 and 22:6v3 in the diet of plaice
ŽPleuronectes platessa larvae. Mar. Biol. 113, 463–468..
Enright, C.T., Newkirk, G.F., Craigie, J.S., Castell, J.D., 1986a. Evaluation of phytoplankton as diets for juvenile Ostrea edulis L. J. Exp. Mar. Biol. Ecol. 96, 1–13.
Enright, C.T., Newkirk, G.F., Craigie, J.S., Castell, J.D., 1986b. Growth of juvenile Ostrea edulis L. fed
Chaetoceros gracilis Schutt of varied chemical composition. J. Exp. Mar. Biol. Ecol. 96, 15–26.
Epifanio, C.E., 1979. Growth in bivalve molluscs: nutritional effects of two or more species of algae in diets
Ž .
fed to the American oyster CrassostreaÕirginica Gmelin and the hard clam Mercenaria mercenaria L. Aquaculture 18, 1–12.
Gallager, S.M., Mann, R., 1981. The effect of varying carbonrnitrogen ratio in the phytoplankter T.
Ž3 .
pseudonana H on its food value of the bivalve Tapes japonica. Aquaculture 26, 95–105. Laing, I., 1987. The use of artificial diets in rearing bivalve spat. Aquaculture 65, 243–249.
Langdon, C.J., Waldock, M.J., 1981. The effect of algal and artificial diets on the growth and fatty acid composition of Crassostrea gigas spat. J. Mar. Biol. Assoc. UK 61, 431–448.
Leonardos, N., 1998. Environmental effects on the growth and biochemical composition of four microalgae, in relation to their use as food for Mytilus edulis larval rearing. PhD thesis, University of Wales, Bangor, 248 pp.
Leonardos, N., Lucas, I.A.N., 2000a. Effects of environmental parameters in the growth and biochemical composition, with emphasis in fatty acid content, of four microalgae. J. Appl. Algae, submitted. Leonardos, N., Lucas, I.A.N., 2000b. The use of larval fatty acids as an index of growth in Mytilus edulis L.
larvae. Aquaculture, in press.
Loosanoff, V.L., Davis, H.C., 1963. Rearing of bivalve molluscs. Adv. Mar. Biol. 1, 1–136.
O’Connor, W.A., Heasman, M.P., 1997. Diet and feeding regimens for larval doughboy scallops,
Mimach-lamys asperrima. Aquaculture 158, 289–303.
O’Connor, W.A., Nell, J.A., Diemar, J.A., 1992. The evaluation of twelve algal species as food for juvenile
Ž .
Sydney rock oysters Saccostrea commercialis Iredale and Roughley . Aquaculture 108, 277–283. Sargent, J.R., Whittle, K.J., 1981. Lipids and hydrocarbons in the marine environment. In: Longhurst, A.R.
ŽEd. , Analysis of Marine Ecosystems. Academic Press, London, pp. 491–533..
Sokal, R.R., Rohlf, F.J., 1987. Introduction to Biostatistics, 2nd edn. Freeman, New York.
Su, H.M., Lei, C.H., Liao, I.C., 1988. The effect of environmental factors on the fatty acid composition of
Ž . Skeletonema costatum, Chaetoceros gracilis and Tetraselmis chuii. J. Fish. Soc. Taiwan 15 1 , 21–34.
Thompson, P.A., Guo, M.-X., Harrison, P.J., 1993. The influence of irradiance on the biochemical
composi-Ž
tion of three phytoplankton species and their nutritional value for larvae of the Pacific oyster Crassostrea
.
gigas . Mar. Biol. 117, 259–268.
Thompson, P.A., Guo, M.-X., Harrison, P.J., 1994. Influence of irradiance on the nutritional value of two
Ž .
phytoplankton species fed to larval Japanese scallops Patinopecten yessoensis . Mar. Biol. 119, 89–97. Thompson, P.A., Guo, M.-X., Harrison, P.J., 1996. Nutritional value of diets that vary in fatty acid
Ž .
composition for larval Pacific oysters Crassostrea gigas . Aquaculture 143, 379–391.
Walne, P.R., 1963. Observations on the food value of seven species of algae to the larvae of Ostrea edulis: I. Feeding experiments. J. Mar. Biol. Assoc. UK 43, 767–784.
Watanabe, T., Kitajima, C., Fujita, S., 1983. Nutritional values of live organisms used in Japan for mass propagation of fish: a review. Aquaculture 34, 115–134.
Webb, K.L., Chu, F.L.E., 1983. Phytoplankton as a food source for bivalve larvae. In: Pruder, G.D., Langdon,
Ž .
C., Conklin, D. Eds , Proceedings of the 2nd International Conference on Aquaculture Nutrition. Biochemical and Physiological Approaches to Shellfish Nutrition. World Mariculture Society Special Publication, Vol. 2, pp. 272–291.
Widdows, J., 1991. Physiological ecology of the mussel larvae. Aquaculture 94, 147–163.
Wikfors, G.H., Twarog, J.R.W., Ukeles, R., 1984. Influence of chemical composition of algal food sources on growth of juvenile oysters, CrassostreaÕirginica. Biol. Bull. 167, 251–263.