Title: Handbook on smart materials in analytical chemistry / edited by Miguel de la Guardia, Francesc A Esteve-Turrillas. Esteve-Turrillas and Miguel de la Guardia 1.1 Role of smart materials in analytical chemistry 1 1.2 Smart materials for sample treatment 2.
List of Contributors
Centro de Investigación en Química de Materiales Departamento de Química y Centro de Excelencia para la Innovación en Química, Universidad Khon Kaen. Departamento de Química de Providencia González-Hernández (Unidad Departamental de Química Analítica) Universidad de La Laguna.
Preface
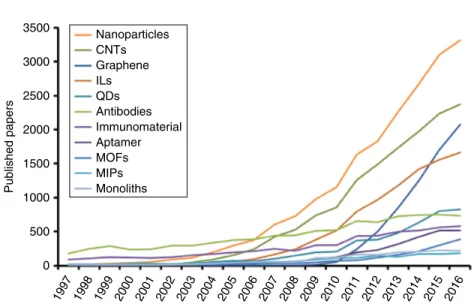
Role of Smart Materials in Analytical Chemistry
Thus, analytical chemistry is a multidisciplinary science in constant development, which must be adapted to face new problems and limitations. In this sense, the design of green analytical chemistry in its 12 principles takes into account aspects such as: i) direct analytical techniques instead of sample processing, (ii) reduction of samples and residues, (iii) automation and miniaturization, and (iv) multi-analyte determination methods [2] .
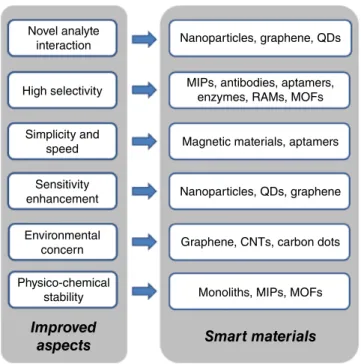
Smart Materials
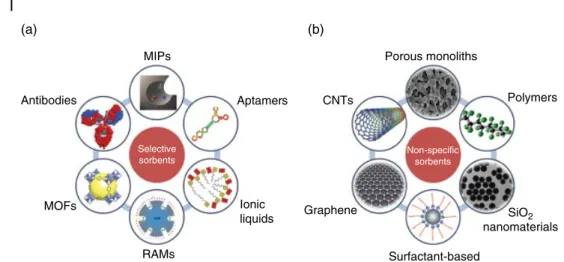
Smart Materials for Sample Treatment
- Solid‐Phase Extraction
- Solid‐Phase Microextraction
- Magnetic Extraction
- Automatization and Miniaturization
However, the use of magnetite (Fe3O4) is usually preferred due to the ease of synthesis, biocompatibility and low cost [38]. However, the use of RAMs, MIPs and immunosorbents has also been reported to increase the selectivity of the procedure [ 45 ].
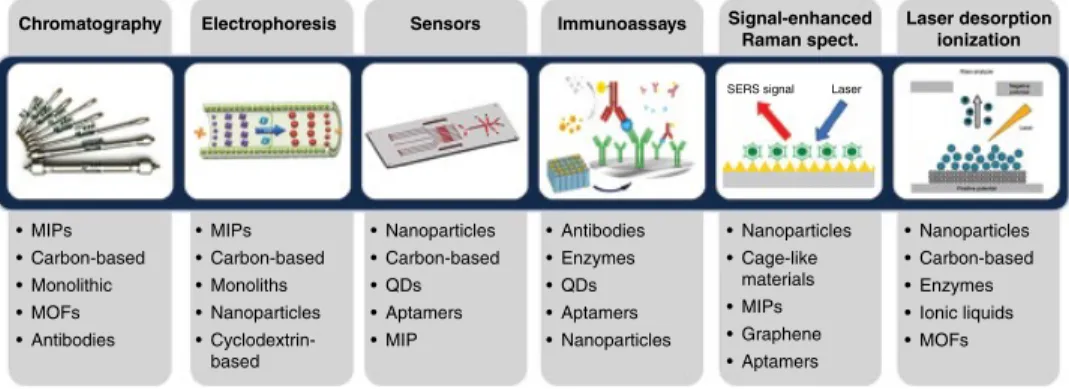
Smart Materials for Analytical Determinations
- Stationary Phases
- Sensor Development
- Immunoassays
- Signal Enhancement
- Laser Desorption/Ionization Mass Spectrometry
Graphene nanosheets, graphene oxide and reduced graphene oxide have been used in the development of chemiluminescence resonance energy transfer and luminescence quenching based sensors [70]. Aptasensors have been used for the analysis of small molecules, proteins and nucleic acids through their optical, electrochemical coupling.
The Future Starts Now
Smart materials based on organic matrices are promising sorbents that offer new functions for MALDI‐MS. Furthermore, the combination of different smart materials and their interfacing with new or classic instruments further expands the possibilities within the reach of current scientific advances.
Acknowledgements
However, some successful progress will be made through the close collaboration of chemists, biologists and materials scientists, working together to develop tailor-made materials that will enable us to improve current analytical procedures and design new ones. In this book, you are sure to find many exciting ideas to improve your daily work.
Introduction
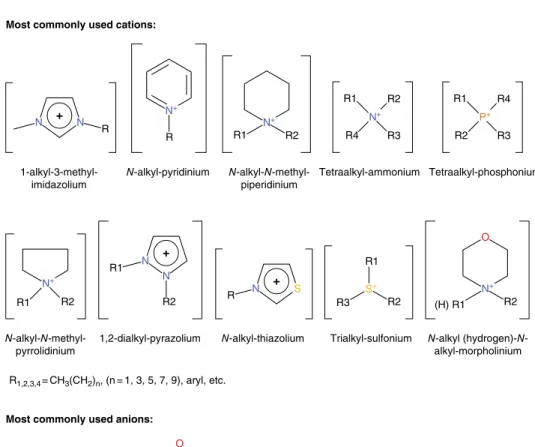
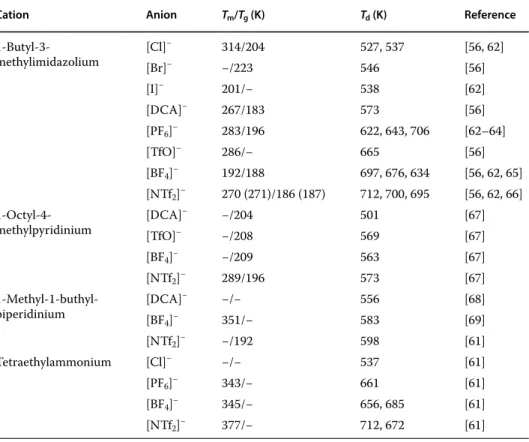
The most commonly used definition of ionic liquids or so-called "molten salts" describes these compounds as salts characterized by a melting temperature below 100 °C. This asymmetry plays a key role in limiting and hindering the crystallization process, resulting in low melting temperatures for ionic liquids.
Nanoconfined Ionic Liquids
Bulk Properties of Ionic Liquids
- Solvation Versatility
- Thermal Properties
- Electrochemical Window
The thermal stability of ionic liquids changes over a very wide range depending on the cation-anion system. The reduction potentials of tertiary sulfonium-based ionic liquids with [NTf2] anion are slightly higher compared to liquids with an imidazolium core [91].
Confinement Effects
- Structure of Confined Ionic Liquids
- Impact of Confinement on Physicochemical Properties of Ionic Liquids
Based on the TEM images, structural models (middle) were constructed and the diameter of the SWNTs suitable for each configuration was determined (middle right). The results of studies on the second IL also show that the degree of melting temperature reduction is influenced by the size of the anion.
![Figure 2.5 (a) Optimized geometry of [C 2 C 1 IM][EtSO 4 ], (b)–(d) outputs for the IL [C 2 C 1 IM][EtSO 4 ] in confinement in different trials, respectively, showing interaction of IL‐anion with Si present on the surface of silica pore wall matrix, IL‐](https://thumb-ap.123doks.com/thumbv2/123dok/10273783.0/55.723.387.626.101.289/figure-optimized-geometry-outputs-confinement-different-respectively-interaction.webp)
Preparation of Ionogels
- In‐Situ Impregnation
- Post‐impregnation
- Other Methods
An analogous consideration can be made in the case of ILs characterized by relatively loose structuring in the bulk phase. One such technique is the "ship-in-a-bottle" technique applied in the case of zeolites.
Analytical Applications of Ionic Liquids Confined in a Solid Matrix
- Solid‐Phase Extraction
- Solid‐Phase Microextraction
- Stir‐Bar Sorptive Extraction
- Biosensors
Under optimized conditions, the limits of detection were in the range of 0.04–0.95 μg l−1, and so was the repeatability and reproducibility of the method (RSD%). High loads of the IL were limited to the hybrid network with sol-gel technology. 185] applying ionic liquid-mediated sol-gel sorbents to solid-phase microextraction of hollow fibers.
The mechanical strength and durability (chemical/thermal stability) of the prepared IL-bonded sol-gel coatings were excellent.
Conclusions
Polymeric ionic liquids as selective coatings for the extraction of esters using solid-phase microextraction.
Introduction
Smart Porous Monoliths for Chromatographic Separations
Temperature Responsive Polymers
- Grafted on Organic Monoliths
- Silica and Hybrid Monoliths
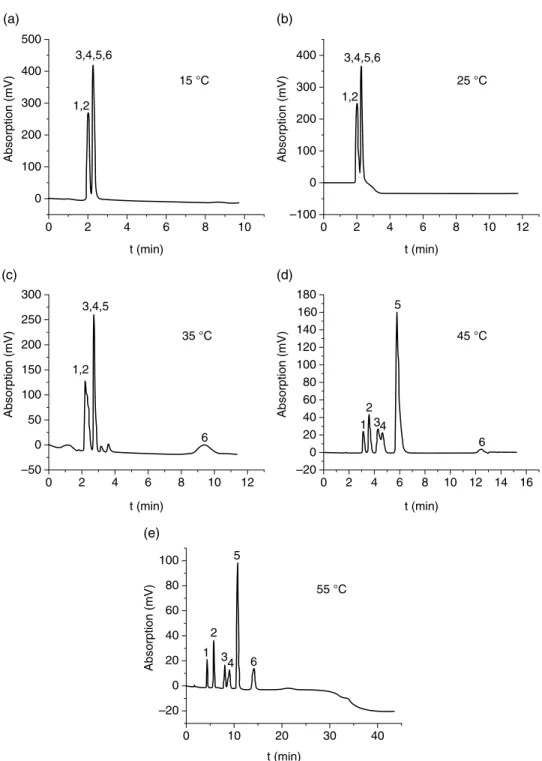
39] investigated the influence of the molar mass of the polymers on the separation performance of grafted silica beads in the separation of steroids. The effect of the density of PNIPAAm on the separation of steroids was investigated (Figure 3.11). Copolymers of MeO2MA and OEGMA were prepared from the commercially available monomers of ATRP in the presence of the initiator N-succinimidyl-2-.
35], which grafted P(MEO2MA-co-OEGMA) onto a methacrylate monolith, the temperature dependence of the separation was not as clear as in the PNIPAAm-grafted columns (Figure 3.12).
Salt Responsive Monoliths
Temperature PNIPAAm Aminated P(GMA‐co ‐EDMA) 10 mm thick membrane Separation of proteins, thermal valves, thermal gates [22]. Separation of sodium benzoate, phenol, methylbenzene, ethyl p-aminobenzoate, ethyl benzoate and methyl hydroxybenzoate by hydrophobic interactions. These monoliths are efficient for the separation of proteins by cation or anion exchange, but they fail to separate small cations and anions.
Baseline separation of cytochrome c, myoglobin, bovine serum albumin (BSA) and bovine thyroglobulin was achieved by a combination of linear and stepwise gradient elution with Na2SO4 from 2.0 to 0 mol l−1 in 0.050 mol l−1 phosphate buffer, pH 7.0.
Dual Mode Stimuli/Response
Monolith P(GMA-co-EDMA) has been exploited to produce ion exchangers by chemical derivatization of epoxy groups of GMA with Na2SO3, ethylene diamine (EDA), chloroacetic acid and iminodiacetic acid (IDA) [9]. The retention factors of several steroids increased with increasing salt concentration (NaCl, Na2SO4 and (NH4)2SO4) at a constant temperature of 28 °C, with the highest retention being found for progesterone, the steroid with one of the highest log big. P (Table 3.1). Therefore, the retention factor of the three hydrophobic steroids (hydrocortisone, dexamethasone acetate, and medroxyprogesterone acetate) increased with pH until the pH of the mobile phase was adjusted to 8.0.
Separation of the three steroids was achieved at mild conditions of pH 7.0 through hydrophobic interactions with the surface of the partially collapsed chains of the deprotonated PDMAEMA.
Temperature Responsive Molecularly Imprinted Monoliths
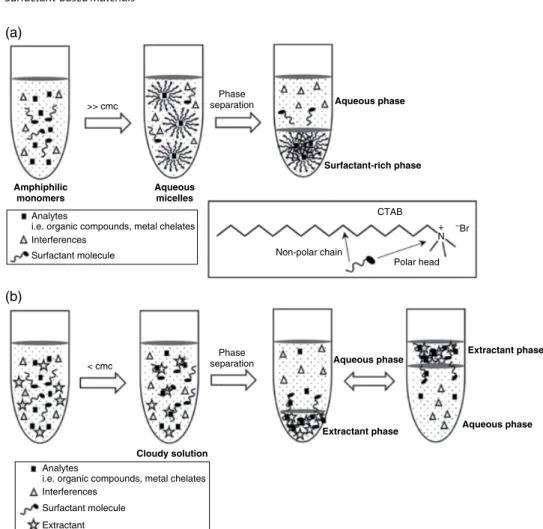
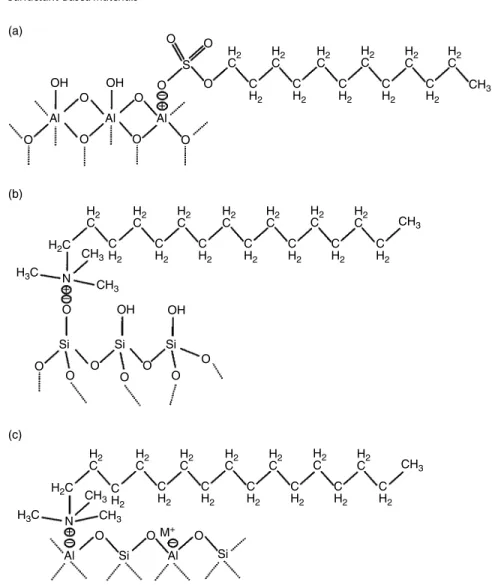
Retention factors increased with the increase in salt concentration because the charges of the protonated tertiary amines were screened by the increased amount of NaCl favoring hydrophobic retention of the steroids, which were separated by elution with 1.5 mol l−1 NaCl, pH 7.0, and flow rate of 1 ml min−1 [54]. At pH 7.0 and a temperature In addition, the number of binding sites of the smart MIP monolith at 35 °C was approximately 76 times greater than that at 25 °C. Scanning electron micrographs show the internal structures of the P(GMA-co-EDMA) monolith (a) and its counterpart grafted with the protein-imprinted PNIPAAm layer (b) with significantly larger non-porous microglobules and denser morphology, confirming successful grafting. of suppressed PNIPAAm. On the other hand, the cis membrane separation factor can be decreased by 455 nm radiation [65]. Thermoresponsive columns for HPLC: effect of chromatographic support and polymer molecular weight on column performance. A thermosensitive monolithic column as an artificial antibody for the online selective separation of the protein. Surfactants Recently, there has been increasing interest in the development of environmentally friendly analytical methodologies following the green analytical chemistry approach. In modern liquid-liquid micro-extraction (LLME) techniques, surfactants are added to the extraction media as an emulsifier to facilitate the dispersion of extraction solvent in aqueous sample solution instead of toxic organic solvent. The addition of surfactant to the extraction system containing an aqueous sample and a water-immiscible organic extractant lowers the interfacial tension between two phases. At low surfactant concentration, the electrostatic interaction between the polar head group of the surfactant and the surface charge is the primary force involved in the adsorption process, and the surfactant molecules are dispersed on solid surfaces. The amount of Triton X-114 affects the final volume of the surfactant-rich phase that is recovered. A wetting-rich phase was separated at the cloud point of the non-ionic wetting agent (Triton X-114) and then determined by ETAAS. The contact angle of water with the carbon surface of the graphite used in ETAAS is 85.7°; the surfactant can reduce the contact angle of an aqueous solution with graphite, so they may offer a possible solution to this inconvenience. The addition of Triton X-114 (as an emulsifier) could increase the rate of mass transfer from the aqueous sample to the extraction solvent. The study shows that surfactant-modified zeolites are effective sorbents for the removal of As(V) from aqueous solution [106]. For example, CTAB-modified zeolite NaY has been used as a sorbent for the preconcentration of carbamate pesticides in vortex-. Surfactant-modified zeolite has been extensively studied for the removal of inorganic anions and other ionizable organic solvents. Several surfactant-modified solid sorbents have been comparatively studied for the retention of carbamate pesticides in aqueous solutions [112]. Ultrasound-assisted surfactant-enhanced emulsification microextraction for the determination of carbamate pesticides in water samples by high-performance liquid chromatography. Ultrasound-assisted surfactant-enhanced emulsification microextraction for the determination of carbamates in ultrahigh-performance liquid wines. Ultrasound-assisted surfactant-enhanced emulsification microextraction combined with HPLC for the determination of estrogens in water. Determination of Phthalate Esters in Beverage Samples by Vortex-Assisted Surfactant-Enhanced Emulsification Liquid-Liquid. Introduction As shown above, a number of MIPs have been developed for the selective adsorption and detection of proteins/glycoproteins. Recent advances for the selective pharmaceutical assays using molecularly imprinted adsorbents and their related techniques: a review. Development and characterization of magnetic molecularly imprinted polymers for selective enrichment of endocrine disrupting chemicals in water and milk samples. A new molecular imprinted electrochemical sensor modified with carbon dots, chitosan and gold nanoparticles for the determination of patulin. He was a visionary in the belief that better methods will be discovered in the near future, which will enable the wider use of enzymes in various fields of application [1]. To realize this vision, the industrial sector was the first to recognize the huge potential of enzymes and use them to speed up production processes, reduce reaction time, temperatures and pressures while using cheaper starting materials [2]. In this general scenario, one of the main fields of application is bioanalysis due to the commercial availability of pure enzymes produced in good quality and in large quantities. The era of enzymatic bioanalysis was entered in 1980 by the combination of enzymes with spectrophotometers, with Boehringer Mannheim being the pioneer company in marketing enzymatic test kits, followed by major companies such as Roche and Merck.Conclusions and Outlook
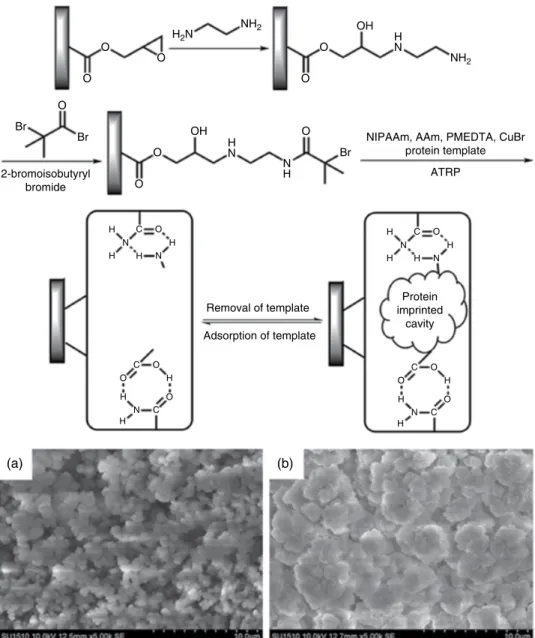
Surfactant‐Based Materials*
Roles of Surfactant in Modern Sample Preparation Techniques
Surfactant‐Based Liquid‐Phase Extraction
Surfactant‐Modified Sorbents
Final Remarks
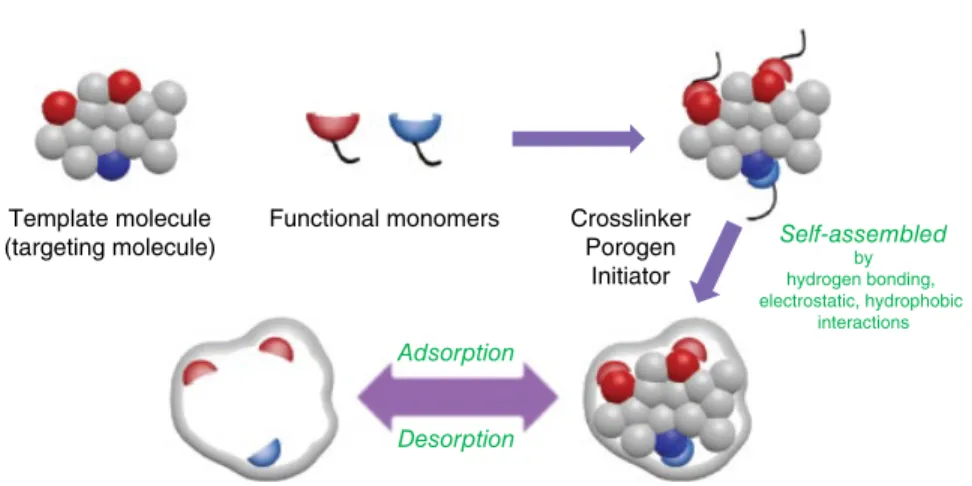
Molecularly Imprinted Materials
Enzyme‐Based Materials
![Figure 2.4 (a) DSC scan for [C 4 C 1 IM][BF 4 ], showing a distinct freezing point upon cooling and a distinct melting point upon heating](https://thumb-ap.123doks.com/thumbv2/123dok/10273783.0/51.723.102.555.46.946/figure-showing-distinct-freezing-cooling-distinct-melting-heating.webp)
![Figure 2.6 (A) Packing arrangement of [Me 3 NC 2 H 4 OH][ZnCl 3 ] inside SWNTs of different nanotube diameters](https://thumb-ap.123doks.com/thumbv2/123dok/10273783.0/57.723.109.620.111.693/figure-packing-arrangement-inside-swnts-different-nanotube-diameters.webp)
![Figure 2.10 EDX spectrum of [C 4 MIM][NTf 2 ]‐ionogel fiber and the corresponding elemental mapping of C, O, F, Si, and S](https://thumb-ap.123doks.com/thumbv2/123dok/10273783.0/73.723.103.642.628.893/figure-edx-spectrum-ionogel-fiber-corresponding-elemental-mapping.webp)