This is to confirm that the research work in the thesis titled "Studies on Poly (Lactic Acid) and Polysaccharide Gum based Bionanocomposites for Adhesive and Gas Barrier Film Applications", has been done by me at the Chemical Engineering Department, Indian Institute of Technology Guwahati, under Dr. The results documented in this thesis have been obtained by me and have not been submitted to any other university or institute for the award of any degree or diploma. Chapter three focuses on the selection of polysaccharide gums among various polysaccharide gums such as gum arabic, guar gum, xanthan gum and gum resin for the desired applications.
Synthesis and Characterization of Microwave Initiated Gum Arabic Grafted Poly L-Lactic Acid
Microwave Initiated Gum Arabic Grafted Poly L-Lactic Acid Bionanocomposite: Adhesive
PLA/Functionalized-Gum Arabic Based Bionanocomposite Films for High Gas Barrier
Thermal Degradation Behaviour of High Gas Barrier Poly (lactic acid)/modified Gum Arabic based
88 Figure 4.9 Contact angle analysis of GA, OLLA and GA-g-PLA. iii) 2000 N, (iv) comparison of shear strength at different compressive loads. 116 Figure 6.6 Mechanical properties of film samples (a) tensile strength,. b) elongation at break and (c) Young's modulus (error bars represent standard deviations).

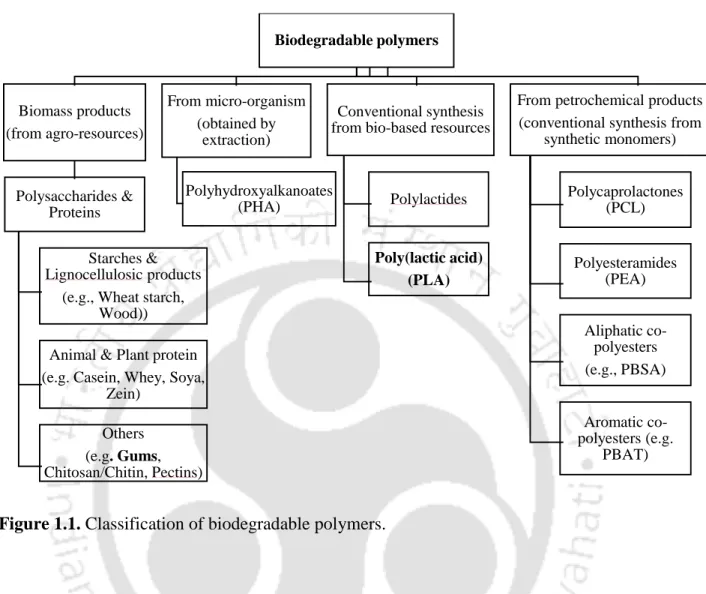
Introduction
Introduction
- Poly (lactic acid)
PLA has great potential to replace conventional non-renewable petroleum-based polymers (Zhang et al., 2014). PLA can be synthesized by various synthesis routes such as direct polycondensation, azeotropic dehydrative condensation, ring opening polymerization (ROP), melt polycondensation (MP) and solid state polymerization (SSP) of low molecular weight PLA (Lunt et al., 1998).
Lactic acid polymerization
- Polysaccharide gums
- Gum arabic
- Guar gum
- Xanthan gum
- Gum rosin
- Literature Review
- Grafting of polysaccharide gums
- Adhesive application of bio-based materials Polysaccharide gum based adhesive
- Packaging applications of bio-based materials Polysaccharide gum based films
- Gaps in the Prior Art
- Objectives of the Present Work
GA has been granted GRAS (Generally Recognized as Safe) status by the United States Food and Drug Administration (USFDA) (Nayak et al., 2012). Manna et al., 2015 studied the synthesis of carboxylmethyl guar gum (CMGG) from the indigenous guar gum (GG).
Materials and Methods
Materials
Methods
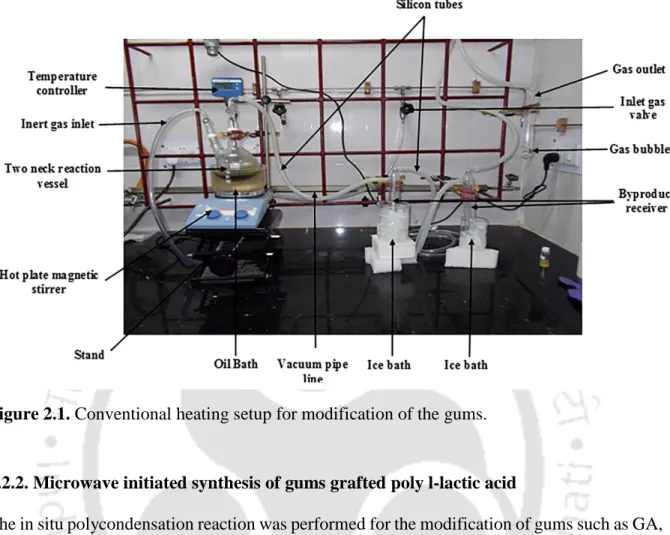
- Conventional heating method for the synthesis of gums grafted poly l-lactic acid Initially, 100 g of lactic acid (LA) was taken in a round bottom flask (RBF) and 10 % (wt/wt)
- Microwave initiated synthesis of gums grafted poly l-lactic acid
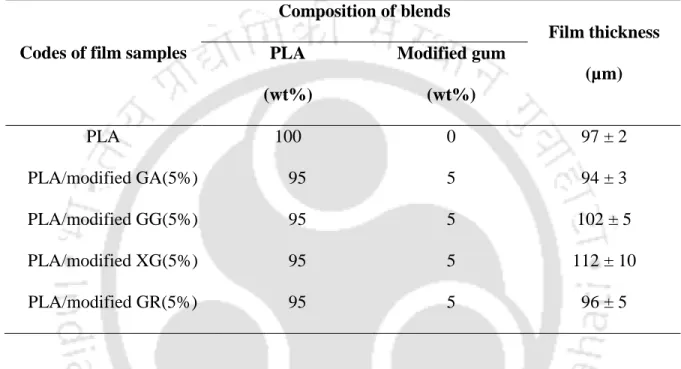
- Fabrication of PLA/modified gums (5%) bionanocomposite films
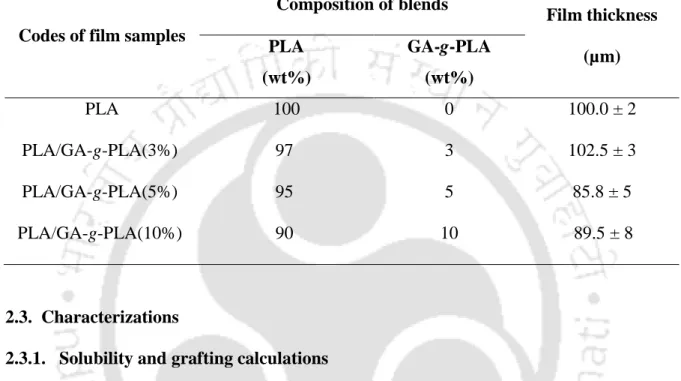
Pristine PLA, neat gum and PLA/modified gum (5%) based bionano composite films were prepared by solution casting method. The different concentrations of PLA/GA-g-PLA bionanocomposite films were prepared by the same procedure as described above.
Characterizations
- Solubility and grafting calculations
- Fourier transform infrared spectroscopy analysis
- Nuclear magnetic resonance analysis
- X-ray diffraction analysis
- Gel permeation chromatography analysis
- Differential scanning calorimetry analysis
- Thermogravimetric analysis
- Thermogravimetric analysis-fourier transform infrared spectroscopy (TGA-FTIR) analysis
- Scanning electron microscopy analysis
- Field emission scanning electron microscopy analysis
- Transmission electron microscopy analysis
- Contact angle analysis
- Single lap shear test analysis of modified gums for adhesive selection
- Mechanical properties
- Oxygen permeability analysis
- Water vapour transmission rate analysis
OLLA and GA-g-PLA at ~10 mg ml-1 concentration were prepared by dissolving the samples in deuterated chloroform (CDCl3). The thermal stability of the samples was determined by thermogravimetric analysis (TGA) with a PerkinElmer thermogravimetric analyzer (TGA 4000). The morphology of the samples was examined under exposure to an accelerating voltage of 10 kV.
The analyzed surface of the samples was prepared by dissolving the samples in chloroform, all samples were filtered using Whatman® syringe filter (0.45 µm pore size).
Selection of Gum Grafted Poly L-Lactic Acid for Adhesive and Food Packaging Application
Introduction
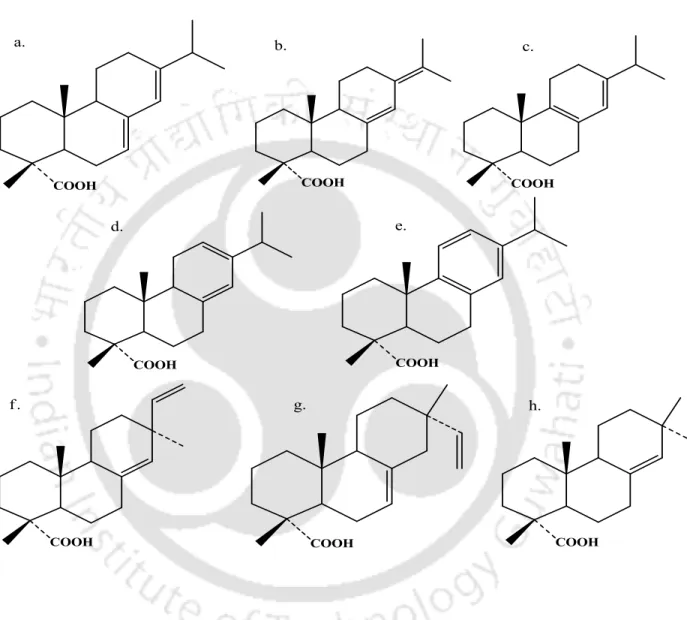
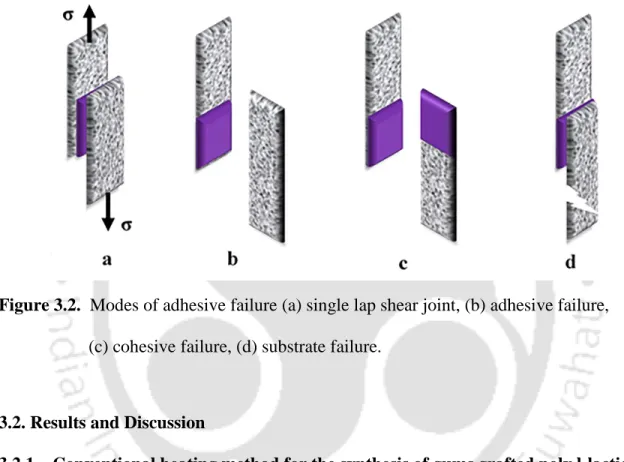
A few advantages of microwave heating over conventional heating are listed in Table 3.1. Adhesives and sealants have many commercial uses, being a 20-million-ton global industry with $20 billion in sales. When performing the test, we encounter three modes of failure to confirm the bonding strength of the adhesives (Figure 3.2).
The highest adhesive strength is achieved when the adhesive and cohesive strength of the adhesive is greater than the strength of the substrate, resulting in failure of the substrate (Dunn, 2003).

Results and Discussion
- Microwave initiated synthesis of gums grafted poly l-lactic acid
The free hydroxyl groups in the GA, GG and XG molecules were grafted with the reducing end groups of the LA molecules. The proposed Scheme 3.1 also shows the reaction of free amino groups in GA with LA molecules. Free radical sites were also generated on the polar groups of the polysaccharide gum backbone and LA monomer upon exposure to microwave radiation.
When the free radical sites were produced in the gum pillar, the LA monomer attached and diffusion of monomeric molecules started to the active sites.

Reaction pathway for the modification of gum arabic
Reaction pathway for the modification of guar gum
Reaction pathway for the modification of xanthan gum
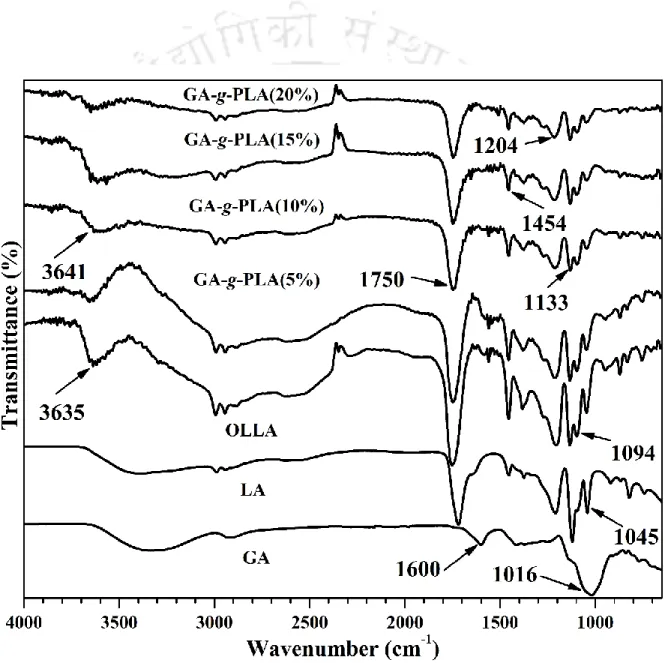
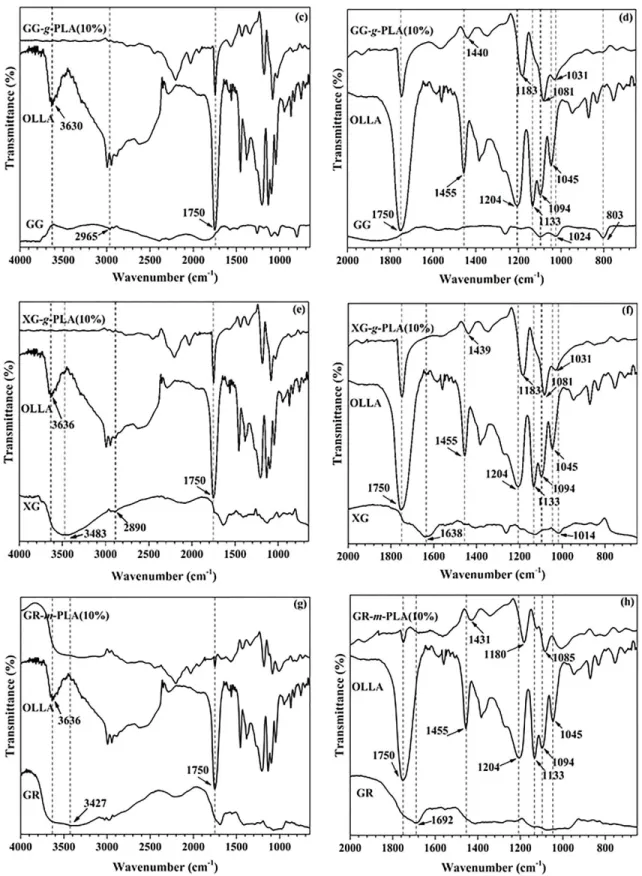
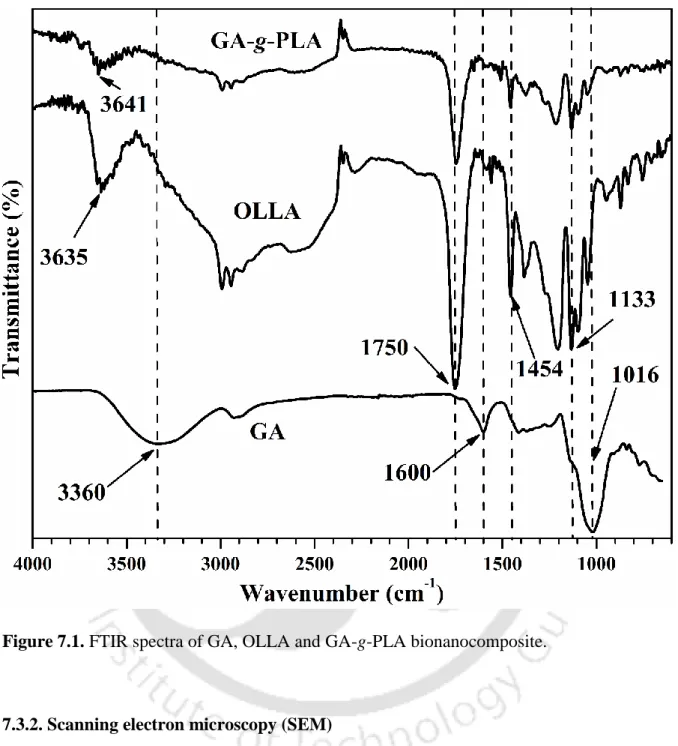
- Grafting by FTIR analysis
- Scanning electron microscopy analysis
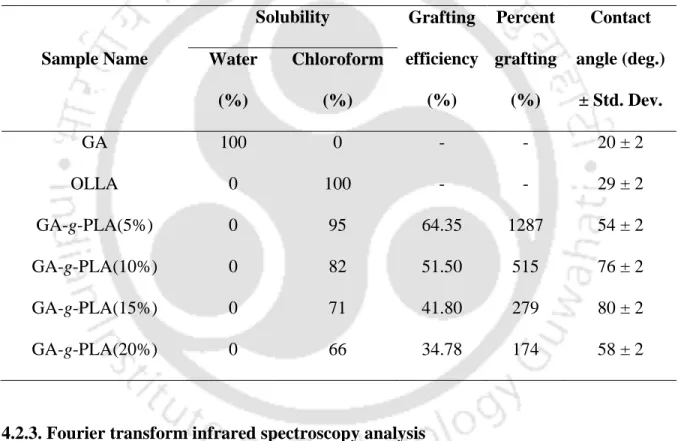
- Solubility
- Single lap shear test of modified gums for adhesive selection
- Fabrication of PLA/modified gums (5%) bionanocomposite films
- Scanning electron microscopy of PLA/modified gum films
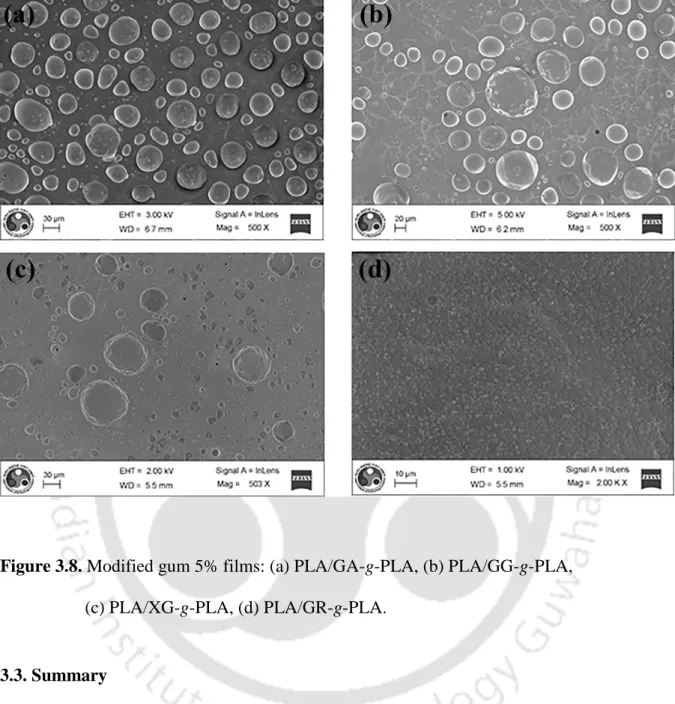
- Summary
The surface topography of pure rubbers and modified rubbers was successfully investigated by SEM analysis. The single lap shear test was performed to check the bond strength of the modified rubbers. On the other hand, the films were easily produced from PLA and modified rubbers together.
As shown in Figure 3.8, when the modified rubbers were introduced into PLA films at a ratio of 1:20, changes in the surface morphology of films were observed.
Synthesis and Characterization of Microwave Initiated Gum Arabic Grafted Poly L-Lactic Acid Bionanocomposite
Introduction
According to the literature, the graft copolymers can be defined as a long chain of a polymer backbone with branched polymer side chains (Rani et al., 2013; Rani et al., 2013). It should be noted that the external agent should not break the structural integrity of the preformed polymer chain (Mahto et al., 2014). Graft copolymerization can be mainly carried out by chemical initiation and radiation techniques (Setia et al., 2014).
Recently, great interest has increased in the synthesis of graft copolymers by microwave radiation due to the ease of operation, high reproducibility and less reaction time (Rani et al., 2013).
Results and Discussion
- Microwave initiated synthesis of gum arabic grafted poly l-lactic acid (GA-g-PLA) In the process LA molecules were grafted on GA backbone on exposure of the microwave
Whereas in microwave-initiated synthesis, cleavage is only triggered by microwave radiation to create free radical sites on polysaccharides. FTIR, NMR, XRD, GPC and contact angle analyzes were performed to determine the various properties of the synthesized bionanocomposite. Free radical sites were also formed on the polar groups of the GA polysaccharide framework and the LA monomer upon exposure to microwave radiation.
The reaction occurred between the free amino groups of protein in GA grafted with the reducing end groups of the polysaccharide or LA molecules (Guan et al., 2011; Guan et al., 2006).
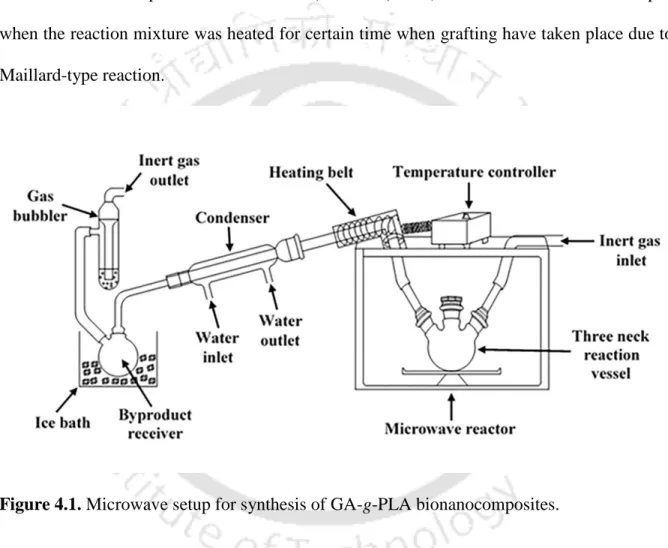
Synthesis of GA-g-PLA bionanocomposite
- Solubility and grafting calculations
- Fourier transform infrared spectroscopy analysis
- Nuclear magnetic resonance analysis
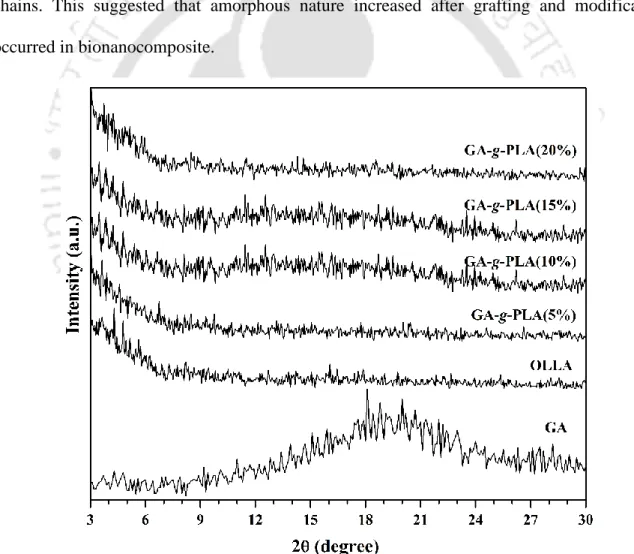
- X-ray diffraction analysis
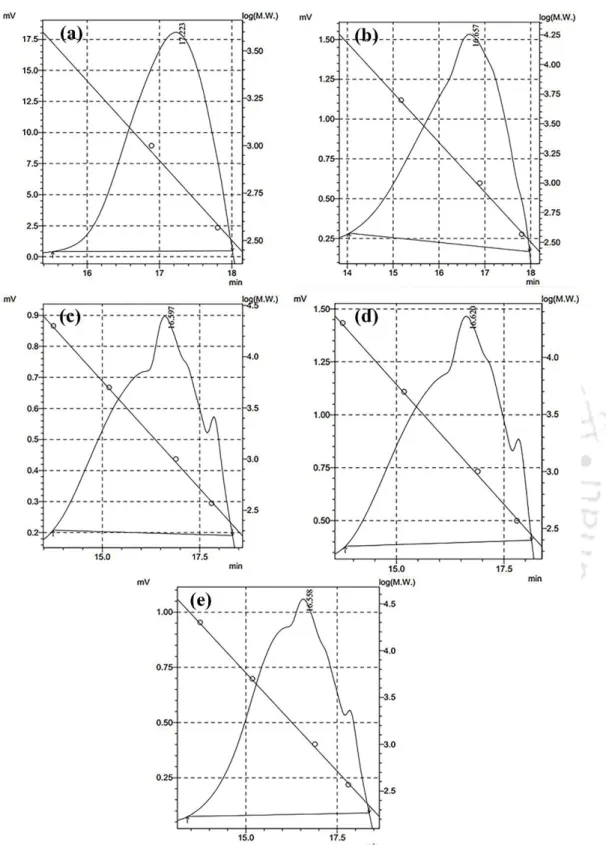
- Gel permeation chromatography analysis
- Differential scanning calorimetry analysis
- Thermogravimetric analysis
- Surface morphology
- Contact angle analysis
- Summary
The 1H-NMR of OLLA and GA-g-PLA was performed to study the grafting as shown in figure 4.3. Furthermore, no such hump or any peak was observed in the synthesized GA-g-PLA bionanocomposites. DSC thermograms (exo up) for (a) first heating scan and (b) second heating scan of GA and GA-g-PLA bionanocomposites.
The surface morphology of GA and GA-g-PLA was determined by SEM, as shown in Figure 4.8(a) and (b).
Microwave Initiated Gum Arabic Grafted Poly L-Lactic Acid Bionanocomposite: Adhesive Application
Introduction
Currently, most adhesives are prepared from precursors obtained from non-renewable sources such as crude oil. Most adhesives also use petroleum-based volatile organic compounds (VOCs) which evaporate from the adhesive compounds and cause environmental pollution. However, some of the bio-based polymers such as starch, soy proteins, lignin, vegetable oils and their derivatives are in the process of exploring its application to adhesive.
One of the important bio-based precursors that has been identified based on its characteristics is lactic acid.
Results and Discussion
- Substrate lamination protocol
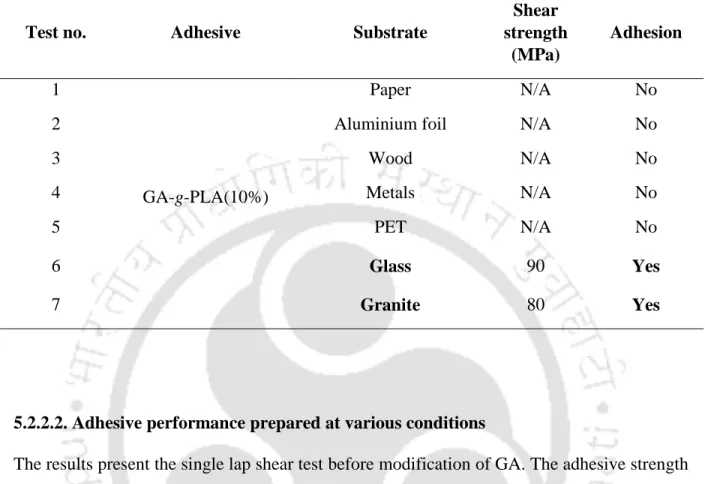
- Single lap shear test
- Adhesive performance at various substrates
- Adhesive performance prepared at various conditions
- Adhesive performance for glass substrates
The study was investigated to check the adhesive properties of GA-g-PLA by single lap shear test. The best shear strength was obtained for GA-g-PLA (15%) when applying a compressive load of 2000 N for 10 minutes. Substrate failure was observed for GA-g-PLA (15%) because the shear strength of the adhesive was greater than the tensile strength of the substrate.
The significant improvement in the shear strength of GA-g-PLA (10%) adhesive was observed with the increase of the compression load and compression time, as shown in Figures 5.1 and 6.2.
Summary
PLA/Functionalized-Gum Arabic Based Bionanocomposite Films for High Gas Barrier Applications
Introduction
Most food packaging materials are made from petroleum-based polymers due to their high availability, good gas barrier and mechanical properties. Therefore, increasing wrap has a great impact on extending the shelf life of products in food packaging applications (Duncan, 2011). Oxygen permeability depends on a number of interrelated factors such as hydrogen bonding properties, degree of branching or cross-linking between polymer chains, molecular weight, polarity and structural characteristics of polymer side chains, processing methodology, degree of crystallinity and synthesis method. A review of the literature reveals that none of the articles presented blends of PLA and GA-g-PLA for the preparation of packaging film.
One of the main objectives of the present investigation is to develop a new and eco-friendly packaging material with improved gas barrier properties.
Results and Discussion
- Chemical interaction analysis
- Crystal structure analysis
- Thermal analysis
- Surface morphology
- Mechanical properties
- Oxygen permeability
- Water vapour transmission rate
A 14% decrease in tensile strength was observed for PLA/GA-g-PLA(3%) films compared to pure PLA films. A 16% increase in elongation at break (εb) was observed for PLA/GA-g-PLA films relative to pure PLA films. A decrease in WVTR × e for PLA/GA-g-PLA (3%) films was observed to be about 7%, and with further addition of filler, which for PLA/GA-g-PLA (5%) was about 27 %.
In contrast, a drastic increase in WVTR × e of around 28% was observed for PLA/GA-g-PLA films (10%) relative to pure PLA films.
![Figure 6.1. FTIR spectra of (a) PLA, PLA/GA-g-PLA(3%), PLA/GA-g-PLA(5%), PLA/GA-g-PLA(10%) bionanocomposite films (b) carbonyl peak at 1755 cm -1 [PLA/GA-g-PLA(3%) film]](https://thumb-ap.123doks.com/thumbv2/azpdfnet/10515648.0/135.918.138.802.126.891/figure-ftir-spectra-pla-pla-bionanocomposite-films-carbonyl.webp)
Summary
A reduction in glass transition temperature indicates that these films are good for packaging applications. However, the magnitude of the reduction in the values of these properties was dependent on the percentage of GA-g-PLA. The main achievement of this work is the significant improvement in the oxygen transmission rate (OTR) of composite films.
A ~10-fold decrease in the OTR value can be considered a very significant improvement in the barrier property of the film, especially for its use as packaging films.
Thermal Degradation Behaviour of High Gas Barrier Poly (lactic acid)/modified Gum Arabic based Bionanocomposite Films
Introduction
- Kissinger method
- Augis and Bennett method
- Flynn-Wall-Ozawa method
- Kissinger-Akhira-Sunose method
Thermal degradation kinetics of the polymeric blends were effectively studied by TGA (Huang et al., 2015). Therefore, understanding the complex degradation mechanism is essential and can be understood through the kinetics (Corres et al., 2011). Kissinger method is applicable where the maximum degradation temperature of the process is known (Lizundia et al., 2015).
In this method, Ea can be estimated without knowledge of the specific reaction function (Achilias et al., 2011).
Results and Discussion
- Fourier transform infrared spectroscopy (FTIR)
- Scanning electron microscopy (SEM)
- Thermogravimetric analysis (TGA)
- Thermal analysis-Fourier transform infrared spectroscopy (TGA-FTIR)
- Degradation pathways
- Thermal degradation kinetics
Consequently, it can be concluded that, with increase in filler loading in PLA matrix, the degradation of the PLA/GA-g-PLA bionano-composite films also increased. The probable mechanism of degradation of PLA/GA-g-PLA bionanocomposite films is presented in figure 7.6. It was observed that the interaction of PLA and GA-g-PLA molecules formed the microcapsules in the PLA film.
In the range 0.1 ≤ ≤ ≤ 0.6, lower Ea values for PLA/GA-g-PLA bionanocomposite films were observed due to the presence of filler.
Summary
Conclusions and Future Scope
Conclusions
PLA was found to be compatible with GA-g-PLA filler, which was confirmed by one Tg of polymer blends obtained by mixing the two materials in different ratios. The presence of nanospheres is confirmed by TEM images, which clearly conclude that mixing GA-g-PLA with PLA significantly changed the morphological characteristics of the film. The most important achievement of this work is the significant reduction of the oxygen transfer rate (OTR) of the composite films.
The increase in activation energy with increase in conversion indicates complex reaction in PLA/GA-g-PLA bionanocomposite films.
Scope for Future Work
Kinetics of thermal degradation of polymers: complementary use of isoconversion and model fitting methods, Journal of Thermal Analysis and Calorimetry. Thermal and thermooxidative degradation of poly(hydroxyether of bisphenol-A) studied by TGA/FTIR and TGA/MS, Journal of Analytical and Applied Pyrolysis. Effect of incorporation of natural and synthetic antioxidants on the gas permeation properties of poly(lactic acid) films, Journal of Food Engineering.
Thermal degradation of poly(vinyl chloride)/poly(ethylene oxide) blends: thermogravimetric analysis, Polymer degradation and stability.