초기 자궁육종 환자의 원격 전이를 예측하기 위한 모델. 자궁육종은 자궁의 중배엽에서 기원하는 매우 드문 악성종양으로 수술 전 예측이 가장 어려운 질환 중 하나이다. 이는 저단계 자궁육종 환자에서도 재발률이 높고 전체 생존율이 낮은 이유이기도 하다.
같은 1기 자궁육종이라도 일부 환자에서는 재발하거나 질병이 빠르게 진행되는 경우가 있는데, 현 병기의 방법으로는 해당 환자군을 걸러낼 수 없다. 따라서 본 연구에서는 자궁육종 환자의 진단 시 생존율 예측 모델과 조기 원격 전이 재발 예측 모델을 개발하여 수술 후 추가 치료가 필요한 군을 선정하여 향후 예후에 기여하는 것을 목적으로 한다. 자궁육종 환자가 그렇습니다. 자궁육종에는 평활근육종, 저등급 자궁내막 간질 육종, 고등급 자궁내막 간질 육종, 악성 혼합형 뮬러관 종양이 포함됩니다.
본 논문에서 제시된 예측 모델은 다양한 유형의 자궁육종 환자에 대한 기존 병기결정 방법보다 5년 생존율을 더 잘 예측할 수 있습니다. 또한, 조기 자궁육종 환자에서 기존 병기결정 방법으로는 예측하기 어려운 원격 전이의 재발을 예측할 수 있습니다. 따라서, 조기 자궁육종 환자 중 추가 치료가 필요한 자궁육종 환자 풀을 구축함으로써 자궁육종 치료 및 예후 개선에 도움이 될 것으로 사료된다.
Introduction
From January 1, 2007 to December 31, 2017, patients who underwent at least one operation for uterine sarcoma (leiomyosarcoma, low-grade endometrial stromal sarcoma, high-grade endometrial stromal sarcoma, malignant mixed Mullerian tumor) at our institution were identified. In the case of unexpected uterine sarcoma after surgery, metastases were evaluated by imaging within one month of the final pathology report. Another endpoint was to develop nomograms to predict 5-year overall survival and distant metastases.
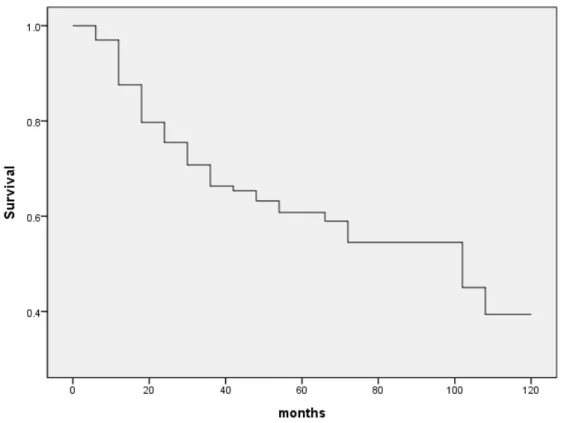
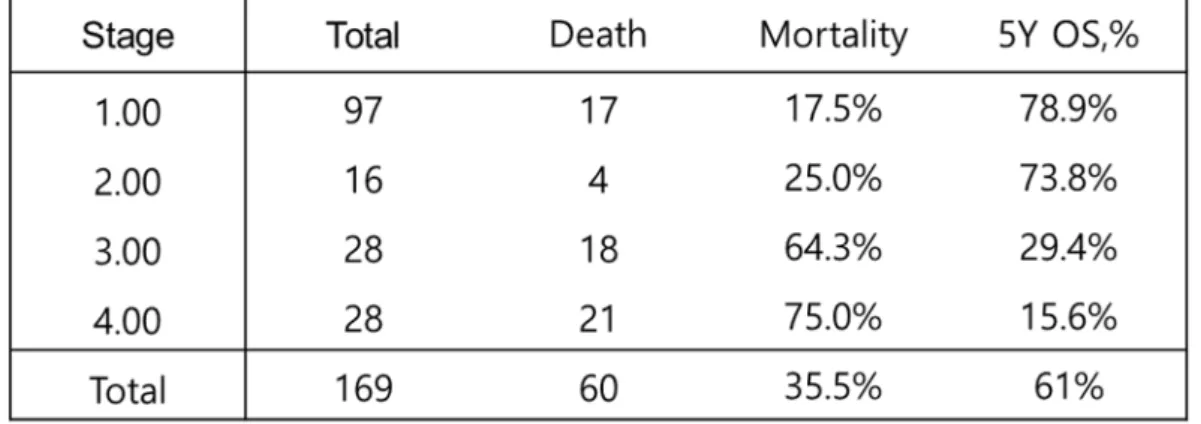
Cox proportional risk regression analysis and multivariable analysis were used to distinguish factors associated with overall survival and recurrence. Five-year overall survival by stage is presented in Table 3 and the Kaplan-Meier plot is presented in Figure 2. Overall Survival of Patients with Uterine Sarcoma This graph shows the median overall survival of the study population.
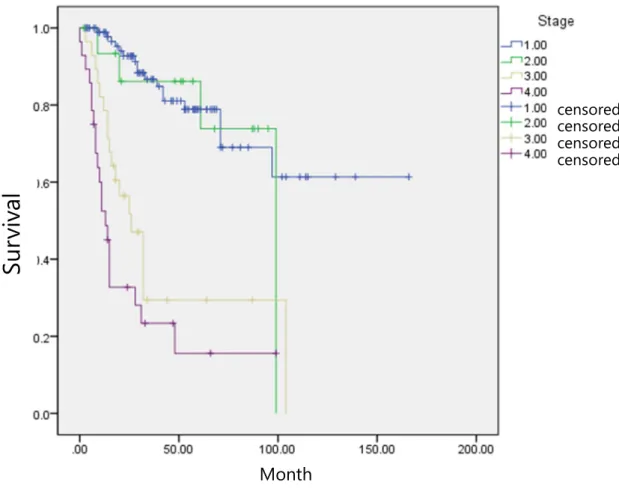
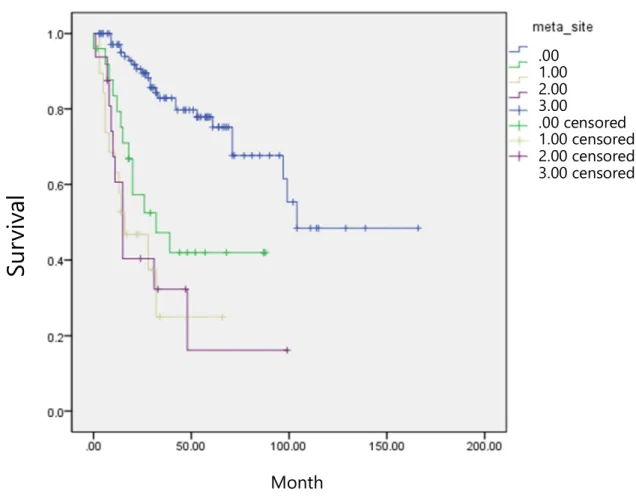
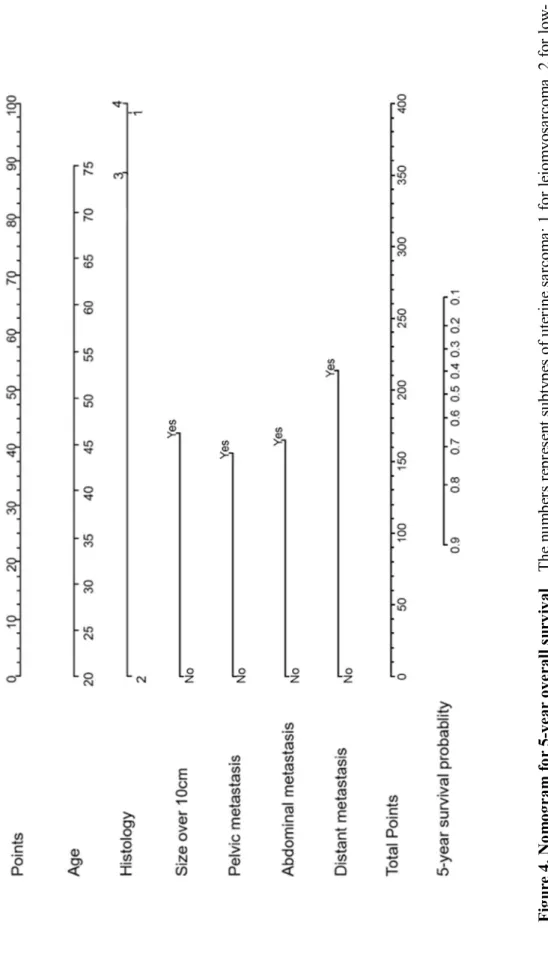
Mortality rate and 5-year overall survival by stage Results show higher mortality rate and lower 5Y OS in more advanced stage. Meier chart for uterine sarcoma by stage Survival is significantly different between early stage (1,2) and advanced stage (3,4). censored censored censored censored. Nomogram for distant recurrence Numbers represent subtypes of uterine sarcomas; 1 for leiomyosarcoma, 2 for low-grade endometrial stromal sarcoma, 3 for high-grade endometrial stromal sarcoma, and 4 for mixed Mullerian malignancy.
One of the most recent drugs for sarcoma, Olaratumab, shows a significant improvement of 11.8 months in median overall survival.(7) Based on several clinical trials, it is becoming increasingly important to distinguish the right patient group for adjuvant treatment. Nomogram is a useful tool for various types of cancer to predict specific event such as nodal metastasis, overall survival or treatment response based on the clinical information that the patients had at the time of diagnosis.(8-10) Because current staging system for uterine sarcoma is poor performance to predict prognosis, a new nomogram to predict survival for uterine leiomyosarcoma has been developed.(11, 12) Also, some nomograms to predict metastasis and survival in retroperitoneal sarcoma or soft tissue sarcoma have been reported.(13, 14) . Moreover, nomogram comparing subtypes' prognosis has not yet been reported, although it is well known that low-grade endometrial stromal sarcoma shows relatively better prognosis, but leiomyosarcoma, high-grade endometrial stromal sarcoma and malignant mixed mullerian tumor are very aggressive.(5) Also, are some types of uterine sarcoma extremely rare, a useful tool covering those rare types is strongly needed to understand the patients.
However, these nomograms cover 4 histology types of uterine sarcoma and demonstrate relative risk of death and distant metastasis by histology. These nomograms may represent a new patient group that requires additional treatment, even in the early stage of uterine sarcoma. With modification through further study and external validation, these nomograms may be an option to improve the prognosis of uterine sarcoma.
Uterine sarcoma is difficult to diagnose before surgery and is sometimes assumed to be a benign mass. And uterine sarcoma shows a high recurrence rate and a low overall survival rate even at an early stage. There is a need for new uterine sarcoma nomograms to predict overall survival and distant metastases at the time of diagnosis to differentiate the group of patients who require further adjuvant treatment, which should improve prognosis.
Material and Methods
Statistical methods
The first predicted end point for our study was 5-year overall survival, which is the time interval from the date of last visit or date of death from the first diagnosis.
Results
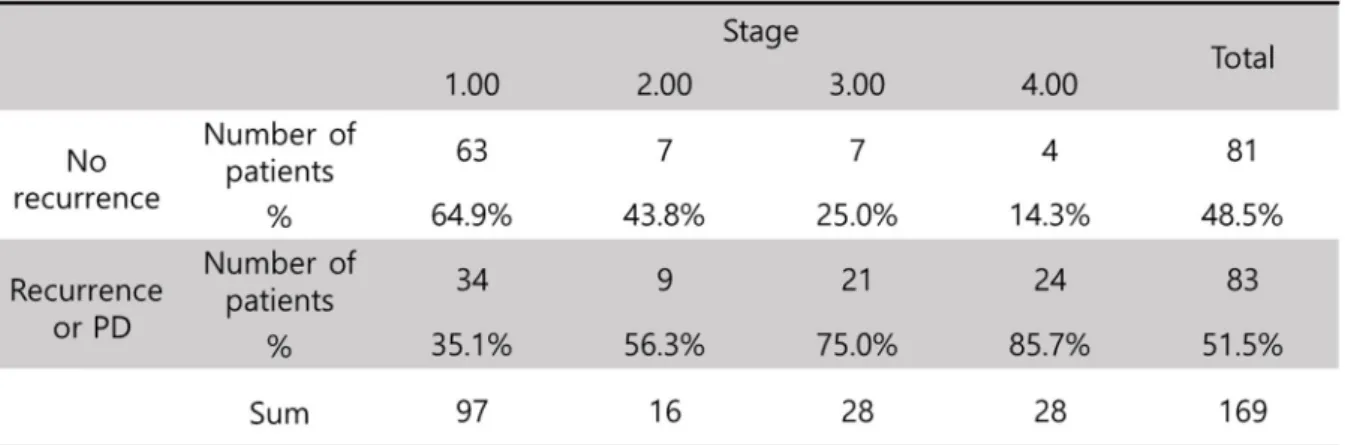
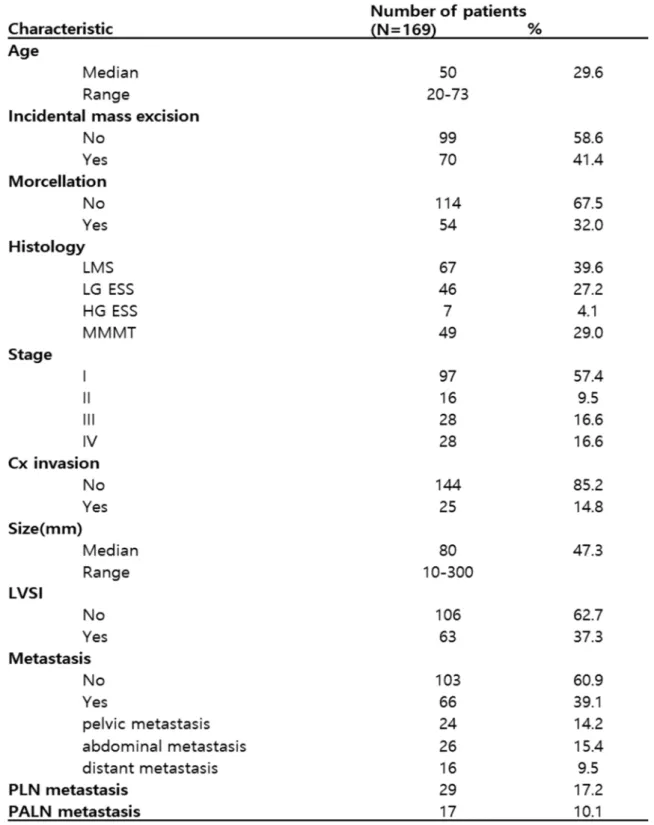
Characteristics of the study population LMS, leiomyosarcoma; LG-ESS, low-grade endometrial stromal sarcoma; HG-ESS, high-grade endometrial stromal sarcoma; MMMT, Malignant mixed Mullerian tumor. Frequency of disease recurrence or progression by stage The rate of disease recurrence or progression is higher in more advanced stages. Kaplan-Meier plot for uterine sarcoma by stage. Survival is significantly different between early stage (1,2) and advanced stage (3,4).
Blue, confined to the uterus; green, pelvic metastasis including pelvic lymph node metastasis; yellow, abdominal metastasis including para-aortic lymph node metastasis; purple, distant metastasis.
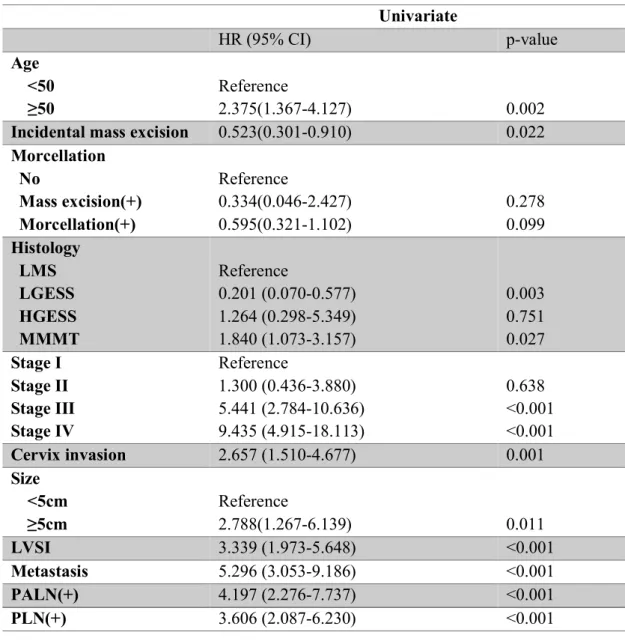
Nomogram to predict 5-year survival
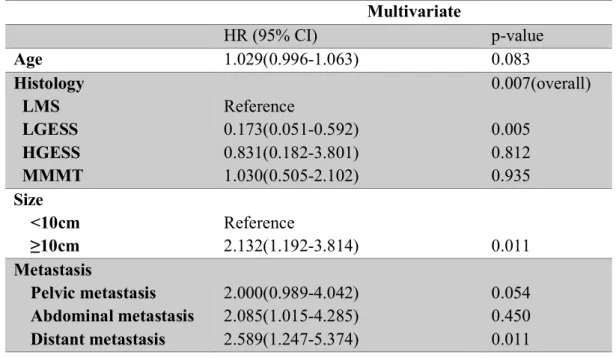
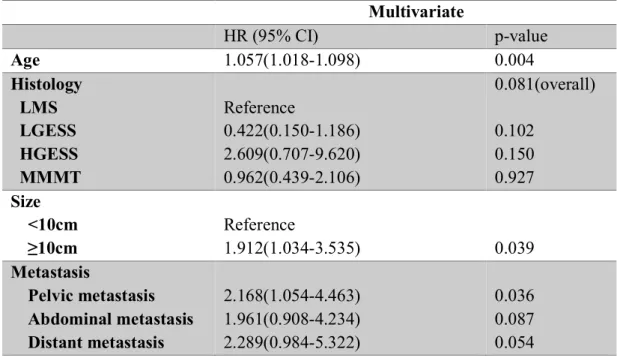
Factors associated with survival and univariate analysis CI, confidence interval; HR, hazard ratio; LMS, leiomyosarcoma; LG-ESS, low-grade endometrial stromal sarcoma; HG-ESS, high-grade endometrial stromal sarcoma; MMMT, malignant mixed Mullerian tumor; LVSI, lymphatic space invasion; PALN, paraaortic lymph node; PLN, pelvic lymph node. Factors associated with survival and multivariate analysis CI, confidence interval; HR, hazard ratio; LMS, leiomyosarcoma; LG-ESS, low-grade endometrial stromal sarcoma; HG-ESS, high-grade endometrial stromal sarcoma; MMMT, Malignant mixed Mullerian tumor. Nomogram for 5-year overall survival Numbers represent subtypes of uterine sarcomas; 1 for leiomyosarcoma, 2 for low-grade endometrial stromal sarcoma, 3 for high-grade endometrial stromal sarcoma, and 4 for mixed Mullerian malignancy.
Calibration curve for nomogram to predict overall 5-year survival Actual 5-year survival was estimated using the Kaplan-Meier method. The dotted line is the ideal line for the nomogram and the solid line is the result of the nomogram's score and prediction.
Nomogram to predict distant metastasis as a recurrence
Recurrence-associated factors and multivariate analysis CI, confidence interval; HR, risk ratio; LMS, leiomyosarcoma; LG-ESS, low-grade endometrial stromal sarcoma; HG-ESS, high grade endometrial stromal sarcoma; MMMT, malignant mixed Mullerian tumor. Calibration curve for nomogram to predict distance recurrence. The actual recurrence distance and the estimated recurrence by the nomogram were compared.
Discussion
Cervical invasion, lymphovascular invasion, lymph node metastasis had been proven as a risk factor through various studies (15, 16) but were not significantly associated with prognosis in this study. 3, 17) However, when analyzing the associated variables, morcellation was mostly performed on the mass expected to be benign uterine mass. Almost of these cases were confirmed to stage 1 or 2 in the final pathological report, I discard morcellation as a variable for fear of bias.
However, the nomogram is developed to overcome the brevity of the current level system and the levels are a sort of categorization of various factors that overlap with the variables already included in the nomogram.
Conclusion
Combined use of T2-weighted and diffusion-weighted 3-T MR imaging to differentiate uterine sarcomas from benign leiomyomas. Olaratumab plus doxorubicin versus doxorubicin alone for the treatment of soft tissue sarcoma: an open-label phase 1b trial and a randomized phase 2 trial. Validation of a nomogram for disease-free survival in colon cancer in a UK population: a prospective cohort study.
Van Zee KJ, Manasseh DM, Bevilacqua JL, Boolbol SK, Fey JV, Tan LK, et al. A nomogram for predicting the probability of additional nodal metastases in breast cancer patients with a positive sentinel node biopsy. Importance of tumor size in soft tissue sarcomas: proposal for a nomogram based on a scoring system for staging soft tissue sarcomas in the postoperative setting.
It means that the current system cannot distinguish between patients with a poorer prognosis and risk of recurrence. Of the 169 patients, patients experienced recurrence or disease progression and 60 (35.5%) patients died of disease.
Abstract