AnimalReproductionSciencexxx (2011) xxx–xxx
ContentslistsavailableatSciVerseScienceDirect
Animal
Reproduction
Science
journalhomepage:www.elsevier.com/locate/anireprosci
Association
study
and
expression
analysis
of
porcine
ESR1
as
a
candidate
gene
for
boar
fertility
and
sperm
quality
Asep
Gunawan
a,
Kanokwan
Kaewmala
a,
Muhammad
Jasim
Uddin
a,
Mehmet
Ulas
Cinar
a,
Dawit
Tesfaye
a,
Chirawath
Phatsara
a,b,
Ernst
Tholen
a,
Christian
Looft
a,
Karl
Schellander
a,∗aInstituteofAnimalScience,AnimalBreedingandHusbandryGroup,UniversityofBonn,53115Bonn,Germany bDepartmentofAnimalandAquaticSciences,FacultyofAgriculture,ChiangMaiUniversity,50200ChiangMai,Thailand
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received13May2011
Receivedinrevisedform12August2011 Accepted24August2011
Available online xxx
Keywords: mRNA
Immunofluorescence Spermatogenesis Epididymis Spermatozoa
a
b
s
t
r
a
c
t
MalefertilityisimpairedthroughthelackofESR1(EstrogenReceptor1)butlittleisknown
abouttheESR1rolesinboarspermatogenesisandfertility.Therefore,thisresearchwas
aimedatinvestigatingtheassociationwithspermqualityandboarfertilitytraitsinatotal
of300boarsbothfrompurebredPietrainandPietrain×Hampshirecrosses.ASNPincoding
regionofESR1g.672C>Tinexon1wasassociatedwithspermmotility(P<0.05)andplasma dropletrate(P<0.01)whilethepolymorphisminnon-codingregionofESR1g.35756T>Cin
inton1wasassociatedwithnon-returnrate(P<0.05).Furthermore,toanalysethemRNA
andproteinexpressionofESR1inboarreproductivetissues,atotalofsixboarsweredivided
intotwogroups[GroupI(G-I)andGroupII(G-II)],whereG-Ihadrelativelybettersperm
quality.ESR1expressionwashigherintissuescollectedfromG-Iboarsthanthoseof
col-lectedfromG-IIboars,andthedifferenceinmRNAexpressionwassignificant(P<0.01)
inheadofepididymis.TheESR1proteinexpressionresultsfromwesternblotcoincided
withtheresultsofqRT-PCR.TheESR1proteinlocalizationobservedastrongstaininginthe
cytoplasmofSertolicellinthetestis,intheepithelialcellsinheadandtailofepididymis,
insmoothmuscleintailofepididymis,andinthepostacrosomalregionandtailofthe
spermatozoa.TheseresultswillimprovetheunderstandingofthefunctionsoftheESR1in
spermatogenesiswithinthereproductivetractandwillshedlightonESR1asacandidate
intheselectionofboarwithgoodspermqualityandfertility.
© 2011 Elsevier B.V. All rights reserved.
1. Introduction
Despitebeingthe‘female’hormone,estrogenispresent inlowconcentrationsinmalebloodandinextraordinarily highconcentrationinsemen(GanjamandAmann,1976).
∗ Correspondingauthor.Tel.:+49228732240;fax:+49228732284. E-mailaddresses:[email protected](A.Gunawan),
[email protected](K.Kaewmala),[email protected] (M.J.Uddin),[email protected](M.U.Cinar),[email protected] (D.Tesfaye),[email protected](C.Phatsara),[email protected] (E.Tholen),[email protected](C.Looft),[email protected] (K.Schellander).
Thehormone estrogenis mediatedthrough thenuclear estrogen receptor which functionsas ligand-dependant transcription factor. The increasing interest in the role ofestrogeninthemalereproductivetractismainlydue to the demonstration that male fertility is impaired in mice lacking estrogen receptor 1 (ESR1) (Eddy et al., 1996). The evidence for ESR1 in different parts of the male reproductive tract suggests a possible physiologi-cal role in spermatogenesis and spermmaturation. The
ESR1knockoutmicehaveprovidedevidencefora signif-icantandcrucialroleofestrogensinmaintainingnormal spermatogenesis(Eddyetal.,1996).ESR1is involvedin the reabsorption of luminal fluid during the transit of
spermatozoa fromthetestistotheheadoftheepididymis whichisimportantfortheirsurvivalandmaturation dur-ing epididymal storage (Couse and Korach, 1999). The absenceofESR1leadstoreducedepididymalsperm con-tent,reducedspermmotilityandfertilizingability(Eddy etal.,1996).Severalstudieshaveshownasignificant asso-ciationofESR1withsemenqualityorfertilityinhumans (Suzukietal.,2002;Guarduccietal.,2006;Lazarosetal., 2010; Safarinejad et al., 2010). Guarducci et al. (2006) reporteda significantassociationofESR1polymorphism with lower sperm count in men. Suzuki et al. (2002) reportedtwosilentpolymorphismsinESR1being associ-atedwithazoospermiaorsevereoligozoospermiainmen. Toourknowledge,onlyonestudywasdevoted to anal-ysetheassociationbetweenESR1polymorphismwithlitter sizeinsowsbutthisfailedtodetectanystatistically sig-nificantassociation(Munozetal.,2007).TheESR1mRNA wasreportedtobehighly expressedinthe epididymis, testis,pituitary,uterus,kidneyandadrenalglandinrats (Kuiper et al., 1996).ESR1 was localized in head, body andtailofepididymisinthebonnetmonkey(Shayuetal., 2004),cat(Nieetal.,2002)andhorse(Hejmejetal.,2005; Parlevlietetal.,2006).Inpigs,ESR1islocatedon SSC1p24-25,whichwasevidencedastheQTLregionfortotalsperm per ejaculate and close to the QTL for sperm motility (Xingetal.,2008).Therefore,ESR1couldbeafunctionalas wellasapositionalcandidategeneformalereproduction in pigs.But nostudyhas yetbeen devotedto unravel-lingitsassociationwithboarspermqualityandfertility traits.
In spermatogenesis, beside the reproductive tissues testisandepididymis, someotheraccessoryglands and tissuesarealsoinvolved.Spermatogenesisisthecomplex processbywhichimmaturegermcellsundergodivision, differentiationand meiosistogiverisetohaploid elon-gated spermatids (O’Donnell et al., 2001). When germ celldevelopmentiscomplete,thematurespermatidsare releasedfromtheSertolicellsintothetubulelumenand proceedthroughtheexcurrentductsystem,knownasthe retetestis,untiltheyentertheepididymisviathe effer-ent ducts. During passage through the epididymis, the spermatids undergoa series of biochemical changes to becomethemotileandmaturespermatozoacapableof fer-tilization(O’Donnelletal.,2001).Failureinanyofthese events leads to disturbances of male fertility. The role ofESR1inboarspermatogenesiswithinthereproductive tractis poorlyunderstood.For thebetterunderstanding ofESR1rolesinspermatogenesisinpigs, theexpression of ESR1 at different parts of reproductive tract, includ-ing non-reproductive tissues, are important. Therefore, theaims of this study were toinvestigate the associa-tionofESR1polymorphismwithspermqualityandboar fertility traits as well as to highlight the roles of ESR1
in boar spermatogenesis within the reproductive tract by mRNA and protein expression and immunoreactive
ESR1localization.Thisworkmightbehelpfultoget fur-ther insight into the roles of ESR1 in spermatogenesis intheboar.Theassociationof ESR1polymorphism with boarsperm qualityand fertility willsupportthe candi-dacy of this gene to be used in selection of breeding boars.
2. Materialsandmethods
2.1. Phenotypes
Samplesand phenotypesfrom200purebredPietrain (PI) and 100 Pietrain×Hampshire crossbred (PI×HA) boars were used for association analysis in this study. TheseanimalswereusedforAIincommercialpigherds inNorth-WesternGermany.Detailsofthepopulationsand phenotypesweredescribedpreviouslybyWimmersetal. (2005), Lin et al(2006) and Kaewmala etal. (2011). In brief,sperm samples frommore than39,000 ejaculates wererepeatedlycollectedfromtheseboars.Whole ejacu-lateswereobtainedfrompurebredPietrainandcrossbred Pietrain×Hampshireboarsagebetweentwoandfiveyears with an average age of 3.5 years. Sperm quality traits included spermconcentration (SCON[×108ml]), semen volumeperejaculate(VOL[ml]),spermmotility(MOT[%]), plasmadroplets rate(PDR[%])and abnormal spermato-zoarate(ASR[%])andwereobtainedfromeachejaculate withlightmicroscopyaccordingtotheguidelinesofthe WorldHealth Organization.Semenwascollectedbythe vinylglovehandmethodtwiceperweek.Foreachboar, therepeatedmeasurementsofspermqualitytraitswere available.Fertilitydata(non-returnratedata[NRR]at42 daysafterinsemination[%]andnumberofpigletbornalive [NBA]perlitter)ofeachboarwereavailableasthe devia-tionfromthepopulationmeanswithinsowbreed,parityof sow,farmandseasonclassesasdescribedearlierbyLinetal (2006)andKaewmalaetal.(2011).Thegeneraldescription ofspermqualitytraitsandboarfertilitytraitsareshownin Table1.
2.2. Polymorphismstudy
Table1
Means,standarddeviation(S.D.),samplesize,rangesoftraitsinPietrainandPietrain×Hampshire.
Population Traits Samplesize Mean S.D. Minimum Maximum
Pietrain(PI) SCON(108/ml) 29,161 3.03 0.94 1 6
VOL(ml) 30,772 237.03 57.32 25 522
MOT(%) 30,118 84.72 4.37 65 95
PDR(%) 30,239 5.41 3.33 0 15
ASR(%) 30,528 6.53 4.18 0 20
NRR42(%)a 200 0.28 7.06 −24.07 17.68
NBA(perlitter)a 200 0.02 0.57 −1.69 1.37
Pietrain×Hampshire(PIHA) SCON(108/ml) 9123 2.95 0.97 1 6
VOL(ml) 9673 297.50 81.62 56 546
MOT(%) 9395 85.46 4.03 70 95
PDR(%) 9409 5.76 3.14 0 15
ASR(%) 9543 4.95 4.00 0 20
NRR42(%)a 100 0.97 4.18 −12.23 13.79
NBA(perlitter)a 100 0.05 0.52 −2.97 0.83
PIandPIHA SCON(108/ml) 38,284 3.00 0.95 1 6
VOL(ml) 40,445 256.87 75.54 25 546
MOT(%) 39,513 85.01 4.37 65 95
PDR(%) 39,648 5.51 3.28 0 15
ASR(%) 40,071 5.99 4.26 0 20
NRR42(%)a 300 0.41 6.23 −24.07 17.67
NBA(perlitter)a 300 0.01 0.55 −2.97 1.37
aFertility(NRR42,NBA)correctedwithfactors:parity,farm,seasonandbreed.
AluIforg.672C>Tandg.35756T>C,respectively(BioLabs). The digestionwascarried out in10l ofreaction mix-tureofeachsampleandincubatedovernightat37◦Cfor g.35756T>Cand65◦Cforg.672C>T.DetectionofRFLPsof 300boarswascarriedoutbyelectrophoresisin3%(w/v) agarosegels.
2.3. Statisticalanalysisforspermqualitytraits
TheassociationofESR1withboarspermqualitytraits wasanalysedbyvarianceanalysis(PROCMIXED)usingthe SASsoftwarepackage(ver.9.2;SASInstituteInc.,Cary,USA) asdescribedearlierbyKaewmalaetal.(2011).
yijklm=+breedi+seasonj+genotypek+agel
+ejaculationm+εijklm [Model 1]
whereyijklmarethespermqualitytraits(SCON,VOL,MOT, PDR,ASR);istheoverallpopulationmean;breediisthe fixedeffectof thei-th breed(i=PI;PIHA, PIand PIHA); seasonjisthefixedeffectofthej-thseason(j=1through8; fourseasonsperyear,intotaleightseasonswithin2years fromJanuary2000toDecember2001);genotypekisthe fixedeffectofthek-thgenotype(k=1,2and3);agelisthe effectofboarage(covariable);ejaculationmisthe perma-nentenvironmentaleffectofthem-thboar(random)and εijklmistheresidualerror.
2.4. Statisticalanalysisforfertilitytraits
TheassociationanalysisbetweenESR1andthefertility traitswascarriedoutusingthefollowinggeneralizedlinear model(PROCGLM)inSAS(Kaewmalaetal.,2011). yijkl=+breedi+genotypej+yeark+εijkl [Model 2] whereyijk istheboarfertilitytraits(NRRandNBA);is theoverallpopulationmean;breediisthefixedeffectof
thei-thbreed(i=PI,PIHA,PIandPIHA);genotypejisthe fixedeffectofthej-thgenotype(j=1,2and3);yearkisthe fixedeffectofthek-thboaryearofbirth(k=1through3: boarbornbefore1996,in1996–97andin1998–99),εijkis theresidualerror.
Achi-square(2)testwasconductedtotestthe popula-tionsforHardy–Weinbergequilibrium.Leastsquaremean valuesfortheESR1genotypeswerecomparedbyt-testand
P-valueswereadjustedbytheTukey–Kramercorrection.
2.5. SelectionofanimalsformRNAandprotein
expression
The reproductive(testis, head,body and tail of epi-didymis, vas deferens, bulbourethral gland, vesicular glandsandprostategland),non-reproductive(brain, mus-cleandliver)tissuesfrom6breedingboarswithdivergent phenotypeswere collectedfromtheAI station(SuisAG, Sempach,Switzerland)formRNAandproteinstudy.For differentialexpression study between reproductiveand non-reproductivetissuesbysemi-quantitativePCRstudy, mRNAfromall6boarswaspooledtogetheraccordingto thetissues.Ontheotherhand,thedifferentialmRNAand proteinexpressionstudyindifferentreproductivetissues fromtwodivergentgroupsofanimalswasperformedby
Table2
Means,standarddeviation(S.D.),numberofboarsandrangesoftraits selectedformRNAandproteinexpressionstudy.
Traits Selected
animals (n=6)
G-I (n=3)
G-II (n=3)
Mean S.D. Mean S.D. Mean S.D.
SCON(106/ml) 262.32 87.97 335.94 50.78 188.70 22.54
SVOL(ml) 215.24 34.93 185.07 16.33 245.40 7.42
[image:3.561.38.507.73.281.2] [image:3.561.277.505.613.694.2]semi-quantitativePCR,qRT-PCRandwesternblot, respec-tively.Forthesepurposes,the6boarsweredividedinto twogroups:groupI(G-I)andgroupII(G-II),eachgroup containing3boarsonthebasisofSCON,SVOLandSMOT (Table2).TheSCON (averagespermconcentration) was highly negatively (r=−0.8)correlated withSVOL (aver-agesemenvolume),whereasSCONwashighlypositively (r=0.7)correlated withSMOT(average spermmotility). Moreover,SVOLwashighlynegatively(r=−0.8)correlated withSMOT.For theanimalsofG-ItheSCONwashigher thanthemean(262.32×106ml),SMOTwashigherthan themean(76.59%)and SVOLwaslower thanthemean (215.24ml/ejaculation)ofrespectiveparameters.Forthe animalsofG-IItheseparameterswereviceversa.The sig-nificantdifferencebetweenthetwogroupswascalculated usingproct-testinSAS.ThereweredifferencesforSCON (P<0.05) and for SVOL (P<0.01) between G-Iand G-II, whereasfortheSMOTthedifferencewasnotsignificant (P=0.12).
2.6. Semi-quantitativePCR
TotalRNAwasisolatedusingTRIreagent(Sigma)from different reproductive and non-reproductive tissues of breedingboarsmentioned above(Section2.5).RNAwas purified using RNeasy Mini Kit (Qiagen) according to the manufacturer’s instructions. Total RNA was treated using on-column RNase-Free DNase set (Promega) and quantifiedsphectrophotometrically(NanoDrop,ND8000). Furthermore,RNAintegritywascheckedby2%agarosegel electrophoresis.First-strandcDNAweresynthesizedfrom individualRNAusingSuperscriptIIenzyme(Invitrogen).
cDNA amplificationwas performedby usingspecific forward and reverse primers (forward: 5′ -agggagagg-agtttgtgtg-3′ and reverse: 5′-tctccagcagcaggtcatag-3′) derivedfromporcineESR1sequence(GenBankaccession AF035775).Amplificationwasperformedwithaninitial heatingat95◦Cfor5minfollowedby35cyclesof95◦Cfor 45s,annealingtemperatureat60◦Cfor1minand72◦Cfor 1min,onthePCRThermalCycler(BioRad).PCRproducts wereelectrophoresedona1.5%agarosegelandvisualized upon staining with ethidium bromide. Amplification of Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) servedashousekeepinggene.
2.7. QuantitativeReal-TimePCR(qRT-PCR)
ForqRT-PCR,totalRNAwasisolatedusingTRIreagent (Sigma) from different reproductive tissues of the two divergentgroupsofanimals(G-IandG-II)aspreviously described (Section 2.5). Total RNA isolation and cDNA synthesiswasdescribedintheprevioussection(Section 2.6).Thesameprimerpairusedinsemi-quantitativePCR was also used in qRT-PCR. Nine-fold serial dilution of plasmidDNA wasprepared and usedas a template for the generation of the standard curve. In each run, the 96-wellmicrotiterplatecontainedeachcDNAsample, plas-midstandardsforthestandardcurvesandano-template control.Toensurerepeatabilityoftheexperiments,each sample wasrun in three replications. qRT-PCR was set upusing2lfirst-strandcDNAtemplate,7.6ldeionized
H2O,0.2Mofupstream anddownstream primers and 10l 1× Power SYBR Green Imaster mixwith ROXas reference dye (BioRad). The thermal cycling conditions were3minat94◦Cfollowedby40cyclesof20sat94◦C and 1min at 60◦C. Experiments were performed using theABIprism®7000(AppliedBiosystems)qRT-PCRsystem. An amplification-based threshold and adaptive baseline were selected as algorithms. The housekeeping gene
GAPDH(forward:5′-acccagaagactgtggatgg-3′andreverse:
5′-acgcctgcttcaccaccttc-3′)derivedfromporcinesequence (GenBankaccessionNo.AF017079)wasusedforthedata normalization.Finalresultswerereportedastherelative abundancelevelafternormalizingwithmRNAexpression levelofthehousekeepinggene.DifferencesinESR1mRNA expressionwereanalysedwiththesimplet-testinSAS soft-ware(SASInstituteInc.,ver.9.2).ValuesofP<0.05were consideredtoindicatestatisticallysignificantdifferences.
2.8. Westernblotting
Theproteinwasextractedfromdifferentreproductive tissues(testis,head,bodyandtailofepididymis)fromthe twodivergentgroupsofbreedingboarsasusedinqRT-PCR. However,forwesternblotstudy,proteinsfromthethree G-Iboarswerepooledtogetherandproteinsfromthethree G-IIboarswerepooledtogetheraccordingtothetissues. Theproteinextractedfromtissueswasseparatedby SDS-PAGE (gradient4–18%). Subsequentlytheproteinswere transferred onto anitrocellulose membrane (Amersham Biosciences).Afterblockinginblockingbuffer(20mMTris pH7.5,150mMNaCl,0.05%Tween-20and1% Polyvinylpy-rolidone)atroomtemperaturefor1h,themembranewas incubatedwiththeanti-ESR1antibodypurifiedfromrabbit polyclonalantibody(Cat.nr.543;SantaCruz)inthe block-ingmedium(diluted1:500)overnightat4◦C.Non-specific bindingofantibodywaswashedoffwithsixchangesof 0.1% PBST(10minto time).Thehorseradishperoxidase conjugatedgoatanti-rabbitIgGsecondaryantibody(Cat.nr. Sc2004;SantaCruz)wasusedasthesecondaryantibody (diluted 1:5000). The membrane was incubated for 1h atroomtemperaturewithsecondaryantibody,followed by washing with six changes of 0.1% PBST (10min to time).ThechemiluminescewasdetectedbyusingtheECL plus westernblotting detectionsystem(Amersham Bio-sciences)andwasvisualizedbyusingKodakBioMaxXAR film(Kodak).GAPDHwasusedasaloadingcontrolandfor normalization.Themembranewasstrippedbyincubation in2%SDS,100mMTris–HCl,0.1%-mercaptoethanolfor 30minat60◦Candre-probedwithGAPDHantibody(Cat.nr. Sc20357;SantaCruz).
2.9. Proteinlocalizationbyimmunofluorescence
Table3
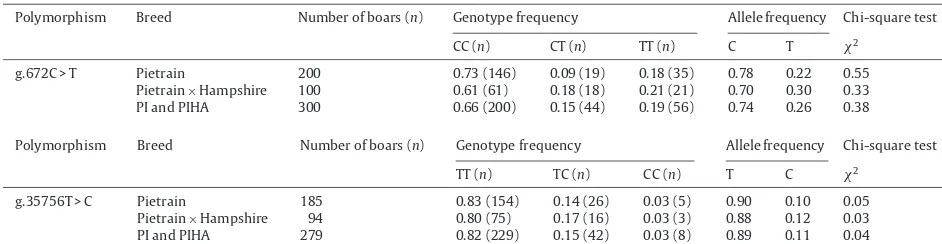
Genotype,allelefrequenciesandthechi-squaretestoftheporcineESR1gene.
Polymorphism Breed Numberofboars(n) Genotypefrequency Allelefrequency Chi-squaretest
CC(n) CT(n) TT(n) C T 2
g.672C>T Pietrain 200 0.73(146) 0.09(19) 0.18(35) 0.78 0.22 0.55
Pietrain×Hampshire 100 0.61(61) 0.18(18) 0.21(21) 0.70 0.30 0.33
PIandPIHA 300 0.66(200) 0.15(44) 0.19(56) 0.74 0.26 0.38
Polymorphism Breed Numberofboars(n) Genotypefrequency Allelefrequency Chi-squaretest
TT(n) TC(n) CC(n) T C 2
g.35756T>C Pietrain 185 0.83(154) 0.14(26) 0.03(5) 0.90 0.10 0.05
Pietrain×Hampshire 94 0.80(75) 0.17(16) 0.03(3) 0.88 0.12 0.03
PIandPIHA 279 0.82(229) 0.15(42) 0.03(8) 0.89 0.11 0.04
temperaturewith5%bovineserumalbumininPBS(50nM sodiumphosphate,pH7.4;0.9%NaCl).Sectionswere incu-bated overnightat 4◦C with theESR1 rabbitpolyclonal primaryantibody(Cat.nr.543;SantaCruz)dilutedat1:50 inPBSTfollowedbysixtimes(10minpertime)washing withPBS.Then,thesectionswereincubated1hatroom temperaturewiththebiotinylateddonkeyanti-rabbit IgG-B asa secondary antibody (Cat.nr.Sc2090; SantaCruz) (dilution 1:200)which wasconjugatedwithfluorescein isothiocynate (FITC).Later onsectionswerewashed six times (10min per time) with PBS. Finally, the samples
werecounterstainedwithvectashieldmountingmedium
(VectorLaboratories)containing40,6-diamidino-2-phenyl indole(DAPI)andcoveredwithacoverglassslip.The stain-ingwasobservedbyconfocallaserscanningmicroscope (Carl Zeiss). In case of negativecontrols, PBSwas used insteadoftheprimaryantibody.
3. Results
3.1. Polymorphismstudy
Twosinglenucleotidepolymorphisms(SNPs)were con-firmedinexon1andintron1ofESR1inthePIandPI×HA populations.Animalsofthesepopulationsweregenotyped atg.672C>Tinexon1andatg.35756T>Cwhichwerethe
SNPsegregating withinthepopulations.The SNPswere
confirmed by PCR-RFLP. The DNA restriction fragments
obtainedforg.672C>TofESR1-BstNIpolymorphismwere: 190,117,50,32and17bpfortheCCgenotype;190,117,50, 32,17and12bpfortheCTgenotype,and190,117,50and 12bpfortheTTgenotype.TheDNArestrictionfragments obtainedforg.35756T>CofESR1-AluIpolymorphismwere: 93,48,40and5bpfortheTTgenotype;133,93,48,40and 5bpfortheTCgenotype,and133,93,and5bpfortheCC genotype.
The calculated genotype and allele frequencies of
porcine ESR1 gene are shown in Table 3. In this study, threegenotypeCC,CTandTTwerefoundforbothSNPsat g.672C>Tandg.35756T>Cinourpopulations. Homozy-goteCCwasmorefrequentandhomozygoteTTwasrare atSNPg.672C>TwhereasincaseofSNPg.35756T>C,the homozygoteTTwasthemorefrequent,andhomozygoteCC wasrareinourpopulations.Thechi-squaretestrevealed thatthelocusofESR1wasinHardy–Weinbergequilibrium inbothPIandPI×HApopulations(Table3).
3.2. Associationanalysis
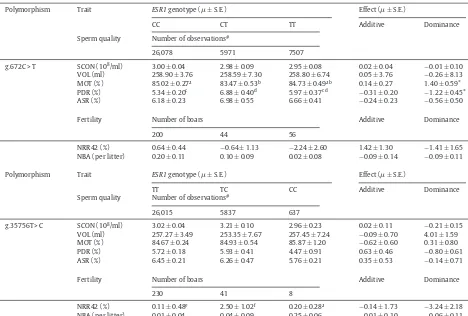
Association analysisofg.672C>Twithspermquality andfertilitytraitsrevealedsignificant(P<0.01)association withMOTandPDR.SemenfromanimalswithgenotypeCT hadsignificantlylowerspermmotilityandhigherplasma dropletratethananimalswithCCandTT(Table4).The resultsalso indicated that there weredominant effects (P<0.05)onMOTandPDRinourpopulation.Inthecase ofg.35756T>C,theassociationanalysisshowedsignificant (P<0.05)associationwithNRR.AnimalswiththeTC geno-typehadasignificantly(P<0.05)higherNRRthanthose withgenotypeTTandCC(Table4).
3.3. mRNAexpressionbysemi-quantitativePCR
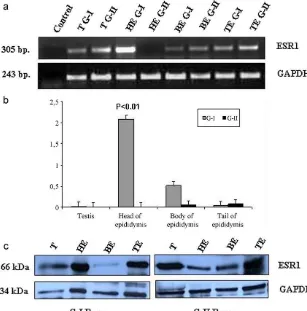
ESR1geneexpressionwashigherin headofthe epi-didymis than in body and tail, moderate in testis, vas deferens,bulbourethralglandsandlowerexpressionwas foundinnon-reproductivetissue(brainandliver)except skeletalmuscle.Thesemi-quantitativereverse transcrip-tionPCRresultofGAPDHshowednoremarkabledifferences amongtissues(Fig.1).
3.4. mRNAandproteinexpressionstudyintissuesfrom
G-IandG-IIboars
TheESR1mRNAwasexpressedintestis,bodyandtail ofepididymis fromboth theG-Iand G-IIboars but the higherexpressionwasshowninheadofepididymisofG-I thanthatofG-IIboarsinsemi-quantitativePCR(Fig.2a). ThesemRNAexpressionresultsofsemi-quantitativePCR appeared to beconsistent with theresults of the qRT-PCR.The ESR1 mRNAexpressionwashigher in headof epididymisofG-Iboars(P<0.01),whereastheexpression
[image:5.561.36.507.72.194.2] [image:5.561.283.503.589.666.2]Table4
AssociationbetweenESR1genotypeswithspermqualityandfertilitytraits.
Polymorphism Trait ESR1genotype(±S.E.) Effect(±S.E.)
CC CT TT Additive Dominance
Spermquality Numberofobservations#
26,078 5971 7507
g.672C>T SCON(108/ml) 3.00±0.04 2.98±0.09 2.95±0.08 0.02±0.04 −0.01±0.10
VOL(ml) 258.90±3.76 258.59±7.30 258.80±6.74 0.05±3.76 −0.26±8.13
MOT(%) 85.02±0.27a 83.47±0.53b 84.73±0.49ab 0.14±0.27 1.40±0.59*
PDR(%) 5.34±0.20c 6.88±0.40d 5.97±0.37cd −0.31±0.20 −1.22±0.45*
ASR(%) 6.18±0.23 6.98±0.55 6.66±0.41 −0.24±0.23 −0.56±0.50
Fertility Numberofboars Additive Dominance
200 44 56
NRR42(%) 0.64±0.44 −0.64±1.13 −2.24±2.60 1.42±1.30 −1.41±1.65
NBA(perlitter) 0.20±0.11 0.10±0.09 0.02±0.08 −0.09±0.14 −0.09±0.11
Polymorphism Trait ESR1genotype(±S.E.) Effect(±S.E.)
TT TC CC Additive Dominance
Spermquality Numberofobservations#
26,015 5837 637
g.35756T>C SCON(108/ml) 3.02±0.04 3.21±0.10 2.96±0.23 0.02±0.11 −0.21±0.15
VOL(ml) 257.27±3.49 253.35±7.67 257.45±7.24 −0.09±0.70 4.01±1.59
MOT(%) 84.67±0.24 84.93±0.54 85.87±1.20 −0.62±0.60 0.31±0.80
PDR(%) 5.72±0.18 5.93±0.41 4.47±0.91 0.63±0.46 −0.80±0.61
ASR(%) 6.45±0.21 6.26±0.47 5.76±0.21 0.35±0.53 −0.14±0.71
Fertility Numberofboars Additive Dominance
230 41 8
NRR42(%) 0.11±0.48e 2.50±1.02f 0.20±0.28a −0.14±1.73 −3.24±2.18
NBA(perlitter) 0.01±0.04 0.04±0.09 0.25±0.06 −0.01±0.10 −0.06±0.11
#Repeatedmeasurements.
a,bP<0.01;c,dP<0.001;e,fP<0.05.*P<0.05.
differenceswere non-significantin the testis,body and tailofepididymisinbetweenG-IandG-IIboars(Fig.2b).
ESR1proteinwith66kDamolecularweightwasdetected intestis,head,bodyandtailoftheepididymisinbothof G-IandG-IIboars(Fig.2c).Thewesternblotresultshowed thattheESR1proteinexpressionwashigherinheadofthe epididymisinG-Iboars.Thisproteinexpressionseemedto beconsistentwiththeresultsoftranscriptionlevels.
3.5. LocalizationofESR1proteininboarreproductive
tissuesbyimmunofluorescence
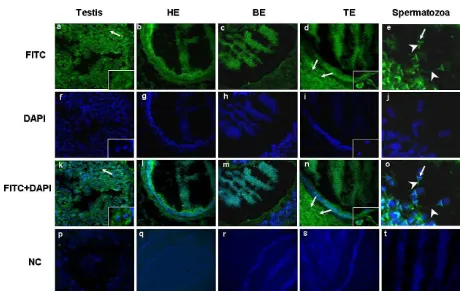
Sectionsof testis, head,body and tailof epididymis werestainedthroughthesameopticalpanelfortheESR1
proteinexpression(Fig.3).ImmunoreactiveESR1protein wasobservedas strongstainingin cytoplasmof Sertoli cells but poorly in germcells and Leydig cells in testis (Fig.3a).Spermatozoawithinthelumenoftheepididymis headwerefoundtobepositivelyimmunostainedwithESR1
(Fig.3b).Similarly,spermatozoawithinthelumenofthe epididymisbodyandtailwerepositivetoimmunoreactive
ESR1protein(Fig.3candd).Theimmunofluorescenceof
ESR1proteinwasspecificallylocalizedinthepost acro-somalregion(arrow)andtailofthesperm(arrowhead) whenspermatozoawithintheepididymiswereexamined
(Fig.3e).Moreprecisely,theepithelialcellsintheheadand tailoftheepididymisexpressedhigherimmunoreactive
ESR1protein(Fig.3bandd),whereastheepithelialcells intheepididymisbodypoorlyexpressedthetarget pro-tein(Fig.3c).Inthecaseoftheepididymisheadandbody, thesterociliacellswereremarkablyhigherimmunostained whileintheepididymistailtheproteinwasstrongly local-izedinthesmoothmuscle(Fig.3b–d).
4. Discussion
4.1. AssociationofSNPwithboarspermqualityand
fertilitytraits
[image:6.561.43.511.71.387.2]Fig.2.mRNAandproteinexpressionofESR1inreproductivetissues(testis,head,bodyandtailoftheepididymis).(a)ESR1mRNAexpressionindifferent reproductivetissuesfromG-IandG-IIboarsbysemi-quantitativePCR.(b)ESR1mRNAexpressionindifferentreproductivetissuesfromG-IandG-IIboars byqRT-PCR.(c)ProteinexpressionindifferentreproductivetissuesfromG-IandG-IIboarsbywesternblotting.(T:Testis,HE:Headofepididymis,BE: Bodyofepididymis,TE:Tailofepididymis.)
estrogensinmalegametematurationandmotility.Ithas beenreportedthatestrogenproducedlocallyisinvolvedin theregulationofspermmotility(Lazarosetal.,2010).ESR1
polymorphism mayinfluence theselocally acting estro-genlevelswitheffectonspermmotility(Carreauetal., 2002). Abnormal sperm production and reduced fertil-ity have beenreportedin transgenicmale micelacking
ESR1 (Eddyet al.,1996).Infertility fromlackingESR1 is reportedmainlyduetothedisruptionoffluidreabsorption inefferentductuleswhichincreasedthebackpressureof theaccumulatingluminalfluidsthatleadstoaprogressive degenerationofthetesticulartissueandseverelyimpaired spermatogenesis (Eddy et al., 1996; Hess et al., 1997; CouseandKorach,1999).TheidentifiedSNPisconfirmed in exonwhich mayplayimportantrole in transcription process.Polymorphisminthecodingregioncouldhavea directeffectthroughchangingthenucleotidesequenceand structureofgene,possiblyleadingtochangesinmRNA syn-thesis,splicing,maturation,transportation,translationor degradation(IidaandAkashi,2000).
TheintronicSNPg.35756T>Cwassignificantly associ-atedwiththefertilitytraitnon-returnrate(NRR)(Table4). Reproductive efficiencyofboars is usuallymeasuredby non-returnrate,which isdefinedaspercentageofsows notappointedforasecondinseminationwithinaperiod
ofdaysafterthefirstinsemination.Malefertilitycanbe regardedasaresultofbothfertilizingabilityofthesperm cellsaswellasthenumberoffertilizedoocytesandthe viabilityof theembryo. But, underthe field conditions onlytheoutcomeoftheinseminationscanberecorded,i.e. whetherornotthefemaleshavereturnedforrepeat insem-ination(Stalhammaretal.,1994).However,littleisknown aboutthepolymorphismandassociationofporcineESR1
[image:7.561.118.425.55.366.2]Fig.3. LocalizationofESR1proteinindifferentpartsofboarreproductivetissues.(a)ImmunofluorescencedetectionofESR1incytoplasmofSertolicells andgermcells.SertolicellswerestainedwithESR1(arrows)andthenucleiwerecounterstainedwithDAPI(arrow).(bandc)ESR1proteinlocalization inepithelialcellsintheheadandbodyoftheepididymisandinthespermatozoawithinthelumenoftheepididymis.(d)LocalizationofESR1proteinin epithelialcells,inspermatozoawithinthelumenoftheepididymisandremarkableexpressionwasinsmoothmuscleinthetailoftheepididymis.Smooth muscleinthetailoftheepididymiswasstainedwithESR1(arrows).(e)TheESR1localizedinacrosomalregion(arrow)andtailofspermatozoa(arrowhead) withinepididymis.(f–j)ThecellnucleiwerecounterstainedwithDAPI.(k–o)Mergedimages.(p–t)Negativecontrol.(T:Testis,HE:Headofepididymis, BE:Bodyofepididymis,TE:Tailofepididymis.)
moderate to medium heritability (h2=0.19–0.37) (See, 2000).However,twoSNPsofESR1areidentifiedinboth exonandintronwhichmighthaveanimportanteffectson spermqualityandfertilitytraitsinpig.
4.2. ESR1mRNAandproteinexpressioninboar
reproductivetissues
Thephysiologicalroleofestrogensinmale reproduc-tion is now extensively revisited (Carreau et al., 2002) butexpressionstudyisrareinboars.Inthisstudy,mRNA expressionanalysisbysemi-quantitativePCRandqRT-PCR demonstratedthattheporcineESR1geneexpressionwas higherin reproductivetissuesthan innon-reproductive tissues(Fig.1).Importantly,ESR1expressedhigherinthe headofepididymiscollectedfromG-Iboarsthanthatof G-IIboars. The highest expression of ESR1 in thehead of theepididymis was in agreement withthe previous reportsdescribingthatthehighestconcentrationof estro-genreceptorwasfoundintheepidydimisheadinmiceand macaques(Schleicheretal.,1984).Itiswelldocumented thatmaleestrogenisinhighpresenceintheefferentducts intheheadof epididymis(Hess andCarnes,2003).The presenceofESR1intheheadoftheepididymishasimmense importancesinceestrogenplaysacrucialfunctioninfluid
reabsorptioninthisregioninthemale(Hessetal.,1997). Thisfluidreabsorptionisimportantforspermmaturation andformaintainingthenumberofspermintransit(Janulis etal.,1996).Anydisruptionoftheestrogenreceptorgenein malemiceresultedwithdecreasedfertilityandtesticular weightandverylowspermcounts(LubahnandGolding, 1993).Moreover, ESR1 abundanceis detected in differ-entpartsofmalereproductivetractandspermatozoain mice(Zhouetal.,2002),humans(Aquilaetal.,2002)and rodents(Pelletieretal., 2000)whichcoincides withour findings.Theevidenceofestrogenindifferentpartsofthe malereproductivetractsuggestsapossiblephysiological role ofestrogenin spermatogenesisand sperm matura-tion.Furthermore,thelowerESR1transcriptabundancein theheadoftheepididymisofG-IIboarsinthisstudymay indicatea lackofestrogenactionwhichmaycontribute tothepoorsperm qualityandfertility.It isknownthat spermanditsmembraneproteinsundergoseveral modifi-cationsduringmaturationwithintheepididymis.Estrogen antagonist treatment in miceshowedthat lack of ESR1
[image:8.561.46.507.46.337.2]degenerationofthetesticulartissueandseverelyimpaired spermatogenesis(Eddyetal.,1996;Hessetal.,1997;Couse andKorach,1999).Moreover,swellinganddamageof sem-iniferoustubulesandthedilutionoftheessentialproteins secretedfromepididymisalsocontributedtothe infertil-ity(Eddyetal.,1996;Hessetal.,1997;CouseandKorach, 1999).
Inthisstudy,theproteinexpressionanalysisby west-ernblottingshowedthatESR1antibodyrecognizedaband at66kDainalltissues.Thebandissimilartothe molecu-larweightofhuman(66kDa)andmouse(66kDa)ESR1and veryclosetothatofthestallion(65kDa).Although,the lev-elsofproteinindifferenttissuesfromG-IandG-IIwerenot asdistinguishableasmRNA,theESR1proteinexpression wasremarkablyhigherintheheadoftheepididymisofG-I boarsthanthatofG-IIboars(Fig.2b).Thevasdeferensinthe headoftheepididymisarethemajorsitesforESR1 expres-sionwhichregulatesthetesticularfluidreabsorptionand increasesthespermcellconcentrationastheyenterthe epididymis(Hessetal.,1997).Moreover,theepididymisis involvedindifferentproteinabsorptionaswellasin pro-teinsecretion(Syntinetal.,1999).Theheadandbodyofthe epididymisarereportedtosecrethigheramountsof differ-entproteinsinboarsincomparisontootherfarmanimals (Syntinetal.,1999).Theseproteinsecretionsareregulated byestrogenand theproteinsareinvolvedin thesperm membraneremodelling,intheinitiationofspermmotility, andespeciallyinsperm-egginteraction(Pearletal.,2007). Ithasbeencalculatedthat50–90%ofthetotalprotein leav-ingthetestisisabsorbedbytheefferentductsinthehead oftheepididymis(Clulowetal.,1994).
4.3. ProteinlocalizationofESR1
ESR1wasfoundtobeexpressedstronglyinthe cyto-plasmofSertolicells,expressedongermcellsandLeydig cells in testis (Fig. 3a). These resultsare in agreement withpreviousresultslocalizingESR1inSertoli and Ley-digcells(HessandCarnes,2003)andinLeydigandgerm cellsinrat(Pelletieretal.,2000).Pelletieretal.(2000) sug-gestedthatintheLeydigcellstheESR1mightbeinvolved in thesecretionand maturationof germcells. Estrogen arereportedtobeinvolvedinmaintainingtheSertolicell function(O’Donnelletal.,2001)aswellas in establish-ingSertoli–germcelladhesion(MacCalmanandBlaschuk, 1994).However,thecellulardistributionpatternsofESR1
proteinbetweenspeciescouldbedifferent.
We localized ESR1 in thehead, body and tailof the epididymis.Whenspermarereleasedfromtheir‘nurse’ (Sertoli)cells inthetestis,theyaretransported in fluid secretedbytheSertolicellstoacollectingarea,therete testis.Fromthere,thedilute suspensionofsperm enter thethin-walledefferentductsintheepididymisheadand theepitheliaofefferentductsexpressenormousestrogen receptors(Fisheretal.,1997).Thespermpassthroughthe epididymiswheretheymatureandprogresstowardthe vasdeferens. Moreover, theESR1in theepididymis are reportedtomodulatesecretionofproteinssuchasoscilin thatpromotesthematurityandviabilityofthe spermato-zoa(MowaandIwanaga,2001).Themostintensesignals ofESR1arereportedinepithelialcellsintheheadandtail
oftheepididymisinratsandmonkeysandsuggestedtobe responsibleforsemenconcentration(Fisheretal.,1997; Hessetal.,1997).Ourstudyidentifiedastrongsignalof
ESR1inthesmoothmusclelayerinthetailoftheepididymis whichisinaccordancewithaprevious immunohistochem-icalstudyinhumanandrabbitwhichstatedthatestrogen isessentialfortransportingthespermatozoaby influenc-ingepididymalsmoothmusclecontractilityaswellasfor ejaculationwiththehelpofoxytocin(Filippietal.,2002). ItisimportanttonotethattheexpressionofESR1in dif-ferentpartsoftheepididymiscouldvaryamongspecies sinceithasbeenlocalizedinallpartsofepididymisinthe rat(Pelletieretal.,2000),mouse(Zhouetal.,2002),bonnet monkey(Shayuetal.,2004),horse(Parlevlietetal.,2006), cat(Nieetal.,2002)anddog(Nieetal.,2002).
We found that the ESR1 is remarkably localized in spermwithinthelumenoftheepididymis.ESR1was espe-ciallyimmunolocalizedinthepostacrosomalregionand tail of the sperm in this study (Fig. 3e). This result is ingoodagreementwithpreviousstudieswhichlocalized immunoreactiveESR1inthepostacrosomalregionofthe spermhead(Solakidietal.,2005)andinthetailpieceofthe sperm(Durkeeetal.,1998)inhumans.Thepost acroso-malregionofspermisinvolvedinthesperm–eggplasma membranefusion(Liuetal.,2008).ThelocalizationofESR1
inthepostacrosomalregionimplies itsinvolvementin thefertilizationprocess(Ramalho-Santosetal.,2002).Itis importanttonotethatthereareotherproteinsidentified inthepostacrosomalregionofspermsuchasequatorin andoscilinwhichareimportantforsuccessfulfertilization (Montagetal.,1998;Solakidietal.,2005).Ourlocalization ofESR1inthetailofporcinespermcoincidedwithprevious reportsinhumans(Durkeeetal.,1998;Aquilaetal.,2002). Moreover,Aquilaetal.(2002)reportedthattheESR1might beinvolvedinspermsurvivalandmotility.Impaired motil-ityofspermhasbeenreportedinmicelackingfunctional
ESR1insperm(Eddyetal.,1996)andinESR1knockoutmice (HessandCarnes,2003).Lowfertilizationrateshavebeen reportedincaseofimmotilespermatozoa(Nijsetal.,1996).
5. Conclusion
Associations of ESR1gene polymorphismswith boar spermqualityandfertilitytraitshavebeendescribedfor thefirsttime,providingevidencethatESR1mightbean importantcandidategeneforboarspermqualityand fertil-ity.However,thisstudyhastobevalidatedinotheranimal populationsinordertoevaluateitspotentialinselective breeding.Finally,theESR1mightplayarolein spermatoge-nesisvalidatedthroughassociationstudyandbyprofiling ofmRNAandproteinexpressioninnon-reproductiveand reproductivetissuesincludingspermatozoa.
Acknowledgments
Hofer,Dr.H. LutherandDr.K.Caspari(SuisAGArtificial Insemination Station, Sempach, Switzerland) for their support during the sample collection and the arrange-mentsofthereproductionrecords.Wearealsothankful to Prof. Dr. C. Knorr (Institute of Veterinary Medicine, Georg-August-University, Goettingen, Germany) for his co-operation.
References
Aquila,S.,Sisci,D.,Gentile,M.,Middea,E.,Siciliano,L.,Ando,S.,2002. Human-ejaculatedspermatozoacontainactiveP450aromatase.J. Clin.Endocrinol.Metab.87,85–90.
Capon,F.,Allen,M.H.,Ameen,M.,Burden,A.D.,Tillman,D.,Barker,J.N., Trembath,R.C.,2004.AsynonymousSNPofthecorneodesmosin geneleadstoincreasedmRNAstabilityanddemonstrates associa-tionwithpsoriasisacrossdiverseethnicgroups.Hum.Mol.Genet.13, 2361–2368.
Carreau,S.,Bourguiba,S.,Lambard,S.,Galeraud-Denis,I.,Genissel,C., Lev-allet,J.,2002.Reproductivesystem:aromataseandestrogens.Mol. Cel.Endocrinol.193,137–143.
Clulow,J.,Jones,R.,Hansen,L.,1994.Micropunctureandcannulation stud-iesoffluidcompositionandtransportintheductuliefferentestestis oftherat:comparisonswiththehomologousmetanephricproximal tubule.Exp.Physiol.79,915–928.
Couse,J.,Korach,K.,1999.Estrogenreceptornullmice:whathavewe learnedandwherewilltheyleadus?Endocr.Rev.20,358–417. Durkee,T.,Mueller,M.,Zinaman,M.,1998.Identificationofoestrogen
receptorproteinandmessengerribonucleicacidinhuman spermato-zoa.Am.J.Obstet.Gynecol.178,1288–1295.
Eddy,E.,Washburn,T.,Bunch,D.,Goulding,E.,Gladen,B.,Lubahn,D., Korach,K.,1996.Targeteddisruptionoftheestrogenreceptorgene inmalemicecausesalterationofspermatogenesisandinfertility. Endocrinology137,4796–4805.
Filippi,S.,Vannelli,G.B.,Granchi,S.,Luconi,M.,Crescioli,C.,Mancina, R.,Natali,A.,Brocchi,S.,Vignozzi,L.,Bencini,E.,Noci,I.,Ledda,F., Forti,G.,Maggi,M.,2002.Identification,localizationandfunctional activityofoxytocinreceptorsinepididymis.Mol.Cel.Endocrinol.193, 89–100.
Fisher,J.,Millar,M.,Majdic,G.,Saunders,P.,Fraser,H.,Sharpe,R.,1997. Immunolocalisationofoestrogenreceptor1withinthetestisand excurrentductsoftheratandmarmosetmonkeyfromperinatallife toadulthood.J.Endocrinol.153,485–495.
Ganjam, V., Amann, R., 1976. Steroid content of fluids and sperm enteringand leaving the bovine epididymis, in epididymal tis-sue, and in accessory sex gland secretions. Endocrinology 99, 1618–1630.
Guarducci,E.,Nuti,Becherini,L.,Rotondi,M.,Balercia,G.,Forti,G.,Krausz, C.,2006.Estrogenreceptor2promoterpolymorphism:stronger estro-genactioniscoupledwithlowerspermcount.Hum.Reprod.21, 994–1001.
Hejmej,A.,Gorazd,M.,Kosiniak-Kamysz,K.,Wisniewska,B.,Sadowska, J.,Bilinska,B.,2005.Expressionaromataseandandrogenreceptorsin reproductivetissueofthestallionandasinglecryptochidvisualized bymeansofimmunohistochemistry.Domest.Anim.Endricinol.29, 534–547.
Hess,R.,Carnes,K.,2003.Theroleofoestrogenintestisandmale repro-ductivetract:areviewandspeciescomparison.Anim.Reprod.1,5–30. Hess,R.,Gist,D.,Bunick,D.,1997.Estrogenreceptor(alphaandbeta) expressionintheexcurrentductsoftheadultmaleratreproductive tract.J.Androl.18,602–611.
Iida,K.,Akashi,H.,2000.Atestoftranslationalselectionat‘silent’sitesin thehumangenome:basecompositioncomparisonsinalternatively splicedgenes.Gene261,93–102.
Janulis,L.,Hess,R.,Bunick,D.,Nitta,H.,Janssen,S.,Asawa,Y.,Bahr, J.,1996.MouseepididimalspermcontaintactiveP450aromatase whichdecrease asspermtraverse theepididymis. J. Androl.17, 111–116.
Jiang,W.,Le,B.,2000.StructureandexpressionofthehumanMEP1Agene encodingthealphasubunitofmetalloendopeptidasemeprinA.Arch. Biochem.Biophys.379,183–187.
Kaewmala,K.,Uddin,M.J.,Cinar,M.U.,Grosse-Brinkhaus,C.,Jonas,E., Tesfaye,D.,Phatsara,C.,Tholen,E.,Looft,C.,Schellander,K.,2011. Association study and expression analysis of CD9 as candidate geneforboarspermqualityandfertilitytraits.Anim.Reprod.Sci., doi:10.1016/j.anireprosci.2011.02.017.
Kuiper,G.,Enmark,E.,Puelto-Huiko,M.,Nilsson,S.,Gustafsson,J.,1996. Cloningofanovelestrogenreceptorexpressedinratprostateand ovary.Proc.Natl.Acad.Sci.U.S.A.93,2925–2930.
Lazaros,L.A.,Xita,N.V.,Kaponis,A.I.,Zikopoulos,K.A.,Plachouras,N.I., Georgiou,I.A.,2010.Estrogenreceptoralphaandbetapolymorphisms areassociatedwithsemenquality.J.Androl.31,291–298. Lin,C.L.,Tholen,E.,Jennen,D.,Ponsuksili,S.,Schellander,K.,Wimmers,K.,
2006.Candidategenemarkersforspermqualityandfertilityofboar. Anim.Reprod.Sci.92,349–363.
Liu,G.Q.,Zhu,J.J.,Wang,Z.Y.,Jiang,X.P.,Dafalla,M.M.,2008.Analysisof spermstorageabilityusingdurationoffertilityinhens.Br.Poult.Sci. 49,770–775.
Lubahn,D.B.,Moyer,J.S.,Golding,T.S.,1993.Alterationofreproductive functionbutnotprenatalsexualdevelopmentafterinsertional dis-ruptionofthemouseestrogenreceptorgene.Proc.Natl.Acad.Sci. U.S.A.90,11162–11166.
MacCalman,C.D.,Blaschuk,O.,1994.GonadalsteroidsregulateN-cadherin mRNAlevelsinthemousetestis.Endocr.Rev.2,157–163. Montag,M.,Parrington,J.,Swann,K.,Lai,F.,VanderVen,H.,1998.
Pres-enceandlocalizationofoscillininhumanspermatozoainrelationto integrityofthespermmembrane.FEBSLett.423,357–361. Mowa,C.N.,Iwanaga,T.,2001.Expressionofestrogenreceptor-alphaand
-betamRNAsinthemalereproductivesystemoftheratasrevealed byinsituhybridization.J.Mol.Endocrinol.26,165–174.
Munoz,G.,Ovio,C.,Estell,J.,Silio,L.,Fernadez,A.,Rodriguez,C.,2007. Association with litter sizeof new polymorphismonESR1 and ESR2genesina Chinese–Europeanpigline.Genet.Sel.Evol.39, 195–206.
Nie,R.,Zhou,Q.,Jassin,E.,Saunders,P.,Hess,R.,2002.Differential expres-sionofoestrogenreceptorsalphaandbetainthereproductivetract ofadultmaledogsandcats.Biol.Reprod.66,1161–1168.
Nijs,M.,Vanderzwalmen,P.,Vandamme,G.,1996.Fertilizingabilityof immotilspermatozoaafterintracyctoplasmicsperminjection.Hum. Reprod.11,2180–2185.
O’Donnell,L.,Robertson,K.M.,Jones,M.E.,Simpson,E.R.,2001.Estrogen andspermatogenesis.Endocr.Rev.22,289–318.
Pagani,R.,Song,M.,McEnery,M.,Qin,N.,Tsien,R.W.,Toro,L.,Stefani, E.,Uchitel,O.D.,2004.Differentialexpressionofalpha1andbeta subunitsofvoltagedependentCa2+channelattheneuromuscular junctionofnormalandP/QCa2+channelknockoutmouse. Neuro-science123,75–85.
Parlevliet,J.,Pearl,C.,Hess,M.,Famula,T.,Roser,J.,2006. Immunolocaliza-tionofestrogenandandrogenreceptorsandsteroidconcentrations inthestallionepididymis.Theriogenoloy66,755–765.
Pearl,C.,Berger,T.,Roser,J.,2007.Estrogenandandrogenreceptor expres-sioninrelationtosteroidsconcentrationsinadultboarepididymis. Domest.Anim.Endocrinol.33,451–459.
Pelletier,G.,Labrie,C.,Labrie,F.,2000.Localizationofoestrogen recep-toralpha,oestrogenreceptorbetaandandrogenreceptorsintherat reproductiveorgans.J.Endocrinol.165,359–370.
Ramalho-Santos,J.,Schatten,G.,Moreno,R.,2002.Controlmembrane fusionduringspermiogenesisandtheacrosomereaction.Biol.Reprod. 67,1043–1051.
Rozen,S.,Skaletsky,H.,2000.Primer3ontheWWWforgeneralusersand forbiologistprogrammers.MethodsMol.Biol.132,365–386. Safarinejad,M.R.,Shafiei,N.,Safarinejad,S.,2010.Associationof
polymor-phismsintheestrogenreceptorsalpha,andbeta(ESR1,ESR2)with theoccurrenceofmaleinfertilityandsemenparameters.J.Steroid Biochem.Mol.Biol.122,193–203.
Schleicher,G.,Drew,U.,Stumpt,W.,Sar,M.,1984.Differentialdistribution ofdihydrosteroneandestrodialbindingsiteintheepididymisofthe mouse:anautoradiographicstudy.Histochemistry,1139–1147. See,M.,2000.SelectionforAIstudtraits.In:ProceedingsofNationalSwine
ImprovementFederation,Nashville,Tennessee,USA,7–8December, p.25.
Shayu, D.,Chennakesava,C.,Soundarajan,R.,Rao, J.,2004. Effectof ICI182780onestrogenreceptorexpression,fluidabsorptionand spermmotilityintheepididymisofthebonnetmonkey.Reprod.Biol. Endocrinol.3,3–10.
Solakidi,S.,Psarra,A.,Nikolaropouluos,S.,Sekeris,C.,2005.Estrogen receptoralphaandbetaandandrogenreceptor(AR)inhumansperm: localizationofEstrogenbetaandARinmitochondriaofthemidpiece. Hum.Reprod.20,3481–3487.
Stalhammar,E.,Janson,L.,Philipsson,J.,1994.Geneticstudiesonfertility inAIbulls.II.Environmentalandgeneticeffectsonnon-returnrates ofyoungbulls.Anim.Reprod.Sci.34,193–207.
Syntin,P.,Dacheux,F.,Druart, X.,Gatti,J.,Okamura,N.,Dacheux,J., 1999. Characterizationand identification of proteins secretedin thevariousregionsoftheadultboarepididymis.Biol.Reprod.55, 956–974.
Wimmers,K.,Lin,C.,Tholen,E.,Jennen,D.,Schellander,K.,Ponsuksili,S., 2005.Polymorphismsincandidategenesasmarkersforspermquality andboarfertility.Anim.Genet.Sel.Evol.36,152–155.
Xing,Y.,Ren,J.,Ren,D.,Guo,Y.,Wu,Y.,Yang,G.,Mao,H.,Brenig,B.,Huang, L.,2008.Awholegenomescanningforquantitativetraitlociontraits relatedtospermqualityandejaculationinpigs.Anim.Reprod.Sci. 114,210–2118.