Vol 10, No 2,
Apil
-
June 2001 Menstruatio,t andmenorrhagia
121Mechanism
of
normal
menstruation
and abnormality
associated
with
menorrhagia
Jeanne
.{diu inau
Pari itanAbstrak
Pada Inid nonnal terjadi peLuruhan jaringan endometium dan perdarahan, diikuti oLeh hemostasis dan 'repair'. Kelainan dapat terjadi pada berbagai tahap dari proses di atas, dan dapat mengakibatkan perubahan jumlah darah haid. Haid dengan jumlah darah
lebih dari 80 ml disebut menorhagia. Sari pustaka
ini
membicarakan meknnisme haid normal, dan berbagai faktor yang berkaitatt dengan menorhagia. Faktor tersebut adalah 'endometrial bleeding associatedfactor
(ebaf)', peran berbagai sel (lekosit migran, makrofag, dan sel mnst), peran berbagai zat (enzim lisosom, berbagai prostaglandin, endotelin, faktor pertumbuhan, dan reseptor-nya), gangguan padafibrinolisis dan proses hemostasis, dan perubahan aliran darah endometrium. (Med J Irulones 2001; 10: 12I-6)Abstract
Notmal menstruation involves endometrial tissue breakdown and bleeding, followed by hemostasis and repair. Abnormality of this
Process at any stage may resub in changes in the quantity of menstrual blood loss. When menstrual blood loss is greater thnn 80 ml,
it
is called menorhagia. This review discuss the mechanism of normal menstruation, and factors associated with menorrhagia. Thosefactors are the endometrial bleeding associatedfactor (ebaf), the role ofvarious cells (migratory leucocytes, macrophages, and mast
cells), the role of various substances (lysosomal enzymes, prostaglandins, endothelins, growthfactors and its receptors), impairment of fibrinolysis and hemostatic proces, and changes in endometrial bloodflow. (Med J Indones 2001; 10: 12l-6)
Keyw ords : e baf, prostaglandin, endothe Lin, fibrinolysis
\ormal menstmation
inr olr
es
endometnal tissue
breakdo*n
and bieeding.
follo*ed br
hemostasis andrepair.
-{br.crr:li:r
aa:hrs licae
s-i3:
3:\
s::_È-:-:',
rcluli t: Jl-.r:.i:i:::.-.i
J-:t.-'.', -:--:-.::-r.:.
-lo ss.
Excessive
raei.:::-.- :-=;---:
for
consultatltrr.
rr'
.3..:.:-of
menstruai
b.i.:
.-:-
.,bleeding
ma\
ii:- :
::':.
Sevefe CaseSrna\
L3-;::.-:.
bleedin-e
is
consii:r:;
:'r.:-::,i :
and constitutes rr'b:a,-:;\3 :--,--.--:r-i
m\
osis.
fibromr
omata. endomernal
pol)p,
endo-xrei:rosls,.
crrntÉ.-€Dtiveuse
rintra-uterine device)
orJi sa-i.J:;r':tr-
;:er:e
:jeedin,s
,DLB
r.'Foq
to 6OVcof
::,::.-:l:-;:.:
;:S3r
::
afinbuted
tO
DLB.) DUB
iS a
t
-5-:,:t: -.: e\;.usion. and reiers
to excessivel)
.-.3É''.,.::-
-rrsÈc
o:
trequent bleeding (mostcommonly
:.-;..-..
.i.
ir.€ltrrrhagia
I
\\hich
is
not
caused
by
::=_i--.-.-'_.
ar
ùit\
recocnizable
pelvic
or
systemic l,:-r'=I::s
:e'.rerl ri.ill
discuss
the
mechanism
of
normal
r:-.:
! ir.r.ili Lrn and factors as soc iatedwith
menorrhagia.\TECTL{\N\T
OF
NORMAL
MENSTRUATION
\rrnnal
menstruation
is
triggered by
the
fall
in
tresîradiol
and
progesteron concentrations
following
lu:eolvsis.
It ts
preceeded
by
endometrial
tissuef.eakdo\\'n
andbleedtng.
and followedby
hemostasrs,repair
and
angiogenesis.These
processesare largely
.L'rniroiiedbi
local factors
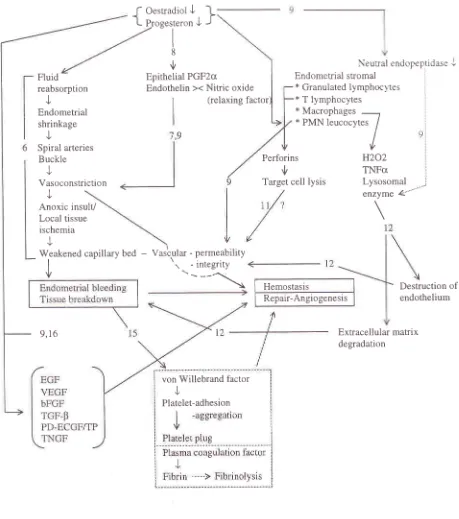
(Figure
1).Menorrhagra
it:.,1i
:.- ---
--(coaguiation
di:.-rJ.:: '-.::
-:
\-Willebrand
Jr:::.:' .
t22
PawitanFigure
I.
Mechanism of normal mensîruationMed J Indones
Fluid
reabsorption
J
Endometrial
shrinkage
J
Spiral arteries
Buckle
J
Vas o con stric ti o n
Epithelial PGF2cr
Endothelin ><
Nitric
oxide (relaxing factorf
Oestradiol J-l
t
P-rog"r,..onJ
J
I 8
ù
Neurral endopeptidaseJ
Endometrial stromal
*
Granulatedlymphoclres
:x
T
lymphocytes x Macrophages-*
PMN leucocytes
/
I
,1,Anoxic insult/
Local tissue ischemia
J
J
9,t6
Perforins
ù
Target cell lysis
Hemostasis
Repair-Angiogenesis
enzyme &""'
H202
TNFctLysosomal
:Extracellular matrix
degradation Weakened
caoillarv
bedI
v
-
Vascrular - permeability1
.--_'\o
-lntetrlt)
Destruction
of
endotheliumvon Willebrand factor I
v
Menstruation and
menorrhagia
123_sranules
of
a variety
of
cytotoxic lymphocytic
cellscontain perforins
that
mediate
target
cell
lysis.
However
it
is
not clear,
if
granulated lymphocytes
ever
mediate such
toxicity
against
blood
vesselslocally.lr
Macrophages
can
becytotoxic to
tissueby
releaseof
H2O2,
TNF
alfa, and various
lysosomal
enzymes.rlThe
secretionof hydrolytic
(lysosomal)
enzymesinto
the
extracellular
space
is a
mechanism
that
could
initiate
endometrial
bleeding,
either directly
by
dissociation
or
destruction
of
endothelial
cells
or
perivascular
tissue,or
indirectly by
degradationof the
extracellular
matrix. I 2The
role
of
proteolytic
enzymesand its
inactivator
Several
proteolytic
enzimes have been
implicated
in tissuebreakdown.
They
are presentin
the
lysosomes(aspartic lysosomal proteinase cathepsin
D
and lysosomal cysteine proteases),or
secretedeither
at thecell
surface
or
in
extracellular matrices
or
fluids
(metallo-
and serine proteinases). The nonsedimentablefraction
of
ll,sosomal enzymes
from
endometrial
homogenates has been reported
to
increasefrom
about 25% at theproliferatire
phaseto
about50%
at the latesecreton'phase.'-Tl^.
:
:^"-::r
:.-r3:.1llop rrr:et:rases,
\ I\ IP
:
co ila_eenase,,::-.:-:.r.;
.:.:
S::r=elr
SIS;
are Capable Of de_erading:--
::
:-':-'.
rfrrl:trS.
tn;luCing interstitial
collagens,--
:!
: rr:ir-. :-:::-'::.:
:
c c ll a gen.proteogll
c ans, I amtnin:--.-
-.::
:.:::-:.
" \l.r".t
metalloproteinases
are:"-.::=!::: -:- ::.3
Sl:rrti.âl
and epithelial
CellS
Of= .4.,-.,a,a--=.
-
Htr'.\Èrer.
decreasing
concentrationsIn
the
n..;:-t-
:--re:,.::-.-
:',-.-
- .== : : :-l:-'--: '
-: ::-.:i:::Cle
feSUlt
in
a
fall in
the
endOmetfialpre-menslnJ..
-:,::;;:: ..
:--.1-:=-1
:, ::,:--:
:-::--
J-:..-3:.lltliu-rn
Oi
neUtral
endOpeptidaSe
(agranulated
1r::r::.-.,.::. - -'.--:-
'!.::
j :--- r--;-r,-
:.'.c..'.
l::ed
zinc metallopeptidase
capable
of
in-ptug"r.
utriie
^;:--.:,--..
.--.1-
:-r,-,:;.=:
:,-.
.-',.'..,4r.g
a
number
of
endocrine
and
paracrine appeafln
Utenneli:i-::
:-
--:
- -=--, t--ct:t-:---
I
at::,je
=Polymorphonuci:a:
.=-:-,:-,.=.
.
= .:-=,
-,-::=:
Vol 10, No 2, April-
June 2001Endometrial
tissue
breakdorrn
and bleeding
Markee
1ascited from Brenner.
1996)noted that
thefirst
and mosrreliable sign of impending
mensrmarion*as
aslou.ing
of
blood
flow in
the upper endometrialregions. accompanied
by
a reduction
in
endometrial thickness dueto
fluid
reabsorption.This
shrinkage,or
premenstrual regression,
began2
days before bleedingand continued
throughout the
bleeding episode.
Thesecond
key
event
was
compression
of
the spiral
arteries. Thesecoiled
vesselsbuckled
to
accommodateshrinkage
of
the
endometrium.
As a
consequence,blood
flow
through
these vessels wasgreatly
reduced.The
third
event was an extensive vasoconstriction
of
thespiral
arterieswhich
further
reducedblood
flow
tothe upper
zones.The
vascular
stasisinduced
by such
compression
andvasoconstriction led to anoxic insult
that weakened the
capillary
bed.6The role of
l\mph(]ic]tti.
rngcr,rphag€s
andp o l1rn
orphon
u clear
leuctr,r--te:
LD tL\: u e b reakd,r u nblood
r essels::
::.3
;r.-
-rr:-r
-r--
.;:
r:=
-,-.a'-\.:osether.
these events leadsto
tissue shedding. TheJ:squamatir
e processprobably
follows
atopographic
sequence and the process
might not
have
reached thesame stage
in
each
segment
of
the
uterine
cavity.
There
is still controversy
about the extent
of
tissueshedding
at
menstruation. Some investigators
statedthat
at no
time
during
menstruation
was
thereextensive denudation
of
surface epithelium.
Thelargest breaks were
not
more than
2 mm
in
diameterand
most
were less
than
100
um.
Some
other
statedthat
most
part
of surface
lining epithelium
are
shed,with only
the
epithelium at
the glandular stumps
left
Granuiated
hmphr\\
iù!
areendometrial
blood
ressels
in
rhe124
Pawitanand, adjacent
to it,
naked endometrial
stromaforming
the
surface.However,
isles
of
non-shedendometrium
may be
preserved
within
thefundal
cavity.r4Hemostasis,
repair and
angiogenesisThe normal
hemostatic process
can be divided
into
three
phases.
The
first
phase,
primary
hemostasis,consist
of
platelet
adhesion
and
aggregation.
After
vascular
injury,
proteins in
the
subendothelium
areexposed
and
promote platelet
adhesion.
Plateletadhesion
is
uniquely
dependent
on
von Willebrand
factor,
a
plasma protein that
serves
as
a
molecular
bridge
between componentsof
the vesselwall
and theplatelet glycoprotein Ib/IX
receptor. Activation of
adherent
platelets
promotes
additional
platelet
recruitment, culminating
in
the
formation
of
theplatelet
plug.'t
In
the
next
phase, secondary
hemostasis,the
plasmacoagulation
factors are
sequentially activated, which
lead
to
fibrin
formation
and
cross-linking.
Thesereactions
take
place
primarily
on the
surface of
activated
platelets
and are essentialin
maintaining
thestability of
theinitial
platelet plug.
Finally, in
thefinal
phase,
fibrinolysis
occurs.
The
fibrin
clot
undergoèsàn
orderly
processof
degradation. 15In
addition,
hemostasisrelies
to
a considerable extenton the timely and
adequateconstriction
of
bleeding vessels. These events arecontrolled by
prostaglandlnsand
endothelins, which appears
to
be
pouerful
vasoactive substances,
and
their
production
isregulated
by
circulating
oestrogen and
progesteroneconcentratlons.
Endothelins,
locatedpredominantly
at theendometrial-myometrial
junction,
act aspowerful
vasoconstrictors'Tissue endothelin concentration
may
interact with
relaxing factors
such asendothelium-derived relaxing
factor IEDRF),
alsoknown
asnitric
oxide.eFor
menstrual bleeding
to
cease, theremust
berepair
of
the epithelium
and vascular endothelium initiated
from the remaining
basal layers
of
endometrium. In
addition,
local
factors
arelikely to play
acentral
rolein
this
reparative
and
angiogenesis process.
It
is suggested,that cells
or cellular
products
from
residual areasof
the
functional
layer
areimportant
in
normal
endometrial remodelling
and
repair'
Regenerationbegins ',vithin
24
hours and
is
complete within
4-5davs
of
the
onset
of
menstruation'
Growth
factors in
Med
I
Inàc*t:
the
endometrium
may
be
crucial to
the
speed
andefficacy
of
the
reparative
and
angiogenesis
process-These growth factors are: epidermal growth
factor
(EGD, transforming growth factors
(e.g.
TGF-beta)-platelet-derived endothelial growth
factor/thymidine
phosphorylase
(PD-ECGFÆP), tumor
necrosisgrowth
factors,
and vascular
endothelial
growth
factor
(VEGD.t6 The
secretionof potent endometrial growth
factors,
such asVEGF,
basic
fibroblast growth
factor
(bFGF) and EGF, is influenced
by ovarian
steroidhormonesn'lu
Ho*"u"t,
it
is
unclear how
important
any direct effect
of
oestradiol may be on
epithelial
prÀliferation
and repair.eFACTORS
ASSOCIATED WITH MENORRHAGIA
Endometrial bleeding
associatedfactor
(ebaf)
A
novel
human gene,endometrial bleeding
associatedfactor (ebaf),
was reported.
This gene is
located
onhuman chromosome
I
at
band
q42.1.11In
normal
human
endometrium,
this
gene
is
transiently
expressed
before
andduring
menstrualbleeding
in
thestroma
without
any
significant expression
in
the endometrial glands or endothelial cells.In
endornemurn-strong expression
of
this
gene.is
a-isociared
u.ithabnormai endometrial
bleeding.'
Therefore.
ebai
mrght have a
role
in
menorrhagia.\tigratory
leucocges. macrophages and mast
cells\{igratory
leucocrtes.
macrochages.
ard
mast
cellsmav contribuæ to the
mechanismof
excessive blood
loss.
Àn
ercessire
leuce-ye
inÏ-rltrate
has
been associatedu'ith
the copperintra-utenne
device. oneof
the
several causesof
menonhagia.
\lacrophages
marrelease
platelet-activating
factor
rPAF
anoprostaglandin
E,
both
of
*
hich are
potentvasodilators,
and
mast
cells may
secrete
a
range
of
vasoactive
substances,all of which could
au-ementmenstrual
blood
loss.Mast cells may also
play
arole
in
the defective
haemostatic process.They
have beenshown
to
degranulate pre-menstrually
to
secreteheparin,
histamin
and other
substances. Heparin
stimulates
endometrial
fibrinolysis
via
the secretionof
tissue plasminogen activator,
and
histamin
causesendothelial
cell
contraction, creating gaps
betweenvascular endothelial
cells to allow
increased vascularVol 10, No 2, April
-
June 2001Lysosomal
enzymesEndometrial
lysosomal enzyme
activity
in
womenwith
ovulatory dysfunctional uterine bleeding
andmenorrhagia secondary
to
intrauterine device
(IIID)
use
is
increased. Excessive
or
prolonged
tissue degradation dueto
lysosomal
enzymeactivity
such asmatrix metalloproteinases could result
in
increased orprolonged
menstrual bleeding.eProstaglandins
and endothelins
Ovulatory dysfunctional
bleeding
is
associatedrvith
ashift in
the
ratio of
endometrial
vasoconstnctin-s PGF2cr
to
vasodilatory PGE2,
and an increasein
thetotal
concentration
of
prostaglandins. There
is
also
anincreased capacity
for
myometrial generation of
prostacyclin
(PGI2)
from
endometrial
precursors.PGI2
is a
vasodilatory prostaglandin and
a
potenttnhibitor of
platelet aggregation.
Successful treatmentof
ovulatory
dysfunctional
uterine
bleeding
with
prostaglandin
sythetaseinhibitors
leadsto
areduction
in
endometrial concentrations
of
PGF2U and
PGE2,and inhibits the binding
of
PGE
to
its
receptor.
In
addition,
in
women
with
menorrhagia,
there
is
anincrease
in
prostaglandin receptor
concentrations,Therefore, endometrial
tissue may be more responsiveto the action of the increased vasodilatory protaglandins.
Endothelin has potent vasoconstrictor
and
growth
factor properties, and
has been proposed
to
have
aparacrine
role in
the regulation
of
uterineblood
flow.
In
menorrhagia.
endothelin
is
reduced
in
glandular
epithelium.ls
Its
concentration
in
the endometrium
of
women
with ovulatory
dysfunctional uterine
bleedingmay be
depressedsubstantially. Disrupted
patternsof
endothelin production may prolonged
or
increasedmenstrual
blood
loss.eIn
addition,
its
metabolizing
enzyme,
neutral
neuropeptidase,
is
increased
in
theglandular epithelium
of
women
with
menorrhagia.Therefore, endothelin
and
neutral
neuropeptida-'emight
be afactor
in
menorrhagia.rEGrowth factor receptor
Angiogenic grou'th iactors play
a
cniical role in
thecyclic
gro*rh and
va-.cularizatron
of
normal
endometrium.
Fibrobla-:t
grourh
Îàctor
receptor-l
(FGF-R1) colocalized r'.'ith
ba-sici-ibroblast cro*th
factor
(FGF-2t.
FGF-RI
is
markedll' reduced
or
absent
in
u'omen
*.ith
menorrhagra
throughout
thecycle relative to that
of
normal clclin_e u'omen. This
suggests
that
FGF-Rl
is
critical
for
endometrial
Menstruation and
menonhagia
125'maturation'
and
regeneration
of
the
normal
endometrium
followrng
menstruation. leFibrinolysis
and hemostatic
processActive
fibrinolysis
within
the normal
endometrium
and
uterine
cavity
at menstruation prevents
organizedclot
formation and the
development
of
intrauterine
adhesions.
Ovulatory dysfunctional
uterinebleeding
isassociated
with an
increased
in
endometrialfibnnolysis,
and
an
increasedconcentration
of
tissueplasminogen
activator.
Overactivation
of
thesehbrinolytic
mechanismmay
unbalance the haemostaticsystem,
and may result
in
excessive
blood loss.
In
addition, quantitative
or
qualitative defect
in
either phaseof
hemostatic
processis
important
to
consider when evaluating womenwith
menorrhagia.Changes
in endometrial blood flow
In
women
with
ovulatory
dysfunctional
uterinebleeding
thereis
asignificant
increasein
endometrial
blood
flow
compared
with
normal, and this
is
seenduring the period
of
raised oestradiol secretion
in
thefollicular
phase.
There
is
a
subsequent
decreasein
flow in
the secretory
phase,with a
late
premenstrualrise equivalent
to
that
seen
in
normal cycles.
It
isunclear
whether the increasedendometrial
flow in
thefollicular
phase hasany
direct
influence
on
menstrualblood
loss.eCONCLUSION
The
mechanism
of
normal
menstruation
is
precededby
endometrial tissue breakdou.n
and bleeding,
andfolloq'ed
by
hemostasis
and
repair.
Excessivemenstrual
blood
lossmight
be
dueto
the abnormality
in
the
rhe
role
of
leucoc)'tes,
macrophagesand
mastcells-
in
the
concentration
of
lysosomal
enzymes-prt
snglandins
and
its
receptors,
and
endothelins.Funhermore
it
may
be
due
to
overactivation
of
endomerrial
fibrinolysis
mechanism
and
changes in
endometrial blood
flow.
REFERENCES
I
Rees MCP. Factors controlling menstrual blood volume.In:
d'Arcangues, FraserIS,
Newton FJR,Odlind
V,editors. Contraception
and
mechanismof
endometrial bleeding. New york: Cambridge; 1990:ll'7-39.126
3.
4.
7.
Pawitan
Kouides
PA.
Femaleswith
von Willebrand disease: 72years as silent majority. Haemophilia 1998;4:665-76.
Pepperell
RJ. Management
of
dysfunctional uterinebleeding
in
the reproductive
age. In: Saifuddin AB,Affandi
B, Wiknjosastro
GH, editors. Women's health.Recent advances in the Asia-Oceania region. Jakarta: Bina
Pustaka Sarwono Prawirohardjo; 1995. p145-7.
Fraser
IS. Treatment
of
menorrhagia. Baillere's ClinObstet Gynecol 1989 :3 :39 | - 402.
Brenner RM, Rudolph L, Matrisian L, Slayden OD,
Non-human primate models:
artificial
menstrual cycles,endometrial
matrix
metalloproteinasesand
s.c.endometrial grafts.
In:
FindlayJ,
Affandi
B,
editors.Menstrual disorders and progestin contraception. Human
reproduction 1996, 1
I
suppl 2:150-64.Rice-Evans C, Cooke
B.
Oxygen radicals, bleeding andtissue
injury.
In: d'Arcangues, Fraser
IS,
Newton FJR,Odlind
V,
editors. Contraceptionand
mechanism ofendometrial bleeding.
New York:
Cambndge; 1990.p41 1-30.
Findlay
JK,
ChernyRA, Salamonsen
LA.
Paracrineinteractions amongst
cells
of
the
endometrium.
In:d'Arcangues, Fraser IS, Newton FJR, Odlind
V,
editors.Contraception and mechanism
of endometrial bleeding.
New york; Cambridge; 1990. p253-65.
Fraser
IS,
Hickey
M,
SongJ-Y.
A
comparison ofmechanisms underlying disturbances
of
bleeding causedby
spontaneous dysfunctionaluterine
bleeding
orhormonal contraception. In: Findlay J, Affandi B, editors.
Menstrual disorders and progestin contraception. Human
Reproduction 1996,
1l
suppl 2:165-78.Kamat
BR,
IssacsonPG.
The immunocytochernical
distribution
of
leucocyte
subpopulationsin
humanendometrium. Am J Pathol 1987l,127:66-73.
Clark DA, Daya S. Macrophages and other migratory cells
in endometrium: relevance
to
endometrial bleeding. In:d'Arcangues, Fraser IS, Newton FJR, Odlind
V,
edotors.Contraception and mechanism
of endometrial bleeding.
New york: Cambridge; 1990. p363-382.
Med J Indones
Comillie FJ, Brosens
IA,
Marbaix E, Vael TH, BaudhuinP, Courtoy PJ. A biochemical study of lysosomal enz)'mes
in control and levonorgestrel-treated human
endometna:analysis
of
total activity and evidencefor secretion.
In;d'Arcangues, Fraser IS, Newton FJR, Odlind
V, editors
Contraception and mechanism
of endometrial bleeding
New york: Cambridge; 1990 p383-410,
Eeckhout
Y
Tissue breakdounIn:
d'Arcangues. FraserIS,
Newton FJR, Odlind\/.
editors. Contraception andmechanism
of
endometrial
blealine
\erç
r ork:Cambridge; 1990. p43240
Ludwig
H, Metzger
H,
Frauli\t.
Endorrern-:5- !r:>-.r.remodelling and regeneration ln: d'.*.-engu:s. Fr:-=r IS.
Newton FJR, Odlind
V,
editors
Conraceptir'n i-1rmechanism
of
endometrial
bleedin-e.\eu'
ioik:
Cambridge; I 990. p44l-66.
Ewenstein
BM.
The pathophysiolog]'
oi
bleedingdisorders presenting as abnormal uterine bleedin-e
.{m
JObstet Gynecol 1996:17 5:770-i .
Smith
SK. Angiogenesis, vascular endotheliaJ gr..'.r-:
factor and the
endometrium.Hum
Reprod
Upcl:
1 998;4:509-19.
Kothapalli R, Buytksai I, Wu SQ, Chegini N, Tabibzadeh
S.
Detectionof
ebaf,
a
novel
human geneof
thetransforming growth factor beta superfamily association
of
gene expression with endometrial bleeding.J
ClinInvest I 997;99 :2342-50.
Marsh
MM,
MalakootiN,
Taylor
NH,
Findlay
JK.Salamonsen LA. Endothelin and neutral endopeptidase in
the endometrium
of
womenwith menorrhagia. Hum
Reprod 1997 :12:2036-40.
Sangha
RK,
Li
XF, Shams
M,
AhmedA.
Fibroblastgrowth
factor
receptor-1is
a
critical component
fo.endometrial remodeling: localization and expression of
basic fibroblast growth factor and
FGF-RI
in
humanendometrium during the menstrual cycle and decreased
FGF-R1
expression
in
menorrhagia.t,ab
Invest1997 77:389-402
t2
5
6
t3.
17 t4
15
t6
18
l9
l0