SYNTHESIS OF CHICKEN EGGSHELLS-BASED
β
-TRICALCIUM PHOSPHATE BIOCERAMICS AND
THEIR BIOCOMPATIBILITY TEST AS A TOOTH FILLER
NUR AISYAH NUZULIA
GRADUATE SCHOOL
BOGOR AGRICULTURAL UNIVERSITY BOGOR
STATEMENT ON THESIS
I hereby declare that the thesis of “Synthesis of Chicken Eggshells-Based β-Tricalcium Phosphate Bioceramics and Their Biocompatibility Test as A Tooth
Filler” is my work with the direction of the supervising committee and has not
been submitted in any form to any college. Source of information derived or quoted from the work published or not published by other authors mentioned in the text and listed in the reference at the end of this thesis.
I hereby assign the copyright of my thesis to Bogor Agricultural University.
Bogor, April 2014
Nur Aisyah Nuzulia
RINGKASAN
NUR AISYAH NUZULIA. Sintesis Biokeramik β-Tricalcium Phosphate Berbasis
Cangkang Telur Ayam dan Uji Biokompatibilitasnya sebagai Tooth Filler.
Dibimbing oleh KIAGUS DAHLAN, GUNANTI, dan SRIHADI AGUNGPRIYONO.
Preservasi tulang alveolar setelah ekstraksi gigi merupakan langkah penting dalam praktek gigi sebelum pemasangan implan gigi. Penelitian ini melaporkan tentang sintesis biokeramik β-Tricalcium Phosphate (β-TCP) dan uji
biokompatibilitasnya sebagai tooth filler untuk mempertahankan dimensi alveolar ridge dan merangsang proses penyembuhan tulang di daerah hilangnya gigi.
Biokeramik β-TCP dihasilkan secara langsung melalui metode presipitasi cangkang telur ayam yang telah dikalsinasi sebagai sumber kalsium dan asam
fosfat. β-TCP tersebut dicampur dengan kitosan 2 % untuk mendapatkan β-TCP dalam bentuk pelet yang digunakan dalam pengujian biokompatibilitas. Pengujian
biokompatibilitas β-TCP dilakukan pada gigi seri bawah kelinci dan domba sebagai hewan uji.
Dalam studi ini, hasil x-ray diffraction (XRD) menunjukkan bahwa pola
puncak XRD cangkang telur ayam yang dikalsinasi pada suhu 1000oC bersesuaian dengan fase kalsium oksida (CaO). Adapun fase CaO bertransformasi menjadi fase Ca(OH)2 selama masa penyimpanan lebih dari tiga bulan. Profil XRD juga membuktikan bahwa biokeramik β-TCP yang dihasilkan memiliki pola yang sama dengan referensi data base dan β-TCP komersil yang tersedia. Spektra fourier transform infrared (FTIR) sampel konsisten dan memperkuat hasil karakterisasi
XRD. Selain itu, rasio Ca/P sebesar 1.48 yang diperoleh dari hasil atomic absorption spectroscopy (AAS) menunjukkan kemurnian sampel yang sesuai
dengan standar biokeramik β-TCP. Adapun pola puncak XRD dan spektra FTIR yang berulang jelas menunjukkan sifat reproducibility sampel.
Alveolar socket domba yang diisi dengan produk β-TCP menunjukkan hasil
karakteristik yang diharapkan. Hasil radiografi sinar-x pada rahang domba menunjukkan tidak adanya perubahan mesio-distal di edentulous area. Selain itu,
hasil histologi juga konsisten terhadap hasil radiografi dan gambaran makroskopik yang dibuktikan dengan adanya pembentukan tulang baru. Namun, hasil histologi dari rahang kelinci menunjukkan hal yang berlawanan dengan hasil radiografi dan gambaran makroskopik. Hasil histologi pada rahang kelinci menunjukkan tidak adanya pertumbuhan gigi pada kelinci yang dibuktikan dengan daerah ekstraksi yang terisi penuh oleh lipid dan kolagen. Pengujian biokompatibilitas biokeramik
β-TCP menunjukkan bahwa produk β-TCP mampu meminimalis hilangnya tulang serta mampu mempertahankan dimensi alveolar ridge.
SUMMARY
NUR AISYAH NUZULIA. Synthesis of Chicken Eggshells-Based β-Tricalcium Phosphate Bioceramics and Their Biocompatibility Test as A Tooth Filler. Supervised by KIAGUS DAHLAN, GUNANTI, and SRIHADI AGUNGPRIYONO.
Preservation of alveolar bone following tooth extraction is an important step in dental practices before dental implant placement. This study reported synthesis of β-Tricalcium Phosphate (β-TCP) bioceramics and its biocompatibility test as a tooth filler to maintain the desired alveolar ridge dimension and stimulate the bone healing processes in the area of tooth loss. β-TCP bioceramics was resulted directly from precipitation of calcinated chicken eggshells as calcium source and phosphoric acid. The corresponding β-TCP was mixed with chitosan 2% to get
β-TCP pellet for biocompatibility testing. Biocompatibility testing of β-TCP on lower incisor was performed in rabbits and sheep as animal model.
In this study, the x-ray diffraction (XRD) profiles showed that 1000 oC calcinated chicken eggshells correspond to calcium oxide (CaO). There was also transformation phase of CaO into Ca(OH)2 observed after more than three months stored. The XRD profiles proved that obtained β-TCP bioceramics by precipitation has the same pattern with that of reference data base and commercial
β-TCP. Fourier transform infrared (FTIR) spectra were completely consistent with the result of crystallization processes observed by XRD measurements. Moreover, the purity of the product shown by the atomic absorption spectroscopy (AAS)
resulted in Ca/P ratio of 1.48 which was closed to that of standard β-TCP ceramics. The repeatable XRD peaks patterns and FTIR spectra obviously showed the sample reproducibility.
Sheep’s alveolar socket that is filled with the corresponding β-TCP demonstrated the expected results. The x-ray radiograph of sheep’s jaw indicated
no change on mesio-distal of the edentulous area. Moreover, the histological results were completely consistent with the x-ray radiograph and macroscopic
result which is proved by the formation of new bone. In the contrary with sheep’s
result, histological results showed no tooth growth on rabbit proved by the presence of lipid and collagen on the extraction site. This result did not correspond to the x-ray radiograph and macroscopic results that demonstrated the tooth growth and erupted tooth. It is clear that the corresponding β-TCP could minimize the bone loss and maintain the dimension of alveolar ridge.
© Copyright of IPB, the year 2014
Copyright reserved by the law
Forbidden to quote part or all of these writings without including or mentioning the source. Citing is only for educational purposes, research, writing papers, drafting reports, writing criticism, or review an issue, and citing it does not harm the interests of fair Bogor Agricultural University.
Thesis
submitted in partial fulfillment of the requirement for Master Degree
in
Biophysics Program
SYNTHESIS OF CHICKEN EGGSHELLS-BASED
β
-TRICALCIUM PHOSPHATE BIOCERAMICS AND
THEIR BIOCOMPATIBILITY TEST AS A TOOTH FILLER
GRADUATE SCHOOL
BOGOR AGRICULTURAL UNIVERSITY BOGOR
2014
Thesis title : Synthesis of Chicken Eggshells-Based β-Tricalcium Phosphate Bioceramics and Their Biocompatibility Test as A Tooth Filler Name : Nur Aisyah Nuzulia
ID : G751110071
Approved by
The Commission of Supervisors
Dr Kiagus Dahlan Supervisor
Dr drh Gunanti, MS
Co-Supervisor drh Srihadi Agungpriyono, PhD Co-Supervisor
Certified by:
Head of Biophysics Graduate Program
Dr Agus Kartono
Dean of the IPB Graduate School
Dr Ir Dahrul Syah, MSc Agr
Tanggal Ujian:
PREFACE
Alhamdulillah, all praises be to Allah because by His blessing I could finish this thesis successfully, which is one of biophysics project of Physics Department funded by Decentralization Research Fund Project 2013. This research
synthesized β-Tricalcium Phosphate as a tooth filler from chicken eggshells as the calcium source that is expected to be applied in dental surgery. The result of this research has been presented in International Seminar on Sciences 2013 in Bogor on November 15-17, 2013.
The author would like to express sincere appreciation of Dr Kiagus Dahlan, Dr drh Gunanti, MS, and drh Srihadi Agungpriyono, PhD for the guidances and advices in directing the research, drh Deni Noviana as external examiner for the valuable suggestion and criticism, Dr Akhiruddin Maddu as representative of Biophysics Program for the suggestion, Setia Utami Dewi, M.Si for the help in the laboratory sample preparation and useful discussion. I also thank to drh. Riki Siswandi, drh. Heryudianto Vibowo for the help in biocompatibility test, Biomaterials team and FVM team for the cooperation of this research. The author is also grateful to the Ministry of Education and Culture for awarding her a scholarship under the programme Beasiswa Unggulan and Hibah Desentralisasi for funding this research. So many thankful to Yessie Widya Sari and Husin Alatas for the useful discussion, 2011’s Biophysics, Hafif, Buan, Wisma Intan family, colleagues and students at Nurul Fikri for the support and kindness. To all of these people, I owe its whole-hearted gratitude that impossible to describe.
I am also thankful to Bapak, Ibu, Mas Aix, Mbak Ria, Adek Agus, Adek Aris, Adek Abdu, Kimia and whole families, for the pray and support in bringing about
this thesis. I give all respect that impossible to describe, thank you so much. Nonetheless, I also welcome any critical feedback and advice from readers in order to maintain it as successful project. I do hope this thesis could be useful.
Bogor, April 2014
TABLE OF CONTENS
TABLE LIST vi
FIGURE LIST vi
APPENDICES LIST viii
1 INTRODUCTION 1
Background 1
Hypotheses 3
Objective 3
Benefit 3
2 MATERIALS AND METHODS 3
Place and Time Schedule 3
Materials and Equipments 3
Experimental Method 4
Calcination of Eggshells 4
β-TCP Synthesis 4
Biocompatibility Testing 5
3 RESULTS AND DISCUSSION 6
Calcination of Chicken Eggshells 6
Chicken Egshells-Based β-Tricalcium Phosphate 7
Biocompatibility Test on Rabbits 12
Biocompatibility Test on Sheep 19
4 CONCLUSION AND SUGGESTION 29
Conclusion 29
Suggestion 29
REFERENCES 29
TABLE LIST
1 β-Tricalcium Phosphate Bioceramics Codes 8
2 Precipitate Mass of β-Tricalcium Phosphate Bioceramics 8 3 Lattice parameter and accuracy of β-TCP products 8 4 AAS results of β-TCP products 11
5 Mesio-distal of the edentulous area 23
FIGURE LIST
1 Calcination process of chicken eggshells 5
2 β-TCP synthesis through precipitation method 5
3 X-ray diffraction (XRD) pattern of calcinated chicken eggshells at 1000oC for 5h (a) and peak matching of calcinated chicken eggshells
with database PCPDF 37-1497 (b) 7
4 XRD pattern of β-TCP samples with five repetition and commercial
TCP, RTR Septodont 9
5 FTIR spectra of β-TCP samples with five repetition 10 6 XRD pattern of calcinated chicken eggshells after three months stored 11 7 Temperature of rabbits both control (untreated) and treated with β-TCP
product during the healing process post extraction 12 8 Pulse rate of rabbits both control (untreated) and treated with β-TCP
product during the healing process post extraction 13 9 Respiration rate of rabbits both control (untreated) and treated with β
-TCP product during the healing process post extraction 13 10 The laterolateral (LL) view radiograph of rabbit’s jaw pre-operation.
Scale bar 12mm 14
11 The laterolateral (LL) view radiograph of rabbit’s jaw that unfilled with β-TCP product (control) at day+0 post operation(a). There was tooth growth that is shown by increasing opacity of extracted area at day+7 post operation (b) and the presence of margin and higher opacity of alveolar socket at day+30 post operation indicates the formation of
tooth germ (c). Scale bar 12mm 14
12 The laterolateral (LL) view radiograph of rabbit’s jaw that filled with β -TCP product. There was high opacity at day+0 post operation due to the similar inorganic of sample with bone (a) and gradually decreased because of the resorption of sample by body fluid at day+7 post operation (b). The formation of complete tooth was showed by clear margin on the extracted area at day+30 post operation (c). Scale bar
10mm 15
13 Macroscopic results of rabbit’s mandible at Day+30 harvesting. Sketch
of rabbit’s mandible was shown to describe the anatomy of lower jaw. There was no presence of erupted tooth on control (b) but the erupted tooth on the treatment showed the complete tooth growth (c) 15 14 A lateral section photomicrograph of lower rabbit incisor (filled with β
blood clot in the middle portion of socket indicate blood vessel rupture(b). Hematoxylin Eosin Stained, x40, Scale bar 100μm 17 15 A lateral section photomicrograph of lower rabbit incisor (control) at
Day+30 post operation. Tooth growth was indicated by the presence of pulp chamber with odontoblastic layer ( ) in the apical portion of socket (a) and fully filled with fat cells in the middle portion (b).
Hematoxylin Eosin Stained, x40, Scale bar 100μm 18
16 Temperature of sheep both control (untreated) and treatments (filled
with β-TCP product and commercial β-TCP) during the healing
process post extraction 19
17 Pulse rate of sheep both control (untreated) and treatments (filled with
β-TCP product and commercial β-TCP) during the healing process post
extraction 20
18 Respiration rate of sheep both control (untreated) and treatments (filled
with β-TCP product and commercial β-TCP) during the healing
process post extraction 20
19 The radiograph of dorsoventral (DV) position of sheep ’s jaw pre
-operation. Scale bar 15mm 21
20 The radiograph of dorsoventral (DV) position of sheep’s jaw of control
(unfilled with sample). There was large mesio-distal and low opacity on the extraction site ( ) at (a) day+0 post operation that become narrower at day+7 post operation (b) and there was high alveolar bone resorption shown by significant change on mesio-distal at day+30 post operation
(c). Scale bar (a) 10mm (b) 8mm (c) 11mm 21
21 The radiograph of dorsoventral (DV) position of sheep (filled with
β-TCP product). High opacity on the extracted area ( ) at day+0 post operation showed the alveolar socket filled with sample (a). The same width of mesio-distal indicated there was no alveolar bone resorption day+7 post operation and there was resorption of sample showed by decreasing opacity (b). Resubstituting process of sample and preservation of alveolar ridge was clearly showed by the same width of mesio-distal on the edentulous area at day+30 post operation (c). Scale
bar (a) 3mm (b) 10mm (c) 7mm 22
22 The radiograph of dorsoventral (DV) position of sheep (filled with
commercial β-TCP, RTR Septodont). High opacity on the extracted area ( ) at day+0 post operation showed the the alveolar socket filled with commercial product (a). Decreasing opacity on the extracted area showed the resorption of materials at day+7 post operation (b) and there was alveolar bone resoption on the horizontal direction at day+30 post operation(c). Scale bar (a) 7mm (b) 7mm (c) 10mm 22 23 Macroscopic results of sheep’s mandible at day+30 harvesting.
Narrower distal occurred on control (a) but the width of
mesio-distal remained same on the treatment filled with β-TCP product (b). There was alveolar bone resorption in the horizontal direction on the
treatment filled with commercial β-TCP, RTR Septodont (c) 23 24 A lateral section photomicrograph of alveolar bone healing (control) at
and a cavity in the middle portion (b). Hematoxylin Eosin Stained, x40,
Scale bar 100μm 24
25 A lateral section photomicrograph of alveolar bone healing (filled with
β-TCP product) at Day+30 post operation. Biocompatibility of tooth
filler seen from infiltration of fibrous connective tissue within β-TCP product in the middle portion of extracted area (a). Bone remodeling occurs in the formation of woven bone (b) via intramembranous bone development under layer of connective tissue (c) in the upper portion near gingiva. Hematoxylin Eosin Stained x40 (a, b) x100 (c). Scale bar
100μm 26
26 A lateral section photomicrograph of alveolar bone healing (filled with
commercial β-TCP) at Day+30 post operation. Biocompatibility of tooth filler seen from the presence of vessel in the upper portion of extracted area but the presence of blood clot indicated vessel rupture(a). Alveolar bone resorption occurs in horizontal direction shown by the presence of collagen and fibrous connective tissue near gingiva (b). Repairing process by connective tissue in the middle portion shown by the presence of vessel (c). Hematoxylin Eosin Stained x40. Scale bar
100μm 28
APPENDIX LIST
1 Flow chart of the research 34
2 Joint committee on powder diffraction standards (JCPDS) database of
Calcium Oxide 35
3 Joint committee on powder diffraction standards (JCPDS) database of β-Tricalcium Phosphate 35 4 Joint committee on powder diffraction standards (JCPDS) database of
Calcium Phosphate (Ca2P2O7) 36
5 Joint committee on powder diffraction standards (JCPDS) database of
1
INTRODUCTION
Background
Oral health is essential to general health and quality of life. It means a condition of being free from any diseases that may impact to our whole body. Based on the report of World Health Organization (WHO) website on April 2012, the most common oral diseases are dental cavities, periodontal (gum) disease, oral cancer, oral infectious diseases, trauma from injuries, and hereditary lesions. Worldwide, dental cavities happen to 60–90% of school children and nearly 100% of adults and severe periodontal (gum) disease is found in 15–20% of middle-aged (35-44 years) adults. Complete loss of natural teeth is widespread and particularly affects older people (WHO 2013). In Indonesia, health survey conducted by Ministry of Health Republic of Indonesia in 2001 showed that about 70% of Indonesian aged over than 10 years old have tooth decay (Pratiwi et al. 2013).
Among these diseases, dental cavities and periodontal disease are the major causes of tooth loss. Physiologically, there is the loss of alveolar bone as a result of human tooth loss process. The rate of alveolar bone resorption is fairly high as a reduction of about millimeters happens in the first 6 months of the healing process and two-thirds of reduction occurs in the first 3 months (Brkovic et al.
2008, Kokovic and Todorovic 2011). It is observed that over 12 months the reduction is about 50% in both horizontal and vertical directions (Brkovic et al.
2008). A very progressive bone resorption is able to reduce the dimensions of the socket and lead to the bone athropy and cause difficulty on implant or prostheses placement (Calixto et al. 2007). Therefore, preservations of bone volume in the
area of tooth loss are necessary to maintain the dimensions of the alveolar ridge. In general, grafting procedures are also necessary for improving the bone tissue ability to regenerate. For this purpose, several approaches have been attempted for defect filling and subsequent regeneration, including autogeneous and xenogeneous bone grafting and synthetic biomaterials (Coimbra et al. 2009).
Compared to autogeneous and xenogenous procedures, biomaterials eliminate problems of donor scarcity, supply limitations, pathogen transfer, and immune rejection. In the field of dental surgery, biomaterials have been used include filling of pockets and maxillary ridge augmentation, deficiencies caused by loss of dentition with advancing age or various diseases (Costa et al. 2008). The use of
filling biomaterials is one way to preserve the adequate bone volume that commonly used by clinicians.
Nowadays, bioceramics have been widely studied for orthopedic and dental applications due to their good biocompatible and osseoconductive properties (Coimbra et al. 2009). Then, because the mineral phase of bones and
teeth is composed of calcium phosphate salts, the use of some Ca-P materials for bone substitution, augmentation, and repair has gained clinical acceptance in many aspects of orthopedics and dentistry (Dai et al. 2004). Among calcium
2
polymorphs in the field of bioceramics (Shi 2004) with Ca/P ratio of 1.5 that has been shown to exhibit good biocompatibility and osteoconductivity (Ain et al.
2008 & Sader et al. 2009) in both animal and clinical studies (Oi et al. 2009)
while HA is considered as bioactive and nonbiodegradable bone replacement
materials. Moreover, β-TCP appears to be advantageous in comparison to HA for the property of absorption (Tazaki et al. 2009). The faster dissolution/resorption
of β-TCP may allow a gradual biological degradation over a period time and a progressive replacement by the natural host tissue (Coimbra et al. 2009).
β-TCP itself can be obtained from several methods in which the method of wet precipitation is one of the widely used methods (Boilet et al. 2013,
Nasiri-Tabrizi and Fahami 2013, Farzadi et al. 2011, Koç et al. 2004). In this method,
synthetic materials such as CaCO3 and Ca(NO3)2 are commonly used as calcium
source for obtaining β-TCP ceramics (Park and Bronzino 2000, Wei and Akinc 2007). Previous study reported that β-TCP can not be synthesized directly in aqueous solution (Belouafa et al. 2006). Wet precipitation in aqueous solution
would synthesize apatitic TCP, Ca9(HPO4)(PO4)5(OH). Following precipitation from aqueous solution, sintering at high temperatures above 750 C was required to transform apatitic TCP into β-TCP (Kannan et al. 2006, 2009). Different
synthesis approach applying natural resources material could obtain bioactive
β-TCP that might elicit a direct bond between the bioceramics and living host tissue. The chicken eggshells are one of natural resources that commonly considered as waste, contain much of calcium in the form of CaCO3 that potentially could be used as calcium source in synthesizing β-TCP bioceramics. The previous study showed that chicken eggshells as the corresponding calcium source could obtain the other kind of calcium phosphate (hydroxyapatite, hydroxyapatite/carbonate apatite) by precipitation method (Dahlan et al. 2012).
Therefore, it is clear that β-TCP might be produced from natural resources and synthesized directly by precipitation method.
Prior to the clinical use, biocompatibility and mechanical stability of new materials should be test under both initial in vitro and in vivo conditions (Pearce
2007). In vitro testing is used primarily as a first stage test for acute toxicity and
cytocompatibility while in vivo testing is used to demonstrate the tissue response
to materials (Arora et al. 2011). Previous study reported that calcium phosphates
derived from eggshells were nontoxic and performed good adhesion interaction to the host cells in vitro (Solihat 2011). Therefore, advanced research under in vivo
3 Objectives
1. Synthesizing β-TCP bioceramics obtained from chicken eggshells by precipitation method
2. Observing the tooth growth and the bone healing using β-TCP bioceramics and tissue response to materials by in vivo testing in rabbits and sheep
Hypotheses
1. Chicken eggshells can be used as calcium donor in synthesizing β-TCP bioceramics that biocompatible for dentistry
2. Chicken eggshells-based β-TCP bioceramics as tooth filler can accelerate the tooth growth on rabbits and stimulate the bone healing on sheep after tooth extraction
Benefits
This research is expected to obtain natural resource-based β-TCP bioceramics such chicken eggshells that generally known as waste in use as calcium donor in synthesizing of β-TCP bioceramics. It may produce biocompatible biomaterials with economical price for dentistry application in Indonesia. This will subsequently assume to decrease the dependence on importing biomaterial for bone and tooth substitution.
2
MATERIALS AND METHOD
Place and Time Schedule
This research was conducted from January 2013 through September 2013 which took place in Biophysics Laboratory-IPB in sample preparation, while biocompatibilitytesting was done in Faculty of Veterinary Medicine-IPB. Sample characterization was done in Forestry Development and Research Center-Bogor for x-ray diffraction (XRD) characterization, in Laboratory of Material Analysis-IPB for fourier transform infrared (FTIR) analysis, and in Indonesian Soil Research Institute-Bogor for atomic absorption spectroscopy (AAS). Histological test and x-ray radiography analysis was done in Faculty of Veterinary Medicine-IPB.
Materials and Equipments
4
aluminium foil, pipette, magnetic stirrer, hotplate, furnace, digital thermometer, filter paper, mortar, crucible, and vacuum.
Materials and equipments used for biocompatibility testing were rabbits and sheep as animal model, minor surgery set, dental surgery tools, anesthetic material, and surgery room for aseptical insertion. Then, materials used for histopathological examination were formalin 10%, nitric acid 5%, aquadest, aquabidest, alcohol, xylol, Hematoxilin-Eosin (HE), and Permount ®.
Experimental Methods
Calcination of chicken eggshells
The raw eggshells where their surface was initially cleaned were calcinated in an air atmosphere at 1000oC for 5 hours as seen in Figure 1. This calcination aimed to convert calcium carbonate (CaCO3) into calcium oxide (CaO). To synthesize calcium phosphate powder, the corresponding eggshells were crushed and milled in crucible then characterized by using x-ray diffraction (XRD) Shimadzu diffractometer, Cu-Kα radiation, 40 kV, 30 mA and 0,02o s-1 scanning step. The chemical reaction of eggshell calcination is given below:
β-TCP synthesis
β-TCP bioceramics was prepared by precipitation of H3PO4 as the phosphor (P) source. Then it was dropped into CaO as the calcium (Ca) source which was synthesized further at 50oC with molar ratio of Ca:P as 1.2M : 0.8M and the chemical reaction is given below:
5
The cleaned raw eggshells Calcination at 1000 oC Powder of CaO
Figure 1 Calcination process of chicken’s eggshells
Ca and P solution Precipitation Filtering Sintering
Figure 2 β-TCP synthesis through precipitation method
Biocompatibility testing
Biocompatibility test of β-TCP bioceramics as a tooth filler was performed by using rabbits and sheep as animal models. There are two 1 year old rabbits used with 2.5 kg body weight and three 1 year old sheep. All experiments were conducted along the institutional guidelines for the care and use of laboratory animals. Prior to surgery, the animal models were observed closely for a week in order to check their health status. They were maintained under identical environment, management and standard diet with ad libitum supply of drinking
water. Then, surgery was done on the lower incisor. Before surgery, rabbits and sheep were anaesthetized by injection of ketamine 10% and xylazin 10%. Under aseptical conditions, the lower incisor was extracted and immediately after tooth extraction, the alveolar socket was filled with synthesized β-TCP bioceramics that were sterilized initially by exposure to ultraviolet light. As control requirement,
other rabbit’s and sheep’s lower incisor were extracted in the same manner and unfilled with any tooth filler. Each surgery was performed under same veterinary surgeon. Then, the rabbits and sheep were housed under a climate-controlled environment in stall of animal used of FVM Bogor Agricultural University.
6
out, decalcified, and processed for paraffin embedding. The decalcification process was carried out using 10% EDTA solution and the specimens was dehydrated in ascending grades of ethyl alcohol, cleared in xylene and embedded in paraffin wax. The histopathological examination was done for observing the degree of tooth growth on rabbits and new bone formation on sheep. The histological analysis was carried out with some parameters comprised the presence and type of inflammatory infiltrate, reaction to foreign body, fibrous connective tissue, osteoblastic activity, formation of osteoid and new bone. Detailed flow chart of the research was shown in Appendix 1.
3
RESULTS AND DISCUSSION
Calcination of Chicken Eggshells
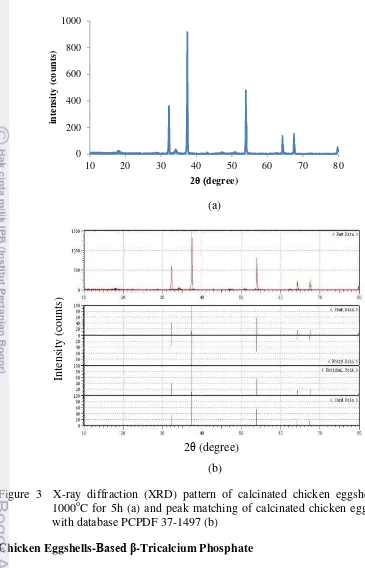
It was found that the calcinated chicken eggshells contain pure calcium oxide with mass decomposition of 48.40%. The characteristics of the corresponding calcium oxide atomic structure were examined by using XRD where its pattern is shown in Figure 3a. It was observed that the highest peaks
were on 2θ = 32.31, 37.47, 53.98, 64.27, 67.51, and 79.76. Based on data from joint committee on powder diffraction standards (JCPDS) database number
37-1497 (Appendix 2), these results clearly prove the presence of calcium oxide. Figure 3b shows that the sample pattern was match to the database, since they share similar peaks pattern. The absence of other phases in the sample pattern indicates the high purity of CaO powder. The result in this study had higher accuracy of CaO pattern compared to that of other studies (Saeed et al. 2011,
Balazsi et al. 2007). The XRD pattern of 950 oC calcined chicken eggshell
corresponds to CaO with Ca(OH)2 as the majority phase according to Miller indices of (1 0 0), (1 0 1), and (1 0 2) (Saeed et al. 2011). In addition to Ca(OH)2,
7
(a)
(b)
Figure 3 X-ray diffraction (XRD) pattern of calcinated chicken eggshells at 1000oC for 5h (a) and peak matching of calcinated chicken eggshells with database PCPDF 37-1497 (b)
Chicken Eggshells-Based β-Tricalcium Phosphate
Synthesis of chicken eggshells-based β-Tricalcium phosphate (β-TCP) by precipitation method was performed five times with the sample codes are shown
in Table 1. The sintering temperature used to obtain β-TCP bioceramics was 1000oC. Referring to the previous study it was reported that the required temperature to start phase transformation of calcium phosphate into β-TCP is 760oC and get stable at 1125oC (Salahi and Heinrich 2003, Shi 2004). The result of this process was a white powder with mass precipitates are shown in Table 2.
8
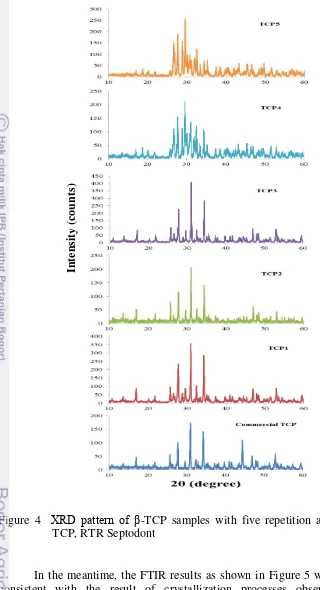
In contrary with Belouafa et al. (2006), β-TCP could be synthesized
directly in aqueous solution as shown from its XRD profile (Figure 4). The three important highest peaks pattern of the first three samples (TCP1, TCP2 and TCP3) were completely similar to the peaks of β-TCP database number 09-0169 (Appendix 3). It was proved by high intensity on 2θ around 27.84, 31.06, and
34.41. The XRD peaks pattern of the corresponding β-TCP samples was the same with that found by heating the apatitic TCP at temperature above 750oC (Kannan
et al. 2006, 2009). Moreover, it was similar with that one of commercial TCP
(RTR Septodont France) and sintered commercial TCP powder (Sader et al. 2009).
The determined lattice parameters of the corresponding β-TCP samples are shown in Table 3 by fitting the peaks of identified reflections from the XRD pattern using Cohen Method. It is clearly showed the formation of β-TCP bioceramics proved by rhombohedral structure with lattice parameters correspond to the reference a=b=10.42Å and c=37.38Å (Shi 2004).
Table 1 β-Tricalcium Phosphate Bioceramics Codes
Sample Code
Table 2 Precipitate Mass of β-Tricalcium Phosphate Bioceramics
Sample Precipitate Mass (g)
Table 3 Lattice parameter and accuracy of β-TCP samples
Sample Lattice parameter (Å) Accuracy (%)
a c a c
TCP1 10.44 37.44 99.78 99.84
TCP2 10.39 37.22 99.71 99.57
9
Figure 4 XRD pattern of β-TCP samples with five repetition and commercial TCP, RTR Septodont
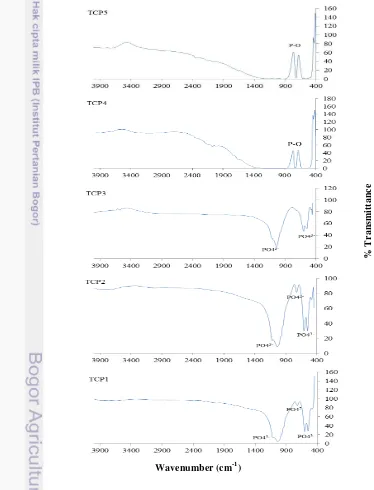
In the meantime, the FTIR results as shown in Figure 5 were completely consistent with the result of crystallization processes observed by XRD measurements given by Figure 4. The broad IR band of FTIR spectra around 540-580 cm-1, 690 cm-1 and 1010 cm-1 indicates the formation of a typical TCP structure containing PO43- band. The absence of hydroxyl band in corresponding samples around 3500 cm-1 shows better characteristics compared to Salahi and Heinrich (2003) that used synthetic materials. The FTIR result of β-TCP obtained
In
te
n
sity (
cou
n
10
from synthetic materials was not pure that clearly showed by the existence of hydroxyl band in sample sintered in 1000 oC (Salahi and Heinrich 2003). Other study done by Ain et al. (2008) showed the presence of another phase
(hydroxyapatite) on the XRD results. It means that β-TCP bioceramics has been formed directly in aqueous solution by precipitation method which also been proved with a better characteristics compared to Salahi and Heinrich (2003) and Ain et al. (2008) that used synthetic materials. Since the chicken eggshells are considered as a raw material, the corresponding β-TCP samples should be more economical. It is obvious that the corresponding β-TCP samples is reproducible due to the repeatable XRD peaks patterns and FTIR spectra.
Figure 5 FTIR spectra of β-TCP samples with five repetition
%
T
ra
ns
mitt
a
nce
11 On the other hand, it should be noted that the last two samples (TCP4 and TCP5) correspond to the other phase of calcium phosphate (Ca2P2O7) with database number 09-0346 as seen in Appendix 4 which is occured probably due to
the different calcium source used in synthesizing β-TCP. The corresponding calcium source was calcinated chicken eggshells powder that had been stored for more than three months. This result is obviously depicted by its XRD profile as shown in Figure 6 that refers to the phase of Ca(OH)2 with database number 44-1481 (Appendix 5). It is exhibited that there is phase transformation caused by interaction of CaO with the environment during the storage at non-vacuum condition. The existence of OH- functional group on Ca(OH)2 tends to release and to form another phase of calcium phosphate as also been observed in the previous study that synthesized biphasic calcium phosphate (BCP) with CaO and Ca(OH)2 as raw materials (Dahlan et al. 2011). Their research also showed that TCP only
appeared on samples synthesized with CaO that probably caused by the difference binding type of each precursor.
Furthermore, to verify the purity of the products, the β-TCP samples were characterized by using atomic absorption spectroscopy (AAS). Based on the AAS results, it is shown that Ca/P ratio of TCP3 corresponded to β-TCP bioceramics as shown in Table 4. This result confirmed the XRD results and in accordance with the theory of Ca/P ratio of β-TCP bioceramics (Shi 2004). Nevertheless, the AAS measurements of TCP1 and TCP2 are not consistent with the XRD results. This inconsistency probably occured due to the limited number of samples powder that used for the AAS analysis.
Figure 6 XRD pattern of calcinated chicken eggshells after three months stored
0
Sample Percentage Ca/P ratio
12
Biocompatibility Test on Rabbits
Biocompatibility is the ability of a material to perform with an appropriate host response in a specific application (Shi 2004). Biocompatibility test of β-TCP as tooth filler on rabbits was performed to observe the tooth growth post extraction. Biocompatible properties of new materials could be analyzed from several examinations such as physical examination, x-ray radiographic analysis, and histological evaluation.
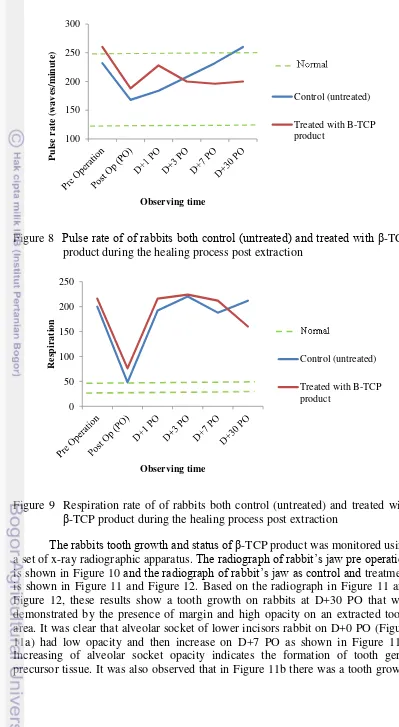
The physical examination on rabbits that is observed for 30 days showed that β-TCP product was biocompatible and non-toxic. There were no significant influence on rabbits filled with β-TCP product and unfilled (control) in temperature and pulse rate post operation. The results of physical examination on rabbits were in normal range of each parameter (Figure 7 and Figure 8). The normal temperature of rabbit is 38.9-40.5oC and its normal pulse rate is 120-250 waves/minute (Kelly 1984). There was temperature decreasing on rabbit filled with β-TCP product post operation that might be influenced by the injection of anaesthetic material. Thus, the temperature was in normal range on D+1 PO and days after. It is worth to note that filling β-TCP product do not interfere the physiological body temperature of rabbit. Nevertheless, the respiration of rabbits as seen in Figure 9 both control and treatment were not in normal range that is 30-45 (Kelly 1984). It might be caused by condition of rabbits that are stress during data collection process and made them excited. An increased demand for oxygen after exercised might increase respiratory frequency (Kelly 1984). Moreover, the longer period of expiration in healthy animals depends upon whether the animal is relaxed and resting, or has recently been excited or excercised. In short, postextraction preservation by β-TCP as a tooth filler do not interfere the physiological condition of rabbit.
Figure 7 Temperature of rabbits both control (untreated) and treated with β-TCP product during the healing process post extraction
13
Figure 8 Pulse rate of of rabbits both control (untreated) and treated with β-TCP product during the healing process post extraction
Figure 9 Respiration rate of of rabbits both control (untreated) and treated with
β-TCP product during the healing process post extraction
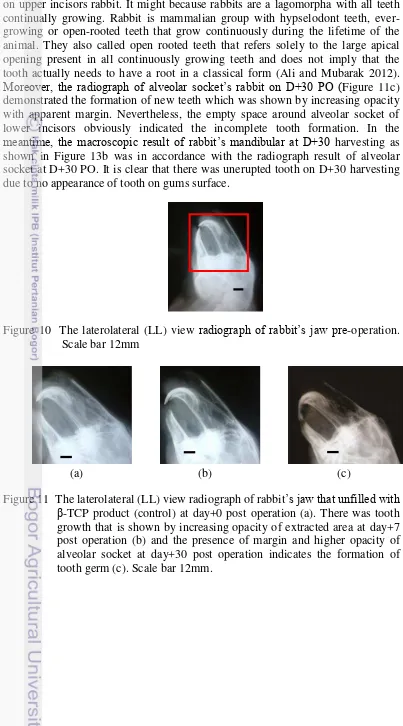
The rabbits tooth growth and status of β-TCP product was monitored using a set of x-ray radiographic apparatus. The radiograph of rabbit’s jaw pre operation
is shown in Figure 10 and the radiograph of rabbit’s jaw as control and treatment is shown in Figure 11 and Figure 12. Based on the radiograph in Figure 11 and Figure 12, these results show a tooth growth on rabbits at D+30 PO that was demonstrated by the presence of margin and high opacity on an extracted tooth area. It was clear that alveolar socket of lower incisors rabbit on D+0 PO (Figure 11a) had low opacity and then increase on D+7 PO as shown in Figure 11b. Increasing of alveolar socket opacity indicates the formation of tooth germ precursor tissue. It was also observed that in Figure 11b there was a tooth growth
14
on upper incisors rabbit. It might because rabbits are a lagomorpha with all teeth continually growing. Rabbit is mammalian group with hypselodont teeth, ever-growing or open-rooted teeth that grow continuously during the lifetime of the animal. They also called open rooted teeth that refers solely to the large apical opening present in all continuously growing teeth and does not imply that the tooth actually needs to have a root in a classical form (Ali and Mubarak 2012).
Moreover, the radiograph of alveolar socket’s rabbit on D+30 PO (Figure 11c) demonstrated the formation of new teeth which was shown by increasing opacity with apparent margin. Nevertheless, the empty space around alveolar socket of lower incisors obviously indicated the incomplete tooth formation. In the
meantime, the macroscopic result of rabbit’s mandibular at D+30 harvesting as shown in Figure 13b was in accordance with the radiograph result of alveolar socket at D+30 PO. It is clear that there was unerupted tooth on D+30 harvesting due to no appearance of tooth on gums surface.
Figure 10 The laterolateral (LL) view radiograph of rabbit’s jaw pre-operation. Scale bar 12mm
(a) (b) (c)
15
(a) (b) (c)
Figure 12 The laterolateral (LL) view radiograph of rabbit’s jaw that filled with
β-TCP product. There was high opacity at day+0 post operation due to the similar inorganic of sample with bone (a) and gradually decreased because of the resorption of sample by body fluid at day+7 post operation (b). The formation of complete tooth was showed by clear margin on the extracted area at day+30 post operation (c). Scale bar 10mm.
(a) (b) (c)
Figure 13 Macroscopic results of rabbit’s mandible at Day+30 harvesting. Sketch
of rabbit’s mandible was shown to describe the anatomy of lower jaw. There was no presence of erupted tooth on control (b) but the erupted tooth on the treatment showed the complete tooth growth (c).
On the contrary, the radiograph of alveolar socket filled with β-TCP as shown in Figure 12 demonstrated the tooth growth on rabbit. As seen in Figure 12a, the alveolar socket was filled with β-TCP and there was higher opacity than one in control at D+0 PO, which might be caused by the similar inorganic
component of β-TCP with bone. At the same time, there was jaw bone fracture during tooth extraction shown by unclear margin that is different with the origin. Figure 12b clearly shows resorption of β-TCP by the body fluids that is indicated
by volume decreasing of β-TCP pellet in alveolar socket. In addition, there was a tooth growth of upper incisors rabbit at D+7 as well as at the control. However,
there were differences on rabbits alveolar socket filled with β-TCP at D+30 PO. It
16
indicates the complete tooth formation of rabbits lower incisors due to the erupted tooth of 15 mm. it demonstrated a rapid growth of rabbits lower incisors and relevant to the other studies that reported lower incisors growth is approximately 2-3 mm per week (Capello 2005, Meredith 2007). Furthermore, the upper incisors growth of rabbit was shorter than that of control. It might be caused by the presence of erupted tooth and makes slower growth on the rabbit upper incisors. It is obvious that β-TCP could accelerate the formation of complete tooth and stimulate the tooth growth. In short, it could be applied clinically in case of premature tooth loss that commonly happen in children.
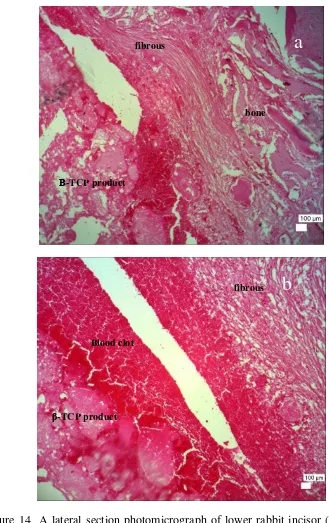
On the other hand, the histological results of treated extraction site that filled with β-TCP product inconsistent with the x-ray radiograph and macroscopic result. It is clearly proved by Figure 14 that showed the presence of fibrous connective tissue at the ground portion lining the sample as seen in Figure 14a. As seen in Figure 14a, blood clot filled the socket post extraction and was replaced with fibrous connective tissue. Then, in the middle portion of the socket was fully filled with mass of blood clod. Figure 14b showed that β-TCP product was partly resorbed and degraded. It was clearly showed by distributed β-TCP product that interacted with blood then will be replaced with fibrous connective tissue. The The existence of blood clot might indicate the incomplete healing processes and means no tooth growth on rabbit mandible. The erupted tooth in the macroscopic
result might be a remnant of unextracted tooth that forced by the blood clot and β -TCP product.
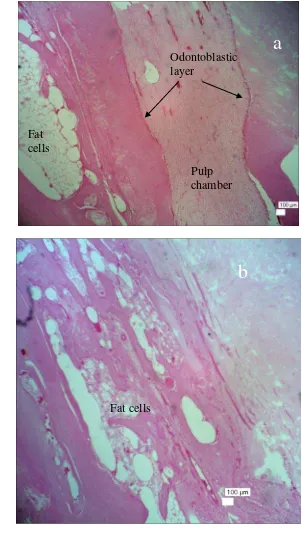
The opposite microscopic result was showed by control (unfilled) rabbit in Figure 15 that demonstrate the formation of tooth germ. It was showed by the presence of pulp chamber as shown in Figure 15a. It was obvious from wide pulp chamber that lined by odontoblastic layer in the apical region of rabbit incisor. Odontoblastic layer was not covered by dentine and enamel on its external side. These results showed the incomplete tooth formed proved by no cervical loop at the labial side in rabbit incisor that has function to generate ameloblasts. Ameloblast is cells that produce enamel on the labial tooth surface (Ali and Mubarak 2012). Moreover, the extracted socket was also filled with fat cells as seen in Figure 15b. These cells occur as single cells, clusters of cells, or as massive sheets of cells associated with connective tissues, especially loose connective tissue (Banks 1986). In addition, these cells appear as large empty spaces surrounded by the peripheral portion of the cell and a flattened nucleus (Copenhaver et al. 1978). These results support the radiograph result of untreated
rabbit (control) at day+30 post operation that indicated the formation of
17
Figure 14 A lateral section photomicrograph of lower rabbit incisor (filled with
β-TCP product) at Day+30 post operation. The presence of fibrous connective tissue in the ground portion of socket (a) and the presence of blood clot in the middle portion of socket indicate blood vessel rupture(b). Hematoxylin Eosin Stained, x40, Scale bar 100μm.
Β-TCP product
β-TCP product
fibrous
Blood clot fibrous
bone
a
18
Figure 15 A lateral section photomicrograph of lower rabbit incisor (control) at Day+30 post operation. Tooth growth was indicated by the presence of pulp chamber with odontoblastic layer ( ) in the apical portion of socket (a) and fully filled with fat cells in the middle portion (b).
Hematoxylin Eosin Stained, x40, Scale bar 100μm.
a
Pulp chamber Odontoblastic layer
Fat cells
Fat cells
19 Biocompatibility test on sheep
Biocompatibility test was also done in sheep to observe the alveolar bone healing process by resubstituting process of the corresponding β-TCP. It is clear that the corresponding β-TCP was biocompatible and non-toxic. Based on the clinical examination, there was no inflammation on the extracted area of each sheep. The result of physiological examination in sheep was the same with that in rabbits. The results showed the biocompatibility properties of β-TCP product. It is well described by Figure 16 and Figure 17 that showed there were no influence of filling β-TCP product and commercial β-TCP on temperature and pulse rate. The normal temperature and pulse rate of sheep are 38.9-40oC and 70-90 waves/minute, respectively (Kelly 1984). There was a little decrease of temperature on control rabbit at D+30 PO that might be caused by some factors such as environmental temperature and diurnal variations (Swenson 1984). In addition, the temperature of all healthy animals varies during the day (Kelly 1984). Then, higher pulse rate frequency of sheep filled with β-TCP product at D+1 PO might be caused by increased activity due to the handling process that trigger stress condition. It agreed with Adisuwirdjo (2001) that reported some factors such as activity, CO2 levels, body weight, and age are some that influence the pulse rate of animals. The respiration of sheep was fluctuative as seen in Figure 18
both control and treatment that filled with β-TCP product. The results were not in normal range that is 20-30 (Kelly 1984). It might be caused by stress condition of sheep during data collection process that made them excited. It is agreed with Kelly (1984) that reported increased respiratory frequency occurs whenever there is an increased demand for oxygen after exercise, exposure to high environmental temperature or humidity, and in obesity. Moreover, the longer period of expiration in healthy animals depends upon whether the animal is relaxed and resting, or has recently been excited or excercised. In short, postextraction preservation by
β-TCP product and commercial β-TCP as a tooth filler performs good response to the host and do not interfere the physiological condition of sheep.
20
Figure 17 Pulse rate of sheep both control (untreated) and treatments (filled with
β-TCP product and commercial β-TCP) during the healing process post extraction
Figure 18 Respiration rate of sheep both control (untreated) and treatments (filled
with β-TCP product and commercial β-TCP) during the healing process post extraction
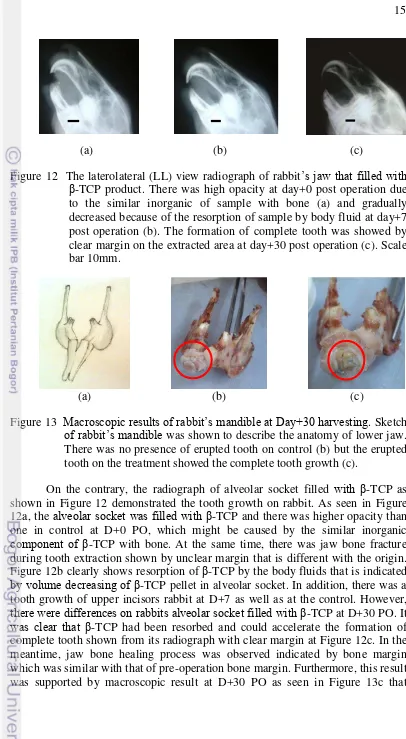
X-ray radiograph analysis was done to observe the healing process of extracted area. As a requirement, sheep jaw’s radiograph pre-operation was done as shown in Figure 19. The radiograph of control (sheep’s alveolar socket that unfilled with β-TCP) in Figure 20 demonstrate the alveolar bone resorption that could be seen from the narrower mesio-distal of edentulous are along the observing time. Mesio-distal is horizontal width (in mm) of edentulous area determined by measuring distance from mesial to distal side of adjacent tooth.
21
Figure 19 The radiograph of dorsoventral (DV) position of sheep ’s jaw pre-operation. Scale bar 15mm.
(a) (b) (c)
Figure 20 The radiograph of dorsoventral (DV) position of sheep’s jaw of control (unfilled with sample). There was large mesio-distal and low opacity on the extraction site ( ) at (a) day+0 post operation that become narrower at day+7 post operation (b) and there was high alveolar bone resorption shown by significant change on mesio-distal at day+30 post operation (c). Scale bar (a) 10mm (b) 8mm (c) 11mm.
Mesial is anatomical term for showing location closest to medial line and distal is anatomical term for showing location far from medial line. Thus, edentulous area is the area without tooth or artificial tooth inside mouth. As seen in Figure 20a, there was a larger distance of the alveolar ridge after tooth extraction with low opacity due to the unfilled socket. The dimension of the alveolar ridge had changed at D+7 PO (Figure 20b) which is became narrower and clearly followed by significant change at D+30 PO (Figure 20c). This result was clearly related to the macroscopic result at D+30 harvesting day in Figure 23a. It exhibited the faster resorption of alveolar bone in control shown by no space in the edentulous area.
In contrast, sheep’s alveolar socket that filled with the corresponding
β-TCP showed the expected goal. Figure 21 indicates resubstituting processes of
β-TCP product and showed preservation phenomena on the alveolar ridge. It was obvious from mesio-distal of the edentulous area that it remains in the same width. High opacity on D+0 PO as shown in Figure 21a demonstrated the alveolar socket
filled with β-TCP after tooth extraction. The same alveolar ridge width was
22
horizontal direction. It is clearly demonstrated on Figure 22 that showed the narrow mesio-distal at day+30 PO and supported by macroscopic results on Figure 23c. The same alveolar ridge width in vertical direction indicates no alveolar resorption and there was resubstituting process by commercial β-TCP. Macroscopic result of D+30 harvesting day confirms the bone healing process on post extraction socket as shown in Figure 23b. The detailed mesio-distal of the edentulous area is shown in Table 5. It is worth to note that β-TCP as a tooth filler could stimulate the alveolar bone healing process and maintain the desired alveolar ridge. This preservation could be done before prostheses or implant placement.
(a) (b) (c)
Figure 21 The radiograph of dorsoventral (DV) position of sheep (filled with
β-TCP product). High opacity on the extracted area ( ) at day+0 post operation showed the alveolar socket filled with sample (a). The same width of mesio-distal indicated there was no alveolar bone resorption day+7 post operation and there was resorption of sample showed by decreasing opacity (b). Resubstituting process of sample and preservation of alveolar ridge was clearly showed by the same width of mesio-distal on the edentulous area at day+30 post operation (c). Scale bar (a) 3mm (b) 10mm (c) 7mm.
(a) (b) ( c)
Figure 22 The radiograph of dorsoventral (DV) position of sheep (filled with
23
Biocompatibility properties of β-TCP bioceramics as a tooth filler was also proved by histological study in sheep. As seen in Figure 24, the untreated
extraction site of sheep’s alveolar socket (control) showed the alveolar bone
resorption. It was clearly seen from the decreasing of alveolar bone that is replaced by connective tissue shown in Figure 24a. Connective tissue here serves some functions such as insulate organs from mechanical damage, serve nutritive function as well and replace damaged tissues (Banks 1986). In addition, there was cavity on the extracted site as seen in Figure 24b. It is obvious that there was faster alveolar bone resorption in the horizontal direction. Moreover there was no bone healing on the socket post extraction. These results are completely consistent with the radiograph and macroscopic results proved by the narrow mesio-distal at day+30 PO.
Table 5 Mesio-distal of the edentulous area
24
Figure 24 A lateral section photomicrograph of alveolar bone healing (control) at Day+30 post operation. Alveolar bone resorption occurs that was showed by connective tissue in the upper portion of extracted area (a) and a cavity in the middle portion (b). Hematoxylin Eosin Stained,
x40, Scale bar 100μm.
On the other hand, the treated extracted site that filled with β-TCP product shows the bioactive properties as tooth filler. It well demonstrated by the interaction of sample with the living host tissue and the presence of connective tissue within β-TCP as seen in Figure 25a. At D+30 PO, alveolar socket was filled with newly formed fibrous connective tissue. A few of the implanted β-TCP product remained within the sockets, surrounded by numerous fibroblast cells that indicate the healing process of extracted area. Fibroblasts are the principal cells
Connective tissue
Gingiva
a
Cavity in extracted area
25 responsible for fiber formation (Copenhaver et al. 1978). The fibroblast is the
primary cell responsible for regenerative and reparative processes. Old and/or damaged cells are removed and replaced by new fibroblasts (Banks 1986). There was also neovascularization in the fibrous connective tissue that showed the osteoconductive properties of the corresponding β-TCP. As explained by Kalfas (2001) that osteoconduction is the physical property of the graft to serve as a scaffold for viable bone healing, allows the ingrowth of neovasculature and the infiltration of osteogenic precursor cells into the graft site. Moreover, Figure 25b showed small amount of new bone formation which was shown by the formation of woven bone on the upper portion of defect that surrounded by connective tissue. It agreed with Banks (1986) that explained woven bone is the first bone that forms at the site of the fracture repair. Normally, it occurs in alveolar bone associated with teeth and at points of attachment of ligaments and tendons to bone. The type of bone development here is intramembranous in which the bone develops under or within a layer of connective tissue (Copenhaver et al. 1978) as seen in Figure
25c. It is clear that β-TCP could minimize the bone loss and maintain the dimension of alveolar ridge as well as showed by the same width mesio-distal of extracted site shown in x-ray radiograph result.
Fibrous connective tissue
β-TCP product
Neovascularization
26
Figure 25 A lateral section photomicrograph of alveolar bone healing (filled with
β-TCP product) at Day+30 post operation. Biocompatibility of tooth filler seen from infiltration of fibrous connective tissue within β-TCP product in the middle portion of extracted area (a). Bone remodeling occurs in the formation of woven bone (b) via intramembranous bone development under layer of connective tissue (c) in the upper portion near gingiva. Hematoxylin Eosin Stained x40 (a, b) x100 (c). Scale bar
100μm.
Woven bone
Gingiva
b
Fibrous and osteogenetic layers of periosteum
Osteocytes
Osteoblast
27 The treated extracted site that filled with commercial β-TCP (RTR Septodont) shows different result with that did with β-TCP product. Figure 26a demonstrated alveolar bone resorption as showed by no alveolar bone remains. The upper portion of extracted area was fully filled with connective tissue. In the meantime, there was blood clot that surrounded by collagen. No presence of new bone showed the full resorption of alveolar bone in horizontal direction. Then, the presence of blood clot in the extracted area might be remnant post extraction that has not been replaced by granulation tissue. It was confirmed by Figure 26b that showed connective tissues such as fibrous and collagen in the middle extracted area near gingiva. Nevertheless, Figure 26c showed the biocompatibility of commercial β-TCP. It was clearly demonstrated by the presence of vessel in the middle portion of extracted area. The presence of vessel showed the vascularized site that indicates the healing process proved by the presence of connective tissue. These results showed that alveolar bone resorption only happened in horizontal direction. It might be caused by less dense β-TCP that is filled in the extracted area due to the granule form of commercial β-TCP used as a tooth filler.
Blood clot
vessel
Collagen Fat cells
28
Figure 26 A lateral section photomicrograph of alveolar bone healing (filled with
commercial β-TCP) at Day+30 post operation. Biocompatibility of tooth filler seen from the presence of vessel in the upper portion of extracted area but the presence of blood clot indicated vessel rupture(a). Alveolar bone resorption occurs in horizontal direction shown by the presence of collagen and fibrous connective tissue near gingiva (b). Repairing process by connective tissue in the middle portion shown by the presence of vessel (c). Hematoxylin Eosin Stained x40. Scale bar
100μm.
Gingiva
Fibrous connective tissue Collagen
b
Vessel
Fibrous
connective tissue
29 purity of the products is shown by the AAS results with Ca/P ratio of 1.48.
Biocompatibility test of β-TCP bioceramics as a tooth filler on postextraction preservation of alveolar socket shows a promising challenge in dental practices. The rabbit’s alveolar socket filled with β-TCP indicates no tooth growth showed by histological analysis that demonstrate blood clot and sample mass in the extracted site. The alveolar bone healing process is obviously
30
Abdurrahim T, Sopyan I. 2008. Recent progress on the development of porous bioactive calcium phosphate for biomedical applications. Recent Patents on Biomedical Engineering. 1: 213-229.
Adisuwirdjo D. 2001. Buku ajar dasar fisiologi ternak. Purwokerto (ID): Fakultas
Peternakan, Universitas Jenderal Soedirman.
Ain RN, Sopyan I, Ramesh S. 2008. Preparation of biphasic calcium phosphate ceramics powders and conversion to porous bodies. ICCBT. 23: 245-256.
Ali ZH, Mubarak R. 2012. Histomorphological study of dentin pulp complex of continuously growing teeth in the rabbits. Life Science Journal. 9 (3):
1554-1564.
Arora H, Nafria A, Kanase A. 2011. Rabbits as animal models in contemporary implant biomaterial research. WJD. 2(2): 129-134.
Balázsi C, Kõvér Z, Horváth E, Németh C, Kasztovszky Z, Kurunczi S, Wéber F. 2007. Examination of calcium-phosphate prepared from eggshell. Mater Sci Forum. 537-538: 105-112.
Banks WJ. 1986. Applied veterinary histology. USA (US): Williams and Wilkins.
Belouafa S, Chaair H, Chroqui W, Digua K, Britel O, Essaadani A. 2006. Central composite design and optimization by response analysis of β-tricalcium phosphate elaboration. Phosphorus, Sulfur Silicon Relat. Elem. 181:
779-786.
Boilet L, Descamps M, Rguiti E, Tricoteaux A, Lu J, Petit F, Lardot V, Cambier F, Leriche A. 2013. Processing and properties of transparent
hydroxyapatite and β tricalcium phosphate obtained by HIP process.
Ceram Int. 39(1): 283-288.
Brkovic BMB, Prasad HS, Konandreas G, Milan R, Antunovic D, Sándor GBK, Rohrer MD. 2008. Simple preservation of a maxillary extraction socket using beta-tricalcium phosphate with type I collagen: preliminary clinical and histomorphometric observations. JCDA. 74 (6): 523-528.
Calixto RFE, Teófilo JM, Brentegani LG, Lamano-Carvalho TL. 2007. Grafting of tooth extraction socket with inorganic bovine bone or bioactive glass particles: comparative histometric study in rats. J Can Dent Assoc. 16 (3):
260-269.
Capello V. 2005. Rabbit and rodent dentistry handbook. United State of America
(US): Zoological Education Network, Inc.
Coimbra ME, Salles MB, Yoshimoto M, Allegrini Jr. S, Fancio E, Higa O, Suzuki M, Coelho PG. 2009. Physico/chemical characterization, in vitro, and in vivo evaluation of hydroxyapatite/PLGA composite and tricalcium phosphate particulate grafting materials. Titanium. 1 (1): 16-28.
Copenhaver WM, Kelly DE, Wood RL. 1978. Bailey’s textbook of histology. USA (US): The Williams and Wilkins Company.
Costa HS, Mansur AAP, Barbosa-Stancioli EF, Pereira MM, Mansur HS. 2008. Morphological, mechanical, and biocompatibility characterization of macroporous alumina scaffolds coated with calcium phosphate/PVA. Mat Sci. 43: 510-524.
31 Dahlan K., Dewi SU, Nurlaela A, Soejoko DS. 2012. Synthesis and characterization of calcium phosphate/chitosan composite. IJBAS-IJENS.
12(1): 50-57.
Dai H, Li S, Yan Y, Cao X, Lu X, Leng Y. 2004. Ultrastructural analysis on the
osteogenesis and transformation of calcium phosphate ceramics in vivo. J. Mater. Sci. Technol. 20(1): 59-62.
Kalfas IH. 2001. Principles of bone healing. Neurosurg Focus. 10 (4):7-10.
Kannan S, Goetz-Neunhoeffer F, Neubauer J, Ferreira JMF. 2009. Synthesis and structure refinement of zinc-doped β-tricalcium phosphate powders. J. Am. Ceram. Soc. 92 (7): 1592-1595.
Kannan S, Pina S, Ferreira JMF. 2006. Formation of strontium-stabilized β -tricalcium phosphate from calcium-deficient apatite. J. Am. Ceram. Soc.89(10): 3277-3280.
Kelly WR. 1984. Veterinary clinical diagnosis. London (UK): Baillière Tindall.
Koç N, Timuçin M, Korkusuz F. 2004. Fabrication anf characterization of porous tricalcium phosphate ceramics. Ceram Int. 30(2): 205-211.
Kokovic V, Todorovic L. 2011. Preimplantation filling of tooth socket with β
-tricalcium phosphate/polylactic/polyglicolic acid (β-TCP/PLGA) root analogue: clinical and histological analysis in a patient. Vojnosanit Pregl.
68 (4): 366-371.
Meredith A. 2007. Rabbit dentistry. EJCAP. 17 (1): 55-62.
Nasiri-Tabrizi B, Fahami A. 2013. Mechanochemical synthesis and structural characterization of nano-sized amorphous tricalcium phosphate. Ceram Int.
39(8): 8657-8666.
Oi Y, Ota M, Yamamoto S, Shibukawa Y, Yamada S. 2009. Beta-tricalcium phosphate and basic fibroblast growth factor combination enhances periodontal regeneration in intrabony defects in dogs. Dent Mater J. 28
(2): 162-169.
Park JB, Bronzino JD. 2000. Biomaterials Principles and Applications. Florida
(US). CRC Press.
Pearce Al. 2007. Animal models for implant biomaterial research in bone: A review. Eur Cell Mater. 13: 1-10.
Pratiwi NL, Basuki H, Suprapto A.2013. Hubungan perilaku oral hygiene, status dan konsumsi gizi, sosbud, akses pelayanan kesehatan terhadap besaran indeks DMFT. Research report [Internet]. [diunduh 2013 Maret 21]; Tersedia
pada:http://grey.litbang.depkes.go.id/gdl.php?mod=browse&op=read&id=jkpkbppk--ninieklpra-3743
Sader MS, LeGeros RZ, Soares GA. 2009. Human osteoblast adhesion and proliferation on magnesium-substituted tricalcium phosphate dense tablets.
J Mater Sci: Mater Med. 20: 521-527.
Saeed AM, Hassan RA, Thajeel KM. 2011. Synthesis of calcium
hydroxyapatite powder from hen’s eggshell. Iraqi Journal of Physics.
9(16): 24-28.
Salahi E, Heinrich JG. 2003. Synthesis and thermal behavior of β tricalcium
phosphate precipitated from aqueous solutions. Brit Ceram T.
32
Shi D. 2004. Biomaterial and Tissue Engineering. Germany (DE): Department of
Chemical and Materials Engineering, University of Cincinnati.
Solihat R. 2011. In vitro analysis of biphasic calcium phosphate and hydroxyapatite as bone implants [thesis]. Bogor. Bogor Agricultural University.
Swenson MJ. 1984. Duke’s physiology of domestic animals. London: Cornell University Press.
Tazaki J, Murata M, Akazawa T, Yamamoto M, Ito K, Arisue M, Shibata T, Tabata Y. 2009. BMP-2 release and dose-response studies in hydroxyapatite and β-tricalcium phosphate. Biomed MaterEng. 19:
141-146.
Wei X, Akinc M. 2007. Crystal structure analysis of Si- and Zn-codoped tricalcium phosphate by neutran powder diffraction. J. Am. Ceram. Soc. 90
33
34
Appendix 1 Flow chart of the research
Biocompatibility test
Materials and equipment preparation
Solution preparation
Precipitation of β-TCP bioceramics
Analysis data
Report arrangement
No
XRD, FTIR and AAS Characterization
Y Yes
Ready
35
Appendix 2 Joint committee on powder diffraction standards (JCPDS) database
of Calcium Oxide
Appendix 3 Joint committee on powder diffraction standards (JCPDS) database
36
Appendix 4 Joint committee on powder diffraction standards (JCPDS) database
of Calcium Phosphate (Ca2P2O7)
Appendix 5 Joint committee on powder diffraction standards (JCPDS) database
37
CURRICULUM VITAE
The author was born in Jombang on May 15, 1987 as the second child of five children of the couple of Abu Hasan Sholeh and Rahayu Suprapti. Pursued graduate education in Physics Program at Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, graduating in 2010. In 2011, the author received at Biophysics Program at Graduate School of Bogor Agricultural University with Beasiswa Unggulan DIKTI supported by National Education
Ministry of Republic Indonesia. The author worked as a freelance teacher of
Bimbingan dan Konsultasi Belajar Nurul Fikri since 2009.