1
Clinical Characteristics and The Selection of Initial ARV Regimen
Therapy for HIV-Infected Patients
in VCT-CST RSUP Sanglah,
Denpasar
I G. A. Putu Candradewi, I Ketut Agus Somia
Departement of Internal Medicine, Medical Faculty of Udayana University/RSUP Sanglah
ABSTRACT
Number of new cases of HIV infections in Bali is still high and ARV becomes the one and only therapy. There are many ARV drugs used globally. The selection of ARV regimens are closely related to the patients clinical characteristics. So that, in this study, the author investigates the clinical characteristics of initial ARV regimen therapy in VCT-CST RSUP Sanglah so does the factors related to the selection of initial ARV regimen. This study is retrospective study, using descriptive methods. Data of the patients are distributed based on initial ARV regimen and other variables related to therapy and patients clinical characteristics. The significance of differences of patients clinical characteristics among the initial ARV regimens are also tested using ANOVA and chi-square test to find out the factors related to the selection of inital ARV regimen for the patients. Based on the study of 127 patient’s medical records, 69.3% (n=88) of patients receive NVP based regimen (AZT/3TC/NVP), 21.3% (n=27) of patients receive EFV based regimen (AZT/3TC/EFV), and 9.4% (n=12) of patients receive TDF based regimen (TDF/3TC/NVP or TDF/3TC/EFV). Characteristics that are significantly different among the three regimens are patients opportunistic infections (p=0.000), initial hemoglobin (p=0.009), side effect appearance (p=0.022) and change of regimen during 6 months of therapy (p=0.048). Initial ARV regimens commonly used for HIV-infected patients at VCT-CST RSUP Sanglah are NVP based regimen (AZT/3TC/NVP), EFV based regimen (AZT/3TC/EFV), and TDF based regimen (TDF/3TC/NVP or TDF/3TC/EFV). Selection of initial ARV regimens is related to the opportunistic infections, initial hemoglobin of the patients, the appearance of side effect, and tendency to change ARV regimen.
Keywords. HIV infection, initation of ARV regimen therapy, ARV regimens, characteristics of ART, treatment onset, change of regimen.
INTRODUCTION
Human Immunodeficiency Virus (HIV) infection has been an endemic worldwide. An estimation of 35.3 (32.2–38.8) million people were living with HIV all over the world in 2012. New HIV infection cases in 2012 was 2.3 (1.9–2.7) million. However, it declines if compared to the number of
2 government, health care providers, or community organization. However, the number of new HIV infection cases is repication and progression which called antiretroviral (ARV). Suppression of HIV replication is important in prolonging life and improving the quality of life in patients with HIV infection. Patients undergoing treatment for HIV infection have to take a daily regimen of at least three antiretroviral drugs.2
Antiretroviral (ARV) therapy has been used widely around the world to treat people who are already infected by HIV. The number of ART receiver all over the world is increasing year by year.1 However, there are still many issues about starting antiretroviral therapy such as the timing and regimen Antiretroviral Guidelines for Adults and Adolescents has classified many kinds of ARV regimens into 3 groups: preferred regimens, alternative
regimens, and other regimens.4 Among many kinds of initial ARV regimen, patients clinical characteristics are important to determine which initial ARV regimens will be given to patients. As the effectiveness of therapy and outcome are very closely related to the factors mentioned before, the author would like to discuss about the clinical characteristics of ARV therapy and selection of initial ARV regimen for HIV-infected patients in VCT-CST clinic of RSUP Sanglah, Denpasar. METHODS
This study is a retrospective study using descriptive method to presents the clinical characteristics of patients among the initial ARV regimens used in
HIV-infected patients who receive their initial ARV regimen therapy in VCT-CST RSUP Sanglah in 2012. Sample is taken from the population after excluding some patients whose medical records cannot be found.
3 after 6 months of therapy, side effect of
initial ARV regimen, change of regimen during 6 months of therapy, CD4+ change after 6 months of therapy, and mortality.
RESULTS
Distribution of Data Based on the Initial ARV Regimen
Based on the study of 127 patient’s medical records, there are 3 different regimens of antiretroviral (ARV) used in VCT-CST RSUP Sanglah to initiate the treatment of HIV-infected patients in 2012. All of the regimens contain lamivudine (3TC), but the other two
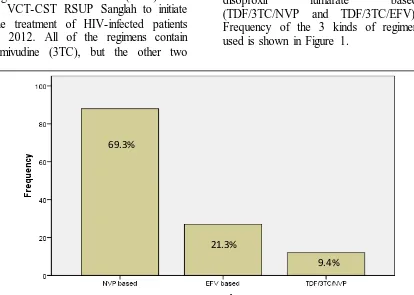
drugs are different based on the patient’s characteristic and doctor’s consideration. Other ARV drugs used as initial treatment are zidovudine (AZT), nevirapine (NVP), efavirenz (EFV), tenofovir disoproxil fumarate (TDF). Data of 127 patients medical record is distributed based on 3 ARV regimens, which are nevirapine based (AZT/3TC/NVP), efavirenz based (AZT/3TC/EFV), and tenofovir disoproxil fumarate based (TDF/3TC/NVP and TDF/3TC/EFV). Frequency of the 3 kinds of regimen used is shown in Figure 1.
Figure 1. Frequency of initial ARV regimen used in VCT-CST RSUP Sanglah in 2012 (n=127)
Patients Clinical Characteristics among the Three Initial ARV Regimens Therapy
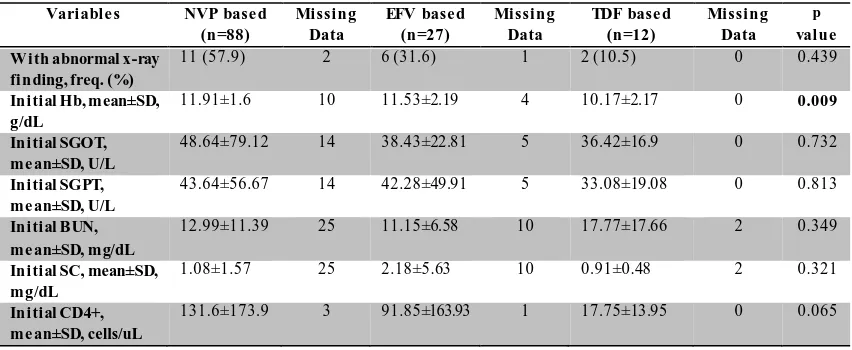
Based on Table 1, the mean age of patients using NVP based therapy is 32.66 years, while the EFV based is 31.63 years and TDF based is 35.92 years. Most of the patients are male. There are 5 patients among females who are pregnant. Means of body weight of the patients are 52.78 kg in NVP based
regimen, 52.41 kg in EFV based regimen, and 52.83 kg in TDF based regimen. Most of the patients initiate therapy before 6 months after the date of firstly diagnosed as positive HIV-infected. Almost all of the patients show some clinical manifestations when initiating therapy. Patients with
69.3%
21.3%
4 opportunistic infections mostly receive
NVP based therapy. Opportunistic
infections are related to the selection of initial ARV regimen (p<0.05).
Table 1. Sociodemographic Characteristic of patients of Each Initial ARV Regimen Therapy
Table 2. Characteristics of Initial Laboratory Results of patients of Each Initial ARV Regimen Therapy
Abbreviation: SD, standard deviasion; Hb, hemoglobin; SGOT , Serum Glutamic Oxaloacetic T ransaminase; SGPT , Serum Glutamic Pyruvic T ransaminase; BUN, Blood Urea Nitrogen; SC, Serum Creatinine.
5 Table 3. Characteristics of Patients Outcome after 6 Months of Therapy
Variable s NVP base d (n=88)
Missing Data
EFV base d (n=27)
Missing Data
TDF base d (n=12)
Missing Data
p value C D4+ after 6 months
of the rapy, me an±SD, cells/uL
252.66±201.76
30 275.94±230.93 9 137±48.5 5 0.291
Side effect appears, fre q. (%)
37 (84.1) 0 6 (13.6) 0 1 (2.3) 0 0.022
Toxicity appears, fre q. (%)
1 (100) 0 0 (0) 0 0(0) 0 0.8
C hange of regimen, fre q. (%)
32 (84.2) 0 5 (13.2) 0 1 (2.6) 0 0.048
Afte r 6 months CD4+ change, freq. (%)
55 (69.6) 33 17 (21.5) 10 7 (8.9) 5 0.958
Mortality, freq. (%) 4 (57.1) 0 1 (14.3) 0 2 (28.6) 0 0.202
After 6 months of therapy, the means of patients CD4+ count are 252.66 cells/µL in patients receiving NVP based regimen, 275.94 cells/µL in patients receiving EFV based regimen, and 137 cells/µL in patients receiving TDF based regimen. Side effects are common in patients using NVP based regimen. The appearance of side effects is significantly different among the three regimens (p<0.05) means that it is related to the selection of initial ARV regimen. Toxicity is very rarely happen. One case of toxicity in the table above
is mild toxicity caused by nevirapine (NVP).
Some patients change the component or drug of their initial ARV regimen during the first 6 months of therapy. The difference is significant among the three regiems, means that the tendency to change ARV regimen are related to the selection of initial ARV regimen. 57.1% of patients who died during the first 6 months of therapy are from NVP based receivers, 14.3% are from EFV based receivers, and 28.6% are from TDF based receivers.
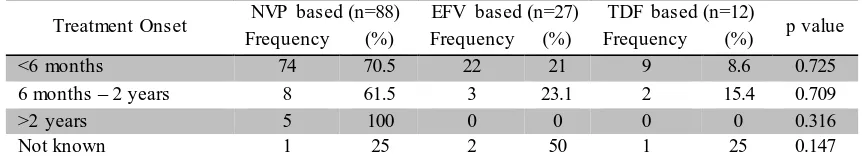
Table 4. Distribution of Data of Each Initial ARV Regimen Based on Treatment Onset from Firstly Diagnosed until Initiation of Therapy
Treatment Onset NVP based (n=88) EFV based (n=27) TDF based (n=12) p value
Frequency (%) Frequency (%) Frequency (%)
<6 months 74 70.5 22 21 9 8.6 0.725
6 months – 2 years 8 61.5 3 23.1 2 15.4 0.709
>2 years 5 100 0 0 0 0 0.316
Not known 1 25 2 50 1 25 0.147
Based on the table above, most of the patients initiate ARV regimen therapy in the first 6 months after they are firstly diagnosed as positive for HIV- infection. Among them, 70.5% receive NVP based regimen, 21% receive EFV based regimen, and 8.6% receive TDF
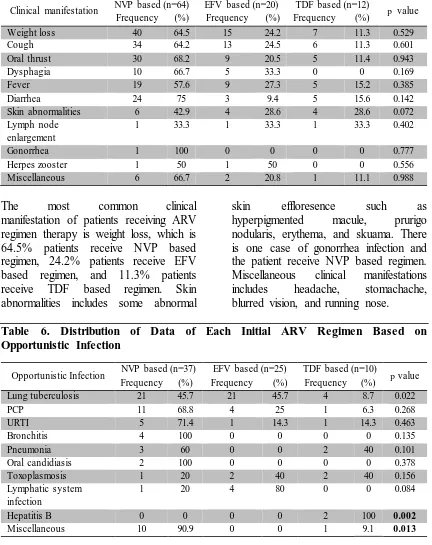
6 Table 5. Distribution of Data of Each Initial ARV Regimen Based on Patients Clinical Manifestation
Clinical manifestation NVP based (n=64) EFV based (n=20) TDF based (n=12) p value Frequency (%) Frequency (%) Frequency (%)
Weight loss 40 64.5 15 24.2 7 11.3 0.529
Cough 34 64.2 13 24.5 6 11.3 0.601
Oral thrust 30 68.2 9 20.5 5 11.4 0.943
Dysphagia 10 66.7 5 33.3 0 0 0.169
Fever 19 57.6 9 27.3 5 15.2 0.385
Diarrhea 24 75 3 9.4 5 15.6 0.142
Skin abnormalities 6 42.9 4 28.6 4 28.6 0.072
Lymph node enlargement
1 33.3 1 33.3 1 33.3 0.402
Gonorrhea 1 100 0 0 0 0 0.777
Herpes zooster 1 50 1 50 0 0 0.556
Miscellaneous 6 66.7 2 20.8 1 11.1 0.988
The most common clinical manifestation of patients receiving ARV regimen therapy is weight loss, which is 64.5% patients receive NVP based regimen, 24.2% patients receive EFV based regimen, and 11.3% patients receive TDF based regimen. Skin abnormalities includes some abnormal
skin effloresence such as hyperpigmented macule, prurigo nodularis, erythema, and skuama. There is one case of gonorrhea infection and the patient receive NVP based regimen. Miscellaneous clinical manifestations includes headache, stomachache, blurred vision, and running nose.
Table 6. Distribution of Data of Each Initial ARV Regimen Based on Opportunistic Infection
Opportunistic Infection NVP based (n=37) EFV based (n=25) TDF based (n=10) p value
Frequency (%) Frequency (%) Frequency (%)
Lung tuberculosis 21 45.7 21 45.7 4 8.7 0.022
PCP 11 68.8 4 25 1 6.3 0.268
URTI 5 71.4 1 14.3 1 14.3 0.463
Bronchitis 4 100 0 0 0 0 0.135
Pneumonia 3 60 0 0 2 40 0.101
Oral candidiasis 2 100 0 0 0 0 0.378
Toxoplasmosis 1 20 2 40 2 40 0.156
Lymphatic system infection
1 20 4 80 0 0 0.084
Hepatitis B 0 0 0 0 2 100 0.002
Miscellaneous 10 90.9 0 0 1 9.1 0.013
The most common opportunistic infection occurs in patients receiving ARV regimen therapy is lung tuberculosis. 45.7% of patients with lung tuberculosis receive NVP based regimen, 45.7% others receive EFV
7 includes lymphadenitis tuberculosis and
lymphangitis tuberculosis.
Miscellaneous finding includes herpes zooster, retinitis CMV, diarrhea, condiloma accuminata, herpes simplex genital, recurrent varicella, and blepharoconjunctivitis. The difference
of lung tuberculosis, hepatitis B, and miscellaneous infections percentage among the three regimens is significant (p<0.05). It indicates that the appearance of those opportunistic infections are related to the selection of initial ARV regimen and vice versa.
Table 7. Distribution of Data of Each Initial ARV Regimen Based on X-ray Abnormalities
X-ray finding NVP based (n=11) EFV based (n=6) TDF based (n=2) p value Frequency (%) Frequency (%) Frequency (%)
Pneumonia 3 60 2 40 0 0 0.647
Bronchitis 3 100 0 0 0 0 0.274
Pleural efusion 2 100 0 0 0 0 0.444
Suspect BE 1 50 1 50 0 0 0.779
Infiltrate 1 50 1 50 0 0 0.779
Lung tuberculosis 0 0 3 75 1 25 0.031
PCP 1 100 0 0 0 0 0.681
Miscellaneous 1 50 0 0 1 50 0.133
The most common abnormality found on patients x-ray result is pneumonia. 60% of patients with pneumonia receive NVP based regimen and 40% of them receive EFV based regimen. All patients with bronchitis, pleural effusion, and PCP findings receive NVP based regimens. Suspect BE reffers to suspect bronchiectasis and PCP reffers to
Pneumocystis Carinii Pneumonia. Miscellaneous finding includes cardiomegaly and toxoplasmosis. In the table, p value of lung tuberculosis is less than 0.05. It indicates that x-ray finding of lung tuberculosis is related to the selection of initial ARV regimen for the patient.
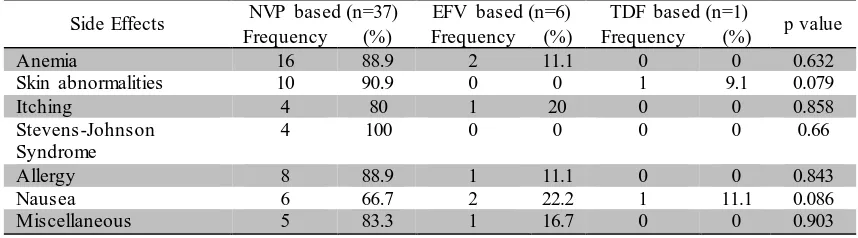
Table 8. Distribution of Data of Each Initial ARV Regimen Based on Side Effects
Side Effects NVP based (n=37) EFV based (n=6) TDF based (n=1) p value
Frequency (%) Frequency (%) Frequency (%)
Anemia 16 88.9 2 11.1 0 0 0.632
Skin abnormalities 10 90.9 0 0 1 9.1 0.079
Itching 4 80 1 20 0 0 0.858
Stevens-Johnson Syndrome
4 100 0 0 0 0 0.66
Allergy 8 88.9 1 11.1 0 0 0.843
Nausea 6 66.7 2 22.2 1 11.1 0.086
Miscellaneous 5 83.3 1 16.7 0 0 0.903
Anemia is the most common side effect occurs on patients receiving initial ARV regimen therapy. 88.9% cases of anemia occurs on patients receiving NVP based regimen and 11.1% occurs on patients
8 abnormality in TDF based regimen
reffers to erythema multiforme. Miscellaneous side effect includes
vomiting, tinitus, dizziness, and dyspepsia.
Table 9. Distribution of Data of Each Initial ARV Regimen Base d on After 6 Months CD4+ Change
After 6 Months CD4+ Change
NVP based (n=88) EFV based (n=27) TDF based (n=12) p value Frequency (%) Frequency (%) Frequency (%)
Increase 51 69.9 15 20.5 7 9.6 0.974
Decrease 4 66.7 2 33.3 0 0 0.597
Not known 33 68.8 10 20.8 5 10.4 0.958
Table 10. Pattern of Average Increase in CD4+ count after 6 Months of Therapy
NVP based (51) Average increase, mean±SD, cells/µL
EFV based (15) Average increase, mean±SD, cells/µL
NVP based (7) Average increase, mean±SD, cells/µL
p value
125.73±85.25 205±231.87 121.14±49.32 0.102
Most of the patients develop an incerase in their CD4+ count after 6 months of therapy. 69.9% of patients whose CD4+ count increases are patients receiving NVP based therapy, 20.5% are from EFV based and 9.6% others are from TDF based one. Decrease in CD4+ count also occurs in NVP based regimen (66.7%) and EFV based regimen (33.3%). None of the classification of after 6 months CD4+ change has p<0.05 indicates that those changes are not significantly different among the three initial ARV regimens. The average of increase of CD4+ count after 6 months of therapy among the three regimens are not significantly different as shown in the Table 10. DISCUSSION
Based on 127 patients who initiate their ARV regimen therapy at VCT-CST RSUP Sanglah in 2012, 69.3% of them receiving NVP based regimen (AZT/3TC/NVP) for initial therapy. Only 21.3% of patiens receive EFV based regimen and 9.4% receive TDF based. Compared to the guidelines from
HHS Panel on Antiretroviral Guidelines for Adults and Adolescents,4 none of those three regimens are included in the preferred or alternative regimens. They all belong to the other regimens for initial therapy. The initial regimens used in VCT-CST RSUP Sanglah are same as the initial regimens used in resource-limited settings.5 Other related study conducted in Kenya, Uganda, and Tanzania stated that the most common NRTI component used in the initial ARV combination is stavudine (d4T). Higher WHO stage of the patients is associated with a higher chance of d4T use in the first regimen in these countries.6
9 square analysis. Lung tuberculosis and hepatitis B are also related to initial ARV regimen selection, so are some other miscellaneous opportunistic infections (Table 4 and 5).
10 most appropriate initial ARV regimen for the patients and also improving the clinical condition of patients who seek medical attention after suspected to be infected by HIV through a good socialization. The author realizes that this study is still far from perfection. Limitations of this study especially caused by limited time in collecting and patients come to the clinic.
CONCLUSIONS hemoglobin count and opportunistic infections, such as lung tuberculosis and However, few other patients experience a decrease in CD4+ count. Mortality happens to 7 out of 127 patients
receiving initial ARV regimen therapy in 2012.
The author suggests that the newest guidelines for initial ARV regimen can be used as consideration in cilical setting. Patients initial condition should be recorded completely before receiving ARV therapy. Socialization for early seeking of medical attention if suspected as HIV infected should be increased in order to improve the outcome of HIV-infected patients after ARV therapy. Monitoring of patients outcome during initial ARV regimen therapy should be done closely and HIV/AIDS (UNAIDS). Global report: UNAIDS report on the global AIDS epidemic 2013. NLM. 2013:4-11.
2. Wood E, Hogg RS, Harrigan PR, Montaner JSG. When to initiate antiretroviral therapy in HIV-1-infected adults: a review for clinicians and patients. Lancet Infect Dis. 2005;5:407-14.
3. Jain V, Hartogensis W, Bacchetti P, et al. Antiretroviral therapy initiated within 6 months of HIV infection is associated with lower T-cell activation and smaller HIV reservoir size. J Infect Dis. 2013;208:1202-11. 4. Panel on Antiretroviral Guidelines
for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Department of Health and Human
Services. Available at
11 accessed [20 November 2013]
[F1-29, Table 5a, Table 5b, Table 6, Table 7].
5. Emmelkamp JM, Rockstroh JK. CCR5 antagonists: comparison of efficacy, side effects, pharmacokinetics and interactions – review of the literature. Eur J Med Res. 2007;12:409-17.
6. Geng EH, Hunt PW, Diero LO, et al. Trends in the clinical characteristics of HIV infected patients initiating antiretroviral therapy in Kenya, Uganda and Tanzania between 2002
and 2009. Journal of the International AIDS Society. 2011;14:46.
7. Kasper DL, Fauci AL, editors. Harrison’s principles of internal medicine. United States of America: The McGraw-Hill Companies, Inc.; 2005.