Sustainable Livestock Production in the Perspective of
Food Security, Policy, Genetic Resources, and Climate Change
A
A
P
Proceedings
Full Papers
Ministry of Agriculture Indonesian Society of Animal Sciences Gadjah Mada University
th
The 16 AAAP Congress
th
The 16
AAAP
Congress
Sustainable Livestock Production in the Perspective of
Food Security
, Policy
, Genetic Resources, and Climate Change
Proceedings
!
"
#
$"
% & '&
# () $ & * ' & * *
* + && ,
& ! - * * , * - '
+ * . & * . * / * . 0& 0
(1$(2 3 - 41(2. 5 . 6 7
4849 , && 7 4( : 4;.< *
= > * ' & * *
0 && ? ' * &
+ * & ' & * * @ - ?
A3 ;<8$)14$82<9$8<$<
( ! - * 4 + * B & *
2 * / * 9 0& 0
Scope of AAAP: AAAP is established to devote for the efficient animal production in the
Asian-Australasian region through national, regional, international cooperation and academic conferences.
Brief History of AAAP: AAAP was founded in 1980 with 8 charter members representing 8
countries-those are Australia, Indonesia, Japan, Korea, Malaysia, New Zealand, Philippines and Thailand. Then, the society representing Taiwan joined AAAP in 1982 followed by Bangladesh in 1987, Papua New Guinea in 1990, India and Vietnam in 1992, Mongolia, Nepal and Pakistan in 1994, Iran in 2002, Sri Lanka and China in 2006 , thereafter currently 19 members.
Major Activities of AAAP: Biennial AAAP Animal Science Congress, Publications of the
Asian-Australasian Journal of Animal Sciences and proceedings of the AAAP congress and symposia and Acknowledgement awards for the contribution of AAAP scientists.
Organization of AAAP:
∙ President: Recommended by the national society hosting the next biennial AAAP Animal Science Congress and approved by Council meeting and serve 2 years.
∙ Two Vice Presidents: One represents the present host society and the other represents next host society of the very next AAAP Animal Science Congress.
∙ Secretary General: All managerial works for AAAP with 6 years term by approval by the council
∙ Council Members: AAAP president, vice presidents, secretary general and each presidents or representative of each member society are members of the council. The council decides congress venue and many important agenda of AAAP
Office of AAAP: Decided by the council to have the permanent office of AAAP in Korea.
Currently # 909 Korea Sci &Tech Center Seoul 135-703, Korea
Official Journal of AAAP: Asian-Australasian Journal of Animal Sciences (Asian-Aust. J.
Anim. Sci. ISSN 1011-2367. http://www.ajas.info) is published monthly with its main office in Korea
Current 19 Member Societies of AAAP:
ASAP(Australia), BAHA(Bangladesh), CAASVM(China), IAAP(India), ISAS(Indonesia),
IAAS(Iran), JSAS(Japan), KSAST(Korea), MSAP(Malaysia), MLSBA(Mongolia),
NASA(Nepal), NZSAP(New Zealand), PAHA(Pakistan), PNGSA(Papua New Guinea),
PSAS(Philippines), SLAAP(Sri Lanka), CSAS(Taiwan), AHAT(Thailand), AHAV(Vietnam).
Previous Venues of AAAP Animal Science Congress and AAAP Presidents
I 1980 Malaysia S. Jalaludin II 1982 Philippines V. G. Arganosa III 1985 Korea In Kyu Han IV 1987 New Zealand A. R. Sykes V 1990 Taiwan T. P. Yeh VI 1992 Thailand C. Chantalakhana VII 1994 Indonesia E. Soetirto VIII 1996 Japan T. Morichi IX 2000 Australia J. Ternouth X 2002 India P. N. Bhat XI 2004 Malaysia Z. A. Jelan XII 2006 Korea I. K. Paik XIII 2008 Vietnam N.V. Thien XIV 2010 Taiwan L.C. Hsia
XV 2012 Thailand C.Kittayachaweng XVI 2014 Indonesia Yudi.Guntara.Noor
(1)
A 15 ID Effects of Estrous Synchronization of Bali Cattle Using PGF2α
Indira P N, Ismaya and Kustono
1
A 34 IN Prediction of 305 Days Lactation Milk Yield from Fortnightly Test
Milk Yields in Hill Cattle under Field Conditions R K Pundir
5
A 42 ID Development of Technology Production of Frozen of Swamp
Buffalo (Bubalus bubalis) in the Kampar Regency
Yendraliza, C. Arman and J. Handoko
9
A 116 ID Analysis of Reproductive Efficiency in Peranakan Ongole (PO)- and
its Crosses with Limousin (LIMPO) Cattle in East Java, Indonesia S. Suyadi and H. Nugroho
13
A 135 ID Performance Test and Genetic Potency of Bali Cattle Using Animal
Recording Software
Luqman Hakim and V.M. Ani Nurgiartiningsih
17
A 141 ID Application of Genetic Marker Technology for Predicting Twinning
Trait in Ongole Cattle
Endang Tri Margawati, Indriawati and Muhamad Ridwan
21
A 201 ID Membrane Status, Acrosome and Sperm Quality of Ongole Cross
Bred Bull after Sexing Using Percoll Density-Gradient Centrifugation and Albumin Separation
Trinil Susilawati, Sri Rahayu, Herni Sudarwati, Eko Nugroho, Setiabudi Udrayana and Lieyo Wahyudi
25
A 246 ID Phylogenetic Analysis of Simeulue Buffalo Breed of Indonesian
through Mitochondrial D-loop Region
Eka Meutia Sari, M. Yunus and Mohd. Agus Nashri Abdullah
29
A 339 JP Genetic Polymorphisms and Their Association with Growth and
Carcass Traits in Japanese Black Steers
F.N. Jomane, T. Ishida, K. Morimoto, T. Tokunagaand H. Harada
33
A 413 ID The Effect of Straw Position in Nitrogen Vapour During
Equilibration on Post-Thawing Motility and Membrane Integrity Following Quick Freezing in Maduran Cattle Sperm
H. Ratnani, MN. Ihsan, G. Ciptadi and S. Suyadi
Sustainable Livestock Production in the Perspective of Food Security, Policy, Genetic Resources and Climate Change
(4)
Code Title Page
A 951 ID Supplementation of Gonadotrophin in Culture Media in Vitro on
Matured of Goat Oocyte
Sri Wahjuningsih and Nurul Isnaini
132
A 1052 TW Heritability of Cytometric Measurements for Boar Sperm C. C. Chang, H. L. Chang, T. Y. Kuo and M. C. Wu
135
A 1114 ID Comparison of Two Different Method for Sperm Concentration
Measurement of Ram and Buck Semen R Iis Arifiantini, Ririn Riyanti and WM Nalley
138
A 1124 ID Determained Types of Intra Celullar Cryoprotectant (Cp) of Ultra Rapid Method Freezing Method on Survival of Goat Embryo
Agung Budiyanto
142
Poultry
A 5 IR Likelihood Method Estimation of Genetic Parameters of Fars Native
Chicken
Beigi Nassiri M.T, Jafari F, Fayazi, J and Longhair M. A
146
A 96 ID Contribution of Insulin-Like Growth Factor Binding Protein 2 Gene
on Growth Rate and Parameter Genetic of Kampung Chicken in Indonesia
Sri-Sudaryati, J.H.P. Sidadolog, Wihandoyo and W.T. Artama
150
A 119 TW Study on Genetic Diversity in Germplasm-Preserved White Tsaiya
Ducks by Microsatellite Markers
Y. Y. Chang, J. F. Huang, L. Y. Wei, M. C. Hsiao and H. C. Liu
154
A 182 ID KUB Chicken: “The First Indonesian Kampung Chicken Selected
for Egg Production”
Sofjan Iskandar and Tike Sartika
157
A 425 ID Polymorphisms of Growth Hormone (GH|MspI) Gene in Indonesia
Local Chicken and the Crossbred Using PCR-RFLP Ria Putri Rahmadani, Cece Sumantri and Sri Darwati
161
A 441 ID The Effect of Centrifugation Time on the Quality of Domestic
Chicken Spermatozoa Maintained at 5°C Yosephine Laura, Tri Yuwanta and Ismaya
165
A 675 ID Indigenous Chicken Breeds in Indonesia: Extinction Risk Status,
Driving Factors and Implications for Conservation
Indrawati Y. Asmara, Romy Greiner and Adam G. Drucker
169
A 676 KR Genome-wide QTL analysis of Economically Important Traits in
Korean Native Chicken
Dong-Won Seo, Hee-Bok Park, Shil Jin, Nu-Ri Choi, Muhammad Cahyadi, Chae-Kyoung Yoo, Jae-Bong Lee, Hyun-Tae Lim, Kang-Nyeong Heo, Cheorun Jo and Jun-Heon Lee
(21)
Code Title Page
E 265 ID Factors Affecting the Fattening Efficiency of Cull Bali Cows
Offered Local Complete Feeds
I G.N. Jelantik, G. E.M. Malelak, M. R. Deno-Ratu and C. Leo-Penu
870
E 411 ID Correlation Carcass Weight and Carcass Length with Fleshing Index
in Bali, Ongole Cross and Australian Commercial Cross Cattle Undang Santosa, Irlandia Ginanjar and Maria Yosita
874
E 508 ID Identification of Feeding, Physiology States and Hematology of
Deliver Twin Calves Bali Cows
AS Dradjat, TS Panjaitan LA Zainuri and Sasongko
877
E 686 TH Performance and Carcass Traits of Beef Steers Fed Crude Glycerin
in the Diet
P. Chanjula, S. Yimmongkol, T. Raungprim, S. Poonko, S. Majarune, and W. Maitreejet
881
E 705 ID Life Cycle Assessment of Local and Crossbred Cattle Production
Systems in Central Java, Indonesia
T.S.M.Widi, H.M.J. Udo, K. Oldenbroek, I.G.S.Budisatria, T. Viets and A.J. van der Zijpp
885
E 737 TH Comparative Study on Conjugated Linoleic Acid in Meat from Thai
Native Beef and Swamp Buffalo
Suthipong Uriyapongsan and Danupastra Chanapia
890
E 748 TH Study on Fatty Acid Profiles and Fatty Acid Concentration in Meat
from Thai-native cattle, Brahman-Native and Holstein-Friesian Suthipong Uriyapongson and Doungkamol Kusanteay
893
E 768 ID The Effect of Organic Selenium Supplemented Duration on the
Production Performance of Brahman Cross
Endang Yuni Setyowati, Undang Santosa, Denny Widaya Lukman and U. Hidayat Tanuwiria
896
E 787 ID Performance Ongole Grade and Simmental Ongole Crossbred Cow
at Village Breeding Center and Non Village Breeding Center at Special Region Yogyakarta
E. Baliarti, F. Ariyanti, Ismaya, N Ngadiyono,I Gede S Budisatria and Panjono
900
E 829 ID Morphometric Analysis of Bali Cattle in Jambi Province
Eko Wiyanto, Gushairiyanto dan Iskandar
904
E 912 TH Effect of Krabok Oil Supplementation on Feed Intake and Growth
Performance of Beef Cattle
C. Yuangklang, K. Vasupen, S. Bureenok, S. Wongsuthavas and B. Saenmahayak
908
E 998 ID Carcass Characteristics of Bali and Ongole Crossbreed Cattle Fed
With Sorghum Base
E.L. Aditia, R. Priyanto, M. Baihaqi, B.W. Putra and M. Ismail
Sustainable Livestock Production in the Perspective of Food Security, Policy, Genetic Resources and Climate Change
(24)
Code Title Page
F 229 VN Impacts of Socio-Cultural Factors on Beef Cattle Value Chain: a Case Study of Producers in the Northwest Region of Vietnam
Duong Nam Ha, Pham Van Hung, Nguyen Thi Thu Huyen, Laurie Bonney and Stephen Ives
1000
F 323 VN Policies and Institutions Governing the Beef Cattle Value Chain in the North-West Highlands of Vietnam
G. Duteurtre, Hoang Xuan Truong, Dang Thi Hai, L. Bonney and S. Ives
1005
F 433 ID Implementation of NLIS on Supply Chain Imported Cattle in West
Java Indonesia
Tawaf Rochadi and Rachmat Setiadi
1009
F 511 ID The Effect of Country of Design and Country of Manufacturing on
Perceived PRODUCT Quality: Empirical Study on UHT Milk Product Suci Paramitasari Syahlani, Rindang Matoati, Mujtahidah Anggriani Ummul Muzayyanah, Sudi Nurtini, Rini Widiati, and Tri Anggraeni Kusumastuti
1012
F 530 ID Techno-Economics Analysis of Complete Feed from Sugar Cane
Waste Product for Onggole Beef Cattle Adrizal, Fauzia Agustin and Welpriadi
1016
F 564 LK Influence of Socio Economics Status on Milk Production at Small-Scale Dairy Farmer’s Level
Senanayake S. R. L. I. B. , De Silva P.H.G.J. and Thakshala Seresinhe
1019
F 926 ID Characteristics of End Users in the Beef Supply Chain in East Java,
Indonesia
Atien Priyanti, D. Andrayani, I. G.A. P. Mahendri, and R. A. Cramb
1023
F 1135 LA Trans-Boundary Cattle and Beef Trade Flows in the Mekong Region: Implications on Sustainable Livestock Production for Smallholders in Vietnam and Laos
Luong Pham and Aloun Phonvisay
1027
H 95 LK Achieving Practice Change and Adoption in Small Holder Dairy Farms in Sri Lanka
D. E. Burrell
1033
H 287 ID Institutions Hindering the Sustainable Adoption of Supplementation Technology for Bali Cattle Calves in West Timor, Indonesia J.A. Jermias, C.L.O. Leo Penu, I.G.N. Jelantik, and A.C. Tabun
1037
H 351 ID Risk Perception Analysis of Dairy Farmers in the Southern Slope of Merapi Volcano Post Eruption 2010
S. Andarwati, R. Rijanta, R. Widiati and Y. Opatpatanakit
(33)
Code Title Page
K 727 ID Agronomic Performance of Leucaena leucocephala cv. Tarramba in
Tropical Environment of Sumbawa
Tanda Panjaitan, Muhammad Fauzan, Dahlanuddin, Michael Halliday, and Max Shelton
1365
K 745 ID Productivity and Species Diversity of Domestic Forage Based on
Altitude in Malang Regency, East Java
Iwan Prihantoro, Fransiska Rahmadani, Agustinus Tri Aryanto and M. Agus Setiana
1369
K 885 ID Effects of Land Type on Vegetative Character (Germination, Leaves,
Stems) and Rooting (Heavy, Long, Nodule) of Peanut (Arachis
hypogaea)
Bambang Suwignyo, S. Al - Kautsar and Bambang Suhartanto
1373
K 941 ID The Effect of Legumes Mulch as Fertilizer on Growth Characteristics
and Production of Rumput Benggala (Panicum maximum)
Lizah Khairani and Iin Susilawati
1377
POSTER PRESENTATION
Code Title Page
Genetic and Reproduction
Large Ruminant
A 63 BT Effect of Traditional Inter-Species Crossing (Bos indicus x Bos
frontalis) on Cattle Productivity in Bhutan Nar B Tamang, Tashi Samdup and John Perkins
1383
A 107 KR Molecular Genetic Evaluation of Korean Native Cattle Breeds
Using Microsatellite Markers
Sangwon Suh, Mi-Jeong Byun, Chang-Yeon Cho, Seong-Bok Choi, Young-Sin Kim, Yeoung-Gyu Ko and Jae-Hwan Kim
1387
A 163 ID Reproductive Performance of Brahman Cows Kept in Individual or
Group Pens in East Java, Indonesia
D. Ratnawati, L. Affandhy, D.A. Indrakusuma, D.E. Mayberry and D.P. Poppi
1390
A 167 LK Genetic Parameters and the Effect of Production and Type Traits on
Productive Life of Korean Holsteins at First Lactation
Nidarshani Wasana, Gwang Hyun Cho, Su Bong Park, Si Dong Kim, Jae Gwan Choi, Byung Ho Park and Chang Hee Do
1394
A 171 KR An Analysis of Monthly Measured Acetone and β Hydroxybutyrate
Acid in Milk of Holstein Cows
Yang Shin Chul, Gwang Hyun Cho, Chan Hyuk Park, Hyung Jun Song and Chang Hee Do
Proceedings of the 16th AAAP Animal Science Congress Vol. II
10-14 November 2014, Gadjah Mada University, Yogyakarta, Indonesia
(37)
Code Title Page
A 555 ID Genetic and Phenotypic Parameters for Milk Production of
Priangan Sheep Bess Tiesnamurti
1531
A 822 TH Efficacy of Estrus Synchronization Methods with Fixed-Time
Artificial Insemination in Admixture Breed Goat
Jitthasak Maungkhiow, Chanyut Kaphol, and Thunchira Thepparat
1535
A 932 ID Effect of Time after Mating on the Recovery and Motility of
Spermatozoa from the Female Reproductive Tract of Ewes Ismaya and Phillip Summers
1538
A 962 ID Quantitative and Qualitative Characteristics of Kosta Goat
Endang Romjali, Hasanatun Hasinah, Eko Handiwirawan, Bess Tiesnamurti, and Ismeth Inounu
1541
A 971 ID Study Identifcation of GDF9 Gene and Its Relationship with the
Prolific Traits on Four Breeds of Indonesian Local Goats Aron Batubara, R.R. Noor, A. Farajallah and B. Tiessnamurti
1544
A 992 ID Productivity Indices of Composite Breed of Sheep and Their
Contemporary
Subandriyo, Bambang Setiadi, Eko Handiwirawan, and Ismeth Inounu
1548
A 1091 TW Effect of Vitamin E on the Reproductive Performance of Nubian Goats and Barbado Sheep Ewes
Y. W. Chen and L. C. Hsia
1552
A 1092 TW Seasonal Variation of Semen Quality in Nubian Goats and Barbado Sheep
Y. W. Chen and L. C. Hsia
1555
A 1099 TH Estimates of Genetic Parameters for Kleiber Ratio from Birth to Weaning in Thai Native Goats
Sansak Nakavisut and Mongkol Thepparat
1558
Poultry
A 91 ID Identification of Avian Influenza Resistance Using 3 Primers Mx
Gene at Merawang Chicken from South Sumatera Island, Indonesia Tike Sartika
1562
A 100 TW Impact of Environmental Factors on Eggs at Late Stage of
Incubation in the Shipping Container
C. H. Cheng, C. H. Su, J. H. Lin, and J. F. Huang
1566
A 102 TW Study on Muscovy Semen Stored in Different Temperature
L. Y. Wei, H. C. Liu, Y. C. Chen, Y. Y. Chang, Y. A. Lin, and J. F. Huang
(38)
Code Title Page
A 299 ID Grouping of Alabio, Mojosari and Crossbred of Peking X White
Mojosari (PMp) Ducks Based on Their Growth T. Susanti and L.H. Prasetyo
1572
A 445 TH Genetic Evaluation for Reproductive Performance in Thai Native
Cocks (Pradu Hang Dam and Chee)
W. Boonkum, M. Duangjinda, B. Laopaiboon,and T. Wongpralub
1577
A 642 JP Genetic Diversity and Differentiation within Breeds of Native
Japanese Chickens Based on Microsatellite DNA Analysis T. Oka and M. Tsudzuki
1580
A 750 TH Comparative Study on Live Weight and Growth Performance of
Thai Synthetic Chickens
T. Buasook, S. Siripanya, B. Laopaiboon, M. Daungjinda and S. Kunhareang
1584
A 1007 IT A Logistic Model to Describe the Growth of a Nondescript Chicken
Breed From Apulia, Italy
Maria Selvaggi, Vincenzo Tufarelli, Francesco Pinto, Federica Ioanna, and Cataldo Dario
1588
A 1078 ID The Effects of Diluents and Cryoprotectants on Sperm Motility of
Native Chicken Frozen Semen
W. Asmarawati, Kustono, D. T. Widayati, S. Bintara and Ismaya
1592
Others
A 185 KR The Effect of Ultrasound Live Body Composition and Structure
Traits on Carcass Traits in Crossbred Pigs of Korea
ChangheeDo, Chanhyuk Park, Nidarshani Wasana, Jaegwan Choi,Su Bong Park, Sidong Kim, Gyuho Cho, Incheol Kim and Donghee Lee
1596
A 222 KR Selection Response of Production Traits in the Closed Herd in
Swine
ChangHee Do, JaeGwan Choi, YoungGuk Joo, ChanHyuk Park, Nidarshani Wasana, HyungJun Song, SeokHyun Lee, HyeongSeop Kim
1600
A 375 KR Production of Alpha1,3-Galactosyltransferase Null Pig Expressing
Membrane Cofactor Protein
Keon Bong Oh, Seongsoo Hwang, Jeong-Woong Lee, Sun-A Ock, Dae-Jin Kwon and Seok Ki Im
1604
A 656 JP Genome-Wide Association Study of Disease Caused by
Mycoplasma hyopneumoniae in Duroc
Tomoshi Yoneno, Shimazu Tomoyuki, Liushiqi Borjigin, Yuki Katayama, Ryosuke Otsu, Hayato Saito, Hiroshi Kunii, Toshimi Matsumoto, Tadahiko Okumura, Hirohide Uenishi, and Keichi Suzuki
Sustainable Livestock Production in the Perspective of Food Security, Policy, Genetic Resources and Climate Change
(46)
Code Title Page
B 192 TH Effect of Soybean Oil Supplementation on Conjugated Linoleic
Acid Contents and Milk Quality in Dairy Goat
Sasipron Cholumyai, Chaianan Racho and Udorn Srisaeng
1881
B 332 ID Usage of Sago Waste as Component of Complete Feed for Growing
Boerka Goats
Kiston Simanihuruk, Antonius and Juniar Sirait
1885
B 402 ID Effect of Different Protein and Energy Levels in Concentrate Diets
on Anglo-Nubian Young Goat Performance
Supriyati, L. Praharani, IGM Budiarsana and I-K. Sutama
1890
B 474 TH Effects of Supplementing Dietary Neem Foliage on Protozoan
Population in the Rumen and Faecal Nematode Egg Excretion in Meat Goats
S. Srisaikham, P. Paengkoum and W. Suksombat
1894
B 532 ID Nutrition Status of Female Bligon Goat Fed Diets Containing
Undegraded Protein Supplement
Ahmad Iskandar Setiyawan, Kustantinah, Subur Priyono Sasmito Budhi, Zuprizal and Nanung Danar Dono
1898
B 743 ID Application of Total Mixture Forages Silage on Sheep Farming:
Bean Sprouts Addition and Controlled Internal Drug Release Vaginal Insertion on Sheep Reproduction
Zaenal Bachruddin, Dodo Ramadhan, Yusuf Candra Kurnia, Edi Suryanto, Ismaya and Lies Mira Yusiati
1902
B 800 TH Effect of Sunflower Oil and Nitrate on Rumen Nutrient
Digestibility in Meat Goats Fed Low Quality Roughage Using Gas Production Technique
Jiravan Khotsakdee, Chalermpon Yuangklang, Thansamay
Vorlaphim, Bhutharit Vittayaphattananurak Raksasiri and Pramote Paengkoum
1906
B 845 DZ Clay in the Feeding of Ewes: Effect on the Quality of Milk and
Blood Parameters
Meredef Aissa, Ouachem Derradji, Soltane Mahmoud and Dehimi Mohamed Laziz
1910
B 881 MY Effect of Different Levels of L. leucocephala and M. esculenta
Leaves on Urinary Purine Derivatives of Goats Liyana, A. H., Alimon, A. R. and Samsudin, A. A.
1914
B 915 IR Fermentation Characteristics and Aerobic Stability of Triticale
Silage Treated with Formic Acid or a Mixture of Formic and Propionic Acids
A. R.Vakili, M. Danesh Mesgaran and A. Hodjatpanah-Montazeri
Effects of Diluents and Cryoprotectants on Native Chicken Sperm Motility
W. Asmarawati, Kustono, D. T. Widayati, S. Bintara and Ismaya
Faculty of Animal Science, Gadjah Mada University Yogyakarta, Yogyakarta, Indonesia Corresponding email: [email protected]
ABSTRACT
This research conducted to determine the influence of various cryoprotectants and diluents on post-thawing semen quality. Two diluent used were buffer phosphate (BP) and egg-yolk buffer phosphate (EYBP). Three cryoprotectants added to the diluents were glycerol (G), dimethyl formamide (DMF) and dimethyl sulfoxide (DMSO). Semen were collected from 4 males chicken aged 1 year to 1.5 years. After examined, 2 different diluents and 3 different cryoprotectants were adding into polled semen that was devided into 6 different treatment tubes (BP+G 7%, BP+DMF7%, BP+DMSO7%, EYBP+G 7%, EYBP+DMF7%, and EYBP+DMSO7%), then prosessed to be frozen with slow freezing method. Motility before dan after equilibration were examined. Frozen semen was stored at temperature -196 °C for a week, then thawed and its motility were examined. Fresh sperm quality were analyzed with mean and standard deviation, whereas data of motility before equilibration, after equilibration, and post-thawing were analyzed by analysis of variance using completely randomized design factorial 2 x 3. The result showed that diluents treatment gave significant
differences (P≤0.05) on sperm motility after equilibration and post-thawing, but not gave
significant differences on sperm motility before equilibration. Cryoprotectant treatments gave
significant differences (P≤0.01) on sperm motility before equilibration and after equilibration,
but not gave significant differences on post-thawing sperm motility. It can be concluded that DMF adding into EYBP diluents gave highest post-thawing sperm motility.
Key Words: Native chicken, Diluent, Cryoprotectant, Sperm motility.
INTRODUCTION
Various methods and procedures for freezing chicken semen have been studied for many years. Technology of artificial insemination face the major problem that is declining of sperm motility just in a few moment after ejaculation andstart losing ability to fertilize
(Tri-Yuwanta et al., 1998). Cryopreservation can be defined as problem solving for future use,
cryopreservation of cock semen remains a challenging obstacle due to low survival and motility rate of frozen-thawed cock semen (Makhafola et al. 2009). Fowl sperm have low resistance to the formation of internal ice crystals and reduce (Seigneurin and Blesbois, 1995), which reduce viability and motility. Motility is used as the most important value to measure sperm fertilizing ability. The critical steps in fowl semen cryopreservation are selection of suitable semen extender, proper cryoprotectant, and thawing method (Suidzinska and Lucaszewics, 2008; Makhafola et al., 2009). Egg-yolk buffer phosphate diluent is a balanced blending of phosphate buffer solution and fresh egg yolk, this diluent maintaining sperm motility and fertility (Bearden and Fuquay, 1997). The addition of glycerol as a cryoprotectant agent in frozen chicken semen as much as 8%, 12%, and 16% gave post-thawing sperm motility 48%, 50%, and 52% respectively (Saleh and Sugiyatno, 2007). Setioko et al. (2002) useglycerol, dimethyl sulfoxide (DMSO) and dimethyl formamide (DMF) in freezing duck semen with the results of post-thawing sperm motility 9.02%; 21.75%; and 32.86% respectively.The purpose of this study was to determine and comparing the effects of twodiluent used for native chicken frozen semen i.e.buffer phosphate (BP) and egg-yolk buffer phosphate (EYBP). Also, combine with three kind of cryoprotectants i.e glycerol (G), dimethyl formamide (DMF) and dimethyl sulfoxide (DMSO).
MATERIALS AND METHODS
Materials of this research were semen from 4 males native chicken aged 1 year to 1.5 years. Males were housed in individual cages under natural light. Males were fed in ad libitum diet with commercial feed for native chicken R-19 produced by PT Japfa Comfeed Surabaya. The extenders for semen was Buffer phosphate (BP) and egg-yolk buffer phosphate (EYBP) that was buffer solution from Na-phosphate monobasis, Na-phosphate dibasis, aquadest, and gentamicine 0.5 mg/100 mL. Adding of chicken egg yolk to phosphate buffer was needed for making EYBP. Three cryoprotectants added in diluents were Glycerol (G), Dimethyl Formamide (DMF) and Dimethyl Sulfoxide (DMSO). Semen was collected by Burrows and Quinn method (1937) that is dorso-abdominal massage (Donoghue and Wishart, 2000).Fresh semen from 4 cockerels was pooled and assessed for semen quality, which include: volume, color, consistency, motility, concentration, viability, and abnormallity. Semen extender was used Buffer phosphate (BP) with Gentamicin 0.5 mg/100 mL. Diluent EYBP was made from BP supplemented with 20% fresh egg yolk. Two kinds of extenderdivided into six different tubes and added with three kinds of cryoprotectants. Six extender treatments were BP+G 7%, BP+DMF7%, BP+DMSO7%, EYBP+G 7%, EYBP+DMF7%, EYBP+DMSO7%, respectively. Pooled semen from 4 cockerels after assessed, then divided into six equal parts. Each parts was diluted into each extender treatment, with ratio of sperm and extenders was 1: 2. Motility observations were carried out for three times i.e before equilibration,after equilibration and post-thawing. After motility observation, samples were equilibrated for 1 hour at 4 °C. After equilibration time, sperm motility were assessed. The semen samples were
directly filed into the straw and placed in pre-freezing unit (steam above LN2 for 8 minutes),
then directly in LN2 container for frozen storage for at least 1 week before being thawed for
post-thawing motility assessed. The semen samples were thawed at temperature 37 ° C for 30 seconds. Sperm motility was evaluated by visual determination of sperm with progressive motions using microscope. Data obtained from fresh sperm (includes volume, color, consistency, motility, concentration, viability, and abnormallity) was analyzed with a mean and standard deviation. Sperm motility before equilibration,after equilibration and post-thawing were analyzed with analysis of variance using completely randomized design factorial 2 x 3 and followed by DMRT test(Astuti, 1980).
RESULTS AND DISCUSSION
Fresh Sperm of Native Chicken
Fresh semen characteristic from four males of native chicken showed in Table 1 below. Table 1. Fresh sperm characteristic of native chicken
Parameter Result (average ± SD)
Volume (mL) Colour Consistency
Concentration (x 109spermatozoa/mL)
Motility (%) consistency, concentration, motility; viability,and abnormality. Results of this research within normal limits, which was volume ranges from 0.1 to 0.9 ml (Etches, 1996); viscous white
color (Utomo, 1997; concentrations between 3.109 to 7.109 spermatozoa/mL (Ax et al., 2000);
viabilityaverage around 96,64% and abnormality around 8% (Blesbois et al., 2005).
3URFHHGLQJVRIWKHWK$$$3$QLPDO6FLHQFH&RQJUHVV9RO,, 1RYHPEHU*DGMDK0DGD8QLYHUVLW\<RJ\DNDUWD,QGRQHVLD
Sperm Motilities before Equilibration
Sperm motilitiesbefore equilibration insix treatments of diluent BP+G 7%, BP+DMF7%, BP+DMSO7%, EYBP+G 7%, EYBP+DMF7%, EYBP+DMSO7% showed in Table 2 below. Table 2.Sperm motilities before equilibration
Extender Cryoprotectant averagens
Glycerol 7% DMF 7% DMSO 7%
BP
EYBP 57,50 ± 2,89 50,00 ± 8,16 68,75 ± 7,50 70,00 ± 4,08 52,00 ± 2,45 51,25 ± 7,50 59,42 ± 8,50 57,08 ± 11,37
Average 53,75 ± 6,94a 69,38 ± 5,63b 51,63 ± 5,18a
a, b : Data with different superscripts within each row were significantly different (P≤0.01) ns : non significant
The result showed that diluents treatment not gave significant differences on sperm motilities
before equilibration. Cryoprotectant treatments gave significant differences (P≤0.01) on
sperm motility before equilibration. The sperm motility before equilibration in DMF 7% treatment was significantly higher than that obtained with glycerol or DMSO. It is not much different from the results obtained by Setioko et al. (2002) thatuse DMF 7% as cryoprotectants in the freezing duck semen provide motility 74.53% after dilution. Under this research condition, DMF 7% was the best cryoprotectant tested for native chicken semen motility before equilibration in BP and EYBP diluents. Observations also indicate that there was significant interaction between diluentsand cryoprotectants on sperm motility.
Sperm Motilitiesafter Equilibration
Sperm motilitiesafter equilibration insix treatments of diluentBP+G 7%, BP+DMF7%, BP+DMSO7%, EYBP+G 7%, EYBP+DMF7%, EYBP+DMSO7% showed in Table 3 below. Table 3.Sperm motilities after equilibration
Extender Glycerol 7% Cryoprotectant DMF 7% DMSO 7% Average
BP
EYBP 48,75 ± 2,50 43,75 ± 4,43 63,75 ± 2,50 63,75 ± 9,46 63,75 ± 2,50 50,00 ± 7,07 58,75 ± 7,72
x
52,50 ± 10,98y
Average 46,25 ± 4,43a 63,75 ± 6,41b 56,88 ± 8,84b
a, b : Data with different superscripts within each row were significantly different (P≤0.01) x, y : Data with different superscripts within each column were significantly different (P≤0.05) ns : non significant
The result showed that diluents treatment gave significant differences (P≤0.05) on sperm
motilities after equilibration. The sperm motility in BP was significantly higher than EYBP.
Cryoprotectant treatments gave significant differences (P≤0.01) on sperm motilities after
equilibration. The sperm motilities after equilibration in DMF 7% and DMSO 7%were significantly higher than obtained with glycerol.Similar result from Setioko et al. (2002), also use DMF 7% as cryoprotectants to freezing duck semenand gave sperm motility 63.64% after equilibration. Under this research condition, DMF 7% was the best cryoprotectant tested for native chicken semen motility after equilibration in BP and EYBP diluents. Observations also indicate that there was significant interaction between type of diluents and type of cryoprotectants in affecting sperm motility after equilibration.
Post-thawing Sperm Motilities
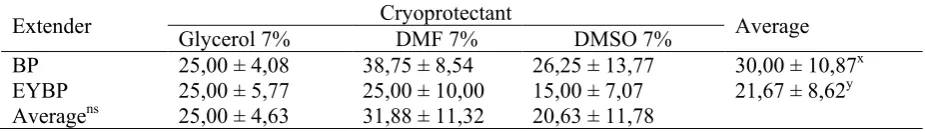
Sperm motilitiesafter thawing insix treatments of diluentBP+G 7%, BP+DMF7%, BP+DMSO7%, EYBP+G 7%, EYBP+DMF7%, EYBP+DMSO7% showed in Table 4 below.
The result showed that diluents treatment gave significant differences (P≤0.05) on
post-thawingsperm motility. Post-thawing sperm motility in BP was significantly higher than that obtained in EYBP. Cryoprotectant treatments not gave significant differences on
thawing sperm motility.Post-thawing sperm motility in DMF 7% was higher than DMSO 7% and Glycerol 7%.
Table 4.Post-thawing sperm motilities
Extender Glycerol 7% Cryoprotectant DMF 7% DMSO 7% Average
BP
EYBP 25,00 ± 4,08 25,00 ± 5,77 38,75 ± 8,54 25,00 ± 10,00 26,25 ± 13,77 15,00 ± 7,07 30,00 ± 10,87
x
21,67 ± 8,62y
Averagens 25,00 ± 4,63 31,88 ± 11,32 20,63 ± 11,78
x, y : Data with different superscripts within each column were significantly different (P≤0.05) ns : non significant
Makhafola et al. (2009) also use DMSO 5% as cryoprotectants to freezing semen from White Leghorn cockerel and gave sperm motility up to 40±4.7%; 20±4.0%; 15±3.7%; and 12±3.6% for 0 minute thawing; 30 minutes thawing; 60 minutes thawing; and 90 minutes thawing, respectively.Avian sperm are more sensitive to the freezing and thawing process and fertility rates of cryopreserved poultry sperm are dramatically lower than any of the domestic mammalian species (Donoghue and Wishart, 2000; Long, 2006; Gerzilov, 2010). Observations of post-thawing sperm motility also showed that there was no significant interaction between the type of diluentsto the type of cryoprotectants in affecting sperm motility.Under this research condition even though was not significantly, DMF 7% was the best cryoprotectant tested for post-thawing native chicken semen motility in BP and EYBP diluents.
REFERENCES
Astuti, J. M. 1980. Rancangan Percobaan dan Analisis Statistik, bagian I. Bagian Pemuliaan Ternak, Fakultas Peternakan UGM, Yogyakarta.
Ax, R.L, M. Dally, B.A. Didion, R.W. Lenz, C.C. Love, D.D. Varner, B. Hafez, and M.E.
Bellin. 2000. Semen Evaluation. In: Reproduction in Farm Animals 7th ed. E. S. E
Hafez. Ed., Lea & Febiger, Philadelphia. Pp. 365-375.
Bearden, H. J., and J. W. Fuquay. 1997. Applied Animal reproduction: Fourth Edition. Prentice-Hall Inc. USA.
Etches, R.J. 1996. Reproductiom in Poultry. UK University Press, Cambridge. Pp. 208 – 232. Gerzilov, V. 2010. Influence of Various Cryoprotectant on the Sperm Motility of Muscovy Semen Before and After Cryopreservation. J. Agri. Sci. and Tech, vol 2 (2) Pp :57 -60. Makhafola, M. B., K. C. Lehloenya, M. L. Mphaphathi, A. Dinnyes, and T. L. Nedambale.
2009. The Effect of Breed on The Survivability and Motility Rate of Cryopreserved Cock Semen. J. Anim. Sci. 39 : 242 - 245.
Saleh, D.M., dan Sugiyatno. 2007. Pengaruh Aras Glycerol terhadap Motilitas dan Fertilitas Spermatozoa Ayam Kampung yang Dibekukan dengan Nitrogen Cair. Animal Production pp: 45 - 48.
Setioko, A.R., P. Situmorang, E. Triwulanningsih, T. Sugiarti, dan D.A. Kusumaningrum. 2002. Pengaruh Krioprotektan dan Waktu Ekuilibrasi Terhadap Kualitas dan Fertilitas Spermatozoa Itik dan Entog. JITV 7 (4) : 237-243.
Seigneurin, F., and E. Blesbois. 1995. Effects of The Freezing Rate on Viability and Fertility of Frozen-Thawed Fowl Spermatozoa. Theriogenology 43 : 1351 – 1358.
Tri-Yuwanta, Kustono, C.H. Wibowo. 1998. Pengaruh Pencucian Sel Sperma dan Penyimpanan Sperma Ayam Kampung Terhadap Fertilitas. Bulletin Peternakan volume 22 : 64 – 72.
3URFHHGLQJVRIWKHWK$$$3$QLPDO6FLHQFH&RQJUHVV9RO,, 1RYHPEHU*DGMDK0DGD8QLYHUVLW\<RJ\DNDUWD,QGRQHVLD