Brain Research 882 (2000) 217–220
www.elsevier.com / locate / bres
Short communication
21
Activation of ATP receptor increases the cytosolic Ca
concentration
in nucleus accumbens neurons of rat brain
*
Masaru Sorimachi , Takashi Moritoyo, Kazuhiko Yamagami
Department of Physiology, Kagoshima University, Faculty of Medicine, Kagoshima 890-8520, Japan
Accepted 8 August 2000
Abstract
21
ATP increased the cytosolic Ca concentration ([Ca] ) in nucleus accumbens neurons acutely dissociated from rat brain. The ATPi
21 1 21
response was dependent on external Ca and Na , and was blocked by voltage-dependent Ca channel blockers. The results suggest
21
that the ATP-induced depolarization increases Ca influx resulting in the increase in [Ca] .i 2000 Elsevier Science B.V. All rights
reserved.
Theme: Neurotransmitters, modulators, transporters and receptors
Topic: Other neurotransmitters
21
Keywords: ATP; Fura-2 microfluorometry; Ca channel; Nucleus accumbens; Purinoceptor
The nucleus accumbens (NAc) has been considered to neurons express P2 type receptor, which is less permeable 21
function in promoting locomotion, in motivation, be- to Ca .
havioral drive, and reward [10,16,24] including the rein- The neurons of NAc were dissociated from 15- to forcing effects of addictive drugs [18], and also in the 23-day-old Wistar rats, which were decapitated under ether pathogenesis of schizophrenia [1,4,21]. The NAc neurons anesthesia after the study protocol was approved by the receive excitatory glutamatergic inputs, which may be Committee on Animal Experimentation, Faculty of Medi-modulated by a wide variety of endogenous neuro- cine, Kagoshima University. The brain was quickly re-modulators [2,11], from the cortical and subcortical limbic moved from the skull and placed in ice-cold saline area [7,8]. For example, dopamine depresses excitatory containing 150 mM NaCl, 5 mM KCl, 2 mM CaCl , 1 mM2 synaptic transmission [13,14,22,23] and thus has been MgCl , 10 mM HEPES and 5.5 mM glucose, the pH of the2 assumed to contribute to the behavioral and rewarding solution was adjusted to 7.4 by adding tris(hydroxy-effects of psychostimulants. The principal medium spiny methyl)aminomethane (Tris-base). The brain was sliced at GABAergic NAc neurons are generally quiescent with a thickness of 400 mm with a microslicer (DTK-1000, highly negative resting potentials [15,28,29], and are Dosaka, Japan), and the core and shell regions of NAc therefore strongly dependent on excitatory input to gener- were micropunched using a sample corer (FST; internal ate output. However, little is known about other excitatory diameter of 0.5 or 1 mm). The tissue pieces were treated inputs. We now report that the excitatory transmitter with saline containing dispase (1600–2500 pU / ml;
21
candidate ATP [3,25] increased the cytosolic Ca con- Goudoushusei, Japan) and DNase (0.02%; Sigma, type 1) centration ([Ca] ) in a subset of acutely isolated NAci for 45 min at 348C under continuous O2 bubbling, neurons, using fura-2 microfluorometry, and that these followed by further incubation in saline at room tempera-ture for 1 h. Finally, the tissues were gently triturated in saline containing 1% bovine serum albumin (BSA) using finely polished glass pipettes in a range of sizes, and the *Corresponding author. Tel.: 181-99-275-5227; fax: 1
81-99-275-dissociated cells were allowed to adhere to poly-L
-lysine-5231.
coated glass coverslips. Cells on the glass coverslip were E-mail address: [email protected] (M.
Sorimachi). loaded with 1 mM fura-2 acetoxymethyl ester (fura-2 /
218 M. Sorimachi et al. / Brain Research 882 (2000) 217 –220
AM), 0.1% dimethyl sulfoxide, and 0.2% BSA for 30 min synthetic enzyme for GABA, using the primary antibody at 378C. The coverslips were then mounted in a superfu- (Chemicon, USA).
sion chamber and placed on the stage of an inverted Although the NAc neurons were reported to be electri-microscope (Diaphot-TMD, Nikon, Japan). Cells were cally quiescent [15,28,29], we sometimes observed sponta-continuously superfused at a rate of 1 ml / min with neous fluctuations in [Ca] , which were markedly inhibitedi standard saline at 25–268C through a polyethylene tube, by adding 10 mM GABA (N57; data not shown). We the tip of which was placed 1–2 mm away from the cells. tentatively assumed that neurons in slice preparations are The changes in fluorescence ratios at 340 and 380 nm tonically inhibited by recurrent axon collaterals of other excitation wavelengths (F340 / F380) were measured using GABAergic neurons [5,17]. The addition of 10 mM a CAM-200 spectrometer (Jasco, Japan). The absolute glutamate increased [Ca] in most cells examined, buti value of [Ca] was calculated using the formula describedi about 34% of these cells responded to 100mM ATP (109 by Grynkiewicz et al. [12]: [Ca]i5b3K (Rd 2Rmin) / of 320 cells). The diameter of ATP-responsive cells ranged (Rmax2R). Calibration constants were determined in between 10 and 12.5mm, and these cells exhibited GAD-separate experiments with the same experimental set-up, as like immunoreactivity (32 of 32 cells; Fig. 1C), showing described previously [27]. Several points on the F340 / that purinoceptors are expressed on the GABA-containing F380 ratio tracing were selected, and the values of [Ca]i neurons.
are shown on the ordinates in the figure. After measuring As shown in Fig. 1A, the response to 10–500mM ATP the ATP-induced [Ca] increase, cells on a coverslip werei was compared with 100mM ATP, and the dose–response fixed with 4% (v / v) paraformaldehyde in 0.1 M phosphate relationship was obtained (Fig. 1B). Both 2-methyl-thio-buffer for 15–30 min at 48C, followed by the conventional ATP (2MeS-ATP) and ATPgS were effective in increasing immunostaining of glutamate decarboxylase (GAD), a [Ca]i (Fig. 1B), but neither a, b-methylene-ATP nor
Fig. 1. [Ca] increases induced by various concentrations of ATP and its analogues. (A) [Ca] increases induced by 10, 20, and 50i i mM ATP were compared with that by 100mM ATP. (B) Dose–response curves of ATP (s), 2-methyl-thio-ATP (MeS-ATP;d), or ATPgS (n). The mean of two responses to 100
M. Sorimachi et al. / Brain Research 882 (2000) 217 –220 219
21
b,g-methylene-ATP had any effect on [Ca] at a con-i Mn entry into chick ciliary ganglion cells [26] when centration of 200–500mM (N57 or 6, respectively). membrane depolarization was minimized by removal of
1
The ATP-induced [Ca] increase was dose-dependentlyi external Na , and considered that the purinoceptor chan-21
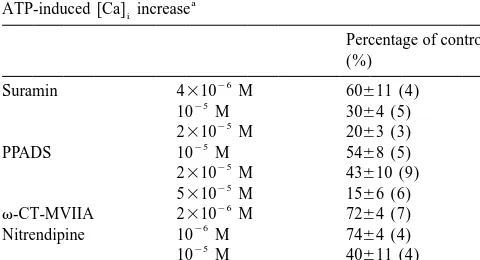
inhibited by P2 purinoceptor antagonists, suramin [9] and nels expressed are permeable to Ca . We therefore pyridoxal-phosphate-6-azophenyl-29,49-disulphonic acid employed the same manipulation to investigate the per-21 (PPADS; [20]) (Table 1). The ATP-induced [Ca] increasei meability of purinoceptor associated channels to Ca .
1 1
in the absence of external Na (replaced by NMDG ) was When the cells were stimulated for 1 min with 100 mM
1 1
only 562% (N55) of the mean of two controls at 150 mM ATP in a Na -free (NMDG ) solution containing 10 mM
1 21
Na and completely nullified in the absence of external Ca , the response was only 1063% (N511) of the mean 21
Ca (N56). These results suggest that ATP increased of two controls obtained with 15 s ATP stimulation at 150
21 1
Ca influx by depolarizing the membrane potential via mM Na . These results suggest that nonselective cationic 21
activation of P2 receptor type. channels on NAc neurons are less permeable to Ca .
21
To determine the types of voltage-dependent Ca Previous studies have demonstrated that P2 receptors are channels involved, we investigated the effects of specific situated at both the dopaminergic terminals of ventral antagonists on the ATP response. Individual antagonists tegmental area (VTA) neurons and the glutamatergic for L-type (nitrendipine), N-type (v-conotoxin-M VIIA), terminals of corticofugal pyramidal neurons, and that these
21
and P/ Q-type (v-agatoxin IVA) Ca channels caused receptors play an essential role in modifying the release of slight inhibitions (Table 1), the magnitudes of which were dopamine [19]. The present results add a new finding that comparable to that found in electrophysiological studies ATP acts on P2 receptors present on a subset of NAc [6]. The combined addition of three antagonists involving neuron as an excitatory neurotransmitter. The origin of
26
10 M nitrendipine reduced the ATP responses to ATP has not been investigated in the present study, but it 21
2263% of controls (N510). Cd at 0.2 mM alone or in is possible that ATP is released from axon terminals of 21
combination with three Ca channel antagonists reduced VTA neurons, where it is co-stored with dopamine [19]. the responses to 1463% (N59) or 962% (N55),
respec-tively. The responses in the presence of 10 and 50 mM
D-600 (methoxyverapamil) were also reduced to 3665% References (N56) and 564% (N53), respectively. On the other hand,
an L-type agonist Bay-K-8644 at 1mM markedly poten- [1] N.C. Andreasen, The mechanisms of schizophrenia, Curr. Opin. tiated the response to 50 mM ATP (439664%; N54). Neurobiol. 4 (1994) 245–251.
These results suggest that the ATP-induced membrane [2] J.A. Angulo, B.S. McEwen, Molecular aspects of neuropeptide regulation and function in the corpus striatum and nucleus accum-depolarization via the activation of nonselective cationic
21 bens, Brain Res. Rev. 19 (1994) 1–28.
channels activates Ca channels including L-, N-, P/ Q-,
21 [3] G. Burnstock, The past, present and future of purine nucleotides as and possibly R-types, and that the resultant Ca influx is
signalling molecules, Neuropharmacology 36 (1997) 1127–1139. mainly responsible for the observed [Ca] increase.i [4] M. Carlsson, A. Carlsson, A schizophrenia: a subcortical
neuro-We previously found that ATP increased [Ca]i and transmitter imbalance syndrome?, Schizophr. Bull. 16 (1990) 425– 432.
[5] H.T. Chang, S.T. Kitai, Projection neurons of the nucleus accum-Table 1
bens: an intracellular labeling study, Brain Res. 347 (1985) 112–
21
Effects of purinoceptor antagonists and Ca channel blockers on the
116.
a
ATP-induced [Ca] increasei [6] D. Churchill, B.A. MacVicar, Biophysical and pharmacological 21
Percentage of controls characterization of voltage-dependent Ca channels in neurons (%) isolated from rat nucleus accumbens, J. Neurophysiol. 79 (1998)
635–647.
26
Suramin 4310 M 60611 (4)
[7] M.J. Christie, R.J. Summers, J.A. Stephenson, J.C. Cook, P.M.
25
10 M 3064 (5)
Beart, Excitatory amino acid projections to the nucleus accumbens
25
2310 M 2063 (3) 3
septi in the rat: a retrograde transport study utilizing D-[ H]
25
PPADS 10 M 5468 (5) 3
aspartate and [ H]GABA, Neuroscience 22 (1987) 425–439.
25
2310 M 43610 (9)
[8] J.F. DeFrance, R.W. Sikes, R.B. Chronister, Dopamine action in the
25
5310 M 1566 (6)
nucleus accumbens, J. Neurophysiol. 54 (1985) 1568–1577.
26
v-CT-MVIIA 2310 M 7264 (7)
[9] P.M. Dunn, A.G.H. Blakeley, Suramin: a reversible P2-purinoceptor
26
Nitrendipine 10 M 7464 (4)
antagonist in the mouse vas deferens, Br. J. Pharmacol. 93 (1988)
25
10 M 40611 (4)
243–245.
27
v-AT-IVA 2.5310 M 6866 (7)
[10] H.C. Fibiger, A.G. Phillips, Reward, motivation, cognition:
psycho-a
The experimental protocol was the same as that shown in Fig. 1A. The biology of mesentelencephalic dopamine systems, in: V.B. Mount-cells were treated for 1–3 min with the indicated agent before 15 s castle, Bloom F.E. (Eds.), Handbook of Physiology, Intrinsic stimulation with 100mM ATP. The mean [Ca] increases induced by firsti Regulatory Systems of the Brain. Section 1: The Nervous System, and third stimulations were referred to as 100% and the response in the Vol. IV, American Physiological Society, Bethesda, MD, 1986, pp. presence of an agent was expressed as a percentage of the control. 647–675.
Numbers of experiments are indicated in parentheses. Abbreviations: [11] C.R. Gerfen, Synaptic organization of the striatum, J. Electron
220 M. Sorimachi et al. / Brain Research 882 (2000) 217 –220
21
[12] G. Grynkiewicz, M. Poenie, R.Y. Tsien, A new generation of Ca [21] H.Y. Meltzer, The mechanism of action of novel antipsychotic drugs, indicators with greatly improved fluorescence properties, J. Biol. Schizophr. Bull. 17 (1991) 263–287.
Chem. 260 (1985) 3440–3450. [22] S.M. Nicola, S.B. Kombian, R.C. Malenka, Psychostimulants de-[13] J. Harvey, M.G. Lacey, Endogenous and exogenous dopamine press excitatory synaptic transmission in the nucleus accumbens via depress EPSCs in rat nucleus accumbens in vitro via D1 receptor presynaptic D1-like dopamine receptors, J. Neurosci. 16 (1996) activation, J. Physiol. (Lond.) 492 (1996) 143–154. 1591–1604.
[14] J. Harvey, M.G. Lacey, A postsynaptic interaction between dopa- [23] S.M. Nicola, R.C. Malenka, Dopamine depresses excitatory and mine D1 and NMDA receptors promotes presynaptic inhibition in inhibitory synaptic transmission by distinct mechanisms in the the rat nucleus accumbens via adenosine release, J. Neurosci. 17 nucleus accumbens, J. Neurosci. 17 (1996) 5697–5710.
(1997) 5271–5280. [24] C.M. Pennartz, H.J. Groenwegen, F.H. Lopes da Silva, The nucleus [15] K. Higashi, K. Inanaga, S. Nishi, N. Uchimura, Enhancement of accumbens as a complex of functionally distinct neuronal ensem-dopamine actions on rat nucleus accumbens neurones in vitro after bles: an integration of behavioral, electrophysiological, and ana-methamphetamine pre-treatment, J. Physiol. (Lond.) 408 (1989) tomical data, Prog. Neurobiol. 42 (1994) 719–761.
587–603. [25] V. Ralevic, G. Burnstock, Receptors for purines and pyrimidines, [16] S.D. Iversen, Interactions between excitatory amino acids and Pharmacol. Rev. 50 (1998) 413–492.
dopamine systems in the forebrain: Implications for schizophrenia [26] M. Sorimachi, Y. Abe, K. Furukawa, N. Akaike, Mechanism
21
and Parkinson’s disease, Behav. Pharmacol. 6 (1995) 478–491. underlying the ATP-induced increase in the cytosolic Ca con-[17] Y. Kawaguchi, C.J. Wilson, S.J. Augwood, P.C. Emson, Striatal centration in chick ciliary ganglion neurons, J. Neurochem. 64
interbneurons: Chemical, physiological and morphological charac- (1995) 1169–1174.
terization, Trends Neurosci. 18 (1995) 527–535. [27] M. Sorimachi, K. Yamagami, J.-S. Rhee, H. Ishibashi, N. Akaike,
21
[18] G.F. Koob, Drugs of abuse: anatomy, pharmacology, and function of Excitatory effect of Cd on cat adrenal chromaffin cells, Brain Res. reward pathways, Trends Pharmacol. Sci. 13 (1992) 177–184. 832 (1999) 23–30.
[19] U. Krugel, H. Kittner, P. Illes, Adenosine 59-triphosphate-induced [28] N. Uchimura, H. Higashi, S. Nishi, Hyperpolarizing and depolariz-dopamine release in the rat nucleus accumbens in vivo, Neurosci. ing actions of dopamine via D-1 and D-2 receptors on nucleus Lett. 265 (1999) 49–52. accumbens neurons, Brain Res. 375 (1986) 368–372.
[20] G. Lambrecht, T. Friebe, U. Grimm, U. Windscheif, E. Bungardt, C. [29] F.J. White, R.Y. Wang, Interactions of cholecystokinin octapeptide Hildebrandt, H.G. Baumert, G. Spatz-Kumbel, E. Mutschler, and dopamine on nucleus accumbens, Brain Res. 300 (1984) 161– PPADS, a novel functionally selective antagonist of P2 purinocep- 166.
![Fig. 1. [Ca] increases induced by various concentrations of ATP and its analogues. (A) [Ca] increases induced by 10, 20, and 50 mM ATP were comparedwith that by 100ii mM ATP](https://thumb-ap.123doks.com/thumbv2/123dok/3139473.1382779/2.612.129.464.334.671/increases-induced-various-concentrations-analogues-increases-induced-comparedwith.webp)