Fitoterapia 75(2004)360–363
0367-326X/04/$ - see front matter䊚2004 Elsevier B.V. All rights reserved. doi:10.1016/j.fitote.2003.12.023
Short report
Antibacterial and brine shrimp lethality tests
of biflavonoids and derivatives of
Rheedia gardneriana
Luiz G. Verdi , Moacir G. Pizzolatti *,
a a,Ana Beatriz P. Montanher , Ines M.C. Brighente ,
aˆ
aArtur Smania Junior , Elza de F.A. Smania ,
ˆ
´
bˆ
bEdesio L. Simionatto , Franco Delle Monache
c dDepartamento de Quımica, Universidade Federal de Santa Catarina, Florianopolis, SC, Brazil
a ´ ´
Departamento de Microbiologia, Universidade Federal de Santa Catarina, Florianopolis, SC, Brazil
b ´
Departamento de Quımica, Fundacao Universidade Regional de Blumenau, Blumenau, SC, Brazil
c ´ ¸˜
Centro Chimica dei Recettori, C.N.R., Instituto di Chimica, Largo F. Vito 1, 00168 Roma, Italy
d
Received 18 November 2003; accepted 11 December 2003
Abstract
The hydroalcoholic extract of the leaves from
Rheedia gardneriana
yielded volkensiflavone
(
1
)
, fukugetin
(
2
)
, fukugiside
(
3
)
, GB2a-I-7-
O
-glucoside
(
4
)
and epicatechin
(
5
)
.
Com-pounds 1–5, and some derivatives of 1 and 2 were evaluated for lethality to brine shrimp
larvae and for antibacterial activity.
䊚
2004 Elsevier B.V. All rights reserved.
Keywords: Rheedia gardneriana; Biflavonoids; Antimicrobial activity
Plant.
Rheedia gardneriana
Planch et Triana
(
Guttiferae
)
, leaves collected in
February 1999 in Blumenau. A voucher specimen is deposited at the Dr Roberto
Miguel Klein Herbarium
(
Departamento de Ciencias Naturais, FURB, Blumenau
ˆ
)
under numbers 534–540.
*Correspondingauthor. Fax:q55-48-331-9711.
361
L.G. Verdi et al. / Fitoterapia 75 (2004) 360–363
Uses in traditional medicine. This species has been reported as folk medicine for
the treatment of inflammations of urinary tract, arthritis and to relieve pain
w1x.
Previously isolated classes of constituents. Biflavonoids
w2–4x, xanthones
w5–8x,
triterpenes
w5,9x, and 4-substituted coumarins
w7,10,11x.
New-isolated
constituents.
GB2a-I-7-
O
-glucoside
(
0.02%
)
and
epicatechin
(
0.004%
)
.
Tested material. Hydroalcoholic extract, partitioned with ethyl acetate resultingin
the EtOAc-fraction
(
3.42%
)
and aqueous fraction
(
5.97%
)
. Si-gel CC of the
EtOAc-fraction yielded volkensiflavone
(
1
) (
0.085%
)
, fukugetin
(
2
) (
0.15%
)
, fukugiside
(
3
) (
0.007%
)
, GB-2a-I-7-
O
-glucoside
(
4
) (
0.02%
)
, epicatechin
(
5
) (
0.004%
)
.
Derivatives: volkensiflavone peracetylated
(
6
)
, tetramethylated
(
7
)
, tetrabenzoylated
362
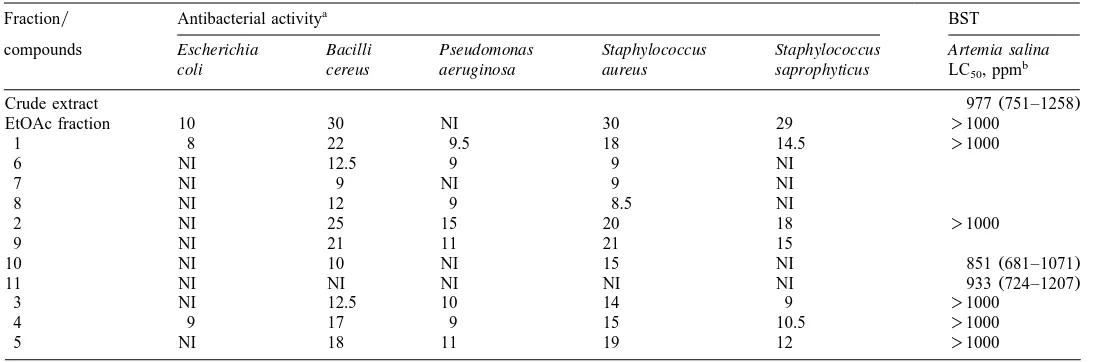
Antibacterial activity and Brine Shrimp Lethality Test(BST)of extract, fraction, isolated compounds and derivatives ofR. gardneriana
Fractiony Antibacterial activitya BST
compounds Escherichia Bacilli Pseudomonas Staphylococcus Staphylococcus Artemia salina
coli cereus aeruginosa aureus saprophyticus LC , ppmb
50
Crude extract 977(751–1258)
EtOAc fraction 10 30 NI 30 29 )1000
1 8 22 9.5 18 14.5 )1000
Values(mean of three replicates)are diameter of zone of inhibition(mm):-9, inactive; 9–12, partially active; 13–18, active;)18, very activew13x. a
wCompoundsxs1 mgy50ml;wFractionxs3 mgy50ml; NIsno inhibition. Confidence intervals 95%.
363
L.G. Verdi et al. / Fitoterapia 75 (2004) 360–363
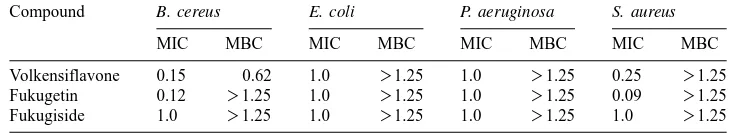
Table 2
Minimum inhibitory (MIC)and minimum bactericidal concentrations (MBC) from compounds ofR. gardnerianaa
Compound B. cereus E. coli P. aeruginosa S. aureus
MIC MBC MIC MBC MIC MBC MIC MBC
Volkensiflavone 0.15 0.62 1.0 )1.25 1.0 )1.25 0.25 )1.25
Fukugetin 0.12 )1.25 1.0 )1.25 1.0 )1.25 0.09 )1.25
Fukugiside 1.0 )1.25 1.0 )1.25 1.0 )1.25 1.0 )1.25
Concentrations in mgml .
a y1
Studied activity. Brine shrimp lethality test
w
12
x
, antimicrobial activity by the
agar-well diffusion method and determination of minimum inhibitory and minimum
bactericidal concentrations
w
13
x
.
Used microorganisms. Listed in Table 1.
Results. Reported in Tables 1 and 2.
Conclusions. The extract and methylated and benzoylated fukugetin showed toxicity
to brine shrimp. Compounds 1–5 were active against all Gram-positive bacteria
tested. Derivatives of compound 1 and 2 were less active than the startingmaterial.
References
w1x Cordeiro R, Nunes V do A, Almeida CR de. Plantas que Curam. Ed. Tres, 1996.ˆ
w2x Botta B, Mac-Quhae M, Delle Monache F, Delle Monache G. J Natl Prod 1984;47:1053.
w3x Luzzi R, Guimaraes CL, Verdi LG, Simionatto EL, Delle Monache F, Yunes RA, Floriani AEO,˜ Cechinel Filho V. Phytomedicine 1997;4:141.
w4x Cechinel Filho V, Da Silva KL, De Souza MM. Z Naturforsch 2000;55c:820.
w5x Braz Filho R, Magalhaes GC, Gottlieb OR. Phytochemistry 1970;9:673.˜
w6x Delle Monache F, Delle Monache G, Marini-Bettolo GB. J Natl Prod 1983;46:655.
w7x Delle Monache F, Delle Monache G, Waterman PG, Crichton EG, Alves de Lima R. Phytochem-istry 1984;23:1757.
w8x Delle Monache F, Botta B, De Mello JF, Coelho JSB, Menichini F. J Natl Prod 1984;47:620.
w9x Santos MH, Nagen TJ, Oliveira TT, Braz Filho R. Quimica Nova 1999;22:654.
w10x Hegnauer R. Chemotaxonomie der Pflanzen 1966;4:216.
w11x Basle BV, Rezende CMA, Gottlieb OR. Biochem Syst Ecol 1973;1:111.
w12x Meyer BN, Ferrigni NR, Putman JE, Jacobson LB, Nichols DE, McLaughlin JL. Planta Med 1982;45:31.