ISSN: 2087-3940 (print)
Vol. 3, No. 2, Pp. 64-67 ISSN: 2087-3956 (electronic)
July 2011
Oral squamous cell carcinoma patients which human papilloma virus
infection: a case control study in Muwardi Hospital Surakarta, Central
Java, Indonesia
ADI PRAYITNO1,♥, ELYANA AZNAR2, POERNOMO2, SUHARTONO TAAT PUTRA2
1Department of Dental and Oral Disease, Faculty of Medicine, Sebelas Maret University (UNS), Jl. Ir. Sutami 36A, Surakarta 57126, Central Java,
Indonesia. Tel. +62-271-664178. Fax. +62-271-637400. ♥Email: drgadiprayitno@yahoo.com
2 Faculty of Medicine, Airlangga University, Surabaya 60131, East Java, Indonesia.
Manuscript received: 27 February 2011. Revision accepted: 4 June 2011.
Abstract. Prayitno A, AznarE, Poernomo, Putra ST. 2011. Oral squamous cell carcinoma patients which human papilloma virus infection:
a case control study in Muwardi Hospital Surakarta, Central Java, Indonesia. Nusantara Bioscience 3:64-67. Annual incidence rates
for oral and pharyngeal cancer are estimated at 25 cases per 100,000 in developing countries. Human papilloma virus (HPV) was implicated in pathogenesis of Oral Squamous Cell Carcinoma (OSCC). Aims of this research were to know the incidence of OSSC patient which realized HPV infection. Women OSCC (15) and Benign Oral Squamous Cells (BOSC) (40) tissue biopsy frozen sections were from Departement of Oral and Dental, Muwardi Hospital in Surakarta from January to December 2007. Tissue was cut into two parts. To ascertain the type of neoplasm was subsequently stains with HE. To amplify the L1-HPV gene for 450bp long. The collected data was analyzed by Chi Square Test. The result of this experiment showed nine patients from 40 patients BOSC identified have HPV infections(9/40 = 23%). Eleven patient from 15 patient OSCC identified have HPV infections(11/15 = 73%). From Chi Square analysis have significant differences between BOSC and OSCC. HPV is a factor for OSCC pathogenesis.
Key words: developing countries, HPV, pathogenesis, OSCC, Moewardi Hospital.
Abstrak. Prayitno A, Aznar E, Poernomo, Putra ST. 2011. Pasien oral squamous cell carcinoma dengan infeksi virus papiloma manusia:
studi kasus kontrol di Rumah Sakit Muwardi Surakarta, Jawa Tengah, Indonesia. Nusantara Bioscience 3: 64-67. Tingkat insiden
tahunan untuk kanker mulut dan faring diperkirakan mencapai 25 kasus per 100.000 di negara-negara berkembang. Human papilloma virus (HPV) terlibat dalam patogenesis Oral Squamous Cell Carcinoma (OSCC). Tujuan penelitian ini adalah untuk mengetahui kejadian pasien OSSC yang mengalami infeksi HPV. Irisan beku biopsi jaringan OSCC (15) dan Benign Oral Squamous Cells (BOSC) (40) wanita diperoleh dari Bahian Mulut dan Gigi Rumah Sakit Muwardi Surakarta, dari bulan Januari sampai Desember 2007. Jaringan dipotong menjadi dua bagian. Untuk memastikan jenis neoplasma, maka diwarnai dengan HE. Untuk gen L1-HPV digunakan penanda berukuran 450bp. Data yang dikumpulkan dianalisis dengan Chi Square Test. Hasil penelitian ini menunjukkan sembilan pasien dari 40 pasien BOSC diidentifikasi mengalami infeksi HPV (9/40 = 23%). Sebelas pasien dari 15 pasien OSCC diidentifikasi mengalami infeksi HPV (11/15 = 73%). Dari analisis Chi Square terdapat perbedaan yang signifikan antara BOSC dan OSCC. HPV merupakan faktor patogenesis OSCC.
Kata kunci: negara berkembang, HPV, patogenesis, OSCC, Rumah sakit Muwardi.
INTRODUCTION
Factor that known implicated as a potential cock and or promoter cancer were tobacco, alcohol, radiation of sunrise, ionization radiation, carcinogen related work, environment pollutant, medicines, nutrition and infectious agent. Another factor is life in village, social-economic factor, age, gender and response immune mechanism. Information about another factor was little. The followed factor is periodontal disease chronic, bed oral hygiene, diseases of tooth, sharp of set teeth, electrogalvanism and edentulism. Another researcher found that human papilloma virus (HPV), especially 16 and 18 type, implicated in oral squamous cell carcinoma (OSCC) pathogenesis (Bsoul et al. 2005). Risk Factors (account for 75% of cases) are tobacco abuse confers 6 fold risk, smokers represent 90% of oral cancer
patients, alcohol abuse or heavy use, combined risk of heavy alcohol and tobacco use (women: 100 fold risk of oral cancer, men: 38 fold risk of oral cancer), other risks are sunlight exposure, poor dentition and viral infection (HSV, HPV) (Ravi and Yadav 2006; Ord et al. 2007).
The prevalence of oral cancer is also on the increase in Africa. Annual incidence rates for oral and pharyngeal cancer are estimated at 25 cases per 100,000 in developing countries. The rapid urbanisation and increasing access to, and utilization of tobacco in its various forms as well as alcohol, is leading to an increase in the incidence of oral pre-cancer and cancer. Epidemiology of oral cancer are squamous cell represents 90% of oral cavity tumors,
incidence increases with age and oral cancer is 9th most
r
research were which realized
M
Kind of th and the desig
group design.
Distric Hospit All patient of Benign Ora fiveteen biop Carcinoma (O Dental Clinic Java, Indonesi
Parrafin bl subsequently ascertain the t DNA isolatio was made by S up to 25 mg o microfuge tub buffer. Add 2 vortexing, and
The DNA Polymerase Ch fixed the HPV by Schmits (1 method with Ready To Go mixed with 5’GC(A/C)CAG
5’CGTCC(A/C)
AB) and 2 µ amplifications
seconds, 720C
amplification that present in The collec (SPSS for Win
R
The result Table 1-2.
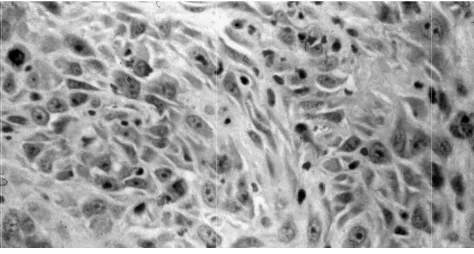
Figure 1. Histop
e to know th d HPV infectio
MATERIALS
his research is gn of this res Ethical clear tal Surakarta te
ts was women al Squamous C
sy frozen se OSCC) tissue p
of Muwardi ia from Januar
locks were m stains with H type of neopla on. Dioxyribo Schmits (1994 of tissue into be volume, and
0 µL of Prote d incubate at 5 A isolation
hains Reactio V. Diagnose re
994) and McM some modific PCR Bead (
2 µL HPV GGG(A/T)CATA
)A(A/G)(A/G)GGA µL DNA tem
s are 940C f
C for 50 s very conserve n all HPV subt
cted data was ndows 15).
RESULTS AN
of this experi
pathology view
P
he incidence on.
S AND METH
s observasiona search is post rance was don
eam and sign ns Fourty bio Cells (BOSC) ection of Ora patients collec Hospital in ry to Decembe made from cut
Haematoxylin asm. Cutting I onucleic Acid 4) with some small pieces, d add 200 ul o einase K stock
550 C overnigh
results we n (PCR) to am elated HPV in Millan and Fo
cations (25 µ (Amersham P V consensus
AA(C/T)AATG A(A/T)ACTAT mplate. PCR for 50 secon seconds and ed region of H types produce s analyzed by
ND DISCUSS
iment showed
w from HE stain
PRAYITNO et
of OSSC p opsy frozen se ) tissue patien al Squamous cted from Ora
Surakarta, Ce er 2007. tting I, which ne Eosine (HE
II was subject d (DNA) isol modifications place in 1.5 of DNA extra k solution, m ht.
ere subjected mplify L1-HP nfections are owler (1998). P µL microfuge Pharmacia Bio
primers (M GC3’ and M TC3’) (Cyber protocol for
nds, 590C fo
40C soak.
HPV the L-1 d 450 bp long y Chi Square
SION
d in Figure 1-3
n of OSCC.
al. – OSCC wit
atient
tional ontrol wardi 2008. ection nt and Cell al and entral
h was E) to ted to lation s. Cut mL a action mix by
th HPV infectio
re 2. Polymera C tissue frozen
re 3. Polymera C tissue frozen
el 1. The data 42.6years and m 40 patient
0 = 23%). E tified have HP Dental caries sidered to be t
ld. In developi her as commo
on
ase Chains Rea section, amplif
ase Chains Rea section, amplif
women patien
No. Age
he experiment i
HPV posit
11 (73%) 9 (23%) 20
this experime d OSCC was
BOSC ident Eleven patien PV infections
and periodo the major oral
ing countries on nor of the
action (PCR) L fied 450 bp lon
action (PCR) L fied 450 bp lon
nt with OSCC
Benign
No. Age
tive HPV nega
4 (27%) 31 (77%) 35
ent showed m 50 years. An tified have H nt from 15
(11/15 = 73% ontal disease l health proble
of Africa, the same order of
6
L1-HPV gene i g.
L1-HPV gene i g.
(Malignant) an
No. Age
ative Total
15 40 55
mean for BOS nd nine patien HPV infection patient OSC %).
3 (2): 64-67, July 2011
66
the developed world. An epidemiological description of a given health problem usually includes its prevalence, severity (morbidity, mortality) and age-adjusted distribution in the population. Oral diseases known to exist in each community must be assessed in this way in order to develop programmes appropriate to community needs. Based on this form of analysis, the most prominent oral health problems in Africa amongst low socio-economic communities include Noma, ANUG (Acute Necrotising Ulcerative Gingivitis), oral cancer, the oral manifestations of HIV and AIDS, oro-facial trauma, and dental caries. The highest global prevalence of HIV and AIDS is found in Africa. Studies have shown that the oral manifestations of HIV/AIDS are common. Candida infections, necrotizing gingivitis and oral hairy leukoplakia are the most common.
The prevalence of oral cancer is also on the increase in
Africa. Annual incidence rates for oral and pharyngeal cancer are estimated at 25 cases per 100,000 in developing countries. The rapid urbanisation and increasing access to, and utilisation of tobacco in its various forms as well as alcohol, is leading too (WHO/AFRO 2008).
In the case of high risk HPV infection and under favorable conditions, the viral genome is integrated into the host genome which is the necessary event for the keratinocytes immortality. During this process of integration the circular form of viral genome breaks at the level of the E1 and E2 regions, never at the level of the E6 or E7 region. Different studies have shown that the integrated part of the genome corresponds to E1, E6 and E7 while the regions from E2 to E5 are lost and are not transcribed in the tumours. The loss of E2 during this process of integration produces the loss of E6 and E7 control. Therefore, the sequences E6 and E7 are directly involved in the cellular cycle by inhibiting the normal functions of p53 and pRb respectively. The protein p53 is known as the "genome's guard" and in the case of DNA damage, the p53 can provoke the arrest of cellular division and assure the time necessary for DNA repair. If damage can’t be repaired, p53 is able to induce the programmed cellular death and prevent the propagation of DNA damage in subsequent generations of cells. In the case of other types of tumors p53 is usually mutated and acts as a real oncogene. In the case of HPV infection, E6 suppresses the properties of p53 gene product achieving the functional equivalent of the two hits required to knock out both alleles of a tumor suppressor gene. The mutations of p53 are normally not found. The E7 protein interacts with retinoblastoma protein (pRb), which is the crucial factor for the cellular cycle control. This interaction causes the release of the transcription factor E2F, which is now free to act and can stimulate the cellular division. E7 is also able to bind and inactivate the protein kinase inhibitors p21 and p27 and can interact with different proteins whose significance has still not been determined. E6 and E7 can cooperate with cellular oncoproteins like ras and myc which enables the virus to act at the level of growth factors and cellular and nuclear metabolism producing oncogenic cells. E6 and E7 can provoke directly DNA mutations of the host cell, probably by causing alterations of DNA repair mechanisms. This means that certain types of HPV are able
to cause malignant lesions even without the action of other cofactors (González Intxaurraga et al. 2002).
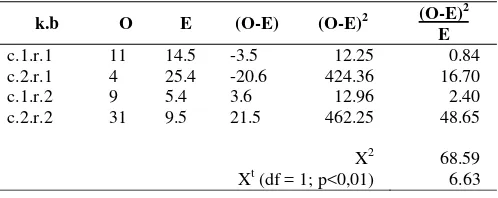
Because Xt (df=1; p<0.01) < X2 (68.59) that showed in
Table 3, so that have significant differences between BOSC and OSCC.
Table 3. Chi Square data analysis of table 1.
k.b O E (O-E) (O-E)2 (O-E)
2
E
c.1.r.1 11 14.5 -3.5 12.25 0.84
c.2.r.1 4 25.4 -20.6 424.36 16.70
c.1.r.2 9 5.4 3.6 12.96 2.40
c.2.r.2 31 9.5 21.5 462.25 48.65
X2 68.59
Xt (df = 1; p<0,01) 6.63
Cancer is widely perceived as a heterogeneous group of disorders with markedly different biological properties, which are caused by a series of clonally selected genetic changes in key tumour-suppressor genes and oncogenes. However, recent data suggest that cancer has a fundamentally common basis that is grounded in a polyclonal epigenetic disruption of progenitor cells, mediated by “tumour-progenitor genes”. Furthermore, tumour cell heterogeneity is due in part to epigenetic variation in progenitor cells, and epigenetic plasticity together with genetic lesions drives tumour progression. This crucial early role for epigenetic alterations in cancer is in addition to epigenetic alterations that can substitute for genetic variation later in tumour progression. Therefore, non-neoplastic but epigenetically disrupted progenitor cells might be a crucial target for cancer risk assessment and chemoprevention (Bsoul et al. 2005; Feinberg et al. 2006).
The inducible transcription of heat shock genes is the response to a plethora of stress signals, including (i) environmental stresses, (ii) nonstress conditions, and (iii) pathophysiology and disease states (e.g. HPV). Although changes in heat shock protein (Hsp) expression are associated with certain diseases, these observations leave open the question of whether this is an adaptation to the particular pathophysiological state, a reflection of the suboptimal cellular environment associated with the disease, or serves to warn other cells and tissues of imminent danger (Morimoto 1998).
In the face of injury or stress with the use ofvarious
mechanisms for anticipated, including systems of proteins
called molecular chaperones. The typical function of a
chaperone is to assista nascent polypeptide chain to attain a
functional conformationas a new protein and then to assist
the protein's arrival atthe site in the cell where the protein
carries out its functions.It has become increasingly clear
that disruption of chaperoningmechanisms contributes to
aging and disease. This review outlinesthe involvement of
defective chaperones in senescence and inseveral diseases.
Since chaperones are ubiquitous, their deficiencies and
defects are bound to affect diverse tissues and, hence,to be
g
gerontology. O genes that are
arge class o distress cell, folding proce Martin 2004; B
The accum condition to b
which are o
chaperones. N soluble forms dentification nside or aroun manipulate th expression of
aberrant agg
phenotypes or 1996; Li et al Morishima. 20 Gruschus 2008
The result patient identi OSCC patient have significa HPV infection factor for OSC
We thanks Surabaya for c acknowledge his PhD gran University fo Suryanata an collecting sam Prof. Noerhay
Bonnet S, Arche Thompson R 2007. A mito
Only a fractio inducible by
of stress prot
so to make p ess (Fan and
Bonnet et al.2
mulation of be the result o often accomp No matter how or insoluble f
of protein nd dead cells he level of Hs
f chaperones gregation, th
r delaying th
l. 2000; Much 005; Butler an 8; Garyali et a
CONC
of this exper fied have 23 t identified ha ant differences n. The conclu CC pathogene
ACKNOW
s to acknowle chance to stud to Sebelas M d. Biomedica r his laborat nd Tri Darm mple. We also yati Suripto for
REFE
er SL, Allaluni , Lee CT, Lopasc ochondria-K+ chan
P
on of chaperon stressors and teins. OSCC
possible distu Neff 2000; 2007).
protein misf
of anincrease
panied by H
w toxicityis g
fibrils ofthe d
aggregates, has tempted m sp70 to exam
would redu hereby sup he onset of t howski et al. nd Loh 2006 al. 2009).
CLUSION
riment showed 3% HPV inf ave 73% HPV
s between BO usion is HPV
sis.
WLEDGMENT
edge to Airlan dy about cance Maret Universit
al Laboratory tory facilities mani for thei thank Prof. W r their inspirat
ERENCES
is-Turner J, Har chuk GD, Puttagu nnel axis is suppr
PRAYITNO et
nes are encod thus belong t was include urbance in pr Rho et al. 2
folded in di of toxic funct Hsp70 and enerated, eith disease protein including H
manyresearch
mine whether uce the exten
pressing di the diseases 2000; Soto 2 ; Park et al. 2
d that from B fections and V infections. T OSC and OSC V as a signific
TS
ngga Universi er and so than ty in Surakart of Sebelas M s. We thanks
ir cooperatio Widya Asmar
tion.
romy A, Beauli unta L, Bonnet S ressed in cancer
al. – OSCC wit
ded in to the ed in rotein 2002;
stress tions, isease
(Ellis
th HPV infectio
normalization pro Cell 11: 37-51.
l SA, Huber MA he oral tissues: a Contemp Dent P r JS, Loh SN. 20 DNA binding dom
11): 2457-2465. JR. 1996. The c esponse to stress. Z, Neff RA. 2000 tress through m photoirradiation. J berg PA, Ohlsson origin of human ca ali P, Siwach P, Ganesh S. 2009. oxicity of misfold he ubiquitin-prote ales-Intxaurraga nd carcinogenesi chus JM. 2008. proteins? a hypoth
Y, Lee JS, Ko 70 inhibits apopt upstream of casp 25671.
n J. 2004. Ch onfinement. J Mo illan N, Fowler N pproach toward v Training Course, Mada University, moto RI. 1998. R ross talk betwe haperones, and n shima M. 2005. vanescent meetin howski PJ, Schaff FU. 2000. Hsp70 polyglutamine pro USA 97:1589-159 RA, Nikitakis NG of Maryland. Balti SH, Bolender N, DH. 2007. The c misfolded and end degradation via th
1): 153-165. M, Yadav S. pathogenesis and Cancer 43 (2): 60-HW, Choi MJ, L HR. 2002. Cytoto CPAE cells. Life S mits HL. 1994. Sp
cid methods. In: 0. Humana Press C. 2003. Un neurodegenerative O/AFRO. 2008. Strategy 1999-20 Congo.
on
motes apoptosis
A, Terezalmy G. 2 comprehensive r Pract 6 (4): 1-16. 006. Folding and main at physiolo
chaperonins, Indu . Academic Press
. Susceptibility o mediation of ph J Physiol 529 (Pt n R, Henikoff S ancer. Nat Rev G Singh PK, Puri R
The malin-lafori ded proteins by p easome system. H
MA, Stankovic R s. Acta Dermatov Do amyloid olig hesis. Amyloid 15 YG, Kim JI, S tosis downstream pase-3 activation
haperonin functi ol Recognit 7: 46 N. 1998. Typing o vaccine developm At Inter Univers Yogyakarta, Indo Regulation of the h een a family o negative regulator Control of cel ng. J Biochem 137 far G, Sittler A, W and Hsp40 chap oteins into amylo 94
G, Sauk JJ. 2007 imore Maryland. , Eisele F, Kosto cytoplasmic Hsp doplasmic reticul he ubiquitin-prot
2006. Oral squ prognostic valu -66.
Lee JN, Park JW, oxic mechanism Sci 70: 1923-193 pecies diagnostics
Clapp JP (ed). M s Inc., Totowa, NJ
nfolding the r e diseases. Nat Re Oral health in 008. WHO Repo
and inhibits canc
2005. Squamous eview for oral he
misfolding mech ogical temperatu
uction of chaper . San Diego, CA. of ATP-sensitive K hosphoinositides
3): 707-721. S. 2006. The epi Genet 7: 21-33.
R, Mittal S, Seng in complex supp promoting their de Human Mol Gene
R, Sorli R, Trevi venerol 11: 1-8. gomers act as tr 5 (3): 160-165. Seo JS. 2000. H
m of cytochrom n. J Biol Chem
ion – effects o 65-472.
of HPV isolates b ment for viral infe ity Center for Bi onesia, Novembe heat shock transc of heat shock f rs. Gen Develop 1 ll fate by HSP7 7 (4): 449-453 Wanker EE, Haye perones can inhib
oid-like fibrils. P
. Why screening
ova Z, Takeuchi J p70 chaperone m lum import-incom teasome system.
uamous cell car e of genomic al
, Kim JS, Park B m of vibrio vulni
4.
s protocols: PCR Methods in molec
J.
role of protein ev Neurosci 4 (1) the African Re ort office for A
6
cer growth.Canc
cell carcinoma ealthcare provider
hanisms of the p5 re. Protein Sci 1
ronins synthesis .
K+ channels to ce
as examined b
igenetic progenit
gupta S, Parihar R presses the cellul
egradation throug et 18 (4): 688-700 san G. 2002. HP
raps for misfolde
Heat shock prote me c release an
275 (33): 2566
of crowding an
by PCR: molecul ection. Icro-Unesc
iotecnology, Gaja er 1998. criptional respons
factors, molecul 12: 3788-3796. 70: more than a
er-Hartl MK, Har it self-assembly Proc Natl Acad S
works. Universi
J, Coffino P, Wo machinery subjec mpetent proteins Mol Biol Cell 1
rcinoma: etiolog lterations. Indian
BH, Sohn HS, Ki ificus cytolysin
R and other nucle cular biology. Vo
n misfolding ): 49-60.