Study of filtering Ag liquid sample by chitosan biomembrane using
laser-induced breakdown spectroscopy (LIBS)
Ni Nyoman Rupiasih, Hery Suyanto, Made Sumadiyasa, Christine Prita Purwanto, and Rendra Rustam Purnomo
Citation: AIP Conf. Proc. 1555, 32 (2013); doi: 10.1063/1.4820987
View online: http://dx.doi.org/10.1063/1.4820987
View Table of Contents: http://proceedings.aip.org/dbt/dbt.jsp?KEY=APCPCS&Volume=1555&Issue=1
Published by the AIP Publishing LLC.
Additional information on AIP Conf. Proc.
Journal Homepage: http://proceedings.aip.org/
Journal Information: http://proceedings.aip.org/about/about_the_proceedings
Top downloads: http://proceedings.aip.org/dbt/most_downloaded.jsp?KEY=APCPCS
Study of Filtering Ag Liquid Sample by Chitosan
Biomembrane Using Laser-Induced Breakdown
Spectroscopy (LIBS)
Ni Nyoman Rupiasih
1,*, Hery Suyanto
2, Made Sumadiyasa
2,
Christine Prita Purwanto
1and Rendra Rustam Purnomo
11
Biophysics Laboratory, 2Applied Physics Laboratory, Department of Physics, Faculty of Mathematics and Natural Sciences, Udayana University, Denpasar, 80361, Indonesia
*Email: [email protected]
Abstract. The capability of Laser-Induced Breakdown Spectroscopy (LIBS) to resolve filtration process of Ag liquid sample by chitosan biomembrane is demonstrated. The biomembrane was prepared by inversion method used to filter Ag liquid using pressurized technique samples which were then analyzed by monitoring the emission corresponding to Ag (I) at wavelength of 328 nm. The experiment was conducted by varying the laser energy i.e. 80, 120, and 160 mJ, where, subsequently, and its effect on the depth-profile from 20 - 200 μm was characterized by LIBS. The results showed that the physical processes of pressurized filtration led a homogeneous Ag in the membrane from the surface to a depth of 200 μm. The optimum condition was obtained at laser energy of 120 mJ. The adsorption occurred only on the surface of the membrane i.e. 20 μm depth, but there was no inclusion. Improvement of the detection performance of
adsorption process was done by heating the dripped membrane at 35 oC and was resulting in increase in emission
intensity as expected.
Keywords: Chitosan biomembrane; pressurized filtration; liquid Ag; Laser-Induced Breakdown Spectroscopy
PACS:82.35Pq
INTRODUCTION
Chitosan is the most abundant natural polymer after cellulose. It is a partially deacetylated product of chitin, which has many useful features such as non-toxic, biocompatibility, biodegradability, and antibacterial property. Due to its features, chitosan has been widely applied in many industries including weastewater treatment. Chitosan has the ability to form complexes with metals. It exhibits higher adsorption capacity for metal ion compared to that of chitin owing to the amino group content. However, the serious drawback of chitosan for metal ion adsorbent from practical utilization is that it is soluble in acidic media [1-2].
Laser induced breakdown spectroscopy (LIBS) or laser induced plasma spectroscopy is a useful method to determine the chemical composition of a wide range of materials including metals, liquids, aerosols, plastics, minerals, biological tissues, etc. [3]. As an analytical technique, LIBS presents some advantages to other techniques: no sample preparation, real time analysis, contactless analysis, apparatus or experimental simplicity, inexpensiveness, robustness, and quickness are interesting properties which makes LIBS an attractive analytical technique. In many cases is a non destructive technique [1]. In this technique
one requires a pulsed laser for generating micro-plasma on the target surface and elemental analysis is accomplished by studying the emission of the plasma plume. The performance of LIBS depends on several parameters including laser wavelength, pulse energy, pulse duration, time interval of observation, and geometrical configuration of collecting optics, target features, and properties of ambient medium. Extensive work has been devoted to optimize these parameters for the best experimental and commercial realization of this technique.
The main disadvantage of LIBS is the high limit of detection, about 500 ppm but 1ppm can be reached for some elements [4].
LIBS is a very versatile technique and have been used in siderurgy, industrial process control [4-7], environmental geochemistry, biohazard material detection, forensic science, microbiology, odontology, archaeology, cultural heritage and art conservation [8-12].
Several papers dealing with LIBS applications in depth-resolved analysis have also reported [5, 9-13]. In their reports, the laser was focused to a different extent resulting in high irradiances (typically in the order of 109 - 1011 Wcm-2).
(LIBS) to sample by technique. varying la 200 μm d process of carried ou Sample-1; by heating Sample-4;
MAT
The m characteris solubility sodium hy grade and
b. Prep
The ch by dissolv 1% acetic for 8 hou solution. T plate and d membrane 1% sodiu uncharged 3 times hydroxide membrane
In this been treat control (w ppm dropp naturally ( ppm dropp (adsorption membrane ppm (ads membrane filtration (adsorption
o resolve filtr y chitosan bio Depth profile ser energy from depth. Various f Ag solution ut i.e. control (w
natural adsorp g as Sample-3
and adsorption
TERIALS A
a. Ma
materials used a stics i.e. 87,9
in acetic acid ydroxide p.a. A were used with
paration of C
hitosan solution ving 10 g of chi acid solution urs at room te The dope soluti dried at room t e was in the NH um hydroxide d amino form, w
to eliminate , then dried e was around 2
c. Membr
study, the chi ted in 5 diffe without treatm
ped on the me (natural adsorp ped on the mem
n by heatin e to 35°C the sorption after e exposed to A
method with n with pressure
ration process omembrane us e analysis wa m 80 to 160 m s methods of in the membr without treatm ption as Samp 3; adsorption a
n with pressure
AND METH
aterials
are Chitosan p % deacetylate
is 99,4%; ace All reagents we hout further pu
Chitosan M
n of 4% (w/v) w itosan powder n. The mixture emperature to ion was poured temperature for H3+ form. It w
e solution to washed with di the excess d.The thickne
36 μm.
ane Treatm
itosan biomem erent ways i.e ment), Sample-embrane and a ption), Sample mbrane and he ng), Sample-4 n dropped wi heating), and Ag 1000 ppm
h pressure e).
s of Ag liqui sing pressurize s conducted b mJ each for 20 t f the adsorptio ranes were als ment) is called a ple-2; adsorptio after heating a e as Sample-5.
HODS
powder with ed and their etic acid and ere analytical urification.
Membrane
was prepared in 250 ml of e was stirred obtain dope d into a glass r 6 days. The was dipped in o reach the
istilled water of sodium ess of the
ments
mbranes have e. Sample-1: to prob treated the sur uses N there i
FIGUR
pressur scanned 329 nm
d. Laser-In
Spect
ser induced br be the present d chitosan biom
rface. The LIB Nd-YAG Lase width), 200 m spectrometer w spectrum 200-tion of 0,1 n
nt linier silicon
pixels. The ou
OLIBS and OOI
onitor. the present stu es of each sca essary scan). T
yed in the exp he pulse to pul rements perfor laced approxim and a relativel
n the LIBS m entative avera ion spectra w f successive la zed.
RESULTS
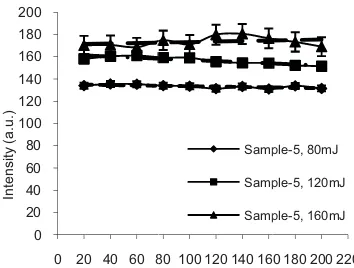
gure 1 shows L pressurized t ple-5) scanned w
327 - 329 nm. nm in Sample
on line of Ag ntrol membran s Ag in the Sam
RE 1. LIBS sp rized treated ch d with laser ene m.
nduced Brea
troscopy (
LI
reakdown spec of Ag elemen membrane at v BS 2500-7 plus
er (1064 nm mJ (max. energ
with specificat -980 nm, with nm using 7 de
n CCD arrays
utput of the spe
ICOR software
udy, the delay t an, and 0 pulse The laser energ periments was
lse energy vari rmed, was no > mately 2 cm in ly wide spot (a measurement in age of the ma ere recorded aser pulses and
S AND DISC
LIBS spectra of treated chito with laser ener It shows a pe e-5 which is
(I), whereas n ne (Sample-1) mple-5 that adm
pectra of contro hitosan biomem ergy of 120 mJ
akdown
IBS
)
ctroscopy was nt in the pressu various depths s model was us (wavelength), gy)) and echell
ions such as, 7 channels, FW etectors with
s that given to ectrometer ana es that displey
time used was e cleaning (to gy density (flu in the range 60 ation througho > ± 5%. The sa n front of the approx. 1 mm n order to obt aterial compos separately for d were subsequ
CUSSION
f control (Samp osan biomem
rgy of 120 mJ ak at waveleng
known as at no peak observ . This explain mitted by press
ol (Sample-1) a mbrane
(Sample-in the range 327
used urized from alyzed yed on sition.
each uently
FIGURE 2
biomembra laser energi spectrum is for the com
Figure chitosan b 329 nm fo 160 mJ. C of 120 mJ that the increasing of electron excited he The spectr signal as l continuum these resu analyzed optimal co
Figure Ag (I) at (with laser of depth fr biomembr emission w Ag has be the higher This mea increase, t process tak
For fu deviation measuring adjacent a standard d 2% for las mJ.
2. LIBS spectra
ane (Sample-5) i ies 80, 120, and s presented (kep mparison.
2 shows LIB biomembrane ( or varies of lase Control (Sampl J was also kept intensity of A g laser energy.
ns from the ato ence more em rum also shows laser energy in m emission of
ults, further ex on laser energ ondition for the
3 shows the in wavelength o r energies 80, 1 from the surfac rane (Sample-5 was observed een evenly dist r laser energy ans that the i the peak becom
ke place. urther examina
of the LIBS g the intensity
areas on the deviation in th ser energy 80 a
a of the pressur in the range 327 d 160 mJ. Contr pt at laser energ
BS spectra of (Sample-5) in er energies 80 m le-1) spectrum t for the comp Ag (I) peak This describe oms and more mission of spec s increasing in ncreases, whic the plasma fo xperiments we gy of 120 mJ e chitosan biom ntensity of atom of 328 nm obt
120, and 160 m ce, of the press 5). The graph for all depths tributed within y, 160 mJ sho intensity of t mes broad and
ation of Fig. intensity was from three ide sample surfac he intensities o and 120 mJ, an
rized chitosan 7 - 329 nm for rol (Sample-1) gy of 120 mJ)
the pressurize the range 327 mJ, 120 mJ, an m at laser energ arison. It show increases wit es more numbe
ions have bee ctrum obtaine
the backgroun ch is due to th orm [14]. From ere focused an
J and stated a membrane.
mic emission o tained by LIB mJ) as a functio surized chitosa shows that th s indicating th n the sample. A
ows fluctuation the backgroun d self adsorptio
3, the standar s calculated b entical series o ce. The overa of Ag (I) was for the
To after h before the las Ag (I) other a
0
RE 3. Intensity ngth of 328 nm 0, and 160 mJ as pressurized chit
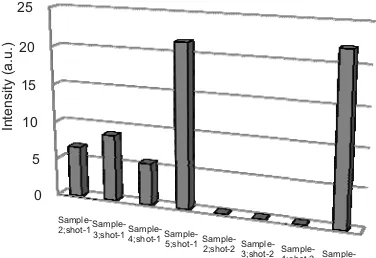
study about th an biomembr
ent are introdu natural (Samp μm) by laser e obtained was second shot) ion process oc mance of t ption by heati d membrane w ated to 35°C
sed to ~ 8.75 a nfluenced by t d shot was also heating method
dripping it ple-4), gave int
ity of Sampl d) was the high ese results des rane properly w ure method).
cally and phy ces strong cova providing a s ser shot and pr at wavelength adsorption met
0 20 40 60 8
y of atomic em obtained by LIB s a variation of d tosan biomembra
he adsorption p rane, four m uced. The resul ple-2) after first energy of 120
~ 6.67 a.u, wh was null, i ccurred. To im the adsorptio ing method w with the same p (Sample-3) an a.u. This indica the temperatur o zero. Further d was used. He with 1000 tensity as low le-5 (adsorpti hest at ~ 21.5 a scribe Ag liqui when pressuriz This causes ysically with t alent bonds in t strong rebound roduced plasma h of 328 nm is h
hods.
80 100 120 140 16
Depth (µm) Samp
Samp
Samp
mission of Ag BS with laser en depth from the s ane (Sample-5).
process of Ag methods mem lts are shown in
t shot (shot-1, mJ, the intens hile at a depth indicating tha mprove the dete on processes was introduced
process of Sam nd the intensi ates that the pr re. Result from rmore an adsor
ating the mem ppm Ag sol w as, ~ 5.5 a.u
ion with pre a.u.
id penetrate int zed (adsorption Ag interact the membrane the membrane d momentum d a emission inte high compared
0 180 200 220 ple-5, 80mJ
ple-5, 120mJ
ple-5, 160mJ
(I) at nergies
urface
in the mbrane n Fig. depth ity of of 40 at no ection
, an d, the mple-2
ity is rocess m the rption mbrane lution u. The essure
to the n with both e and
FIGURE 4
328 nm for
The ad biomembr ions which with N in only a pai bond is ca ion is an donor. Th sufficiently relatively results of carried ou relatively (Sample-2 produced caused a membrane N that acte a low inten
The p
breakdown of Ag ele biomembr was analy (waveleng process re the membr laser ener process oc without in treatment a
0 various membra
dsorption proce rane can be de h were chemica [Ag (NH3)2]+
ir of electrons lled a coordina electron acce herefore, the fo
y strong when low emission f this adsorpt
ut by heatin higher emis 2). The adsor low emission damage to th e and eliminati
ed as Ag adsor nsity.
CONC
present study n spectroscopy ement in the rane at various yzed by monit gth of 328 nm esulted in a hom
rane up to a de rgy was 120 m ccurred only at nclusion. Impro
at 35qC.
Sample-2;shot-1
Sample-3;shot-1Sample-4;shot-1Sa 5;s
mission of Ag (I) ane treatments.
esses occurred escribed as fo ally adsorbed t as a covalent b s shared by at ation covalent b eptor, while N
ormed adsorpt the laser shot
(Sample-2). T tion, the rein ng (Sample-3) sions than w rption after h ns. In this cas he physical s ing or reducing
rption media, t
LUSION
demonstrated y (LIBS) to pr pressurized t s depths from toring the em m). The pressu
mogenously d epth of 200 μm mJ. The chem t the surface o ovements can b
mple-shot-1
Sample-2;shot-2Sample-3;shot-2Sa 4;s
) at wavelength
d in the chitosa llows. The Ag through bondin bond resulted i toms N. Such
bond. Here, Ag N is an electro ion layer is no
and hence gav To enhance th nforcement wa ) and produc without heatin heating metho se, heating ha structure of th g the number o thus resulting i
laser induce robe the presen treated chitosa m the surface. mission of Ag
urized filtratio istributed Ag i m. The optimum mical adsorptio
f the membran be done by he
ample-shot-2
Sample-5;shot-2
e authors w matics and rsity for the LI
REF
R. Popuri, Y. V
ioresour. Techno
. Zieliska, A. Adsorption of ca inetics and equ
hemistry and App olume XV, edite olish Chitin Soci . Ahmed and M.
ioanal Chem385
Maravelaki-Kal . Zafiropulos,
001).
. Ahmed and M
8, No. 8, August, .C. Garcia, J. M
aserna, Spectroch
W.-B. Lee, J. Wu
pectrosc. Rev39, St-OngeU and 99-308 (2000).
St-OngeU a reakdown Spe haracterization”, ational Institute, M. R. Leahy-Ho urnett, Y. Dikm
ensors10, 4342-M. Vadillo, C aserna, J. Anal. A
J. Radziemski
lasma and Appl
989.
WLEDGME
wish to thank Natural S IBS facility.
FERENCES
Vijaya, V. M. B
ol100, 194-199 G. Chostenko admium ions on uilibrium studi
plication of Chit
ed by M. M. Ja iety, 2010, pp. 73 . A. Baig, J. App
A. Brysch, M. K
Sturm, Spectroc
oehne, R. Noll
5, 225–233 (200
laitzaki, D. Angl
Spectrochim. A
M. A. Baig, IEEE
, 2052-2055 (20 M. Vadillo, S. Pal
him. Acta B56, u, Y. –I. Lee and
, No. 1, 27-97 (2
M. Sabsabi, Spe
and M. Sabsa ectroscopy Stu , Ph.D. Thes
2010. ppa, J. Miragli melik, C. McEnn
4372 (2010). C. C. Garc´a, S
At. Spectrom13, and D. A Cre
lications, New Y
ENTS
k the Facult ciences, Uda
S
Boddu and K. A (2009).
and S. Truszko chitosan memb
es” in Progre
tin and Its Deriv
aworska et. al., 3-78.
pl. Phys106, 03
Kraushaar, I. M
him. Acta B 56
and V. Sturm, 6).
los, V. Kilikoglo
Acta B 56, 88
EE. Trans. Plasm
10).
lanco, J. Ruiz an , 923-931 (2001) d J. Sneddon, J.
2004).
ectrochim. Acta
abi, “Laser In udies for M sis, Homi B
iotta, R. Osiand nis and J. B. S
S. Palanco and , 793–797 (1998
emers, Laser In
York: Marcel D
ty of ayana
Abburi,
owski, branes:
ess on vatives
Lodz:
nduced aterial Bhabha
der, J. Spicer,
d J. J. 8).
nduced