Democratic People’s Republic
of Korea 2017
Acronyms
AD Auto disable
AEFI Adverse events following immunization
AFP Acute flaccid paralysis
BCG Bacillus Calmette-Guérin vaccine
CES Coverage evaluation survey
cMYP Comprehensive multi-year plan
CRS Congenital rubella syndrome
DHS Demographic health survey
DT Diphtheria tetanus toxoid, pediatric
DTP Diphtheria – tetanus – pertussis vaccine
DTP-Hib-HepB Pentavalent vaccine
DTP-Hib-HepB3 3rd dose pentavalent vaccine
EPI Expanded programme on immunization
GDP Gross domestic product
HCW Health care worker
HepB Hepatitis B vaccine
Hib Haemophilus influenzae type b
HPV Human papilloma virus
IgM Immunoglobulin M
IPV Inactivated poliovirus vaccine
JE Japanese encephalitis
JE_Live-Atd JE live attenuated vaccine
JRF WHO UNICEF joint reporting form
LB Live birth
M Measles
MCV1 First dose measles containing vaccine
MCV2 Second dose measles containing vaccine
MICS Multiple indicator cluster survey
MMR Measles mumps rubella vaccine
MNT Maternal and neonatal tetanus
MR Measles rubella vaccine
NCIP National committee on immunization practices
NID National immunization day
NTAGI National technical advisory group on immunization
NPEV Non-polio enterovirus
NT Neonatal tetanus
OPV Oral poliovirus vaccine
bOPV Bivalent OPV
tOPV Trivalent OPV
PCV Pneumococcal conjugate vaccine
SEAR WHO South-East Asia Region
SIA Supplementary immunization activities
SNID Subnational immunization day
Td Tetanus diphtheria toxoid; older children, adults
TT Tetanus toxoid
TT2+ 2 or more doses TT
VDPV Vaccine derived poliovirus
VPD Vaccine preventable diseases
WCBA Women of child bearing age
Contents
Impact of rouine immunizaion
Page
No.
EPI history 5
Basic informaion 2016 Table 1 5
Immunizaion schedule 2016 Table 2 5
Naional immunizaion coverage 1980 - 2016 Figure 1 6
Immunizaion system highlights Table 3 6
DTP3 coverage, diphtheria and pertussis cases 1980 - 2016 Figure 2 7 Reported cases of vaccine preventable diseases 2011 - 2016 Table 4 7
DTP-Hib-HepB3 coverage by province 2015 Figure 3 7
DTP-Hib-HepB3 coverage by province 2016 Figure 4 7
Towards measles eliminaion and rubella/congenital rubella
syndrome control
Page
No.
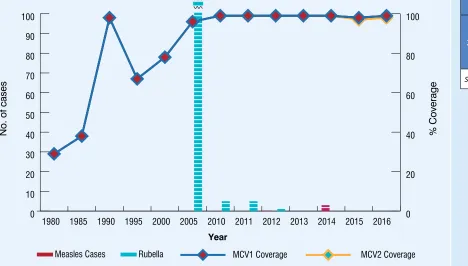
MCV1 and MCV2 coverage, measles and rubella cases, 1980-2016 Figure 10 11
MCV supplementary immunizaion aciviies Table 7 11
MCV1 coverage by province 2015 Figure 11 12
MCV1 coverage by province 2016 Figure 12 12
MCV2 coverage by province 2015 Figure 13 12
MCV1 coverage by province 2016 Figure 14 12
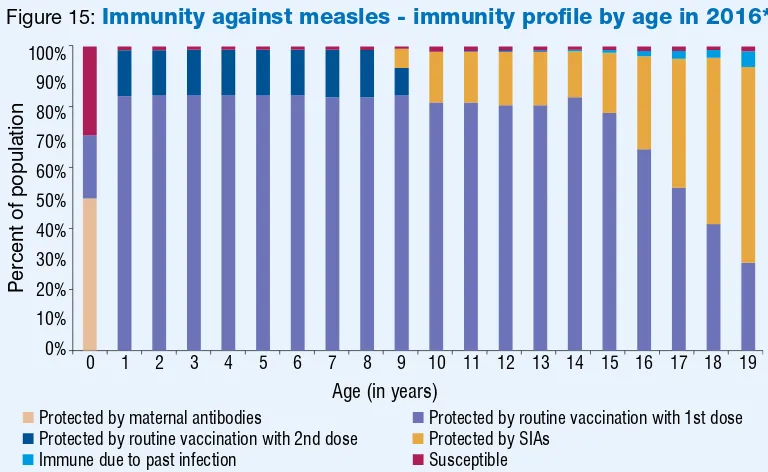
Immunity against measles – immunity proile by age in 2016 Figure 15 12
Subnaional risk assessment for measles and rubella Figure 16 12
Sporadic and outbreak associated measles cases by month 2011 - 2016 Figure 17 13 Immunizaion status of conirmed (laboratory and Epi linked) measles outbreak
associated cases by age 2011 – 2016 Figure 18 13
Quality of ield and laboratory surveillance for measles and rubella 2012 - 2016 Table 8 14
Performance of laboratory surveillance 2012 - 2016 Table 9 14
WHO supported laboratory network for VPD surveillance Figure 19 15
Maternal and neonatal tetanus eliminaion is sustained
Page
No.
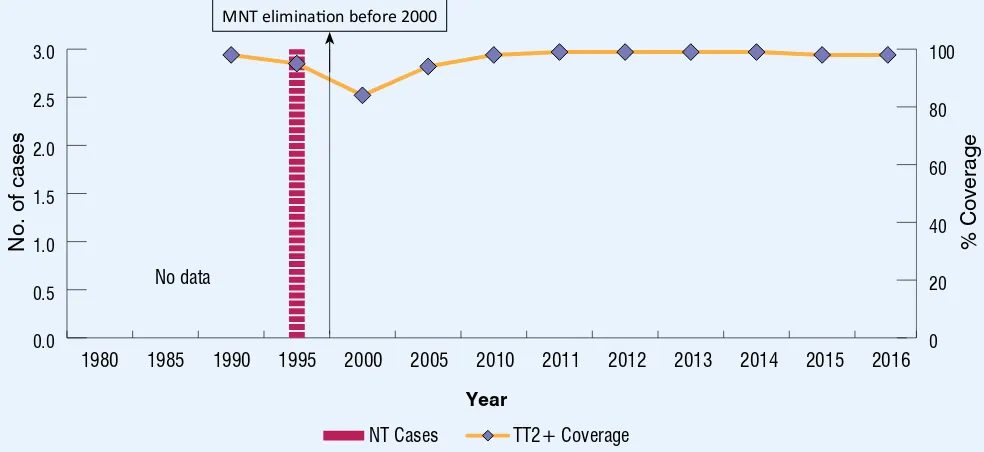
TT2+ coverage and NT cases 1980 - 2016 Figure 5 8
Polio-free status is maintained
Page
No.
AFP surveillance indicators 2011 - 2016 Table 5 9
Non-polio AFP rate by province 2015 Figure 6 9
Non-polio AFP rate by province 2016 Figure 7 9
Adequate stool specimen collecion percentage by province 2015 Figure 8 10 Adequate stool specimen collecion percentage by province 2016 Figure 9 10
Democraic People’s Republic of Korea: province level map
WHO South-East Asia Region
EPI history
• EPI launched in 1980
• HepB vaccine introduced in 2003 • AD syringes introduced in 2003
• HepB birth dose introduced in 2004 • DTP-HepB vaccine introduced in 2006
• MCV2 introduced in 2008 • DTP-Hib-HepB vaccine introduced
in 2012
• IPV vaccine introduced in 2015
• tOPV to bOPV switched on 18 April 2016.
Source: cMYP 2016-2020 and EPI/MOPH
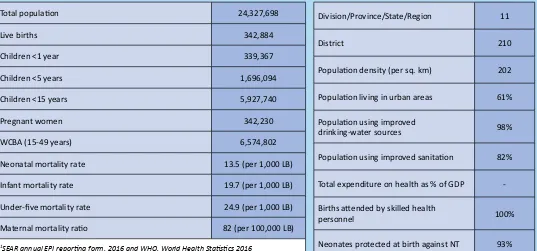
Table 1:
Basic information
12016
Total populaion 24,327,698
Live births 342,884
Children <1 year 339,367
Children <5 years 1,696,094
Children <15 years 5,927,740
Pregnant women 342,230
WCBA (15-49 years) 6,574,802
Neonatal mortality rate 13.5 (per 1,000 LB)
Infant mortality rate 19.7 (per 1,000 LB)
Under-ive mortality rate 24.9 (per 1,000 LB)
Maternal mortality raio 82 (per 100,000 LB)
1SEAR annual EPI reporing form, 2016 and WHO, World Health Staisics 2016
Division/Province/State/Region 11
District 210
Populaion density (per sq. km) 202
Populaion living in urban areas 61%
Populaion using improved
drinking-water sources 98%
Populaion using improved sanitaion 82%
Total expenditure on health as % of GDP
-Births atended by skilled health
personnel 100%
Neonates protected at birth against NT 93%
Table 2:
Immunization schedule, 2016
Vaccine Age of administraion
BCG Birth
HepB Birth
DTP-Hib-HepB 6 weeks, 10 weeks and 14 weeks
OPV 6 weeks, 10 weeks and 14 weeks
IPV 14 weeks
Measles 9 months and 15 months
Td 3 months and 4 months of pregnancy
Vitamin A 6 to 59 months Source: WHO/UNICEF JRF, 2016
Table 3:
Immunization system highlights
cMYP for immunizaion 2016-2020
NTAGI fully funcional
Spending on vaccines inanced by the government no data
Spending on rouine immunizaion programme inanced by the government no data
Updated micro-plans that include aciviies to improve immunizaion coverage 210 districts (100%) Naional policy for health care waste management including waste from immunizaion aciviies in place
Naional system to monitor AEFI in place
Planned for the next 24 months CES and MICS
>80% coverage for DTP-Hib-HepB3 210 districts (100%)
>90% coverage for MCV1 210 districts (100%)
>10% drop-out rate for DTP-Hib-HepB1 to DTP-Hib-HepB3 no district Source: WHO/UNICEF JRF, 2016
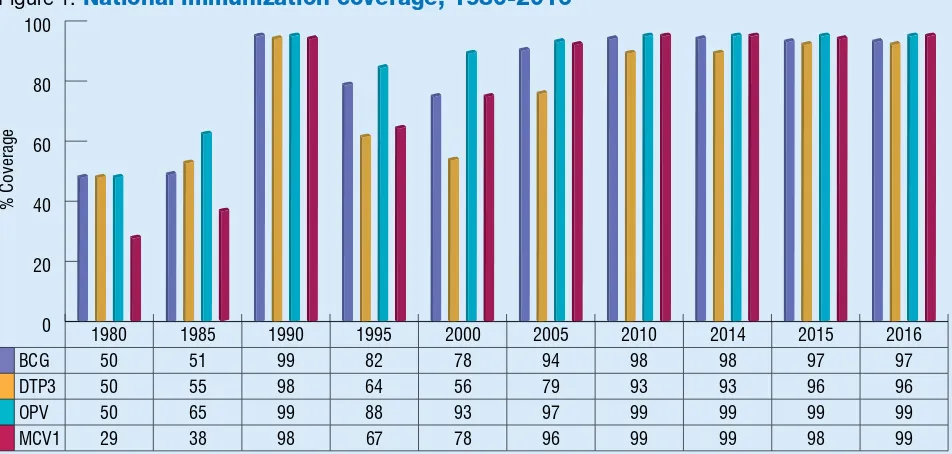
Figure 1:
National immunization coverage, 1980-2016
% Coverage
Source: WHO/UNICEF esimates of naional immunizaion coverage, July 2017 revision
1980 1985 1990 1995 2000 2005 2010 2014 2015 2016
BCG 50 51 99 82 78 94 98 98 97 97
DTP3 50 55 98 64 56 79 93 93 96 96
OPV 50 65 99 88 93 97 99 99 99 99
MCV1 29 38 98 67 78 96 99 99 98 99
Figure 3:
2015
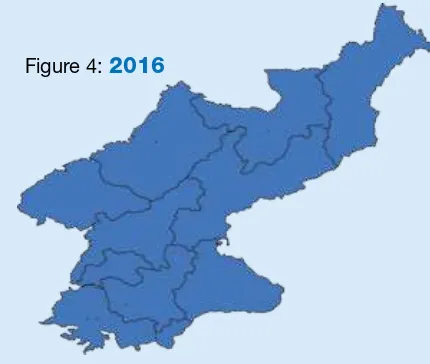
Figure 4:
2016
Source: SEAR annual EPI reporing form, 2016 (administraive data) Source: SEAR annual EPI reporing form, 2015 (administraive data)
Figure 2:
DTP3 coverage
1, diphtheria and pertussis cases
2, 1980-2016
Year
Diphtheria Cases Pertussis Cases DTP3 Coverage
%
1WHO/UNICEF esimates of naional immunizaion coverage, July 2017 revision 2WHO vaccine-preventable diseases: monitoring system 2016
Table 4:
Reported cases of vaccine preventable diseases, 2011-2016
Year Polio Diphtheria Pertussis NT
(% of all tetanus) Measles Rubella Mumps JE CRS
2011 0 0 13 0 0 5 66 0 0
Source: WHO/UNICEF JRF, (muliple years) ND=No data
DTP-Hib-HepB3 coverage by province
1980 1985 1990 1995 2000 2005 2010 2011 2012 2013 2014 2015 2016
1WHO/UNICEF JRF, Country oicial esimates, 1980-2016
2WHO vaccine-preventable diseases: monitoring system 2016 & JRF 2016
Maternal and
neonatal tetanus elimination is sustained
MNT eliminaion before 2000
© Ministry of Public Health/Democraic People’s
Republic of Korea
Table 5:
AFP surveillance performance indicators, 2011-2016
Indicator 2011 2012 2013 2014 2015 2016
AFP cases 129 125 116 115 99 105
Wild poliovirus conirmed cases 0 0 0 0 0 0
Compaible cases 0 0 0 0 0 0
Non-polio AFP rate1 2.25 2.18 2.02 2.00 1.72 1.83
Adequate stool specimen collecion percentage2 100% 100% 100% 100% 100% 98%
Total stool samples collected 270 269 226 184 156 194
% NPEV isolaion 10 11 9 11 5 14
% Timeliness of primary result reported3 99 94 92 99 95 93
1Number of discarded AFP cases per 100,000 children under 15 years of age.
2Percent with 2 specimens, at least 24 hours apart and within 14 days of paralysis onset. 3Results reported within 14 days of sample received at laboratory.
Figure 6:
2015
Figure 7:
2016
Polio-free
status is maintained
The last laboratory conirmed polio case due to WPV was reported in 1996.
Non-polio AFP rate by province
<1 1 – 1.99
>2 No non-polio AFP case
© Ministry of Public Health/Democraic People’s
Table 6:
OPV and JE SIAs
Year Vaccine Geographic
coverage Target age
Target populaion Coverage (%)
Round 1 Round 2 Round 1 Round 2
2000 OPV NID <5 years 2,147,929 99.6 99.7
2001 OPV NID <5 years 2,120,380 98.8 99.5
2002 OPV NID <5 years 2,098,940 99.7 99.8
2009 JE SNID
12 to 23 months and
4 to 5 years 1,500,000 - 99.5
-Source: WHO/UNICEF JRF, (muliple years)
Figure 9:
2016
Figure 8:
2015
Adequate stool specimen collection % by province
<60% 60% - 79%
>80% No AFP
Towards
measles elimination and rubella/CRS control
Figure 10:
MCV1 and MCV2 coverage
1, measles and rubella cases
2, 1980-2016
Measles Cases Rubella MCV1 Coverage MCV2 Coverage
2016
No. of cases
Year
1WHO/UNICEF esimates of naional immunizaion coverage, July 2017 revision 2WHO vaccine-preventable diseases: monitoring system 2016
Table 7:
MCV SIAs
Year Anigen Geographic
coverage Target group Target
Coverage %
2007 M Naionwide 6 months to
45 years 16,123,376 99.9
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19
Percent of population
Age (in years)
Protected by maternal antibodies Protected by routine vaccination with 1st dose
Protected by routine vaccination with 2nd dose Protected by SIAs
Immune due to past infection Susceptible
0%
Figure 15:
Immunity against measles - immunity profile by age in 2016*
* Modeled using MSP tool ver 2 assuming the schedule and MCV coverage remain unchanged and no SIAs in 2015 & 2016.
<80% 80% - 89% 90% - 94% >95%
Figure 12:
2016
Figure 14:
2016
Source: SEAR annual EPI reporing form, 2016 (administraive data)
Figure 11:
2015
Source: SEAR annual EPI reporing form, 2015 (administraive data)
Figure 13:
2015
Source: SEAR annual EPI reporing form, 2016 (administraive data)
Source: SEAR annual EPI reporing form, 2015 (administraive data)
Figure 16:
Sub-national risk assessment - measles and rubella
MCV1 coverage by province
MCV2 coverage by province
Source: developed using WHO risk assessment tool based on JRF & ARF data base
Figure 17:
Sporadic and outbreak associated measles cases* by month, 2011-2016
Outbreak associated measles
0
*Includes laboratory conirmed and epidemiologically linked cases Source: SEAR Monthly VPD reports
Figure 18:
Immunization status of confirmed (laboratory and EPI linked) measles
outbreak associated cases, by age, 2011-2016
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
> 15 years
10-14 years
5-9 years
1-4 years
< 1 year
2011 2012 2013 2014 2015 2016
Immunized Not immunized/ unknown 0
© Ministry of Public Health/Democraic People’s
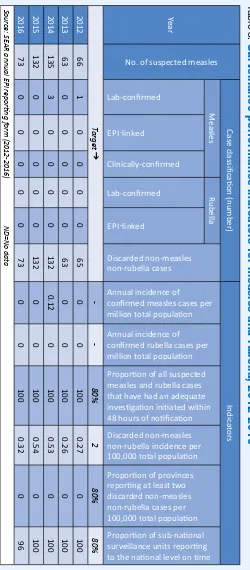
Table 8:
Surveillance performance indicators for measles and rubella, 2012-2016
Year
No. of suspected measles
Case classiic
Discarded non-measles non-rubella cases
Annual incidence of
conirmed measles cases per million total populaion
Annual incidence of
conirmed rubella cases per million total populaion Proporion of all suspected
measles and rubella cases that have had an adequate
invesigaion iniiated within 48 hours of noiicaion
Discarded non-measles non-rubella incidence per
100,000 total populaion Proporion of provinces reporing at least two
discarded non-measles non-rubella cases per
100,000 total populaion Proporion of sub-naional surveillance units reporing to the naional level on ime Lab-conirmed
AR annual EPI reporing f
orm (2012-2016)
ND=No dat
a
Year
Serum specimen collected from suspected measles cases
Serum specimen received in laboratory
within 5 days of collecion
Specimen
posiive for
measles IgM
Specimen
posiive for
rubella IgM
% Results within
4 days of receipt
% Posiive cases
tested for viral
detecion
AR annual EPI reporing f
orm (2012-2016)
ND=No dat
a
Table 9:
Performance of laboratory surveillance, 2012-2016
Figure 19:
WHO supported laboratory network for VPD surveillance
Central hygiene epidemic insituion - Naional polio laboratory
For contact or feedback:
Expanded Programme on Immunizaion Ministry of Public Health, Pyongyang, DPR Korea Phone: +850-2-3814077, Fax: +850-2-44104416 Email: bogon.moph@star-co.net.kp
Immunizaion and Vaccine Development (IVD)
WHO-SEARO, IP Estate, MG Marg, New Delhi 110002, India Tel: +91 11 23370804, Fax: +91 11 23370251
Email: SearEpidata@who.int