*Corresponding author. Tel.:#58-260-52722; fax:#58-260-52707.
E-mail address:[email protected] (J. Charris)
Biochemical Systematics and Ecology 28 (2000) 795}797
(
!
)-Amuronine from the leaves of
Croton
ya
v
ens
L. (Euphorbiaceae)
Jaime Charris
!
,
*
, Jose
H
Dom
m
H
nguez
!
, Catalino de la Rosa
"
,
Carlos Caro
"
!Laboratorio de Sn&ntesis Orga&nica, Facultad de Farmacia, Universidad Central de Venezuela, PO Box 47206, Los Chaguaramos, 1041-A, Caracas, Venezuela
"Facultad de Qun&mica y Farmacia, Universidad del Atla&ntico, Barranquilla, Colombia Received 26 February 1999; received in revised form 10 September 1999; accepted 1 November 1999
Keywords: Crotonyavens; Euphorbiaceae; Proaporphinic alkaloids; (!)-Amuronine
1. Subject and source
CrotonyavensL. (Euphorbiaceae) is a small tree, found throughout the Caribbean
coast of Colombia. The leaves were collected in Santo ToHmas de Villanueva, Colom-bia, in January 1997. The plant was identi"ed by Dr. Edunio CarbonoH de la Hoz. A voucher specimen, No. SC1093, was deposited in the Herbarium of the Pharmacog-nosy Department, Universidad TecnoloHgica del Magdalena, Santa Marta, Colombia.
2. Previous work
Previous studies have reported that species of Croton produce alkaloids of the aporphine, proaporphine and morphinandienone groups (Farnsworth et al., 1969; Southon and Buckingham, 1989) and N-methyltyramine derivatives (Stuart and By"eld, 1971).
The presence of an alkaloid fraction in the leaves ofCrotonyavenswas revealed by general screening methods (Caro, 1998). Proaporphirine alkaloids have been reported
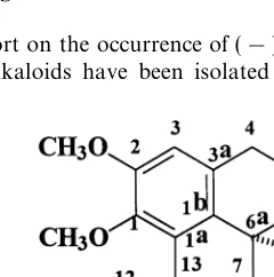
Fig. 1. (!)-Amuronine.
in South American species ofCroton:C. discolorWilld.,C. linearisJacq.,C. Plumieri Urb.,C. sparsiyorusMorong., andC. ruizianusMuell.-Arg (Del Castillo et al., 1996).
3. Present study
The air-dried and powdered leaves (1000 g) were extracted for 72 h with EtOH at room temperature. The crude extract (70 g) was taken up in 5% HCl and"ltered. The
"ltrate was basi"ed with NH
4OH and extracted with CHCl3. Evaporation of the
solvent yielded a crude alkaloidal extract (2 g). A preliminary separation of the alkaloids was carried out using a silica-gel (40}140 mesh, J.T. Baker) column (40]2.5 cm), using CH2Cl2, gradually enriched with acetone as eluent. Fractions
displaying similar alkaloidal composition on silica-gel tlc glass plates were combined and subjected to a second chromatographic separation on a silica-gel (40}140 mesh, J.T. Baker) column (25]1.5 cm) using CH
2Cl2-MeOH (1 : 1 v/v). Final puri"cation
of (!)-amuronine (11.8 mg) was achieved by preparative tlc on silica-gel glass plates.
The identi"cation of (!)-amuronine was supported by the IR (1667, 1619 cm~1) and
UV (243 nm) spectra indicating the presence of a, b-unsaturated carbonyl moiety which is found in some proaporphines. Chemical shifts assignments were determined by the analysis of1H NMR,13C NMR, DEPT 1353, HETCOR and1H-1H COSY 453
data, and NOE studies. Reciprocal NOEs could be detected between H-8 (d6.90) and H-6a (d3.29), H-8 to H-6a (5%) and H-6a to H-8 (7%), pointing to a syn relationship between these hydrogens. The above results were con"rmed by comparison with the chemical shifts reported for known proaporphines (GoKzler et al., 1987; GoKzler, 1990; Guinaudeau et al., 1987; Mukhtar et al., 1997) (Fig. 1).
4. Chemotaxonomic signi5cance
This is the"rst report on the occurrence of (!)-amuronine inCrotonyavens. To date proaporphinic alkaloids have been isolated from the following families and
genera: Annonaceae (gen.Isolona), Berberidaceae (gen.Berberis), Euphorbiaceae (gen.
Croton), Lauraceae (gen.Phoebe,Litsea,Neolitsea, andOcotea), Menispermaceae (gen.
Abuta, Anamarita, Caryomene,Cocculus, Diploclisia, Legnephora, Limacia,
Menisper-mum, Pachygone, Sarcopetalum, Sciadotenia and Stephania), Nelumbonaceae (gen.
Nelumbo), and Papaveraceae (gen.Meconopsis,Papaver, andRoemeria) (Southon and
Buckingham, 1989; GoKzler et al., 1990; Mukhtar et al., 1997). Alkaloids have been proposed as chemotaxonomic markers in the infraspeci"c classi"cation of the genus
Croton(Stuart and Woo-Ming, 1969). This study con"rms the presence of
proapor-phine alkaloids in this species.
Acknowledgements
We thank the IIF, CDCH-UCV, CYTED for their"nancial support and CONICIT grant No LAB-97000665 and the technical assistance of Mr. Rafael Paredes.
References
Caro, C., 1998. Thesis,`Estudio"toqumHmico parcial de la hoja deCrotonyavensafacultad de QumHmica y Farmacia, Universidad del AtlaHntico, Barranquilla, Colombia.
Del Castillo, C.H., De Simone, F., De Feo, V., 1996. Biochem. Syst. Ecol. 24, 463.
Farnsworth, N.R., Blomster, N.R., Messmer, W.M., King, J.C., Persions, G.J., Wilkes, J.D., 1969. Lloydia 32, 1.
GoKzler, B., 1990. Heterocycles 31, 149.
GoKzler, B., Freyer, A., Shamma, M., 1990. J. Nat. Prod. 53, 675. GoKzler, B., GoKzler, T., Mete, E., Freyer, A., 1987. Tetrahedron 43, 1765.
Guinaudeau, H., Freyer, A., Shamma, M., 1987. Tetrahedron 43, 1759, and references listed therein. Mukhtar, M., Martin, M., Domansky, M., Hadi, A., Awang, K., 1997. Phytochemistry 45, 1543. Southon, I.W., Buckingham, J., 1989. Dictionary of Alkaloids. Chapman & Hall, London. Stuart, K.L., By"eld, D., 1971. Phytochemistry 10, 460.
Stuart, K.L., Woo-Ming, R.B., 1969. Phytochemistry 8, 777.