International Conference on Education, Technology, and Science
NETS 2013
Improving The Quality of Education to Face The Impact of Technology
PROCEEDINGS
Universitas Muhammadiyah Purwokerto
!
"
#
$
%
&
' $%&(
)*+,-#
$%& )*+,
#
.
"
/
!
!%0
&
%
1
#
%
#
##
2
#
!
0
.
"
/
#
#
$%& )*+,- %
!
3
$
!
# #
!
$%& )*+,
2
4 !
"
#
$
%
&
'!"$%&(
# 5
!
!
1
%
6
7
&
8
5 3
%
9 &
'"
#
(
#
$
$
&
&
1
8
"
%
9
#
&
%
' &%(
7
9
$:
!
$%& )*+,
#
#
#
1 );* *** *** < 9 #
#
1
# %
#
!
:
#
#
!
:
%
!
#
#
#
8
8
7
$%& )*+,
-.
)= 5
)*+,
1
#
#
#
#
.
7
+>*;
)+; ##
,**
%
#
#
6
# "
/
##
#
#
##
# # "
0
$
#
0
#
1
#
#
!
#
#
#
#
!?
+@+@
7
,++
!
"
#
!
#
#
!
&
A
0
/
&
%
#
B
:
#
%
#
#
#
#
#
%
#
#
# $
$
&
3
"
$
1
8
.
# #
:
#
#
'8! !(
%
:
#
#
&
%
/
!
%
3
!
0
%
!
6
7
0
!
#
# #
:
#
8
B
B
:
#
B
3
#
:
$%&
3
#
C
.
)= 5
)*+,
%
1
4 5 3 &
!
3
'1
#
(
&
"
4
# 5 ! !
1
% '!%&(
# 5 5
&
'
(
# 5 0
'
&(
5 &
!
&3
3 '1
(
5 "
'5
8
(
# 5 5D
&
'A
A
.
E!(
5
' .
/
%
(
# 5 3
5
&
' .
/
%
(
# 5 5
'! 0(
# 5
'
5! (
# 5
'
$&(
# 5 &
&
' !
. 8!& 6 (
5 ! 5
'!%0(
# 5 57
.
&
' 6 (
# 5 1
'8! !(
# 5 A
'& ##
5
6
5A!(
# 5 !
&
& '
& $5(
Prof Dr
#
Jandra
Janan
' % (
5
D
# ' %
(
5 $
&
'
& (
"
4 ! 1
0
%
" <
4 .
&
4
# &%
%
&
3
5
/
&%
%
&
&
A
& &
3
%
&$
&
$
4 &
%
& ' (
& )
%
)
# !
1
'!%& !
(
&
8
'&
(
5 3
%
'%
/
(
' (
& )
$
&
5 '
# &
(
Prof Dr
#
/
/
' %
(
.
'0
7
"
#
0
%
(
5 ! 5
'!%0 !
(
# 5 57
.
&
' 6
!
(
5 "
' 6
!
(
# 5 1
'8! !(
# 5 &
'
!
(
#
1
#
"
$%& )*+,
B
"
A
!
&
$5 " %!
%3$ ! "% F 8%! $5! %$"3 8 6D % $ 68!&3 %$ "3! 6
/ 3 / . 5
+<;
%3$1$ !& $8$ 3 % ! D 1 4"3 88$ 6$& ! %$ "3! 6 8%! 8$ 8$E$8& F 1$ 1$&$ % %! F 1 "3$ !&%1D 1$&$1E!"$ %$ "3$1&
! &
@<+)
5$E$8 ! 6 "3$ !&%1D 6 G! $ & 8$ 1 ! 6 1$& 1"$ F D$ 1<+* &% 5$ %& F &$ ! 1 3!63 &"3 8
$ D &
+,<+=
! 1 E! 6 8$ 1 ! 6 %" $& %31 63 %3$ 8!" %! F
" 88 0 1 %!E$ 8$ 1 ! 6 5$8 5!1$"% 5 " $1 %!E$ %D $ &% 5 "1 $" !" %3$ 1D " 1&$&
8 2 3 &
+H<)I
$F8 %$ "3$1& 5 5!6!% 8 %$ "3! 6 $5! +
& ) # + # )6 7 &
# D 7
);<)H
5$E$8 ! 6 " 1$3$ &! 0 &$5 ! %$1 "%!E$ 1$ 5! 6 5$8 #
,*<,>
5$E$8 $ % F $J $1%!&$ " $%$ "D 0 &$5 3D&!"& 1 "%!" 8 . 1A %3$ F!$85 F & 1D 5 " "1$%$ " &%1 "%! $ 6! $$1! 6 ! E " %! 8 3!63 &"3 8
1 5 & # & $ 5 !
# $
,=<I)
! "1$ &! 6 %3$ 8$ 1 ! 6 %" $& 5 %3$ "1!%!" 8 %3! A! 6 %31 63 %3$ 8!" %! F %3$ 1 08$ & 8E! 6 1 "3
1 B D A &
I,<I>
%3$ 8$ 1 ! 6 F & $ A! 6 5 .1!%! 6 &! 6 !"% ' ! %$1 $% ! %3$ "D0$1 "8 &&1 ( F 1 %3$ &% 5$ %& F &$ ! 1 3!63 &"3 8
& 6 # $ $ 5
I=<;+
F!E$ . D& % 1 %$ "1$ %!E!%D ! %3$ "8 &&1
1 6 # $ $ 5
%3$ E 8 $ $5 " %! 8$ 1 ! 6 ! F 1 ! 6 "3!851$ 0$3 E! 1 6$ ;<@ D$ 1& 85
1 G B
@*<@,
! !%! %! 6 " 8% 1 8<0 &$5 "3 1 "%$1 8$ 1 ! 6 ! 1! 1D &"3 8 & &
@I<>@
! F 1 %! 5 " !" %! %$"3 8 6D ! %$ "3! 6 5
8$ 1 ! 6 ! 3!63$1 $5 " %! 4 ! " "8 &!E$ F! 5! 6&
A 7 1 5 # D " 5
>><=,
%$ "3! 6 5 8$ 1 ! 6 $ 68!&3 %31 63 !F 1 1$& 1"$ 8 " % 1 ' 18( &%1 %$6D
A # & ) A
=I<=>
8 6!" 8 %3! A! 6 ! %3$ %!"& 1 08$ & 8E! 6
! # 6
==<H)
& $$"3 %3$ 0$6! ! 6 F "3!851$ 6$& ; D$ 1&
D &%!A 0
H,<H>
! 1 E! 6 / ! 1 3!63 &"3 8 &% 5$ %& &"!$ "$ 8!%$1 "D 0D &! 6 8% ! $5! ! %$61 %$5<&"!$ "$ 8$ 1 ! 6 ! %3$ $ F %1 &3 5 !%& % "A8! 6 $FF 1%
5 0 A % 6 " 5 %
% $ F # "
H=<+*)
$ 6! $$1! 6
$FF$"% F %! $ 5 %$ $1 % 1$ 8%1 & 5 &&!&%$5 $J%1 "%! F %! J!5 % F1 Coleus tuberosus
& D & 3 " $ 5
F # $ 5
+*,<+*@
. &%$ . %$1 %1$ % $ % ! % F ! 5 &%1D 0D &! 6 !"1 86 $ Nannochloropsis Sp.
" . 5 # " $ 5
+*><++*
! 1 E! 6 %3$ K 8!%D F 01!K $%%$ F1 %!8!G %! F & 8!5 . &%$ F F 1F 1 8 &D %3$&!& 1 "$&& & $FF 1%& ! 1 E!5! 6
8%$1 %!E$ $ $16D 1$& 1"$&
% , 5 # " F # &
& # &
+++<++;
F GGD 8 6!" & %3$ " %1 88$1 ! $8$"%1 !" 8 5 " %1 88$1 &D&%$
! # $ # ! F # $ 5 #
$ $
:
5 0 7
F1 "%! 8 5!&%!88 %! F "8 E$ !8 % E " 1$&& 1$
. 3 3 8 3 L( 0
" L( 3 &
/ % A F % 5 L(/ A F
& 5
+)@<+)H
6 E$1 1 5$8 &! 6 F GGD < !5 " %1 88$1 F 1 8 5 F1$K $ "D " %1 8 6$ $1 % 1
1 +( ! 1 )( / % $ < F%! ! %
& &
+,*<+,,
5$&!6 5$8 F &% %" &! 6 !5 " %1 88$1
3+( 3 )( F 5 ,(1 &I(! 1 ;( ! % &
&
+,I<+,;
$J"!% %! " %1 8 &D&%$ 5$&!6 % ! 1 E$ E 8% 6$ &% 0!8!%D! &! 68$ "3! $ ! F! !%$ 0 & &D&%$ '& !0( $%3 5 &! 6 !5
3 7 F 1 & F 5 !
1 $ $ ! # % &
+,@<+,>
F1$K $ "D &% 0!8!%D ! 1 E$ $ % &! 68$ "3! $ &D&%$ &! 6 F!1&% 15$1 .$1 &D&%$ &% 0!8!G$1
! +( )( ! ,( 3 I(! 1 ;(
'+ ) , ;(
! % & & 'I( 0 %
A
+,=<+I+
5$&!6 5 5$E$8 $ % F %3$ 5"<% <5" " E$1%$1 F 1 3!63 .$1 8$5
1 #M & 1 & $
$ 5 0 7
+I)<+I@
&$%%8! 6 %! $ " %1 8 F &%$ % 10! $ .$1 8 % &! 6 8! $ 1 K 51 %!" 1$6 8 % 1 '8K1(
1 & '+( ! ')( 3 ',( 3 'I( ! 1 ';(
'+ ) I ;(
5 # $ $ ! % & '!%&(
',(
5 # $ $ 5 ? /
+I><+;*
! 8$ $ % %! F $8$"%1 !" 8 5 " %1 88$1 % ! 1 E$ %3$ E 8% 6$ K 8!%D F !"1 3D51 .$1 8 % % 0$ 5 & 1! E!88 6$ <
/ < 0 % F $ &% / E 1 E! "$
1 3 % $ $ 5
0 7
+;+<+;@
3D01!5 .$1 8 % F$ &!0!8!%D &% 5D ! 5 6! !&8 5 &! 6
3 $1 & F%. 1$
& + # B & 2) . , + ) , 5 # $ $
F # $ 0 7
:
8!" %! F " 8 6 &!F!" %! & & 1"$ F F $8 ! %3$ " 10 %! 1 "$&& F 8 " 8 A A & 01!K $%$
. 7 + & )( 5 3 ,(
+ )(
/ % F % & ,(/
% F % &
+@,<+@@
6 E$1 1 " %1 8 5$8 5$&!6 &! 6 !5 " %1 88$1 ! &%$
.$1 8 %
7 F +( 1 & )( 3 ,( ! I( !
1 ;(
+ I ;(
5 # $ $ ! % & &
)(
A & ,( 5 /
+@><+>*
5$&!6 5 5$E$8 $ % F E " 5!&%!88$1F 1 0! $%3 8
1 5 "%!
B B 6 5 $ $ 5
0 7
+>+<+>,
5$8! 6 5 &! 8 %! F &%$$10D .!1$ " %1 8 &D&%$ &! 6 F GGD 8 6!"
F + ! 1 ) & ,
+
$ $ 5 . )$ $ 5 !%&
& , $ 5 !%& &
+>I<+>>
$!6$ E 8 $ &% 0!8!%D 8D&!& 0 &$5 E 8 $ 6$ $1 % 1
$J"!% %! &D&%$ 5$8 & !0
3 '+( ')( 1 ',( ! 1 'I(
'+ ) , I
/ % $ ! % & '!%&(
+>=<+>H
8D&!& F 0!5!1$"%! 8 0 "A<0 &% " &" 5$ " E$1%$1 $1F 1 "$ .!%3 &% %!" 8 5 &! 6 &!
% # 2 1 5 " 1 ! 1 5 #
$ $ F # ! %
!%&
+=*<+=+
" & !5$ %!F!" %! 5 5$&!6 F . %$1 % 10! $ " %1 8 .!%3 8K1 $%3 5
1 &'+( 7 F')(1 & ',(3 'I(! 1 ';(
'+ ) ;(
! # % & & '!%&( ',(A
& 'I(5 ? /
+=)<+=@
5$&!6 F .$1 &D&%$ &% 0!8!G$1 0 &$5 F GGD " %1 88$1 .!%3 &% %$ F$$50$"A F 1 &! 68$ "3! $ ! F! !%$ 0 & &D&%$
% 7 ! 1 5 # $ $ F # !
% & ! # %
+=><+H+
&% 0!8!%D 8D&!& F .$1 &D&%$ 5$8! 6 ) 1$ ! %3$ F 1 F &% %$ & "$
3 1 " 3 1 ! 1 / % $ N
F%! ! % & &
: 86 1!%3
3 +( ! )( 3 ,( ! I( ! 1 ;(
+( )( I( ;(
5 # $ $ F # ! %
!%& ,( 0
5$&!6 5 5$E$8 $ % F 0 !85! 6 $ $16D 5!% & F%. 1$
1 3 3 & . 7 5 # $ $
5 0 7
+H><)*)
5$E$8 $ % F 8!01 1D " 5$& F 1 1 61 ! 6%3$ 3 !5
1 0 %0 &$5 " <;+* 5 8$
F D 5 # $ $
5
)*,<)*>
8D&!& F ! !3D51 .$1 5 3 % E 8% !" ! /$"%! ! % %3$ 61!5 &D&%$
3 & F B $ 3 & 5 # $ $
F # $ 0 7
)*=<)+)
$FF$"% F $%3 8<H@O ! 6 & 8! $ .!%3 !J% 1$ 1 %! F +4H 5 )4= %3$ " 0 &%! 5 $ !&&! F +); F 1<&%1 A$ $ 6! $
5 F A $ & $ D $ $
$ 5 0 7
)+,<)+=
&% 5D %3$ $1F 1 "$ 5 $J3 &% $ !&&! & F 5! "! $ 6! $ F $88$5 .!%3 5!$&$8P8 6 08$ 5&
B K 5 # $ F # $
/
)+H<));
0 !85! 6 5 5$&!6 ! 6 "3$ %$8$" !" %! &D&%$ F 1 %$ 6 A$1/ ! 5 $&! '%A!(
. % $ F % 0 7
))@<),+
5" % 1 & $$5 " %1 8 &! 6 5D !" 8! $ 1 K 51 %!" 1$6 8 % 1 '8K1(
5 7 + ) ! 1 ,
+(
5 # $ $ F # ! % !%& )(5 #
$ $ F $ # 5 ? / ,(5
# $ $ F # ! % !%& &
),)<),I
%! !G %! F !5 " %1 88$1 5 5 8 ! % .$1 &D&%$
&% 0!8!G$1 ! $8$"%1!" 8 &D&%$ &! 6 F!1$F8D 86 1!%3
F ! 1 5 # $ $ F # !
% !%& &
),;<)I)
$1F 1 "$ 8D&!& F %! $ 5!E!&! 8%! 8$J! 6 '%5 (F 1 8 6 &!6 8 %31 63 %!" 8 F!0$1 5 " 5 "% 1 %1 & !&&!
& 3 & & F B $ 5 #
$ $ F # $ 0 7
:
16 !" & 8 1 "$88 0 &$5 $J%1 "%! F D 'Carica papaya( 5 / %1 3 'Ricinus communis( 8$ E$& ! 5&&" '5D$ &$ &!%!G$5 & 8 1 "$88(
& 3 $ / & % 5 # $
$ 0 7
)I=<);+
$FF$"%& F %! $ 5 %$ $1 % 1$ F 8D $%3$1 $%3$1
A$% $& 8F %! ' $$A( %3$ & 8 0!8!%D F $$A !
5! $%3D8 "$% !5$
7 3 3 & 1 E A
F 3 5 # " $
&
);)<);I
&& < & !0 &! 8 %! &! 6 &! Q %8 0 & F%. 1$ +(
! )(3 ,( I(D ;(! 1
+ , I ;(
$ $ 5 !%& )( 0
);;<);>
1$ $. 08$ $ $16D 5!&%1!0 %$5 6$ $1 %! & & 1% & 8 %! F 1 %3$ $ $16D "1!&!& ! ! 5 $&!
1 & + ) ! 1 )
+
5 # $ $ F # $
D )5 # $ $ F # ! % !
% &
);=<)@)
5$&!6 F 5" E 8% 6$ 1$6 8 % 1 " %1 88$5 0D 0 &% " E$1%$1 "8 &$ 8 &D&%$ .!%3 &% %!" 8 5 &! 6 &!
/ 1 A % # 2 1 ! 1
5 # $ $ F # ! % !%&
)@,<)@I
! 8$ $ % %! F 1!%!F!"! 8 $ 1 8 $%. 1A 86 1!%3 5
1$ 1 "$&&! 6 .!%3 & 0$8 86 1!%3
1 A ! # $ " F # $
# /
)@;<)>,
1$5!"%! F %$&% !%$ & E 8!5!%D &! 6 1%!F!"! 8 $ 1 8 $%. 1A
3 # +( & 3 )( 1 . ,( 3 7 I(
+(
! # $ < # '" &
5 & <F ! <6 7 ( )(,(I(" & <F ! <6 7
)>I<)>H
$1F 1 "$ 8D&!& F "8 &&!F!" %! 86 1!%3 F 5 % ! ! 6 % &% 5$ %& &% % & ! 3!63$1 $5 " %!
! % B & 0 5 # ! # & &% !A
!A
)=*<)=;
F1$K $ "D 5 E 8% 6$ " 11$"%! ! &! 68$ "3! $ &D&%$ &! 6 8! $ 1 K 51 %!" 6 &&! F$$5 0 "A
F 5 '+(1 R &')( 7 F',(1 & 'I(! 1 ';(
'+ , ;(
% # ! # % '!%&( ',( " #
&
:
7 3 D . ! 1 5 # $
$ <F # ! % ! #% & '!%&(
8D&!& F D"1 3D51 .$1 8 % &D&%$ &! 6 " &
1 " 3 '+( 3')( 1 ',(3 'I( !
1 ';( $ $ 5 <F%! ! % &
!%&
)H)<)HI
8%" %1 &F 1 $1 &! 6 E1 .!%3 F GGD 8 6!" " %1 8
D . ! 1 / % $ !%& &
)H;<)H@
%! 8 F GGD 8 6!" " %1 88$1 5 1$&& 1!G$5 . %$1 1$ "% 1
' .1( $ 0 6A!% 8!&%1!A %$ 6 A8!1 $ 66 A " "A
&$ 1"3 86 1!%3
5 +( 5 )( ! 1 ,(
+ ,(
/ % $ F % ! ! % &
'!%&( )(/ % $ F % 5 N 0
)H><,*,
/$ $ 8!1 5 D 5 &!&%$ $ 66$1 A $8$A%1!A % 1
! 5 A&! 0$10 &!&A 8%!8$E$8 ! E$1%$1 /$ 0 % <3 0$1%! 6A % % A 8!A &! 0!8 % 3 1! '& 8 1 " 1(
5 " 1 ! 1 % $ <!%&&
,*I<,*H
3$ 8%3 5 $5!"! $
%1 5!%! 8 5 $. " 8$ $ % 1D %3$1 D F 1&! 6
. 0 7 " # 0 %
,+*<,+@
$FF$"%!E$ $&& F " 1$&& 1$ && 6$ % . 15 "3 6$ F 5 5 1! 6 %3$ $ &%1 8 $1! 5&
3 # 3 & <6
,+><,)*
5$&"1! %! F A .8$56$ 0 % 01$ &% F$$5! 6 " %1 "$ %! 5 1! 6 8 "% %! ! !5.!F$ "8! !" F 1& ! 1%! 1! 6 & E!88 6$
1! 6 & 5!&%1!"% &$ 1 6 1$6$ "D
1 $ $ .
,)+<,),
%3$ 5$E$8 $ % F ! %$61 %!E$ $ & 1$ $ % 5$8 F %!$ %
& F$%D "8! %$ F 3 & !% 8 ! 5A! / A 1% )*+, +
1 $ 8 F ! 5 $ + #
# 51 3 A ' 3 A ( /
,)I<,)>
%3$ 5$8 F 68 " &$ F8 "% %! .!%3 5! 0$%$& 0 &$5 ! 6 3 + 5 $ 8!% 5 F 68 " &$ $J" 1&! E 8 $
8 & # + 1 ! ) 0 & , %7 I
+ ,
% 2 F $ 5 ) I
F 0 7
,)=<,,+
:
1$ 8 F !8 1$ %!$ %& F 1 F 51 16 & $A 15/ 08!"
3 & !% 8 1. A$1% &
1$8 %! &3! 0$%.$$ 3D 0!1%3! 6 1$8 J %! %$"3 !K $& 5 "3!850!1%3 5 1 %! F &% 6$ + 0 & D!5 3 A$" A 8!. 6 A 0 A$ 5 8
# A 5
,,@<,,=
%$8$3$ 8%3 ! 8$ $ % %! &! 6 8! $ $$%! 6 8!" %! F 1
%3$ 1$ %$ 1$ 3$ 8%3 &$1E!"$& L
1 B LL
L
5 # $ $ 5 LL5 #
3 5
,,H<,I)
5$E$8 $ % F & 1E<%0 '% 0$1" 8 &!& & 1E$!88 "$ .$06!&(
& + & 0 )
+
A F ! A
& )F A !
,I,<,I@
&% 5D F &%<&%1 A$ 3D $168D"$ ! %3$1 D &%1 A$ ! %!$ %&
! A 3 5!D 3 D 6D A 1% 3 & !% 8
B +(& )(! ,(
+(
D )(F # 6 ,( A
D 3
,I><,;*
.$! "3! +I $1!5! & $J$1"!&$ 5 %!$ % FF!1 "D & %3$
$ %! 8 %3$1 D
& L . LL
L& 4 6 3 & & <! <! "
,;+<,;I
%3$ $FF!"!$ "D F 6 18!" 5 " " 0$1 " & %! ! 5$"1$ &! 6 %3$ 08 5 1$&& 1$ 6 %3$ $85$1& .!%3 3D $1%$ &!
# F # 3 &
,;;<,@*
!& %3$1$ 5!FF$1$ "$ 0$%.$$ F!& 8 ! &% 5 15!G$5 $J%1 "% F1 3D& 8!& 6 8 % 8 5 " %1 8 "1$ %!" F "%! F & 1 6 $ 5 .8$D 1 % ! 5 "$5 0D &%1$ % G % "! < !" %! !5$S
D & & A & 7 # 0
,@+<,@@
3$ $ 8 6D &% 5D F 3$8 8$&& $&& $85$18D $J $1!$ "$ .!%3 3D $1%$ &! ! 0 6 1 "!%D
. D & 3 D & $
,@><,>)
1 F!8$ F / 0 & %!&F "%! 5 !%& $FF$"% " 1! 6 0$3 E! 1 F 3 & !% 8 1&$&
/ & F # 3 & # #
:
& + ! . ) & ,
+ ,
F # 3 & # )
" 11$8 %! 0$%.$$ & "! 8 & 1% 5 &% 1% K 8!%D F 8!F$
F B 0 1 3 Science F , &
,=,<,==
" 11$8 %! 0$%.$$ A$6$8 $J$1"!&$ .!%3 $$58$ $1! $ ! / 1D
3$ 8! 6 % 1 8 &% 1% . ! 10 F! % 3 & 3
0 5 6
1 &%!A 0
,=H<,H)
%3$ " 11$8 %! 0$%.$$ &$8F<" F!5$ "$ 5 5$ 1$&&! 8$E$8 F
%$$ 6$ 1 3 6$& ! %! & 3 D D & & % % !
6$8 6
& & & 5 #3 &
F # #
,H,<,H;
5$%$1 ! %! 5 E 8!5 %! $%3 5 F % % 8 F8 E !5 " %$ %
5 % % 8 3$ 8!" " %$ % F $%3 8!" $J%1 "% F 6 &%$$ 'Garcinia mangostanaL.( 8$ E$& & % 1 8 1$&$1E %!E$& " 5!5 %$ 0D
&! 6 & $"%1 3 % $%1!" $%3 5
5 ! 3 % & F #
#
,H@<I*;
F 1 8 %! 5 %! J!5 % "%!E!%D %$&% F 8! &%!"A F1 6 &%$$ 1! 5 'Garcinia mangostana L.( $%3 8 $J%1 "%
6 L ! & 5 # FA!A /
&
I*@<I++
%3$ 1 8$ F $5! $J & 1$ 5 3$ 8%3 0$8!$F 01$ &% &$8F
$J ! %!
A + 5 )3 3 6 )
+ )
7 7
I+)<I+;
3$ $ 8 6D &% 5D F E 1! & 0&% "8$ $FF 1%& % $8! ! %$ F 5$ 6 $ 3 $ 1 6!" F$E$1 ! 0 D & 5!&%1!"%
! 3 5
I+@<I)*
1$8 %! &3! 0$%.$$ %$ % 8 " 1$ &$1E!"$& 5 $1! % 8 1% 8!%D 1!&A F "% 1& ! 08!" 3$ 8%3 "$ %$1 F 8$ 0 6 ! )*+)
1 $ # 5 5
I)+<I)I
$FF$"% F ! 5 0 5D " 8$ $ % 1D %3$1 !$&4 8!&%$ ! 6 % && 6$& F 8K 1 6 ! &% 8 .$1! 6 08 5 1$&& 1$ % & "! 8 ! &%!% %! F %$1 % ! 1&! 6 3 $& 8$ 0 6)*+,
% # & 5 5
:
$FF$"% F 53!A1 %3$1 D 8 0 1 ! A 8 + % %$1 !%D "8! !"
0 5! ! 5 3 ! 8$ 0 6 ! )*+)
F # & 5 5
I,*<I,@
$%3 8 $J%1 "% F 0! 3 6 8$ E$& $FF$"% 'Anredera cordifolia '% ( & ( 3$ % 8 6D" 8 "3 6$& ! F$ 8$ .!&% 1 1 %&
& A ## + F A + 7 A + 57 57 )
+
8 # <% : F # #
)
8 # " F # #
I,><I,H
%3$ " 1!& F F1$$ 1 5!" 8& &" E$ 6! 6 0!8!%!$& 0$%.$$ $%3 8 $J%1 "%& F 1. "$ 6 'Pimpinella pruatjan ( 3$10& 5 8$ E$&
0 2 3 & 0 . & 1 1 . F
# #
II*<II@
5!FF$1$ "$& ! F8 $ "$ 8D $%1!" $J$1"!&$ .!%3 $ 1 &" 8 1 $8$"%1!" 8 &%! 8 %! ' $&( % ! "1$ &$ %3$ 0!8!%D F E$1%!" 8
/ ! 0 &A$%0 88 8 D$1 &%!A$& 3 5!D 3 8$ 0 6
$ ! 6 0 & 5!!! % &%!A
II><I;I
%3$ " 1$8 %! 0$%.$$ A .8$56$ F $ ! .!%3 $ ! ! "!5$ %
! 1$6 %&
0 1 . 5
I;;<I@*
%3$ !5$ %!F!" %! 0 "%$1! Streptococcus Pneumoniaee ! %3$ !1 1
5 ! &$0$1 6 8 + 8$ 0 6
3 & G 3 % $ 3 # & 5
5
I@+<I@I
%3$ 5!FF$1$ "$ "" 1 "D 1 !& 8 .$!63% $ 01D 8 6D ! 1!& % $%3 5 5 &6 .!%3 .$!63% 0 0!$& .$1$ 0 1 ! 3$ 8%3 "8! !" %$6 81$/ )**H
1 & 5 5
I@;<I>,
%3$ ! F8 $ "$ F $ 18D 01$ &%F$$5! 6 ! !%! %! % %3$ 5 1 %! F
8 0 1 %3!15 $1! 5 ! %$1 !%D %3$1 % 8</ 1!D 3 %$1 !%D
3 & !% 8 8$ 0 6 ! )*+,
1 " 5!!! A &%!A
I>I<I>=
"D% % J!" "%!E!%D %$&% F $J%1 "% 5 F1 "%! F 6 8 6 8 'Alpinia ( 3 8 "$88& 0D ! E!%1
7 & 1 ! & % & &
I>H<I=>
% J!" $FF$"%& F F 1 85$3D5$ A!5 $D ' 8( &.!&& .$0&%$1 8$
1 3 % & %
& 5 !!! A 8 &%!A$& !
:
F 1 $1& 0$3 E! 1 % . 15 1!&A& F % % F 1 ! 6 F 1 61 8 5 %8 %!" E 1!$%!$& ! 0 / 1 $6 1 1$6$ "D
7 F #
IHI<IH>
%3$ $FF$"%!E$ $&& F "8 E$ !8 % " %1 8 & D0$ 1 &% 5!&$ &$
& $ D ! 8 % " 1 !
IH=<;**
E$6$% %!E$ 5 6$ $1 %!E$ $1F 1 "$& F %31$$ & D0$
E 1!$%!$& ! F!E$ 8$E$8& F " 8"3!"! $ 5 & 6$& 5 &$$5 " % $$8! 6 %1$ % $ %&
& 3 A ! 8 % " 1 !
! # 1 5
;*+<;*I
&! 8$ 1%!&! % 1D 6!& F 1 8 3 1 3 G 15 !5$ %!F!" %! ! "! / 1!E$1 & 0 6 5!&%1!"%
1 F ! & 6 F
&
;*;<;*=
%3$ 8D&!& F A 8! <I* 1 5! "8!5$ 88 %! % &$5! $ % &$ . %$1 5 &31! 'Penaeus Merguiensis) ! "!8 " " &% 8 "$
.
;*H<;+)
%3$ $FF$"% F 13!G 0! ! " 8 %! % % 8 F8 E !5 3$ 8!" " %$ %& 5 %! J!5 % "%!E!%D F & D0$ &$$5&
$ D ! 8 % " 1 !
;+,<;+>
&"1$$ ! 6 F 60$ % % 6$ %D $& 1$&!&% % % .5$1D !85$. 'Erysiphe polygoni( 5 8$ F & % 5!&$ &$ 'Cercospora canescens(
& +(D )( $ D +(
+(
! 8 % " 1 ! )(! !
;+=<;)+
F "%! F 61!" 8% 1$ %$"3 8 6D $ E!1 $ % ! " 1! 6 F 1 F 5 &$" 1!%D & 1% ! & 05!&%1!"% 0$8!A F $ 8 6 1$6$ "D
. & 0 F #
;))<;);
$FF$"% F 1!"$ 01 5 16 !" F$1%!8!G$1 5 & 6$& D!$85 F .3!%$ D&%$1 &31 (Pleurotus ostreatus)
5 3 7 7 7 F #
#
;)@<;,,
%!8!G %! F . &%$ !8 8 & % % 8 !J$5 1 %! &!8 6$ .!%3 8 " 8 1$& 1"$& 0 &$5 % "3!$E$5 F 5 &$" 1!%D ! ! 8
1 %$! & 1"$&
! & 5 # & #
;,I<;,>
" 8" 8 %! $ &! 8 &! 6 0$ $F!% 1 1 %$ $%3 5 ' " &
# & 8 $ " (
& . 1 B 1 #2 3
:
1 F!8$ F !"1 $ %$1 1!&$& "1$ %!E$ & E$ !1 1 "$&&$5 " " % . &%$ 6$5 0D 1 1%!& & F 1 $1& ! 1 1 8
5 + % & 7 1 ) & 0 ,
+ ,
8 # & F # #
)
8 # & F # $
#
;I,<;I>
5$E$8 $ % F F & 1! 5!&$ &$ " %1 8 %$"3 8 6D .!%3 0! 8 6!" 8 6$ % ! & " 8%!E 1 0 &$$58! 6& 1$& 8% IN VITRO " 8% 1$&
& # 6
;I=<;;;
& "! 8 $" !"
&$1E!"$& " "!%D F !&8 !" F! "! 8 &$1E!"$& " $1 %!E$ ! %3$
5$E$8 $ % F & 88 5 $5! $ %$1 1!&$& '& $ ( ! 1. 1$/
"$ %1 8 / E ! 5 $&! ' # & K $## 8
& # 0 # ! F &
" ! 5 7 (
F D 5 ! 0 5 & 7 <
7
;;@<;@)
%3$ 1 8$ F & 88 5 $5! $ %$1 1!&$& '& $ ( %
" !%D 0 &! $&& 5$E$8 $ % ! ! 5 $&! ' & #
$ A (
1 5 5 + 3 6 )
+
& # 5 # 0 5 8 # 0
# & ' !( ) & F <
. 0
;@,<;@@
" 11$8 %! 0$%.$$ ! %$ &!%D F 8 D! 6 8! $ 6 $ 5
$ %! 8 ! %$88!6$ "$
# A & D 5 #
# &
;@><;>+
6 ! 5 8 && F " 1 %!E$ 5E % 6$ 6 &$ <@
D ! # ! $ F 5 #
$ < D
;>)<;=+
6 $& 8! $ ! F8 $ "$ % %3$ " F$ 8 6 6$ 5$E$8 $ % !
"3!851$ % A$8 1 3 "$ A %!3 "! % % %! 1 % 6$1 6
&$8 %
A # /
;=)<;=;
8D&!& F $FF$"% F 1!"$ 8 " %! 5 &$1E!"$& % ! "1$ &$ & 8$&
E 8 $
3 5 7 5 # F $ # 5
/
: :
$J!&%$ "$ F 1 1 8 " !%D ! %3$ 5!&&$ ! %! F K !" %$"3 8 6D % 61 % 1!& E!88 6$
! & + ) % # 0 ,
+ )
& & F # & & / &
,
8 # F & F # &
$ / &
;HI<;HH
%3$ 8D&!& F 1!&A 5 1$% 1 %3$ 1 F!%<8 && &3 1! 6 1 5 "%& ! %3$ ! 5 $&! !&8 !" 0 A! 6 ! &%!% %!
D + % ) 3 , & ! I
+ I
8 5 # " # $ '&%!$( #
)
8 # ! F Q $ 6 F #
$ ! ! D ,8 5 #
F # $ / & ' & $5(
Proceedings of International Conference
On Education, Technology and Science (NETS) 2013
Purwokerto, December 28
th, 2013
Universitas Muhammadiyah Purwokerto HM 440 ISBN 978,602,14930,0,7
THE COMPARISON OF FREE RADICALS SCAVENGING ABILITIES BETWEEN
ETHANOL EXTRACTS OF PURWOCENG (
#
impinella pruatjan Molk.) HERBS AND
LEAVES
Balqis Hisyam Saleh Basleman, Wiranti Sri Rahayu, Retno Wahyuningrum
Pharmacy Faculty of Muhammadiyah University of Purwokerto
Abstract
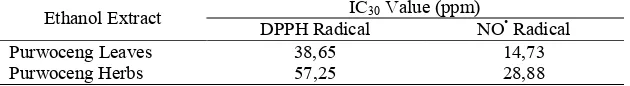
Flavonoids and tannins are potentially as free radicals scavenger. Purwoceng has some of the compounds such as flavonoid, glycosides, and tannins. Research done by using DPPH and Nitric oxide (NO•) radicals. The treatment of ethanol extracts of purwoceng leaves with the scavenging activity of radical DPPH 38,65 ppm and NO• 14,73 ppm results IC30 smaller when compared with the ethanol extract of purwoceng herbs by the scavenging activity of radical DPPH 57,25 ppm and NO• 28,88 ppm. The free radicals scavenging abilities of ethanol extracts of purwoceng leaves better than free radicals scavenging abilities of ethanol extracts of purwoceng herbs.
Keywords: free radicals, Purwoceng, DPPH, nitric
oxide
INTRODUCTION
Free radicals is an atom or group of atoms that has a highly reactive characteristic caused by the presence of one or more free electrons or electrons are not paired, so that it becomes unstable and high, energy (Fessenden and Fessenden, 1982). To avoid the free radicals attack, is by consuming natural antioxidants in sufficient amount each day. Anti, free radical is a compound which in a small amount when compared with the substratum is capable of delaying or preventing or avoiding the oxidation of substrates which are easily oxidized (Halliwell and Gutteridge, 2000).
Flavonoids are natural phenolic compounds are potentially as free radicals scavenger and have bioactivities as a medication. Generally, leaf, root, stem and flower are the part of plant that save many flavonoids as secondary metabolites (Waji et al, 2009). Another natural phenolic compounds that potential as a free radicals scavenger is tannin. Tannins can be found in all parts of the plant, but at some plants, tannin concentrated on specific parts (Hernawan and Setyawan, 2003). One of the plants that contain flavonoids and tannins as secondary metabolites is purwoceng (Haryono, 2013).
Purwoceng (Pimpinella pruatjan Molk.) is a medicinal plant. Commercially, purwoceng’s root
has been widely reported have medicinal properties as aphrodisiac, diuretic, and tonic. This plant is native plant from Indonesia which live in endemic areas, such as mountainous Dieng plateau in Central Java, mount Pangrango, West Java and East Java’s mountainous areas.
According to Caropeboka and Lubis (1975), root extract of purwoceng motor activity, which heightens tonus striated muscles, stimulates central nervous arrangement with capture point work on the medulla oblongata, sexual behavior and to improve male mice (Caropeboka et al., 1979 in S. Usmiati and S. Yuliana, 2010). So it is said that purwoceng’s root extract has an androgenic activity in mice.
Saponin group is the one of chemical compound as secondary metabolites in purwoceng plant. There are also triterpenoid,steroids, sitosterol, and stigmasterol compounds. Purwoceng plant also contains coumarin, alkaloids, flavonoids, glycosides, and tannins. By looking at the content of secondary metabolites of purwoceng plant which are flavonoids and tannins (Haryono, 2013), is needful to do more researchs to find out the potential of the power of free radicals scavenging of purwoceng plant either the leaves or herbs because for specific flavonoid compounds are concentrated in the leaves (the herb Information, 2012; Waji et erlenmeyer flask), micro pipette, Ultrasonic bath, Rotary Evaporator, analytic scales, maceration,
cupboard dryer, exicator, UV,Vis
Spectrophotometer. Materials
1. Material Test
On Education, Technology and Science 2013
Purwokerto, December 28
th, 2013
ISBN 978,602,14930,0,7 HM 441 Universitas Muhammadiyah Purwokerto
2. Chemicals
Ethyl acetate technical degrees, petroleum ether technical degrees, ethanol 96% technical degrees, ethanol p.a., DPPH, sodium nitroprusside, sulfanilic acid p.a., glacial acetic acid p.a., N,(1, Naphthyl),ethylenediamine dihydrochloride, Griess reagent p.a, BHT (butilatedhydroxytoluene), quercetin.
a. Preparation of Test Materials 1) Preparation of simplicia
Cultivation purwoceng plants is
obtained from Dieng’s farmer, Wonosobo, Central Java. Purwoceng plants that have been obtained are cleaned from impurities attached to later washed until clean. The
2) Extraction of simplicia
The dry simplicia powder was added to the container maceration. Done,with use 3 concentrate solution of maceration, namely petroleum ether (PE), ethyl acetate (EA) and ethanol 96%. Each maceration with a different concentrate performed as many as 3x24 hours in order to obtain optimal results maceration. During submersion, stirring for 1 hour was done so that the compounds contained in the simpilisia of purwoceng leaves and herbs can dissolve properly. b. Determining Free Radicals Scavenging
Quantitatively with DPPH Radicals 1) Preparation of DPPH solution 0,4 mM
DPPH solution of 0.4 mM is made by dissolving 15.8 mg powder DPPH in methanol p.a. on pumpkin measures 100.0 mL, then vortex.
2) Determination of Maximum Wavelength of DPPH Solution
Determination of the maximum
wavelength of DPPH 0,4 mM are used in the power of free radicals scavenging test is done as follows: 1000 eL DPPH solution 0,4 mm added to the volume with methanol 5 mL then observed the absorbance in the range of wavelength 400,800 nm using methanol p.a. as blank.
3) Preparation of Ethanol Extract of Purwoceng
Ethanol extracts of purwoceng leaves and ethanol extracts of purwoceng herbs
dissolved in methanol p.a. with a
concentration of 0.01% w/v.
4) Test Power Free Radicals Scavenging As many as 1000 eL DPPH 0.4 mM put in pumpkin measures, added by the test solution with different volume series (500, 1000, 1500, 2000 and 2500 eL) is then added methanol p.a. up to 5.0 mL volume, vortex for 1 minute until the mixture is
homogenous. After 30 minutes, the
absorbance is measure at a maximum wavelength. Measure the negative control absorbance also without the addition of the test solution.
The magnitude of resource free radicals scavenging is calculated using the formula: Resource free radicals scavenging
% = Abs.Control$Abs.Sample
Abs.Control x100%
The acquired absorbance data was created a linear regression equation which expressing the relationship between the concentration of the test substance (x) with the average antioxidant activity (y) of a series of measurements of replication so retrieved IC50 value, is the test solution that necessary scavenge 50% of DPPH radical over the past 30 minutes (operating time), or pause time required by the test solution to reduce the radical DPPH perfectly. After 30 minutes the absorbance obtained will be constant.
c. Determining Free Radicals Scavenging Quantitatively with Nitric Oxide Radicals (NO•)
1) Making a solution of Sodium
Nitroprusside 10 mM
Dissolve 299,12 mg of the powder of sodium nitroprusside in methanol p.a. on pumpkin measures 100,0/250 mL, then divorteks.
2) Griess Reagent Creation
Proceedings of International Conference
On Education, Technology and Science (NETS) 2013
Purwokerto, December 28
th, 2013
Universitas Muhammadiyah Purwokerto HM 442 ISBN 978,602,14930,0,7 3) Determination of Maximum Wavelength
Sodium Nitroprusid 10 mM
Determination of the maximum
wavelength of Sodium Nitroprusside 10 mM used in the power of free radicals scavenging test is done as follows: 2000 eL sodium nitroprusside solution 10 mM added with 500 mL of 7.4 pH phosphate buffer and proceed with incubation at a temperature of 25°C for 150 minutes, then added with 500 eL Griess reagent and measured the absorbance in the range of wavelength 400, 800 nm using methanol p.a. as blank. 4) Determination of Operating Time of
Coupling Compound
Determination of operating time of coupling compound was done by measuring the mixture of 2000 eL sodium nitroprusside 10 mM and 500 eL phosphate buffer (pH 7.4) and the Griess reagent. Then measure the operating time mixture solution at a maximum wavelength by using methanol p.a. as blank.
5) Preparation of Ethanol Extract of Purwoceng
Ethanol extracts of purwoceng leaves and ethanol extracts of purwoceng herbs
dissolved in methanol p.a. with a
concentration of 0.01% w/v.
6) Test Power Free Radicals Scavenging
2.0 mL solution of sodium
nitroprusside 10 mM added with 500 eL phosphate buffer (pH 7.4) and added a test solution with different volume series (500, 1000, 1500, 2000 and 2500 eL) then added methanol p.a. up to 5.0 mL volume, followed by incubation at a temperature of 25°C for 150 minutes. Added by 500 eL Griess reagent and incubated at room temperatures during the operating time. Read absorbance at a maximum wavelength. d. Preparation of Positive Control Solution
1) BHT Stock Solution 100 ppm
The stock solution is made by weighing carefully 0,01 grams of BHT and dissolved in 100 mL of methanol p.a.
2) Quercetin Stock solution 100 ppm The stock solution is made by weighing carefully 0,01 grams of quercetin and dissolved in 100 mL of methanol p.a. 3) Test Power Free Radicals Scavenging
The power free radicals scavenging test for a positive control is carried out with the power free radical scavenging test of ethanol extract of herbs ethanol extract leaves as blank, for quercetin the volume series are 2, 4, 8, 16 and 32 eL of a stock solution of 100 ppm and to measure the absorbance was used same concentration of quercetin on test resource free radicals scavenging as blank. RESULT AND DISCUSSION
1. Determination of Purwoceng Plant
The results of determination for purwoceng plant which is done according to the book the Flora of Java (Backer and Bakhuizen, 1965) is retrieved the key determination stating the plant used is Pimpinella pruatjan Molkenb. with synonym Pimpinella alpina Molk.
2. Extraction of Purwoceng Plant
a. The results of the extraction of Herbs and the leaves of Purwoceng
On Education, Technology and Science 2013
Purwokerto, December 28
th, 2013
ISBN 978,602,14930,0,7 HM 443 Universitas Muhammadiyah Purwokerto
b. Extract Characteristics
Table 1. Purwoceng Viscous Extract Characteristics
Viscous Extract Extract Characteristics Result
Purwoceng Herbs 1. Organoleptic
a. Color b. Smell c. Flavor d. Shape 2. Rendemen
Dark green soupy Typical
,
Viscous solution 7,33%
Purwoceng Leaves 1. Organoleptic
a. Color b. Smell c. Flavor d. Shape 2. Rendemen
Dark green soupy Typical
,
Viscous solution 23,52%
3. Determination of Resource Free Radicals Scavenging Quantitatively
a. Results of The Determination of Maximum Wavelength (λ max)
Results of maximum wavelength
determination with the DPPH radical is 516 nm with maximum absorbance for an ethanol extract of purwoceng leaves is 0,756, ethanol extract of purwoceng herbs is 0,752, quercetin is 1,005 and BHT is 0,756. This means that for power of free radicals scavenging test performed on maximum λ 516 nm.
Capture 1. Scanning (λ max) DPPH Solution 0,4mM
Results of The Determination of Maximum Wavelength (λ max)
Results of maximum wavelength
determination with the NO• radical is 478 nm with maximum absorbance for an ethanol extract of purwoceng leaves is 0,553, ethanol extract of purwoceng herbs is 0,553, quercetin is 0,446 and BHT is 0,443. This means that for power of free radicals scavenging test performed on maximum λ 478 nm.
Capture 2. Scanning (λ max) Sodium Nitroprusside Solution 10mM
b. Power Test Results of FreeRradicals Scavenging
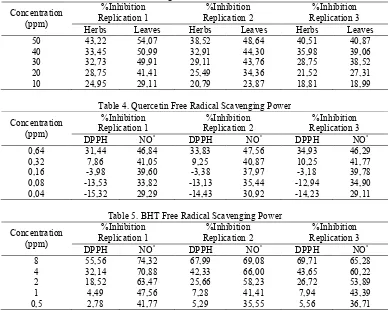
Power test results capture free radicals can be seen from the percent inhibition of free radical DPPH and NO• ethanol extracts of leaves and ethanol extracts of herbs of purwoceng, quercetin and BHT which shown in tables II, III, IV and V. Where the higher concentration of the extract is used so the percent inhibition is also higher which means the better the free radicals scavenging power.
Capture 3. Electron Donation or Hydrogen Radical from Antioxidant to DPPH Radicals (Mun’im, A
Proceedings of International Conference
On Education, Technology and Science (NETS) 2013
Purwokerto, December 28
th, 2013
Universitas Muhammadiyah Purwokerto HM 444 ISBN 978,602,14930,0,7 Table 2. Scavenging Power of Ethanol Extract of Leaves and Ethanol Extract
of Purwoceng Herbs with DPPH Radicals Concentration
Capture 4. The Reaction Between Nitric Ion with Sulphalinamide which Paired with naphtylethylendiamine (Wikipedia en, 2013)
Table 3. Scavenging Power of Ethanol Extract of Leaves and Ethanol Extract of Purwoceng Herbs with NO• Radicals
Concentration Table 4. Quercetin Free Radical Scavenging Power
Concentration Table 5. BHT Free Radical Scavenging Power
Concentration Percent inhibition was analyzed to find out to
know IC30 value, is using curve relationship between percent barrier with a concentration (Gulluce et al, 2006). The magnitude of free radical scavenging power characterized by the IC30 value, is the solution concentration of samples needed to
inhibit 30% of free radicals. The higher of I% value accordingly the IC30 is lower. The lower of IC30 make the better of free radical power inhibition. IC30 is used because if used IC50 the result can have
extrapolation outside the range series of
On Education, Technology and Science 2013
Purwokerto, December 28
th, 2013
ISBN 978,602,14930,0,7 HM 445 Universitas Muhammadiyah Purwokerto
Table 6. IC30 Value of Ethanol Extract of Purwoceng Leaves, Ethanol Extract of Purwoceng Herbs and Positive Controls
Quercetin BHT
1 extract of purwoceng herbs.
4. Comparison of Free Radicals Scavenging Power
The use of different methods for determining the free radicals scavenging power of plant extracts can shown the random results, it is difficult to compare and sometimes give rise to incompatibility (Koleva et al. 2002 in Irawati 2008).
Based on table 7. the IC30 value of ethanol extracts of purwoceng leaves and ethanol extract of purwoceng herbs by DPPH radical greater than by NO• racidal. This is possible because both radical have differences in how forms a stable compound. Another possibility is a compound that plays a role in the free radicals scavenging from ethanol extract of prwoceng leaves and ethanol extracts of purwoceng herbs namely flavonoids less reactive against DPPH radical because of steric hindrance, so that only a small molecule that has a greater opportunity to react with DPPH radical (Irawati, 2008).
Table 7. Comparison of IC30 Value Between DPPH Radical and NO• Radical
Ethanol Extract IC30 Value (ppm) CONCLUSIONS AND SUGGESTIONS
It can be concluded that there is a difference of free radicals scavenging power between ethanol extract of purwoceng leaves and purwoceng herbs by looking at the value of IC30.
It is advisable to commit further research about the free radicals scavenging power for antother purwoceng leaves and herbs extract, both petroleum ether extract or ethyl acetate extract. Types of flavonoids compounds in the leaves and herbs of purwoceng who have power of free radicals scavenging. As well as the use of other methods to test the activity of antioxidants that can support free radical scavenging research of purwoceng plant so it can be known the right dose in order to declare that purwoceng plant is a natural antioxidants.
Reference
Anonim, 1979, Farmakope Indonesia, Edisi III, Jakarta, Departemen Kesehatan Republik Indonesia.
Backer, A.C & van der Brick, B.C.R. 1965. Flora of Java (Spermatophytes only), vol II. Netherlands: N.V.P Noordhoff. Backer, A.C & van der Brick, B.C.R. 1965. Flora of Java (Spermatophytes only), vol II. Netherlands: N.V.P Noordhoff.
Caropeboka, A.M. dan I. Lubis, 1975, Pemeriksaan
pendahuluan kandungan kimia akar
Pimpinella alpine (purwoceng), Dalam Simposium Tanaman Obat I, 8, 9 Desember, Bagian Farmakologi. FKH, Institut Pertanian Bogor.
Caropeboka, A.M, Iskandar dan Paridjo, P, 1979, Pengaruh Ekstrak Akar Pimpinella alpine Koord. Terhadap Reproduksi Hewan, Dalam Usmiati, S dan Yuliani, S, 2010, Efek Androgenik dan Anabolik Ekstrak Akar Pimpinella alpine Molk. (Purwoceng) pada Anak Ayam Jantan, Balai Besar Penelitian dan Pengembangan Pascapanen Pertanian, Bogor. Fessenden, Ralp J. dan Fessenden, Joan S., 1982,
Proceedings of International Conference
On Education, Technology and Science (NETS) 2013
Purwokerto, December 28
th, 2013
Universitas Muhammadiyah Purwokerto HM 446 ISBN 978,602,14930,0,7 Gulluce M, Aslan A, Sokmen M, Sahin F, Adiguzel
A, Agar A & Sokmen A, 2006, Screening the antioxidant and antimicrombial properties of the lichens Parmelia saxatilis, Platismatis glauca, Ramalina pollinaria, Ramalina polymorpha and Umbilicaria nylamderiana, J. Phytomedicine 13 (2006) p.515, 521.
Halliwell, B. & J.M.C. Gutteridge, 2000, Free Radical in Biology and Medicine. Ed 4th, New York, Oxford University Press.
Haryono, Nugroho Tri, 2013, Purwoceng,
http://www.herbaltama.com/?T
anaman_Obat:Purwoceng [12 Mei 2013]. Hernawan, Udhi Eko Dan Setyawan, Ahmad Dwi,
2003, Ellagitanin; Biosintesis, Isolasi, dan Aktivitas Biologi [Review], Surakarta, Jurusan Biologi FMIPA.
UNS Informasi Herbal, 2012, Purwoceng,
http://www.informasiherbal.co m/purwoceng/, [18 Desember 2012].
Irawati, Irma, 2008, Perbandingan Metode
Penentuan Aktivitas Antioksidan Rimpang Temulawak, [skripsi], Bogor, Institut Pertaninan Bogor.
Mun’im, A., 2008, Aktivitas Antioksidan Cendawan Suku Pleurota dan Polyporaceae dari hutan UI, Jurnal Ilmiah Farmasi Vol.5No.1, Jakarta.
Waji, Resi Agestia dan Sugrani, Andi, 2009, Flavonoid (Quercetin) [Makalah Kimia Organik Bahan Alam], Makassar, Program S2 Kimia, Fakultas Matematika dan Ilmu Pengetahuan Alam, Universitas Hasanuddin.
Wikipedia, 2013, Griess Test.