The knowledge learned through direct contact
with the tool
Individual freedom as the basic learning
Anticipating a natural phenom enon
Ex ercising using words and phrases for object
Develop the intellectual and m oral character
of students
Managing lab, lab facility, chem icals, lab
adm inistration
Projecting various purposes of lab
Storage & m aintenance equipm ent &
chem icals
Make adjustm ents to the curriculum
Perform basic t echniques
Planning and organizing t he use of lab for
1 year
Organize dist ribut ion of t ools & m at erials
lab
Prepare regulat ions & safet y lab for
Prepare clear inst ruct ions on how t o use
special equipm ent
Det erm ine t he m obilit y of t raffic flow in t he
pract icum st udent wit h t he benchm ark num ber of st udent s and lab condit ions
Com plet ing P3K box and skilled do first aid
in lab
The room lab
lab facility
Tools and m aterials
Workforce
Format for lab administ rat ion
List of proposed it ems card t ool
List of t ools
Format for lab administ rat ion
• card subst ance
• List of subst ances
• List revenue /
expendit ure subst ances
• List of proposed subst ance
• dat a w orkforce
Room of weight 15 m2
Room of Analysts 20 m2
Room of assistant 20 m2
Room of warehouse 20 m2
Room of computer 20 m2/10 people
Room of practicum 4 m2/ people
Room of workshop 2,5 m2/ people
Sample Format A
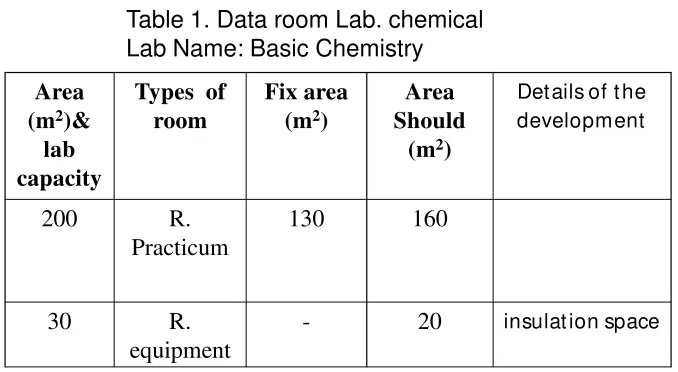
Table 1. Data room Lab. chemical Lab Name: Basic Chemistry
Area
Nam e of equipm ent: m easuring glass
Group : G
Spesification : 100 m L,od.2 cm
Code No. : 5 pyrex
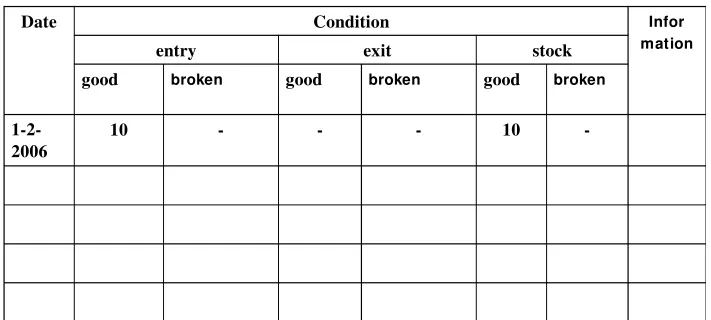
Table 2. Card Tool
Date Condition Infor
mation
entry exit stock
good broken good broken good broken
1-2-2006
- Nam e of chem ical : Silver nitrat (AgNO3)
Group : A
Spesification : 100 gram
Code No. : 5 E Merck
Mr Cu= 63,5; S= 32; O= 16; H= 1
Mr CuSO4.5H2O= (1x 63,5) + (1x 32) +
(4x 16) + 5[(2x 1) + (1x 16)]= 249,5
Mass of 1 m ol CuSO4.5H2O= 249,5 g So t o m ake a 1 M solut ion of copper
sulphat e is needed as m uch as 1 lit er of
In concentrat ed HCl is usually unknown levels
of 36%, density 1.18. Thus the m ass of 1,000 m L of concentrat ed HCl is1.180 g.
In 1180 g of concentrat ed HCl, the am ount of HCl = 36/ 100 x 1180 g = 424.8 g.
In 1 liter (1,000 m L) 1 M HCl solution contained 1 m ol HCl.
The m ass of 1 m ol HCl = 36.5 g.
So to m ake a 1 M HCl solution required 36.5 g HCl, contained in concentrat ed HCl = 36.5 /
Tape m easure Glue/ lem
Epox y/ super glue Plastic cem ent
Brush- teeth
Drinking spook
Term inal block (for joint the cable) Electric tape
Screw driver/ dual screw Ham m er (besi/ plastic)
Mole trips pliers/ Tang clam p key ring
Yank/ obeng getar/ pum p action screw driver Stanley plier/ pem otong kawat
Tanks of various sizes
Metal cutting saws (with silicon carbide cinpade)
Sandpaper
Fuse sizes
1. In t he Lab. available 16M HNO3 solut ion
(concent rat ed). How m any m L of
concent rat ed HNO3 required t o m ake as
m uch as 100m l of 2M aqueous HNO3?
2. In t he Lab. available solut ion of 70%
HNO3, bj = 1.42 g / m L. How m any m L of
concent rat ed HNO3 required t o m ake as
m uch as 100m l of 2M aqueous HNO3?
3. In the Lab. available solution of 96% H2SO4, bj
= 1.84 g / mL. How many mL of H2SO4
required to make dollars. 2M aqueous H2SO4
as 250 ml?
4. How many grams of NaOH are required to
make NaOH solution: a) 0.1 M b) 0.5 M c) 2M?