SUPPLEMENTARY MATERIAL: TABLES III Ð X
An Ab Initio Molecular Orbital Study of the Energies and Conformers of 3,4Dihydro1,2 dithiin, 3,6Dihydro1,2dithiin, 4H1,3Dithiin, and 2,3Dihydro1,4dithiin
Fillmore Freeman, *, Choonsun Lee, Henry N. Po, and Warren J. Hehre,
Department of Chemistry, University of California, Irvine, CA 926972025
Department of Chemistry, California State University, Long Beach, CA 90840
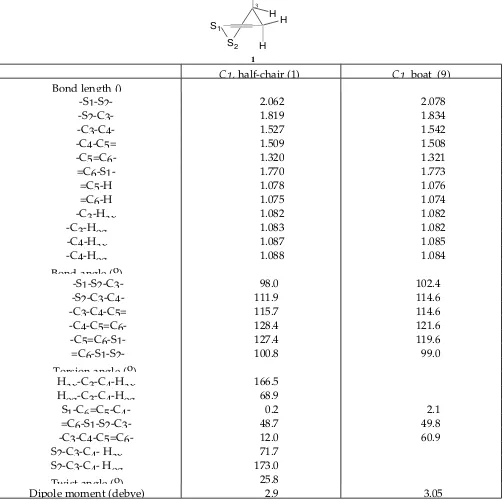
Table III. 631G* Optimized Geometries for the Conformers of 3,4Dihydro1,2dithiin (1,9)
S1 S2
H
H H
H 1
3
C1, halfchair (1) C1, boat (9) Bond length ()
S1S2 2.062 2.078
S2C3 1.819 1.834
C3C4 1.527 1.542
C4C5= 1.509 1.508
C5=C6 1.320 1.321
=C6S1 1.770 1.773
=C5H 1.078 1.076
=C6H 1.075 1.074
C3Hax 1.082 1.082
C3Heq 1.083 1.082
C4Hax 1.087 1.085
C4Heq 1.088 1.084
Bond angle (o)
S1S2C3 98.0 102.4
S2C3C4 111.9 114.6
C3C4C5= 115.7 114.6
C4C5=C6 128.4 121.6
C5=C6S1 127.4 119.6
=C6S1S2 100.8 99.0
Torsion angle (o)
HaxC3C4Hax 166.5
HeqC3C4Heq 68.9
S1C6=C5C4 0.2 2.1
=C6S1S2C3 48.7 49.8
C3C4C5=C6 12.0 60.9
S2C3C4 Hax 71.7
S2C3C4 Heq 173.0
Twist angle (o) 25.8
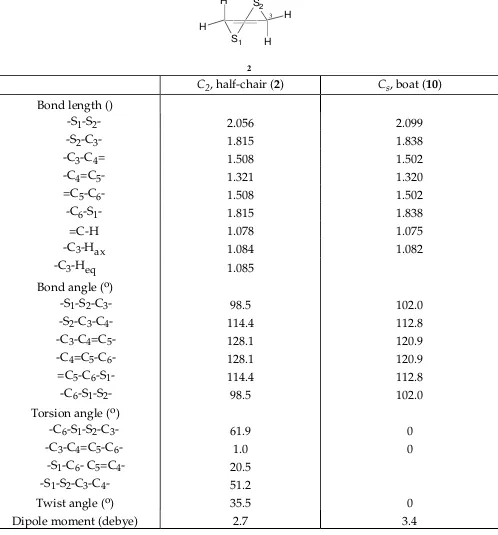
Table IV. 631G* Optimized Geometries for the conformers of 3,6Dihydro1,2dithiin (2, 10)
S1 S2
H
H H
H
2 3
C2, halfchair (2) Cs, boat (10) Bond length ()
S1S2 2.056 2.099
S2C3 1.815 1.838
C3C4= 1.508 1.502
C4=C5 1.321 1.320
=C5C6 1.508 1.502
C6S1 1.815 1.838
=CH 1.078 1.075
C3Hax 1.084 1.082
C3Heq 1.085
Bond angle (o)
S1S2C3 98.5 102.0
S2C3C4 114.4 112.8
C3C4=C5 128.1 120.9
C4=C5C6 128.1 120.9
=C5C6S1 114.4 112.8
C6S1S2 98.5 102.0
Torsion angle (o)
C6S1S2C3 61.9 0
C3C4=C5C6 1.0 0
S1C6 C5=C4 20.5 S1S2C3C4 51.2
Twist angle (o) 35.5 0
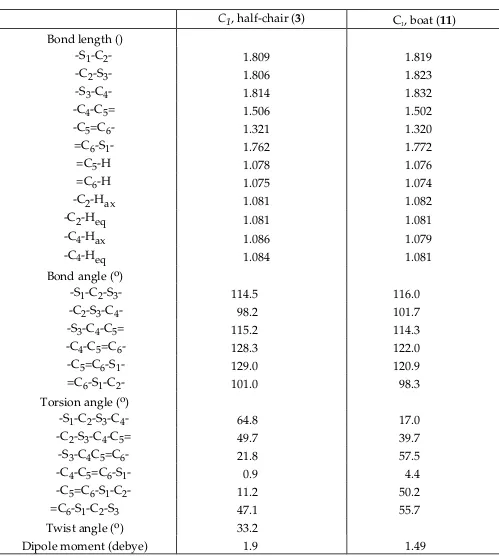
Table V. 631G* Optimized Geometries for the Conformers of 4H1,3dithiins (3,11)
S3 S1 H
H 2
H H
3
C1, halfchair (3) C1, boat (11)
Bond length ()
S1C2 1.809 1.819
C2S3 1.806 1.823
S3C4 1.814 1.832
C4C5= 1.506 1.502
C5=C6 1.321 1.320
=C6S1 1.762 1.772
=C5H 1.078 1.076
=C6H 1.075 1.074
C2Hax 1.081 1.082
C2Heq 1.081 1.081
C4Hax 1.086 1.079
C4Heq 1.084 1.081
Bond angle (o)
S1C2S3 114.5 116.0
C2S3C4 98.2 101.7
S3C4C5= 115.2 114.3
C4C5=C6 128.3 122.0
C5=C6S1 129.0 120.9
=C6S1C2 101.0 98.3
Torsion angle (o)
S1C2S3C4 64.8 17.0
C2S3C4C5= 49.7 39.7
S3C4C5=C6 21.8 57.5
C4C5=C6S1 0.9 4.4
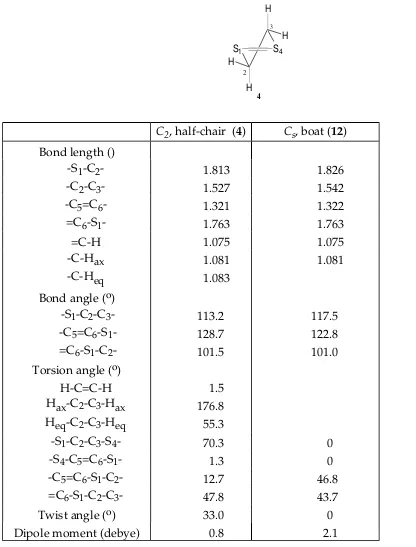
Table VI. 631G* Optimized Geometries for the Conformers of 2,3Dihydro1,4dithiin (4, 12)
S1 S4
H
H
H H
4
2
3
C2, halfchair (4) Cs, boat (12)
Bond length ()
S1C2 1.813 1.826
C2C3 1.527 1.542
C5=C6 1.321 1.322
=C6S1 1.763 1.763
=CH 1.075 1.075
CHax CHeq
1.081 1.083
1.081
Bond angle (o)
S1C2C3 113.2 117.5
C5=C6S1 128.7 122.8
=C6S1C2 101.5 101.0
Torsion angle (o) HC=CH HaxC2C3Hax HeqC2C3Heq
1.5 176.8 55.3
S1C2C3S4 70.3 0
S4C5=C6S1 1.3 0
C5=C6S1C2 12.7 46.8
=C6S1C2C3 47.8 43.7
Twist angle (o) 33.0 0
Table VII. Frequency Calculations (631G*) for 3,4dihydro1,2dithiins (1)
S1 S2
H
H H
H 1
3
half chair Planar boat
normal
modes vibrational frequency, cm1
normal
modes Vibrational Frequency, cm1
normal
modes vibrational frequency, .cm1
A 145.77 A" 259.78 A 115.59
A 263.40 A" 135.34 A 227.19
A 339.77 A' 349.37 A 352.48
A 424.28 A" 420.50 A 392.78
A 467.50 A' 454.67 A 525.80
A 565.19 A' 540.89 A 546.88
A 703.34 A' 710.14 A 660.93
A 730.70 A" 726.56 A 718.54
A 769.38 A' 756.16 A 797.20
A 811.04 A" 872.05 A 814.93
A 931.41 A' 891.08 A 941.50
A 985.19 A' 949.11 A 989.18
A 1084.88 A" 1078.76 A 1079.13
A 1110.29 A" 1107.72 A 1107.06
A 1129.01 A' 1173.23 A 1124.77
A 1291.21 A" 1345.20 A 1271.84
A 1323.21 A' 1352.34 A 1307.51
A 1373.18 A" 1412.42 A 1364.30
A 1454.63 A' 1448.57 A 1450.22
A 1504.46 A' 1510.32 A 1469.02
A 1533.57 A' 1533.18 A 1517.09
A 1610.00 A' 1623.93 A 1630.26
A 1624.44 A' 1656.19 A 1639.70
A 1853.49 A' 1882.00 A 1825.37
Table VIII. Frequency Calculations (631G*) for 3,6dihydro1,2dithiins (2)
half chair planar boat
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
B 156.16 A2 216.17 A" 31.34
A 223.08 B1 130.26 A' 228.09
B 335.75 B2 347.18 A" 318.12
A 391.67 A2 393.38 A' 401.28
A 486.77 A1 438.10 A" 494.04
A 567.57 A1 530.32 A' 535.60
B 704.46 B2 698.54 A" 667.59
B 712.35 B1 708.66 A' 677.45
A 762.41 A1 750.94 A" 792.19
B 902.53 B2 908.10 A' 849.95
A 963.62 A2 979.02 A' 972.74
A 994.81 A1 983.34 A' 1012.37
B 1025.98 B1 1026.87 A" 1043.66
B 1090.23 B2 1123.05 A" 1099.90
A 1122.11 A2 1136.01 A" 1118.14
A 1310.09 A2 1316.71 A' 1269.47
B 1317.73 B1 1328.22 A" 1329.09
A 1341.14 A1 1355.38 A' 1351.67
B 1442.52 B2 1455.11 A" 1405.30
A 1458.83 A1 1476.60 A' 1408.36
B 1545.99 B2 1555.09 A" 1534.35
A 1603.63 B2 1624.11 A" 1621.35
B 1604.16 A1 1624.41 A' 1636.81
A 1894.24 A1 1926.27 A' 1871.18
B 3230.02 A1 3232.65 A" 3249.18
A 3230.05 B2 3232.70 A' 3251.72
B 3273.88 B1 3275.40 A" 3303.10
A 3274.37 A2 3277.26 A' 3305.71
B 3327.12 B2 3323.11 A" 3360.99
Table IX. Frequency Calculations (631G*) for 4H1,3dithiins (3)
half chair planar boat
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
A 121.12 A" 210.74 A 103.00
A 231.44 A" 112.25 A 198.40
A 364.07 A' 381.56 A 359.87
A 378.54 A' 397.23 A 370.71
A 464.36 A" 426.33 A 494.66
A 666.03 A' 628.56 A 637.93
A 732.36 A" 739.28 A 704.39
A 749.22 A' 748.41 A 742.58
A 779.83 A' 767.00 A 801.06
A 845.42 A' 874.53 A 839.65
A 859.41 A' 908.79 A 885.37
A 932.43 A" 931.39 A 941.81
A 1030.17 A" 1007.30 A 1018.56
A 1055.73 A' 1073.27 A 1066.93
A 1099.03 A" 1102.59 A 1099.62
A 1270.80 A" 1274.97 A 1263.47
A 1331.00 A" 1329.81 A 1295.39
A 1341.90 A' 1339.34 A 1343.00
A 1384.24 A' 1405.65 A 1378.12
A 1459.41 A' 1479.13 A 1432.11
A 1514.18 A' 1518.52 A 1496.94
A 1594.75 A' 1631.34 A 1614.03
A 1615.78 A' 1638.95 A 1627.33
A 1855.31 A' 1885.72 A 1832.27
A 3220.16 A' 3230.10 A 3252.81
A 3264.69 A" 3269.21 A 3273.34
A 3272.82 A' 3277.97 A 3307.66
A 3329.07 A" 3333.92 A 3333.68
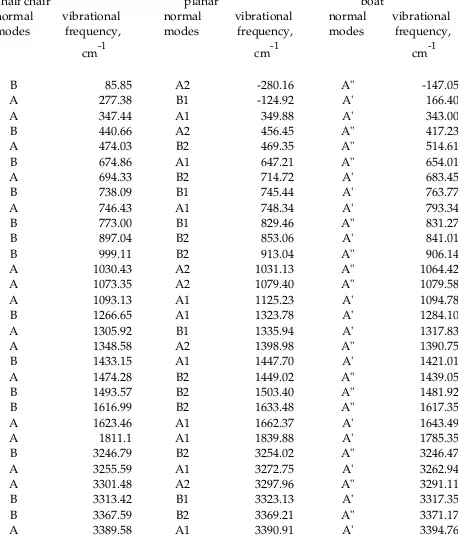
Table X. Frequency Calculations (631G*) for 2,3Dihydro1,4dithiins (4)
half chair planar boat
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
normal
modes vibrational frequency, cm1
B 85.85 A2 280.16 A" 147.05
A 277.38 B1 124.92 A' 166.40
A 347.44 A1 349.88 A' 343.00
B 440.66 A2 456.45 A" 417.23
A 474.03 B2 469.35 A" 514.61
B 674.86 A1 647.21 A" 654.01
A 694.33 B2 714.72 A' 683.45
B 738.09 B1 745.44 A' 763.77
A 746.43 A1 748.34 A' 793.34
B 773.00 B1 829.46 A" 831.27
B 897.04 B2 853.06 A' 841.01
B 999.11 B2 913.04 A" 906.14
A 1030.43 A2 1031.13 A" 1064.42
A 1073.35 A2 1079.40 A" 1079.58
A 1093.13 A1 1125.23 A' 1094.78
B 1266.65 A1 1323.78 A' 1284.10
A 1305.92 B1 1335.94 A' 1317.83
A 1348.58 A2 1398.98 A" 1390.75
B 1433.15 A1 1447.70 A' 1421.01
A 1474.28 B2 1449.02 A" 1439.05
B 1493.57 B2 1503.40 A" 1481.92
B 1616.99 B2 1633.48 A" 1617.35
A 1623.46 A1 1662.37 A' 1643.49
A 1811.1 A1 1839.88 A' 1785.35
B 3246.79 B2 3254.02 A" 3246.47
A 3255.59 A1 3272.75 A' 3262.94
A 3301.48 A2 3297.96 A" 3291.11
B 3313.42 B1 3323.13 A' 3317.35
B 3367.59 B2 3369.21 A" 3371.17