5
4
LAMPIRAN A
DATA BAHAN BAKU
A.1
KOMPOSISI ASAM LEMAK MINYAK DEDAK PADI
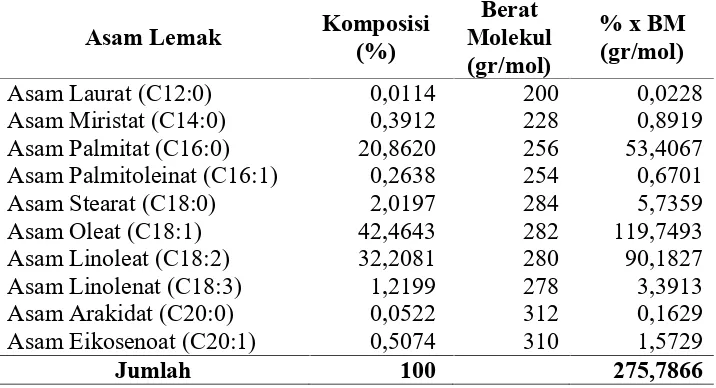
Tabel A.1 Komposisi Asam Lemak Bahan Baku Minyak Dedak Padi
Asam Lemak
Komposisi
(%)
Molekul
Berat
(gr/mol)
% x BM
(gr/mol)
Asam Laurat (C12:0)
0,0114
200
0,0228
Asam Miristat (C14:0)
0,3912
228
0,8919
Asam Palmitat (C16:0)
20,8620
256
53,4067
Asam Palmitoleinat (C16:1)
0,2638
254
0,6701
Asam Stearat (C18:0)
2,0197
284
5,7359
Asam Oleat (C18:1)
42,4643
282
119,7493
Asam Linoleat (C18:2)
32,2081
280
90,1827
Asam Linolenat (C18:3)
1,2199
278
3,3913
Asam Arakidat (C20:0)
0,0522
312
0,1629
Asam Eikosenoat (C20:1)
0,5074
310
1,5729
Jumlah
100
275,7866
Dari perhitungan maka diperoleh berat molekul rata-rata FFA minyak
dedak padi sebesar 275,7866 gr/mol.
A.2
KOMPOSISI TRIGLISERIDA MINYAK DEDAK PADI
Tabel A.2 Komposisi Trigliserida Bahan Baku Minyak Dedak Padi
Asam Lemak
Komposisi
(%)
Molekul
Berat
(gr/mol)
% x BM
(gr/mol)
Asam Laurat (C12:0)
0,0114
638
0,0727
Asam Miristat (C14:0)
0,3912
722
2,8245
Asam Palmitat (C16:0)
20,8620
806
168,1477
Asam Palmitoleinat (C16:1)
0,2638
800
2,1104
Asam Stearat (C18:0)
2,0197
890
17,9753
Asam Oleat (C18:1)
42,4643
884
375,3844
Asam Linoleat (C18:2)
32,2081
878
282,7871
Asam Linolenat (C18:3)
1,2199
872
10,6375
Asam Arakidat (C20:0)
0,0522
974
0,5084
Asam Eikosenoat (C20:1)
0,5074
968
4,9116
Jumlah
100
865,3598
Dari perhitungan maka diperoleh berat molekul rata-rata trigliserida
minyak dedak padi sebesar 865,3598 gr/mol.
5
5
A.3
KADAR
FREE FATTY ACID
(FFA) MINYAK DEDAK PADI
.1
2
CO
3
/ZEOLIT ALAM
!"# $ %& $#$#'( )*+ ,-' $.-/' 01 & , )
jerap
pada Zeolit Alam dari Variasi Konsentrasi K
2
CO
3
No. Konsentrasi Larutan K
(gram dalam 60 ml
aquadest
2
CO
3
)
Kadar Logam Kalium
dalam %
1
0
1,2758
B.2
DATA DENSITAS BIODIESEL
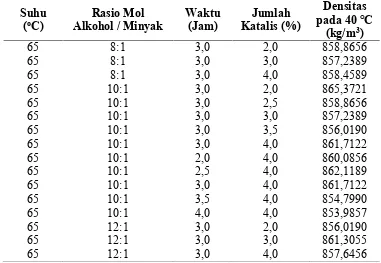
Tabel B.2 Hasil Analisis Densitas Biodiesel
Suhu
(
o
C)
Alkohol / Minyak
Rasio Mol
Waktu
(Jam)
Katalis (%)
Jumlah
Densitas
pada 40 °C
(kg/m
3
)
65
8:1
3,0
2,0
858,8656
65
8:1
3,0
3,0
857,2389
65
8:1
3,0
4,0
858,4589
65
10:1
3,0
2,0
865,3721
65
10:1
3,0
2,5
858,8656
65
10:1
3,0
3,0
857,2389
65
10:1
3,0
3,5
856,0190
65
10:1
3,0
4,0
861,7122
65
10:1
2,0
4,0
860,0856
65
10:1
2,5
4,0
862,1189
65
10:1
3,0
4,0
861,7122
65
10:1
3,5
4,0
854,7990
65
10:1
4,0
4,0
853,9857
65
12:1
3,0
2,0
856,0190
65
12:1
3,0
3,0
861,3055
65
12:1
3,0
4,0
857,6456
57
B.3
DATA VISKOSITAS KINEMATIK BIODIESEL
Tabel B.3 Hasil Analisis Viskositas Kinematik Biodiesel
Suhu
(
o
C)
Alkohol / Minyak
Rasio Mol
Waktu
(Jam)
Katalis (%)
Jumlah
t
(detik)
rata-rata
Viskositas
B.4
DATA
YIELD
METIL ESTER
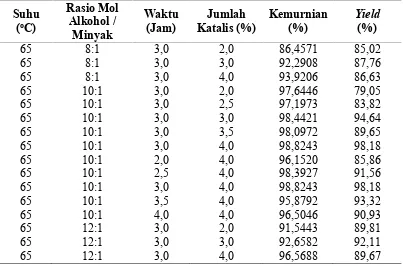
Tabel B.4 Hasil Analisis
Yield
Metil Ester
Suhu
(Jam)
Katalis (%)
Jumlah
Kemurnian
(%)
Yield
(%)
65
8:1
3,0
2,0
86,4571
85,02
65
8:1
3,0
3,0
92,2908
87,76
65
8:1
3,0
4,0
93,9206
86,63
65
10:1
3,0
2,0
97,6446
79,05
65
10:1
3,0
2,5
97,1973
83,82
65
10:1
3,0
3,0
98,4421
94,64
65
10:1
3,0
3,5
98,0972
89,65
65
10:1
3,0
4,0
98,8243
98,18
65
10:1
2,0
4,0
96,1520
85,86
65
10:1
2,5
4,0
98,3927
91,56
65
10:1
3,0
4,0
98,8243
98,18
65
10:1
3,5
4,0
95,8792
93,32
65
10:1
4,0
4,0
96,5046
90,93
65
12:1
3,0
2,0
91,5443
89,81
65
12:1
3,0
3,0
92,6582
92,11
65
12:1
3,0
4,0
96,5688
89,67
ö÷
ø ùúûüý ùþ ÿ
ù üø ù þùø ü ü úû üü ù ù þ ù
úüþ ù ÿ ÿù û ùÿü RICE BRAN OIL
ù ù øü øü ùø ù ú
ÿù þ üÿ ü ø
!
"RICE BRAN OIL#
$%&' %
r D
()* %+ , -A
. %-,s
,s
$C
/ 0&10+ , +,B
%2 %n B
%34 5,ny
%3D
67 %38 %7 ,(
Rice Bran Oil
9:;
<=> ?@A B C @D @CB A BA E@ <@ F CG H@ I E@CB J I KE L M@ < @ ENOGPQRSGCBT@C @I
UVWXV
r D
YZ[V \]X ^V_]A
\ Vt AA
`(
Atomic Absorption Spectroscopy
aUnt
bc d efgbcb ^Vn
[V hVr
io
gVW[(
[ V\]b WadVhV[Vt
V\]s
[j
C
klmne o\]
t A
\V Wpq
r st us
r D
vwx sy z{A
| s{zs
zs AA
}untu
~} st {{ zt A
{st s| s zz~sy z} ny
sw
s
C
r st us
r D
vqxsyz{A
| s{ zs
zs AA
} C
{z
t A
{stD
n
sn
| y ntr
sy zsrut
sn
C
q
r
stptAquadest
¡ ¢ £¤¥ ¦§ ¡ ¢
¨©ª «©
r D
¬ ©® ¯°A
±©°¯s
¯s
²³I
´untuk
µ©mp
¶l
· ¶¸° ¯t A
° ©ª³© ±¹©º¸» ¯fikasi
Senyawa K
2
CO
3
Gambar D.6 Hasil Analisis FTIR K
2
CO
3
/Zeolit Alam dengan
Konsentrasi Larutan K
2
CO
3
45 gram/60 mL
Aquadest
66
¼½¾ ¿ÀÁ  ÃÀÄ ÀàÁ  ÁÅÆÇÈÆÁ  Á ÂÉÂÆ ¼Â ÊÁ ÊÃ
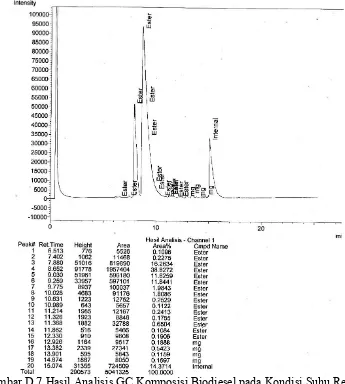
Gambar D.7 Hasil Analisis GC Komposisi Biodiesel pada Kondisi Suhu Reaksi
65
o
C, Rasio Molar Alkohol terhadap Minyak 10 : 1, Jumlah Katalis
K
2
CO
3
/Zeolit Alam 4%, dan Waktu Reaksi 3 Jam
ËÌ
LAMPIRAN E
DOKUMENTASI PENELITIAN
E.1
FOTO PREPARASI ZEOLIT ALAM
G
Ím
ÎÍr
Ï ÐÑFoto
ÒÓol
Ôt
Õl
Ím
G
Ím
ÎÍr
Ï ÐÖFoto
×Ór
Ón
ØÍm
Ín
ØÓn
ÙÍn H
ÚÛ Ú
G
Ím
ÎÍr
Ï ÐÜFoto
×Óm
Ín
Ís
Ín
ØÓn
ÙÍn
×Ón
Ín
ÙÍs
ÕÔr
ÝÞ
G
ßm
à ßr
áâãFoto
äåol
æt
çl
ßm H
ßs
æl
èåm
ßn
ßs
ßn
éån
ê ßn
è ån
ßn
ê ßs
çær
G
ßm
à ßr
áâëFoto
è år
ån
éßm
ßn
é ån
êßn
Aquadest
G
ßm
à ßr
áâÝFoto H
ßs
æl
è år
ån
é ßm
ßn
é ån
êßn
Aquadest
ìí
G
îm
ïîr
ð ñòFoto
ó ôm
în
îs
în
õôn
öîn
÷ øôn
G
îm
ïîr
ð ñùFoto H
îs
úl
ó ôm
în
îs
în
õôn
öîn
÷ øôn
G
îm
ïîr
ð ñíFoto
ó ôn
öúl
ún
öîn
õôn
öîn
ûüýt
îr
þÿ
G m
r
ÿFoto
n
y k n
r
ol
t
l m
G m
r
Foto
r
k
ol
t
l m
E.2
FOTO PROSES IMPREGNASI ZEOLIT ALAM DENGAN
LARUTAN K
2
CO
3
G
m
r
Foto
ros
s Impr
n
s
ol
t
l
m
G
m
r
Foto H
s
l Impr
n
s
ol
t
l
m
G
m
r
!"#Foto
$ %m
n
s
n
&%n
'n
( )%n
G
m
r
!"*Foto
$%ny
r
+n
'n
&%n
'n
$omp
,kum
G
m
r
!"-
Foto H
s
+
l
$ %ny
r
+
n
'
n
&%n
'n
$omp
,
kum
./
G
0m
10r
234.Foto
5ros
6s
70ls
8n
0s
896n
:0n
Furnace
G
0m
10r
234;Foto
70t
0l
8s H
0s
8l
70ls
8n
0s
8G
0m
10r
234 <Foto
56n
::8l
8n
:0n
96n
:0n
=>?t
0r
@A
G
Bm
CBr
DEFGHFoto
IBt
Bl
Js
IKLMNO PQol
Jt
Rl
Bm
E.3
FOTO BAHAN BAKU MINYAK DAN PENGUJIAN KADAR ASAM
LEMAK BEBAS
G
Bm
C Br
SFG TFoto
UBVBn
UBku
WJny
Bk
XQYBk
Z B Y J[Rice Bran Oil
)
G
Bm
CBr
S FGGFoto
ZQn
\u
]JBn
IBYB
r
Rs
Bm
DQm
Bk
U QCBs
^_
E.4
FOTO PROSES TRANSESTERIFIKASI
G
`m
a`r
b cdeFoto
f`n
gh`i `n
jl
`t
kros
ls
mn `ns
lst
lr
i oik
`s
iG
`m
a`r
b cdpFoto
kros
ls
mn`ns
lst
lr
i oik
`s
iG
`m
a`r
b cd_Foto
klny
`r
in
g`n
q`t
`l
is
rln
g`n
komp
`s`kum
tu
G
vm
w vr
xyzuFoto
{ |m
}s
v~ vn H
vs
}l
vns
|st
|r
} }k
vs
} |n
vn
oron
{ |m
}s
v~G
vm
w vr
xyzt
Foto
{
ros
|s
{ |n
u
} vn
}o
} |s
|l
G
vm
w vr
xyzFoto
}o
}|s
|l H
vs
}l
{ |n
u
}vn
E.5
FOTO ANALISIS BIODIESEL
G
m
r
Foto
n
l
s
s
ns
t
s
G
m
r
Foto
n
l
s
s
skos
t
s