Antioxidant Activity of
Syzygium polynthum
Extracts
Mutia Devi Hidayati
1, Taslim Ersam
1, Kuniyoshi Shimizu
2, and Sri Fatmawati
1,* 1Department of Chemistry, Sepuluh Nopember Institute of Technology (ITS) Jl. Arief Rahman Hakim, Sukolilo, Surabaya 60111, Indonesia
2
Department of Agro-environmental Science, Faculty of Agriculture, Kyushu University, Fukuoka, Japan, 812-8581
Received January 20, 2016; Accepted May 6, 2016
ABSTRACT
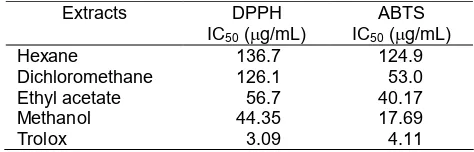
Antioxidant activities ofSyzygium polyanthumleaves extracts (methanol, ethyl acetate, dichloromethane and n-hexane) were evaluated by using DPPH (2,2-diphenyl-2-picrylhydrazyl) and ABTS (2,2’-azinobis (3-ethylbenzothiazole-6-sulfonic acid) methods. The methanol extract showed the highest antioxidant activity of DPPH assay among extract with IC50 value of 44.35 g/mL. In addition, methanol extract also showed the highest antioxidant activity of ABTS assay among extracts with IC50 value of 17.69 g/mL. This study indicated that the
methanol extract ofS. polyanthumleaves is potential as antioxidant.
Keywords:antioxidant; DPPH; ABTS;Syzygium polyanthum; salam leaves
ABSTRAK
Aktivitas antioksidan ekstrak daun Syzygium polyanthum telah dievaluasi menggunakan metode DPPH (2,2-diphenyl-2-picrylhydrazyl) dan ABTS (2,2’-azinobis (3-ethylbenzothiazole-6-sulfonic acid). Ekstrak metanol menunjukkan aktivitas antioksidan yang paling besar dibandingkan ekstrak lain melalui uji DPPH dengan nilai IC50 44,35 g/mL. Selain itu ekstrak metanol juga menunjukkan aktivitas antioksidan yang paling tinggi dibandingkan ekstrak lain melalui uji ABTS dengan nilai IC5017,69g/mL. Hasil penelitian ini menunjukkan bahwa ekstrak metanol daunS. polyanthumberpotensi sebagai antioksidan.
Kata Kunci:antioksidan; DPPH; ABTS;Syzygium polyanthum; daun salam
INTRODUCTION
Role of free radical agents damage to cell and tissues occupies the most important position in the body metabolism. Free radical reactions occur due to the oxidation reaction of stable compound become unstable and also reactive compound. These reactive species can react with other compound in the body and causing tissue damage which will lead to disease such as cancer, alzheimer disease, cardiac reperfusion and abnormalities. These free radicals such as peroxide, hydroperoxide or lipid peroxyl may oxidize nucleic acids, proteins, lipids, DNA, and can initiate the degenerative disease [1]. Physical and chemical factors such as heavy metals, heating, radiation, dyes and preservatives play an important role in the occurrence of excessive oxidation reactions [2].
Antioxidants are chemical compounds that can neutralize free radical agents. These compounds work by donating electron to achieve of stable form, thus inhibit the oxidative mechanism that lead to degenerative disease. Antioxidant compounds can include natural and synthetic compounds. Synthetic antioxidant has some
side effect and become carcinogenic agents [1]. Therefore, many studies are developing antioxidant compounds from natural materials. Most of the antioxidant compounds were obtained from plant such as vitamin C, vitamin E, carotenoids and phenolic acid. Various classes of compounds with wide of physical and chemical properties were isolated, such as gallic acid have strong antioxidant activity [3].
Indonesian biodiversity is one of important asset in the utilization of chemical plants. S. polyanthum which commonly known as salam leaves, are usually used to seasoning because of its rich aroma. Salam leaves has been widely used in Indonesian traditional medicines. It is also known to be effective for keeping the health because it has a wide range of bioactivity such as antihypertensive [4], antimicrobial [5], and antidiarrheal [6].
Materials
Salam leaves (Syzygium polyanthum) that used in this research were obtained from Probolinggo, East Java, Indonesia. Solvent (methanol, ethanol, ethyl acetate, dichloromethane, dimethylsulfoxide and n-hexane) were analytical grade. DPPH (2,2-diphenyl-2-picrylhydrazyl; Sigma-Aldrich, Steinheim, Germany), ABTS (2,2’-azinobis(3-ethyl benzothiazoline-6-sulfonic acid), K2S2O8 (potassium peroxydisulfate), Trolox
(6-hydroxy-2,5,7,8- tetramethylchroman-2- carboxylic acid; Sigma Aldrich) was used as antioxidant standard.
Instrumentation
Incubator EYELA SLI-400 used to process incubation of sample. The reaction was monitored by spectrophotometer (UV Jasco V-530, Japan).
Procedure
Salam leaves extracts
Each of dried salam leaves (25 g) were extracted with MeOH, ethyl acetate, dichloromethane and hexane (200 mL for 24 h) at room temperature. The extracts were filtered through filter paper then concentrated with a rotary evaporator under pressure to give crude extracts.
DPPH assay
DPPH activity was assayed by method described by Brand Williams [9], modified by Dudonne et al. [10] with minor modification. Each of crude extracts (10 mg) was dissolved in 1 mL methanol. The reaction mixture was consisted of 1 mL DPPH solution 6x10-5 M and 33 µL of methanol solution of crude extract. After 20 min incubation for 37 °C, absorbance of the reaction mixture was measured at 515 nm by spectrophotometer (UV Jasco V-530, Japan) to giveAsvalue. Blank sample with 33 µL of methanol in DPPH solution was prepared and
0.70.02 at 734 nm. Each of crude extracts (10 mg) was dissolved in 1 mL DMSO. The reaction mixture was consisted of 1 mL working solution and 10 µL of crude extract and shaken for 10 sec. After 4 min incubation at 30 °C, the absorbance of the reaction
mixture was measured at 734 nm by
spectrophotometer (UV Jasco V-530, Japan) to give As. Ethanol 99.5% was used as a blank and measured at same wavelength (Ab). The experiment was carried out in triplicate and ABTS activity was calculated using formula 1.
RESULT AND DISCUSSION
Salam Leaves Extract
The extraction process was performed by using methanol, ethyl acetate, dichloromethane and n-hexane which provide varying extraction yield (1.75, 1.40, 0.52, and 0.33 g), respectively. The highest extraction yield for various extract was obtained when methanol used as solvent. The results show that extraction yields depend on polarity of the sample. The component in S. polyanthum leaves expected to be polar when extracted with methanol. This result indicated that most compounds in S. polyanthum leaves were soluble in polar solvent.
DPPH and ABTS Assay
Antioxidant activity of four extractsS. polyanthum based on DPPH assay at a concentration of
159.73 μg/mL were presented in Fig. 1. The
Fig 1. DPPH Scavenging activity of S. polyanthum extracts at a concentration of 159.73 g/mL, MeOH, methanol extract; EA, ethyl acetate extract; DCM, dichloromethane extract; Hx, hexane extract and T, trolox (positive control). Each column represents the meanSD, n = 3
Fig 3. ABTS Scavenging activity of S. polyanthum extracts at a concentration of 45.9 g/mL, MeOH, methanol extract; EA, ethyl acetate extract; DCM, dichloromethane extract; Hx, hexane extract and T, trolox (positive control). Each column represents the meanSD, n = 3
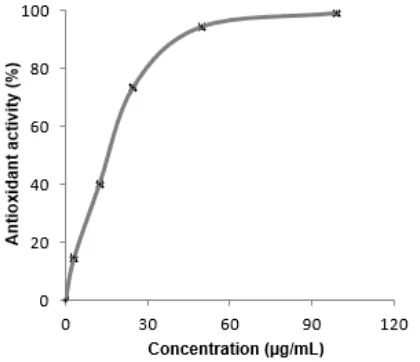
Fig 2. DPPH Scavenging activity of S. polyanthum methanol extract
Fig 4. ABTS Scavenging activity of S. polyanthum methanol extract
Determination of IC50value for each extracts were
determine to perform the doses of extract that can reduce intensity of 50% free radical absorption. Based on the result of interpolation of Fig. 2, the IC50value ofS.
polyanthum MeOH extract was 44.35 μg/mL. Trolox as positive control have higher activity than that of methanol extract with IC50 value of 3.09 μg/mL. Trolox is consist of
aromatic ring with substitution of hydroxy and carboxylate group. The existence of these two group have an important role to determine the efficiency of compound in free radical inhibition.
DPPH (2,2-diphenyl-2-picrylhyrazyl) is a method of measuring the antioxidant activity which is widely used
to test the activity of natural compounds in food and biological system [12]. DPPH is a stable free radical that interacts with antioxidant through the electron transfer. When reacting with antioxidant, DPPH radical is converted to DPPH and its color change from purple to yellow [13]. DPPH assay is one of simple and rapid test. This assay needs UV-vis spectrophotometry to determine the absorbance of compound [14].
Antioxidant activity of salam leaves extracts are also performed by using the ABTS method. The principle of this method is the radical cation decolorization through the transfer of electrons that neutralize free radicals which are marked with a dark blue color change to yellow light [13]. ABTS is soluble in both aqueous and organic solvents. This assay can be used in multiple media to determine both hydrophilic and lipophilic antioxidant capacities of extracts. ABTS reacts rapidly with antioxidant. This assay can be used to determine effects of pH on antioxidant mechanisms and leading to the ABTS wide pH range availability [14].
The results of antioxidant activity of four extracts (MeOH, ethyl acetate, dichloromethane, and hexane) at
a concentration of 45.9 μg/mL were presented in Fig. 3.
The percentages of antioxidant activity were 94.3, 55.9, 45.4, and 13.0%, respectively, while trolox as a standard has antioxidant activity of 98.8%. It indicated that the methanol extract had the highest activity among the other leaves extracts (Fig. 3). ABTS scavenging activity of methanol extract of S. polyanthum was shown in Fig. 4. The IC50 value of methanol extracts is 17.69 μg/mL whereas Trolox is 4.11 μg/mL.
The various extracts of S. polyanthumwere tested for antioxidant activity using DPPH and ABTS. Table 1 showed the IC50 value of DPPH from the methanol
extract was important role in absorption and neutralization of free radicals.
CONCLUSION
S. polyanthumleaves are widely used in traditional medicine in South East Asia. Antioxidant activities of S. polyanthum leaves extract were determined by using DPPH and ABTS. The various extracts was compared with trolox which indicated that only methanol extract which more potential for further isolation process to have bioactive compounds with antioxidant activity. The methanol extract showed the highest antioxidant activity both in DPPH and ABTS assay. This finding support that
Free Radicals and Their Role in Different Clinical Conditions: An Overview,Int. J. Pharma Sci. Res., 1 (3), 185–192.
[2] Fearon, E.R., 1997, Human cancer syndromes: clues to the origin and nature of cancer, Science, 278 (5340), 1043–1050.
[3] Biskup, I., Golonka, I., Gamian, A., and Sroka, Z., 2013, Antioxidant activity of selected phenols estimated by ABTS and FRAP methods, Postepy Hig. Med. Dosw., 67, 958–963.
[4] Ismiyati, 2013, Aktivitas antihipertensi ekstrak etanol daun salam (Syzygium polyantha [wigth] walp) pada tikus wistar, profil kromatografi lapis tipis serta penetapan kandungan fenolik total dan flavonoid totalnya,Thesis, UGM, Yogyakarta. [5] Kusuma, I.W., Kuspradini, H., Arung, E.T., Aryani,
F., Min, Y.H., Kim, J.S., and Kim, Y.U., 2011, Biological activity and phytochemical analysis of three Indonesian medicinal plants, Murraya koenigii, Syzygium polyanthum and Zingiber purpurea, J. Acupunct. Meridian Stud., 4 (1), 75–79.
[6] Nuratmi, B., Winarno, M.W., and Sundari, S., 1999, Khasiat daun salam (Eugenia polyantha Wight) sebagai antidiare pada tikus putih, Media Litbangkes “Obat Asli Indonesia”, 8 (3-4), 14–17. [7] Darusman, L.K., Wahyuni, T.W., and Alwi, F.,
2013, Acetylcholinesterase inhibition and antioxidant activity of Syzygium cumini, S. aromaticumand S. polyanthumfrom Indonesia,J. Biol. Sci., 13 (5), 412–416.
[8] Arumugam, B., Manaharan, T., Heng, C.K., Kuppusamy, U.R., and Palanisamy, U.D., 2014, Antioxidant and antiglycemic potentials of a standardized extract of Syzygium malaccense, LWT Food Sci. Technol., 59 (2), 707–712.
[10] Dudonné, S., Vitrac, X., Coutière, P., Woillez, M., and Mérillon, J.M., 2009, Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays, J. Agric. Food Chem., 57 (5), 1768–1774.
[11] Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., and Rice-Evans, C., 1999, Antioxidant activity applying an improved ABTS radical cation decolorization assay,Free Radic. Biol. Med., 26 (9-10), 1231–1237.
[12] Shalaby, E.A., and Shanab, S.M.M., 2012, Comparison of DPPH and ABTS assays for determining antioxidant potential of water and methanol extracts of Spirulina platensis, Indian J. Geo Mar. Sci., 42 (25), 556–564.
[13] Żurowska, D.M., and Wenta, W., 2012, A
comparison of ABTS and DPPH methods for assessing the total antioxidant capacity of human milk, Acta Sci. Pol., Technol. Aliment., 11 (1), 83–89.
[14] Prior, R.L., Wu, X., and Schaich, K., 2005, Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements, J. Agric. Food Chem., 53 (10), 4290–4302.