49
DAFTAR PUSTAKA
1. Kemendag. Kemendag Mendorong Masyarakat Untuk Beralih Dari Minyak
Goreng Curah Ke Minyak Goreng Kemasan. www.kemendag.go.id.
Published 2013.
2. Ng C, Kamisah Y, Faizah O, et al. Involvement of Inflammation and
Adverse Vascular Remodelling in the Blood Pressure Raising Effect of
Repeatedly Heated Palm Oil in Rats. 2012.
3. Martin J, Joffre F, Vernevaut M. Biochemical and Molecular Action of
Nutrients Cyclic Fatty Acid Monomers from Heated Oil Modify the
Activities of Lipid Synthesizing and Oxidizing Enzymes in Rat Liver 1 , 2.
2000:1524-1530.
4. Adam S, Das S, Jaarin K. A Detailed Microscopic Study of The Changes in
The Aorta of Experimental Model of Postmenopausal Rats Fed with
Repeatedly Heated Palm Oil. Int J Exp Pathol. 2009;90(3):321-327.
5. Leong X, Aishah A, Nor AU, Das S, Jaarin K. Heated Palm Oil Causes
Rise in Blood Pressure and Cardiac Changes in Heart Muscle in
Experimental Rats. Acheives Med Res. 2008;39(6):567-572.
6. Goodman LS, Gilman A. Dasar Farmakologi Terapi. X. (Hardman JG,
Limbird LE, Aisyah C, eds.). Jakarta: ECG; 2012.
7. Wilmana PF, Gunawan SG. Analgesik-Antipiretik Analgesik Anti-Inflamasi
Nonsteroid Dan Obat Gangguan Sendi Lainnya. Dalam: Farmakologi Dan Terapi. V. Jakarta: Balai Penerbit FKUI; 2007.
Faces of Glutathione, A Cellular Protagonist. Biochem Pharmacol. 2003;66
(8):1499-1503.
9. Ulilalbab A. Aktivitas Antioksidan Tablet Effervescent Rosella Ungu
Sebagai Suplement Penghambat Laju Peroksidasi Melalui Pengujian In
Vivo. PKM-P. 2010.
10. Qinna NA, Mallah EM, Arafat TA, Idkaidek NM. Effect Of Licorice and
Grapefruit Juice on Paracetamol Pharmacokinetics in Human Saliva. Int J
Pharm Pharm Sci. 2012;4(4):158-162.
11. Barshop NJ, Capparelli E V, Sirlin CB, Jeffrey B, Lavine JE.
Acetaminophen Pharmacokinetics in Children with Nonalcoholic Fatty
Liver Disease. J Pediatr Gastroenterol Nutr. 2012;52(2):198-202.
12. Kumdi BV, Kolawole JA, Apeh E. The Effect of Yoyo Bitters on The
Pharmacokinetics of Single Oral Dose Paracetamol Tablet in Human
Volunteers. Int J Biol Chem Sci. 2011;5:717-723.
13. Pradana DA, Hayati F, Samudra AG, Setya A. Effect of Curcumin and
Honey to Pharmacokinetics of Paracetamol in Male Wistar Rats. Eksakta.
2013;12(1):193.
14. Simaremare P, Andrie M, Bambang Wijianto. Effect of Durian Fruit Juice
(Durio zibethinus Murr.) to Pharmacokinetic Profile of Paracetamol on
Wistar Male Rats (Rattus norvegicus L.). Tradit Med J.
2013;18(3):178-186.
16. Sartika RAD. Pengaruh Suhu dan Lama Proses Menggoreng (Deep Frying)
Terhadap Pembentukan Asam Lemak Trans. Markara Sains. 2009;13:23-28.
17. Lestari P. Pemanfaatan Minyak Goreng Jelantah Pada Pembuatan Sabun
Cuci Piring. Tesis. 2010.
18. Rukmini A. Regenerasi Minyak Goreng Bekas dengan Arang Sekam
Menekan Kerusakan Organ Tubuh. Semin Nas Teknol 2007 (SNT 2007). 2007:1978-9777.
19. Mulyati S, Meilina H. Pemurnian Minyak Jelantah dengan Menggunakan
Sari Mengkudu. 2007.
http://222.124.186.229/gdl40/go.php?id=gdlnode-gdl-res-2007-srimulyati-1082&node-3517&start=6 .
20. Oktaviani ND. Hubungan Lamanya Pemanasan dengan Kerusakan Minyak
Goreng Curah Ditinjau dari Bilangan Peroksida. J Biomedika.
2009;1:31-34.
21. Lee J, Lee S, Lee H, Park K, Choe E. Spinach (Spinacia oleracea) as A
Natural Food Grade Antioxidant in Deep Fat Fried Products. J Agric Food
Chem. 2002;50:5664-5669.
22. Herawati, Akhlus S. Kinerja (Bht) sebagai Antioksidan Minyak Sawit pada
Perlindungan terhadap Oksidasi Oksigen Singlet. Akta Kim. 2006;2:1-8. 23. Azeredo HMC, Faria JAF, Silva. Minimization of Proxide Formation Rate
in Soybean Oil by Antioxidant Combinations. Food Res Int.
2004;37:689-694.
Lemak Bebas (FFA), Bilangan Peroksida, dan Warna Gelap Minyak
Goreng Bekas. Semin Nas Tek Kim Indones – SNTKI 2009. 2009:1-14. 25. Ghidurus M, Turtoi M, Boskou G, Niculita P, Stan V. Nutritional and
Health Aspects Related to Frying. Rom Biotechnol Lett. 2010;15(6).
26. Castillo’n PG, Artalejo FR, Fornés NS, et al. Intake of Fried Foods is
Associated with Obesity in The Cohort of Spanish Adults from The
European Prospective Investigation into Cancer and Nutrition. Am J Clin
Nutr. 2007;86:198-205.
27. Koch A, KÖnig B, Spielmann J, Leitner A, Stang GL, Eder K. Thermally
Oxidized Oil Increases the Expression of Insulin-Induced Genes and
Inhibits Activation of Sterol Regulatory Element-Binding Protein-2 in Rat
Liver. J Nutr Biochem Mol Genet Mech. 2007;137:2018-2023.
28. Toussaint K, Yang XC, Zielinski MA, et al. What Do We (Not) Know
About How Paracetamol (Acetaminophen) Works? J Clin Pharm Ther. 2010:617-638. doi:10.1111/j.1365-2710.2009.01143.x.
29. Liudmila LM, Sangkuhl K, Caroline FT, Garret AF, Russ BA, Teri EK.
PharmGKB Summary: Pathways of Acetaminophen Metabolism at The
Therapeutic Versus Toxic Doses. Pharmacogenet Genomics. 2015.
30. Katzung BG. Farmakologi: Dasar Dan Klinik Buku 2. I. Jakarta: Salemba Medika; 2002.
31. Ubaldo C, Hall NS, Le B. Postmarketing Review of Intravenous
Acetaminophen Dosing Based on Food and Drug Administration
32. Parkinson A. Biotransformation of Xenobiotics. V. (Klaassen C, ed.). New
York: McGraw-Hill; 1996.
33. Gibson G. Introduction to Drug Metabolism. II. London: Blackie
Academics and Professional; 1994.
34. Mitchell R, McGill, Jaeschke H. Metabolism And Disposition of
Acetaminophen: Recent Advances in Relation to Hepatotoxicity and
Diagnosis. Pharm Res. 2013;30(9):2174-2187.
35. Short Communication. Involvement of Human Cytochrome P450 2D6 in
The Bioactivation of Acetaminophen. 2000;28(12):1397-1400.
36. Winarsi H. Antioksidan Alami Dan Radikal Bebas: Potensi Dan
Aplikasinya Dalam Kesehatan. V. Yogyakarta: Penerbit Kanisius; 2011.
37. Tjay TH, Rahardja K. Obat-Obat Penting : Khasiat, Penggunaan, Dan
Efek-Efek Sampingnya. VI. Jakarta: Penerbit PT. Elex Media Komputindo;
2002.
38. Katzung BG. Basic and Clinical Pharmacology. XII. New York:
McGraw-Hill; 2011.
39. WHO. Research Guideliness For Evaluating The Safety and Efficacy of
Herbal Medicines. Manila: Regional Office for The Western Pasific; 2003.
40. Leong X-F, Mustafa MR, Das S, Jaarin K. Association of Elevated Blood
Pressure and Impaired Vasorelaxation in Experimental Sprague-Dawley
Rats Fed with Heated Vegetable Oil. Lipids Health Dis. 2010;9(66):1-10.
42. Laurence DR, Bacharach AL. Evaluation of Drug Activities:
Pharmacometrics, Ed. by D.R. Laurence and A.L. Bacharach. London: Academic Press; 1964.
43. Rusdiana T, Sjuib F, Asyarie S. Interaksi Farmakokinetik Kombinasi Obat
Parasetamol dan Fenilpropanolamin Hidroklorida sebagai Komponen Obat
Flu. Skripsi. 2009:2-4.
44. Depkes RI. Farmakope Indonesia. IV. Jakarta: Departemen Kesehatan Republik Indonesia; 1995.
45. Sarwono J. Pintar Menulis Karya Ilmiah Kunci Sukses Dalam Menulis
Ilmiah. Yogyakarta: CV. Andi OFFSET; 2010.
46. Hakim L. Farmakokinetik. Yogyakarta: Bursa Ilmu; 2010.
47. Slattery JT, Wilson JM, BS TFK, Nelson SD. Dose-dependent
pharmacokinetics of acetaminophen: Evidence of glutathione depletion in
humans. Clin Pharmacol Ther. 1987;41(4):413-418.
48. Forrest JAH, Clements JA, Prescott LF. Clinical Pharmacokinetics of
Lampiran 6. Tabel konversi perhitungan dosis
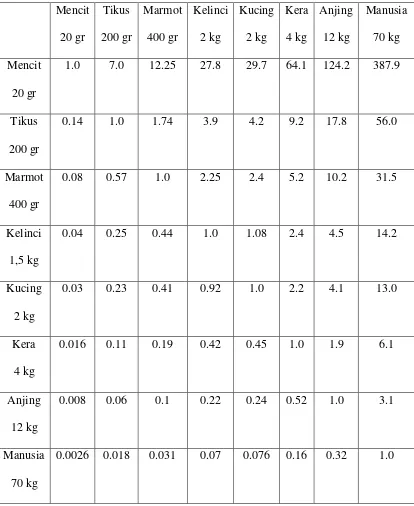
TABEL KONVERSI PERHITUNGAN DOSIS LAURENCE & BACHARACH42
Lampiran 7.Berat badan tikus dan rerata sisa pakan perlakuan
1. Berat badan tikus
No. Kelompok Berat Dosis
1 K1 233 gram 1,45 mL
2 K2 189 gram 1,18 mL
3 K3 249 gram 1,55 mL
4 K4 217 gram 1,36 mL
5 K5 176 gram 1,1 mL
6 P1 255 gram 1,59 mL
7 P2 211 gram 1,31 mL
8 P3 274 gram 1,71 mL
9 P4 300 gram 1,87 mL
10 P5 257 gram 1,60 mL
2. Rerata sisa pakan perlakuan
No. Kelompok Rerata sisa pakan
1 P1 2,83 gram
2 P2 3,38 gram
3 P3 2,36 gram
4 P4 1,87 gram
Lampiran 8.Absorbansi parasetamol pada darah tikus
1. Absorbansi parasetamol pada darah kelompok kontrol
Menit
2. Absorbansi parasetamol pada darah kelompok perlakuan
Lampiran 9. Kadar parasetamol pada darah tikus
1. Kadar parasetamol pada darah kelompok kontrol
Menit
2. Kadar parasetamol pada darah kelompok perlakuan
Lampiran 10. Ln kadar paraseamol pada darah tikus
1. Ln kadar parasetamol pada darah kelompok kontrol
Menit
2. Ln kadar parasetamol pada darah kelompok perlakuan
Lampiran 11. Parameter farmakokinetik
1. Parameter farmakokinetik kelompok kontrol
Parameter Farmakokinetik
Kelompok
K1 K2 K3 K4 K5 Rerata
Kel 0,00445 0,00389 0,00416 0,00416 0,00374 0,00408
t1/2e 155,800 178,392 166,587 166,461 185,155 170,479
Ka 0,123 0,156 0,089 0,205 0,144 0,143
t1/2a 5,657 4,457 7,831 3,386 4,800 5,226
tmaks 28,086 24,335 36,252 19,428 25,972 26,815
Cmax 36,848 35,168 33,419 37,554 35,505 35,699
AUC0-inf 9386,096 9950,508 9341,139 9780,476 10454,477 9782,539
Vd 277,849 244,213 319,101 267,207 224,897 266,653
Cl 1,236 0,949 1,327 1,112 0,842 1,093
2. Parameter farmakokinetik kelompok perlakuan
Parameter Kelompok
Farmakokinetik P1 P2 P3 P4 P5 Rerata
Kel 0,00271 0,00272 0,00292 0,00248 0,00265 0,00269
t1/2e 255,720 254,779 237,329 278,806 261,509 257,629
Ka 0,152 0,107 0,084 0,182 0,126 0,130
t1/2a 4,547 6,448 8,278 3,818 5,495 5,717
tmaks 26,917 35,096 41,533 23,967 31,286 31,760
Cmaks 45,102 43,890 43,555 44,400 45,201 44,430
AUC0-inf 17902,400 17752,205 16839,270 18959,450 18531,310 17996,927
Vd 262,184 217,040 278,215 317,449 260,650 267,108
Lampiran 12. Hasil uji statistik
1. Jumlah sampel tiap kelompok
Case Processing Summary
Tikus
Cases
Valid Missing Total
N Percent N Percent N Percent
K Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
t1/2 e Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
Ka Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
t1/2 a Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
tmax Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
Cp max Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
AUC 0-inf Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
Vd Kontrol 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
Perlakuan 5 100,0% 0 0,0% 5 100,0%
2. Hasil uji normalitas data pengaruh pemberian minyak jelantah terhadap profil
farmakokinetik parasetamol pada darah
Tests of Normality
Tikus
Kolmogorov-Smirnova Shapiro-Wilk
Statistic df Sig. Statistic df Sig.
K Kontrol ,215 5 ,200* ,955 5 ,776
Perlakuan ,242 5 ,200* ,960 5 ,806
t1/2 e Kontrol ,233 5 ,200* ,955 5 ,775
Perlakuan ,224 5 ,200* ,960 5 ,810
Ka Kontrol ,186 5 ,200* ,987 5 ,968
Perlakuan ,143 5 ,200* ,988 5 ,974
t1/2 a Kontrol ,201 5 ,200* ,945 5 ,705
Perlakuan ,151 5 ,200* ,967 5 ,856
tmax Kontrol ,218 5 ,200* ,961 5 ,812
Perlakuan ,158 5 ,200* ,975 5 ,907
Cp max Kontrol ,170 5 ,200* ,968 5 ,860
Perlakuan ,223 5 ,200* ,913 5 ,489
AUC 0-inf Kontrol ,208 5 ,200* ,923 5 ,550
Perlakuan ,181 5 ,200* ,973 5 ,896
Vd Kontrol ,177 5 ,200* ,976 5 ,915
Cl Kontrol ,165 5 ,200* ,963 5 ,827
Perlakuan ,188 5 ,200* ,945 5 ,702
*. This is a lower bound of the true significance.
a. Lilliefors Significance Correction
3. Data statistik tiap kelompok tikus
Group Statistics
Tikus N Mean Std. Deviation Std. Error Mean
K Kontrol 5 ,00407972780 ,000274126637 ,000122593159
Perlakuan 5 ,00269712000 ,000155985487 ,000069758830
t1/2 e Kontrol
5 170,479096609 829920
5 257,628618431 438200
14,8945071830
27645
6,66102611052
1743
Ka Kontrol 5 ,14310600 ,042864090 ,019169404
Perlakuan 5 ,13025000 ,038147003 ,017059858
t1/2 a Kontrol
5 5,22606919674 9445
5 5,71698286521 6155
5 26,8146739056 70844
5 31,7597965211 85734
6,91135484236
3327
3,09085184882
Cp max Kontrol
5 35,6986385271 20620
5 44,4295873633 12020
5 9782,53915586 973400
5 17996,9273378 1966300
5 266,653455326 880360
5 267,107723967 029640
5 1,09323841562 9088
5 ,718606093145 465
,088105330237
085
,039401901518
038
4. Hasil uji homogenitas varians
Independent Samples Test
Levene's Test for
Equality of
Variances
t-test for
Equality of
Equal
variances not
assumed
variances not
assumed
variances not
assumed
variances not
assumed
variances not
Equal
variances not
assumed
variances not
assumed
variances not
assumed
variances not
assumed
3,83
6 5,49
9
5. Hasil uji t test tidak berpasangan pengaruh pemberian minyak jelantah terhadap profil
farmakokinetik parasetamol
Independent Samples Test
t-test for Equality of Means
Sig.
(2-K Equal variances
assumed ,000
,0013826078
00
,0001410509
Equal variances not
t1/2 e Equal variances
assumed ,000
Equal variances not
assumed ,000
Ka Equal variances
assumed ,630 ,012856000 ,025661348
Equal variances not
assumed ,630 ,012856000 ,025661348
t1/2 a Equal variances
assumed ,661
Equal variances not
assumed ,661
tmax Equal variances
assumed ,267
Equal variances not
assumed ,267
Cp max Equal variances
assumed ,000
Equal variances not
AUC
0-inf
Equal variances
assumed ,000
Equal variances not
assumed ,000
Vd Equal variances
assumed ,985
Equal variances not
assumed ,985
Cl Equal variances
assumed ,005
,3746323224
83623
,0976637704
38932
Equal variances not
assumed ,010
,3746323224
83623
,0976637704
Lampiran 13. Dokumentasi
Pencampuran pakan dengan minyak jelantah Pembuatan minyak jelantah
Hewan coba dikandangkan individual Pembuatan sediaan parasetamol
Sentrifuge sampel
darah+EDTA+TCA5% pada 2500 rpm
Pengambilan supernatan
Lampiran 14. Biodata mahasiswa
I. Identitas
1. Nama Lengkap : Faizurrahman Andi Kusuma
2. NIM : 22010112140182
3. Tempat, Tanggal lahir : Grobogan, 14 April 1994 4. Jenis Kelamin : Laki-laki
5. Alamat Rumah : Jl. Ahmad Yani 269 Purwodadi, Kab.Grobogan
6. E-mail : [email protected]
7. Telepon rumah/HP : 08574012498
II. Pendidikan
No. Tingkat Nama & Tempat Tahun Lulus
1. Sekolah Dasar SD Negeri 1 Kuripan 2006
2. Sekolah Menengah Pertama SMP Negeri 1 Purwodadi 2009 3. Sekolah Menengah Akhir SMA Negeri 1 Purwodadi 2012
4. Perguruan Tinggi Fakultas Kedokteran UNDIP 2012 - sekarang
III. Pengalaman Kepanitiaan
No Jabatan Tahun Nama Kepanitiaan
1. Sie Perlengkapan Mei 2014 Scientific Fair
2. Koordinator Sie Lomba September - Desember 2014 Porseni
3. Ketua Panitia September - Oktober 2015 Kemah Bakti
IV. Pengalaman Bekerja
No. Jabatan Tahun Nama Instansi