Study of Glassy Carbon Electrode Preparation Modified by Gold Nanoparticle and Its Application Towards the Determination of Chromium(VI) with the Influence of
Chromium(III), Nickel(II) and Zinc(II) using Voltammetry
Santhy Wyantuti1, Roekmi-ati Tjokronegoro1
1Jurusan Kimia FMIPA Universitas Padjadjaran
Email : [email protected]
Study of Glassy Carbon Electrode Preparation
Modified by Gold Nanoparticle and Its Application
Towards the Determination of Chromium(VI)
with the Influence of Chromium(III), Nickel(II) and
Zinc(II) using Voltammetry
Santhy Wyantuti
1, Roekmi-ati Tjokronegoro
1,Yeni Wahyuni Hartati
1,
Camellia Panatarani
21
Department of Chemistry, University of Padjadjaran, Jl Raya
Bandung-Sumedang km. 21,West Java 40132, Indonesia
2
Department of Physics, University of Padjadjaran, Jl Raya
Bandung-Sumedang km. 21,West Java 40132, Indonesia
Background
Gold nanostructures modified glassy carbon electrode → detect
Cr(VI)
→
stripping voltammetry adsorption (Li et al., 2010)
Highly sensitive Cr(VI
) → cyclic voltammetry and linier voltammetry →
with
boron-doped diamond electrodes (Fierro et al. 2012)
Proposed research
•
develop and modify carbon electrode using gold nanoparticles
•
the influence of Cr(III), Ni(II) and Zn(II) towards the electrochemical analysis
of Cr(VI)
Objective
Process of selecting
reduction agent and
capping agent
Gold nanoparticles
Modifications
process
glassy carbon electrode
Preparation of gold nanoparticles
Gold nanoparticles modified glassy carbon electrode
Characterization
of electrode
processes
Gold nanoparticles modified glassy carbon electrode
Determination process
of Cr(VI)
0.01 M HAuCl4
+ water
+ 0.1 M sodium citrate
+ 0.1 M NaBH4
Size of gold
nanoparticle
about 2.2 nm
Fig 1.
TEM image of gold nanoparticles
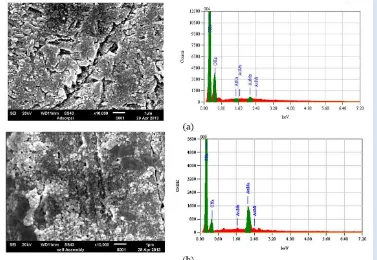
Fig 2
. SEM image of gold nanoparticles modified glassy carbon
electrode surface. Modification was conducted using (a)
adsorption and (b) self assembly process
PREPARATION AND CHARACTERIZATION OF AuNP MODIFIED GCE
Gold
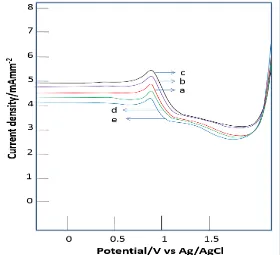
Fig. 3.
Voltammogram of variation of
deposition potential at (a) 600, (b) 700,
(c) 800, (d) 900 and (e) 1000 mV for 0.5
µg/L Cr(VI) solution with deposition
time 180 s, scan rate 50 mV/s and
amplitude modulation 500 mV
Fig. 4
. Voltammogram of variation of
deposition time at (a) 90 s, (b) 120
s, (c) 150 s, (d) 180 s and (e) 210 s
for 0.5 µg/L Cr(VI) solution with
deposition potential 800 mV, scan
rate 50 mV/s and amplitude
modulation 500 mV
OPTIMUM CONDITION OF AuNP-GCE
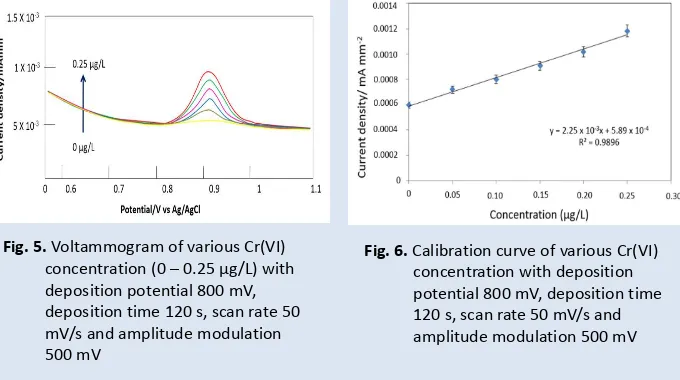
Fig. 5.
Voltammogram of various Cr(VI)
concentration (0
–
0.25 µg/L) with
deposition potential 800 mV,
deposition time 120 s, scan rate 50
mV/s and amplitude modulation
500 mV
Fig. 6.
Calibration curve of various Cr(VI)
concentration with deposition
potential 800 mV, deposition time
120 s, scan rate 50 mV/s and
amplitude modulation 500 mV
LINIER CALIBRATION CURVE OF VARIOUS Cr(VI)
EFFECT OF INTERFERING IONS
Differential pulse voltammogram of 50
ppb Cr(VI) without foreign ions in 0.01 M
hydrochloric acid in optimum condition
The percentage variation of the peak
current induced by the presence of
interfering ions with respect to Cr(VI)
alone are: 0.29% for Zn, 3.70% for Cr(III),
0.07% for Ni(II)
These deviations from Cr(VI)
CONCLUSIONS
From the present study it is found that AuNP colloid with ~2.2 nm
size is formed by reducing 6 mg of chloroauric acid (HAuCl
4
) with
1.102 mg sodium borohydrate (in 290 µL water) as reducing agent
in 0.1 M sodium citrate. The percentage of gold nanoparticles
deposited onto glassy carbon electrode by self assembly process is
11.55% from total surface mass of the glassy carbon (C = 76.38%;
O = 12.07%). The electrode performance for the determination of
Cr(VI) resulted in concentration range of 0.050
–
0.250 µg/L,
sensitivity of 2.25 x 10
-3
and accuracy of 94.00%. Study of the