PROCEEDING
ISBN : 978-979-8969-06-5
CONTENT
iii
PREFACE
iv
WELCOMING SPEECH
v
OPENING REMARK
vi
WELCOMING SPEECH
viii
CONFERENCE COMMITTEE
ix
ACKNOWLEDGEMENT
x
PLENARY SESSIONS
Session 1: Dr. Yam Tim Wing
1
Session 2: Prof. Yasumasa Bessho
9
Session 3: Prof. Christopher M. Austin
17
Session 4: Drs. Langkah Sembiring, M.Sc., Ph.D
25
Session 5: Hao Yu, Ph.D
36
THEMATIC ORAL PRESENTATION
39
Topic 1. Molecular Biology, Genetic and Bioinformatics (O-MB)
39
Topic 2. Ecology and Conservation (O-EC)
139
Topic 3. Systematic and Evolution (O-SE)
209
Topic 4. Physiology and Developmental Biology (O-PD)
293
Topic 5. Biomedics (O-BM)
355
THEMATIC POSTER PRESENTATION
433
Topic 1. Molecular Biology, Genetic and Bioinformatics (O-MB)
433
Topic 2. Ecology and Conservation (O-EC)
465
Topic 3. Systematic and Evolution (O-SE)
517
Topic 4. Physiology and Developmental Biology (O-PD)
557
Topic 5. Biomedics (O-BM)
605
LIST OF STUDENT COMMITTEE
643
LIST OF ORAL AND POSTER PARTICIPANTS
644
CONTENT
ix
CONFERENCE COMMITTEE
ICBS 2011 FACULTY OF BIOLOGY UGM
1. Patron : Dean of Faculty of Biology
2. Steering Committee : Dr. Retno Peni Sancayaningsih, M.Sc. Drs. Langkah Sembiring, M.Sc., Ph.D. Dra. Mulyati, M.Si.
Dr. Endang Semiarti, M.S., M.Sc.
Prof. Dra. Endang S. Soetarto, M.Sc., Ph.D.
Prof. Chris Austin (Charles Darwin University, Australia) Prof. Yasumasa Bessho, Ph.D (NAIST, Japan)
3. Academic Reviewer :
Internal Reviewers : Prof. (ret). Dr. Jusup Subagja, M.Sc Prof. (ret). Dr. Jesmant Situmorang, M.Sc
Prof. (ret). Sukarti Moeljoprawiro, M.App.Sc., PhD Prof. (ret). Dr. Nyoman Puniawati Soesilo, SU. Prof. (ret). Dr. Istiyati External Reviewers : Dr. Sentot Santoso.
(Institut fuer Klinische Immunologie und Transfusionsmedizin Justus Liebig Universität Giessen, Germany
Prof. Yasumasa Bessho, Ph.D. (Graduate School of Biological Science,
Nara Institute of Science and Technology (NAIST), Japan) 4. Chief of Organizing : Dr. Yekti Asih Purwestri, M.Si.
Committee
5. Vice of Chief of : Dr. L. Hartanto Nugroho, M.Agr. Organizing Committee
8. Plenary and Scientific : Dr. Rina Sri Kasiamdari
Session Dr. biol.hom. Nastiti Wijayanti, M.Si. Abdul Rahman Siregar, S.Si., M.Biotech. Dr. Woro Anindito Sri Tunjung, M.Sc Sari Darmasiwi, S.Si, M.Biotech
Aries Bagus Sasongko, S.Si, M.Biotech 9. Publication : Zuliyati Rohmah, S.Si., M.Si.
Donan Satria Yudha, S.Si., M.Sc. Slamet Riyadi, S.Si
Aris Setiawan R. Nur Wigunadi
10. Funding and : Dr. Suwarno Hadisusanto Sponsorship Donan Satria Yudha, S.Si, M.Sc 11. Documentation : Drs. Abdul Rachman, M.Si.
Sudarsono
13. Refreshment : Dra. Siti Susanti, S.U Kodrat Wartini
Rusna Nuraini Prapti
14. Hospitality : Dr. Niken Satuti Nur Handayani Dr. Rarastoeti Pratiwi, M.Sc Drs. Heri Sujadmiko, M.Si
Dra. Upiek Ngesti Wibawaning Astuti, M.Kes Dr. Maryani
15. Accommodation : Slamet Widiyanto, S.Si., M.Sc. Donan Satria Yudha, S.Si., M.Sc. Haryanto
Suharjito Harsono
LIST OF ORAL PRESENTER TOPIC 1: MOLECULAR BIOLOGY, GENETIC AND BIOINFORMATIC
39
46
52
59
67
68
75
76
84
93
101
102
110
116
125
O-MB01
The Continous Function of
KNAT1
gene on Secondary Shoot Growth in
Micropropagation of Indonesian Black Orchid
Coelogyne pandurata
Lindley Transgenic
Endang Semiarti1*, Eggie F. Ginanjar1, Rizqie L.Nurwulan1, Y. Machida2 and C. Machida3
1
Faculty of Biology, Universitas Gadjah Mada, Jl. Teknika Selatan, Sekip Utara, Yogyakarta 55281, Indonesia., 2 Division of Biological Sciences, Graduate School of Science, Nagoya University, Chikusa-ku, Nagoya, Japan, 3
College of Biotechnology and Bioscience, Chubu University, Kasugai, Japan, 4 *Corresponding author: [email protected];
Abstract
Agrobacterium-mediated genetic transformation has become increasingly important tools for improving cultivars and studying gene function in plants. This is particularly true in orchids, which are highly valued ornamental plants that are continually being genetically altered. To improve the quality of Indonesian black orchids, we developed a convenient method for the genetic modification of this orchid using Agrobacterium tumefaciens. The T-DNA of a disarmed Ti plasmid containing the coding region of a neomycin phosphotransferase II gene as a selectable marker was successfully introduced into intact protocorms of the Black Orchid (Coelogyne pandurata L. Form East Kalimantan). The
BREVIPEDICELLUS (BP)/KNAT1 gene was under the control of the Cauliflower Mosaic Virus (CaMV) 35S promoter, and is a member of the family of class 1 KNOTTED-like homeobox (KNOX) genes in Arabidopsis thaliana that is required for the maintenance of indeterminate state of cells. the T-DNA containing BP/KNAT1 was transformed into the black orchid. The protocorms that were transformed with BP/KNAT1 produced multiple shoots, indicating that the BP/KNAT1 gene can be used to improve shoot formation for mass propagation of these orchids. In vitro culture using leaf discs of the 35S::KNAT1 transgenic Black orchid on hormon-free medium also resulted in multishoots production. These data indicate that the KNAT1 gene maintained its function in secondary shoot growth of transgenic black orchid. The method can be applied to the commercial production of orchids in Indonesia for both domestic and international trade.
Keywords: Black orchids, secondary shoot induction, genetic transformation, Agrobacterium tumefaciens.
INTRODUCTION
Techniques on plant tissue culture for orchid micropropagation are useful for mass
production. Since the needs of orchids are always increase by the time for commercial
trades, the conservation efforts should also be elaborated. Mass propagation through in vitro
culture will become a good tool for these efforts. But, in orchids, there are many obstacles to
do tissue culture, due to the slow growth rate and the long life cycle of orchid. Recently, we
developed an efficient technique for orchid micropropagation through Agrobacterium–
mediated genetic transformation of Knotted1-like Arabidopsis thaliana (KNAT1) gene into
genomes of three genera of Indonesian orchids, i.e Phalaenopsis amabilis (L.) Blume,
Vanda tricolor Lindley and Coelogyne pandurata Lindley (1, 2).. The insersion of KNAT1
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
gene into orchid genome resulted in multishoot formation in P. amabilis and C. pandurata,
but not in V. tricolor. In P. amabilis, there was 31-90 shoots emerged from one protocorm
(developing orchid embryo), and in C. pandurata there was 4-7 shoots emerged from one
transformant’s protocorm. There is still a question to be addressed: is there any stable orchid transformant that maintained the function of interest gene in their secondary growth?
In this report, we analyze the continous function of KNAT1 transgene on secondary
growth of black orchid transformant’s shoots in tissue culture condition, to understand the
stability of KNAT1 as a foreign gene in orchid genome. It is worth to elaborate for this orchid
due to the rareness of the black orchid C. pandurata as an Indonesian endemic orchid. The
method could be implemented for other Indonesian natural orchids.
MATERIALS AND METHODS
Plant materials and culture condition
Four developing independent shoots of 35S::KNAT1-black orchid transformants that
are growing up on 100 mg. l-1 Kanamycin-containing New Phalaenopsis (NP) medium were
used as plant materials in this experiments. The shoots as source of explant were cut into
two leaf discs and a stem to induce new shoot formation on regeneration medium in vitro.
Explants were cultivated on half strength of New Phalaenopsis (NP) medium (3), with
addition of 150 ml.l-1 coconut water, and combination of plant growth regulators
2-isopenthenyladenine (2iP) and Naphtalene acetic acid (NAA) with ratio of 1: 1 (0.15 and 3
μM). The cultures were incubated at 25°C with 1000 lux continuous light. The growth of protocorm like bodies (PLBs), and shoots from the explants were examined every week. For
control experiment, a similar set of experiment was also done using non-transformant black
orchid plant.
T-DNA Construct for Genetic Transformation and Detection of Transgene by PCR
Genetic transformation of plasmid 35S::KNAT1 and pGreen vector into orchid was
carried out according to the method of Semiarti et al. (1). The structure of 35S::KNAT1
containing T-DNA construct that inserted into orchid genome is shown in Fig. 1.
Figure 1. Schematic Structure of 35S::KNAT1 containing T-DNA. LB, Left border; RB, Right
Border, 35S: CaMV promoter; KNAT1 gene; HPT: Hygromycin phosphotransferase; Tnos: Nos terminal. Bar: 1.2 kb.
Pnos NPTII Tnos p35S KNAT1 BAR Tnos
RB LB
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
Faculty of Biology UGM - Yogyakarta, Indonesia, September 23rd-24th 2011
Genomic DNA of transformant plants and non transformant were isolated and
extracted using QIAGEN gDNA extraction kit (GmBH, Germany) according to the manual
instruction from the manufacture. Pured gDNA from the emerged shoots from selected
transformant explants were detected by polymerase chain reaction (PCR) method for the
existance of KNAT1 gene using KNAT1 gene specific primer KNAT1F1 (5’
-CTTCCTAAAGAAGC-ACGGCAG-3’) and KNAT1R1 (5’-
CCAGTGACGCTTTCTTTGGT-T-3’), that amplified 1.2 kb DNA fragment.
RESULTS AND DISCUSSION
Phenotypic analyses
Morphology of the shoot and leaves of transformant are normal as the same as non
transformant plant. The growth rate of shoot(s) from transformant and non-transformant
stem explant on half strength NP medium with various concentration of growth regulators
treatment showed that generally, induction of shoot formation in transformant was faster
than that of non-transformant (Table 1).This data indicates that the growth of shoots from
transformant explant may be induced by KNAT1 gene activity that integrated in the orchid
genome, than that of induction by growth regulators endogenously or exogenously.
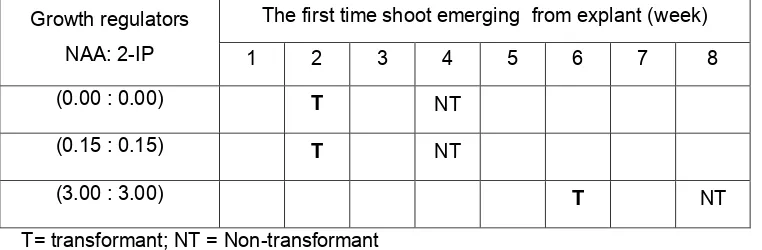
Table 1. The Growth of Shoots from transformant and non-transformant stem explants on
half strength NP Medium and Various Concentration of Growth Regulators Auxin and Cytokinin.
Growth regulators
NAA: 2-IP
The first time shoot emerging from explant (week)
1 2 3 4 5 6 7 8
(0.00 : 0.00) T NT
(0.15 : 0.15) T NT
(3.00 : 3.00) T NT
T= transformant; NT = Non-transformant
The fastest emerged shoot(s) from transformant stem was two weeks after explant
inoculation on ½ NP medium without additional growth regulators and ½ NP+ 0.15 µM NAA
and 0.15 µM 2iP, though in non transformant explant the shoots emerged at 4 weeks after
inoculation. Interestingly, when the higher concentration of growth regulators was added into
medium, the shoot formation delayed up to 6 weeks in transformant explant and 8 weeks for
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
non transformant explant. It is inline with our previous data in P.amabilis, that shoots formed
on leaf discs of 35S::KNAT1 transformant grown on hormon-free NP medium (1).
Multishoots production from 35S::KNAT1 Transformant Explants
In the case of number of shoot production, the transformant stem produced
multishoots from one stem explant. The higher number of shoots were produced in
transformant explants,than that in non transformant stem (Fig.2, Table 2). Multishoot
production were also reported by Yu et al. (4), when Dendrobium Orchid Homeobox1 (DOH1)
introduced into orchid hybrid Dendrobium “Madame Thong In”. Introduction of KNAT 1 gene
into some Dicot was also induced multishoot formation, i.e Chuck et al. (5) observed
multishoot production in Arabidopsis, and Nishimura et al. (6) obtained multishoots in
tobacco transgenic explants. Semiarti et al. (7) reported that the expression of KNAT1 gene
was improved in a leaf mutant of Arabidopsis, assymetric leaves2 mutant, that produced
multishoots on mutant leaf disc in that were cultured on hormone free medium.
Figure 2. Multishoot formation from stem explant of 35S::KNAT1 on ½ NP + 0.15 µM
NAA+ 0.15 µM). 0 ; week 0, starting inoculated explant, 2; Shoot initiation (emerging leaf
primordia) come out from explant, week-2; 7; Week-7, 9 ; Week-9, 11; Week-11, and 13;
multishoots emerged at week-13 (Bar: 1mm)
Multishoots production might also be related to the arrangement of endogeneous
phytohormone biosynthesis pathway, such as cytokinin and gibberelic acid (GA) that
involved in cell division, cell elongation and shoot formation. As described by George et al.
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
Faculty of Biology UGM - Yogyakarta, Indonesia, September 23rd-24th 2011
(8), that in plant tissue culture, during adventif shoot formation the concentration of
cytokinin in cell increase, but the concentration of GA will be decreased. In tobacco
transgenic plants, overproduction of KNAT1 protein suppressed the activity of GA20ox
(Ntc12) that bound to some sequences in the first intron of the GA20ox gene. This
complex reduced the synthesis of GA, in turn it will activate cytokinin synthase gene,that
caused multishoot production.. Overexpression of KNAT1 gene will also eliminate apical
domination in the shoot tip, so that the determinated cells will switch into undeterminated
cells (9, 10). It is reasonable that multishoot production in this experiment has also proved
the activities of overexpressed KNAT1 gene in the black orchid stem.
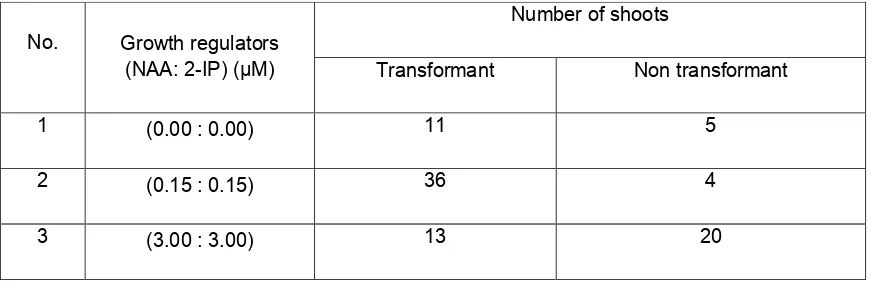
Table 2. Number of Shoot Production from 35S::KNAT1 transformant stem explant after 13
weeks cultivation on ½ NP Medium supplemented with various growth regulators.
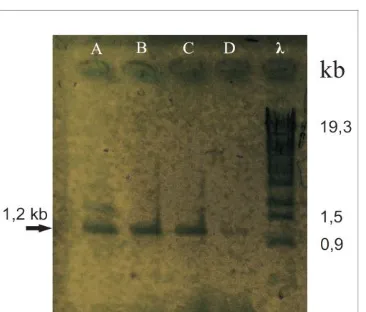
Detection of KNAT1gene in 35S::KNAT1 transformant plant’s genome
Seven shoots of transformants and three non-transformant plants were analyzed to
prove the prescence of 35S::KNAT1 into its genomes. The genomic DNA of each plants
were amplified using specific oligonucleotide primers for KNAT1 genes (KNAT1F1 and
KNAT1R1), that resulted in about 1.2 kb amplified DNA fragment. Four out of seven shoots
showed positive results, but the other three were negative (Fig. 3). These results indicate
that KNAT1 gene still integrated into the genomes of four orchid transformant lines and
maintained its activity for shoot production in these orchids. Using these four lines,
micropropagation of black orchid (C. pandurata) can be improved, as well as the use of the
transgenic technology for other character improvement of this orchid. The use of Green
Flourescent Protein (GFP) as a reporter gene as described previously (11), combine with
KNAT1 gene will improve the orchid quality in both shoot multiplication and flourescence
plant. Hopefully, it will give benefit to support both conservation and commercial trade of
Indonesian natural orchids.
No. Growth regulators (NAA: 2-IP) (µM)
Number of shoots
Transformant Non transformant
1 (0.00 : 0.00) 11 5
2 (0.15 : 0.15) 36 4
3 (3.00 : 3.00) 13 20
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
Figure 3. Detection of KNAT1 gene in 35S::KNAT1 Black Orchid Transformants. Lanes (A-D)
show that 1.2 kb DNA fragment could be amplified from four transformants. λ indicates λ
DNA digested by Sty I enzyme that used as DNA size marker.
CONCLUSION
The Arabidopsis KNAT1 gene can be used for improvement of shoot formation in
micropropagation of Black Orchid (C. pandurata). The KNAT1 gene has stably maintained
its function in secondary shoot growth of black orchid transformant. The method can be
applied to the commercial production of orchids in Indonesia for both domestic and
international trade.
Acknowledgement
The research was supported by Indonesian DGHE Research Competition grant HB XVII
2009-2010 No. LPPM-UGM/604/2009. We thank to Bunga Rintee Orchid Nursery,
Yogyakarta for the gift of fruit of the Black Orchid .
REFERENCES
1. Semiarti, E., A. Indrianto, A. Purwantoro, S. Isminingsih, N. Suseno, T. Ishikawa, Y.
Yoshioka, Y. Machida, C, Machida. 2007. Agrobacterium-mediated transformation
of the wild orchid species Phalaenopsis amabilis. Plant Biotechnology 2, 265-272
2. Semiarti, E., A. Indrianto, E.A. Suyanto, R.L. Nurwulan, R. Restiani, Y. Machida, and C.
Machida. 2010a. Genetic Transformation of Indonesian Black Orchids (Coelogyne
pandurata Lindley) Through Agrobacterium tumefaciens for Micropropagation.
Proceedings of NIOC 2010. Nagoya Dome. Japan
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)
Faculty of Biology UGM - Yogyakarta, Indonesia, September 23rd-24th 2011
3. Islam, M. O., S. Ichihashi and S. Matsui. 1998. Control of Growth and Development of
Protocorm Like Body from Callus by Carbon Sources in Phalaenopsis. Plant
Biotechnology,15 (4): 183-187.
4. Yu, H , S.H. Yang and C.J. Goh. 2000. DOH1, a Class 1 knox Gen, Is Required for
Maintenance of the Basic Plant Architecture and Floral Transtition in Orchid.Plant
Cell Report. 12: 2114
5. Chuck, G., C. Lincoln, and S. Hake. 1996. KNAT1 induced lobeled leaves with ectopic
meristem when overexpressed in Arabidopsis. Plant Cell. 8: 1277-1289
6. Nishimura, A., M. Tamaoki, T. Sakamoto, and M. Matsuoka. 2000. Over-ekspresion of
tobacco Knotted1-type Class 1 homeobox genes alters various leaf morphology.
Plant Cell Physiology. 41(15): 583-590
7. Semiarti, E., Y. Ueno, H. Tsukaya, H. Iwakawa, C. Machida, and Y. Machida. 2001. The
ASYMETRIC LEAVES2 gene of Arabidopsis thaliana regulates formation of a
symmetric lamina, establishment of venation and repression of meristem-related
homeobox genes in leaves. Development.128: 1771-1783
8. George, E.F, M.A. Hall and G-J. de Klerk. 2008. Plant Propagation Tissue Culture 3rd
Edition. Springer. The Netherlands. Pp.1, 2, 175-187, 205-216
9. Davies, P.J. 2004. PLANT HORMONES Biosynthesis, Signal Transduction, Action!.
Kluwer Academic Publishers. London. Pp 588-594
10. Sinha, N.R., R.E.Williams, and S. Hake. 1993. Overekspression of the maize homeobox
gen, KNOTTED1, causes a switch from determinate to indeterminate cell fates
genes. Genes Dev.7: 787-795
11. Semiarti, E., A. Indrianto, A. Purwantoro, N.A. Martiwi, Y.M.L. Feroniasanti, F. Nadifah,
I.S. Mercuriana, R. Dwiyani, H. Iwakawa, Y. Yoshioka, Y. Machida, C, Machida.
2010b. High-frequency genetic transformation of Phalaenopsis amabilis orchid
using tomato extract-enriched medium for the pre-culture of protocom. Journal of
Horticultural Science & Biotechnology 85(3): 205-210.
International Conference on Biological Science Faculty of Biology Universitas Gadjah Mada 2011 (ICBS BIO-UGM 2011)