Bilateral ECT in Schizophrenia: A Preliminary Study
Worrawat Chanpattana, M.L. Somchai Chakrabhand, Wanchai Buppanharun,
and Harold A. Sackeim
Background: This preliminary study examined the effects
of electrical stimulus intensity on the speed of response
and efficacy of bilateral electroconvulsive therapy (ECT)
in the treatment of schizophrenia.
Methods: Sixty-two patients with schizophrenia received
combination treatment with bilateral ECT and
flu-penthixol. Using a randomized, double-blind design, the
effects of three dosages of the ECT electrical stimulus
were examined. Patients were treated with a stimulus
intensity that was just above seizure threshold, two-times
threshold, or four-times threshold. Assessments of
out-come used the Brief Psychiatric Rating Scale, Global
Assessment of Functioning, and the Mini-Mental State
Exam.
Results: Thirty-three of sixty-two patients met remitter
criteria, including maintaining improvement over a
3-week stabilization period. The dosage groups were
equivalent in the number of patients who met remitter
criteria. The low-dose remitter group (n
5
11) received
more ECT treatments and required more days to meet
remitter status than both the twofold (n
5
11) and fourfold
remitter groups (n
5
11). There was no difference among
the groups in change in global cognitive status as assessed
by the Mini-Mental State Exam.
Conclusions: This preliminary study indicates that
treat-ment with high-dosage bilateral ECT speeds clinical
re-sponse in patients with schizophrenia. There may be a
therapeutic window of stimulus intensity in impacting on the
efficacy of bilateral ECT, which needs further study. A more
sensitive battery of cognitive tests should be used in future
research. Biol Psychiatry 2000;48:222–228 © 2000
Soci-ety of Biological Psychiatry
Key Words: Electroconvulsive therapy, method, efficacy,
schizophrenia
Introduction
S
ince its inception, electroconvulsive therapy (ECT) has
been used to treat schizophrenia. Neuroleptic
medica-tions rapidly replaced ECT after their introduction in the
1950s. During the 1970s, when limitations in the efficacy
of neuroleptics and adverse effects from prolonged use
were recognized, interest in ECT as a treatment for
medication-resistant schizophrenia returned (Fink and
Sackeim 1996). A number of surveys in several countries
found that from 2.9% to 36% of patients receiving ECT
had a diagnosis of schizophrenia (Krueger and Sackeim
1995). This rate was reported to be 60% and 75% in Czech
(Baudis 1992) and Indian patients (Shukla 1981),
respectively.
Research on the use of ECT in schizophrenia has
been characterized by a variety of methodologic
limi-tations, including uncertain diagnostic criteria,
nonran-dom assignment to treatment groups, and lack of blind
and reliable clinical assessment (Krueger and Sackeim
1995). Nonetheless, the conclusions suggested in this
literature are that 1) ECT is effective in the treatment of
schizophrenia, especially among patients with acute
exacerbations, relatively short duration of illness, or
both; and that 2) combined ECT and neuroleptic
treat-ment is more effective than either ECT alone or
neuroleptic treatment alone (Chanpattana et al 1999a,
1999b; Fink and Sackeim 1996).
The interactive effects of stimulus intensity and
elec-trode placement on the efficacy of ECT in major
depres-sion is established (Sackeim et al 1987a, 1987b, 1993, in
press; Krystal et al 1998; McCall et al, in press). With
right unilateral ECT, the likelihood of clinical response in
major depression is highly dependent on the degree to
which stimulus dosage exceeds seizure threshold. With
both right unilateral and bilateral ECT, higher stimulus
intensity results in faster clinical improvement ( Nobler et
al 1997; Sackeim et al 1993, in press). The impact of
electrical dosage in moderating the efficacy of bilateral
ECT in schizophrenia is unknown. We hypothesized that
high-dosage bilateral ECT would enhance the speed of
clinical response in patients with schizophrenia.
From the Departments of Psychiatry (WC) and Preventive Medicine (WB), Srinakharinwirot University, Bangkok, and the Department of Mental Health, Nonthaburi (MLSC), Thailand, and the Department of Biological Psychiatry, New York State Psychiatric Institute and Departments of Psychiatry and Radiology, College of Physicians & Surgeons, Columbia University, New York, New York (HAS).
Address reprint requests to Worrawat Chanpattana, M.D., Department of Psychia-try, Srinakharinwirot University, 681Samsen, Dusit Bangkok 10300, Thailand. Received October 1, 1999; revised January 10, 2000; accepted January 19, 2000.
© 2000 Society of Biological Psychiatry 0006-3223/00/$20.00
Methods and Materials
Subjects
Sixty-seven patients (30 men, 37 women) with acute psychotic exacerbations and who met the DSM-IV criteria for schizophre-nia (American Psychiatric Association 1994) were referred for ECT because of failure to respond to neuroleptic treatment. Psychiatric diagnosis was based on the consensus of three psychiatrists and also had to concur with the patients’ medical records. Diagnosis in the medical records had to be consistent throughout the episode of illness. Other inclusion criteria were a minimum pretreatment score of 37 on the Brief Psychiatric Rating Scale (BPRS, 18 items, rated 0 – 6; Overall and Gorham 1962), and subjects had to be between ages 16 and 50 years. Patients were excluded if they received treatment with depot neuroleptics or ECT during the previous 6 months, psychotic disorders due to a general medical condition, neurologic illness, alcohol or other substance abuse, or serious medical illness. All patients had normal results of complete blood count, serum electrolytes, and electrocardiography. This study was approved by the Ethics Committee of the Faculty of Medicine of Srina-kharinwirot University and the National Review Board of Re-search Studies in Humans of Thailand. After complete descrip-tion of the study and the opportunity to ask quesdescrip-tions, voluntary written informed consent was obtained from the patients or their guardians.
Twenty-three patients (10 men, 13 women) were randomized to receive low-dosage bilateral ECT (dose just above the seizure threshold), 23 patients (11 men and 12 women) were assigned to the twofold seizure threshold group, and 21 patients (9 men and 12 women) were assigned to the fourfold seizure threshold group. Five patients dropped out from this double-blind compar-ative study—two from the low-dose group, two from the twofold seizure threshold group, and one from the fourfold seizure threshold group—leaving 62 patients in the study.
Procedures
Neuroleptic medications prescribed before the study were dis-continued without a washout period. Flupenthixol was started before the first ECT session and was continued throughout the study. The dosage schedule of flupenthixol was fixed at 12 mg/day ($600 mg chlorpromazine [CPZ] equivalents) during the first week, 18 mg/day ($900 mg CPZ equivalents) for the days 8 –10, and 24 mg/day ($1200 mg CPZ equivalents) thereafter, depending on tolerability. All patients were treated between 18 –24 mg/day of flupenthixol, which has a half-life of approx-imately 35 hours, leading to steady state at 7 days. Benzhexol (4 –15 mg/day) was used to manage extrapyramidal symptoms, with dosage titrated on a clinical basis. Diazepam (10 mg/day) was prescribed to control agitation on a PRN basis and was withheld at least 8 hours before each ECT treatment.
Electroconvulsive therapy was administered three times per week. The ECT devices were a Thymatron DGx and MECTA SR 1. Each patient was treated with only one ECT device (two different hospitals participated in the study, each using a different device). Thiopental (2– 4 mg/kg) was used at the lowest dosage to induce general anesthesia, minimizing effects on seizure
threshold. Succinylcholine (0.5–1 mg/kg) served as the muscle relaxant. Atropine (0.4 mg intravenously) was administered approximately 2 min before the anesthetic. Patients were oxy-genated from anesthetic administration until postictal resumption of spontaneous respiration. Bitemporal bilateral electrode place-ment was used throughout. The tourniquet method and two channels of prefrontal electroencephalogram (EEG) were used to assess seizure duration. An adequate seizure was defined as a tonic-clonic motor convulsion occurring bilaterally for at least 30 sec, plus EEG evidence of a cerebral seizure. Reliance on the duration of motor manifestations to define adequacy was con-servative because EEG ictal expression typically outlasts motor expression for several seconds (Sackeim et al 1991; Warmflash et al 1987). Grossly suprathreshold stimulation can result in reduced seizure duration, below conventional cutoffs for “ade-quacy” (American Psychiatric Association Task Force, in press; Sackeim et al 1991); however, it was felt that the stimulus intensities used here, all adjusted relative to initial seizure threshold, would be insufficient to result in a marked shortening of seizure duration. In each treatment one adequate seizure was elicited.
Using the empirical titration technique (Sackeim et al 1987c), seizure threshold was quantified at the first two treatment sessions and was defined as the minimal electrical stimulus (in millicoulombs) to produce a motor seizure duration of at least 30 sec. For the MECTA SR 1, the titration schedule followed that used by Coffey et al (1995). With the Thymatron DGx, the first stimulus at the first treatment session was 10% of total charge. If this failed to produce an adequate seizure, electrical dosage was increased in 10% steps. This approach roughly matched the charge delivered by the MECTA SR 1 and Thymatron DGx at each step in the titration schedule. There was a limit of four stimulations in a session, but the maximal number administered in this sample was three. A 40-sec interval elapsed between each step in the titration, and no patient required additional thiopental. At the second treatment session, patients were stimulated with an intensity 5% below the first treatment’s threshold value. If this failed to produce an adequate seizure, the first session’s value was adopted as the initial seizure threshold (ST). Patients were then stimulated with the assigned electrical stimulus charge (either 1, or 2, or 43ST). If the decreased intensity produced an adequate seizure, this new value was taken as the ST, and there was no further stimulation in that session. At subsequent sessions in the 1 ST group, if needed, electrical stimulus dosage was increased by one step to achieve an adequate seizure. In the 2 ST and 4 ST groups, the duration of motor seizures was ignored during the three subsequent sessions. Afterward, if the seizure was shorter than 30 sec, the delivered charge was increased by one step. At the 10th treatment session, all patients were reassessed for ST. The stimulus charge was then adjusted based on the assigned ST group or to the maximal charge settings of the device.
Clinical Evaluation
on to a 3-week stabilization period (Chanpattana 1998, 1999; Chanpattana et al 1999a, 1999b). The stabilization period com-prised the following treatment schedule: 3 ECT treatments in the first week, then once a week for the second and third weeks (during which BPRS scores of 25 or less had to be consistently maintained). If BPRS scores rose above 25 at any time during this period, and the total number of ECT treatments was less than 20, patients returned to the acute ECT treatment regimen and repeated the acute phase and stabilization schedules again. Patients with BPRS scores above 25, who had already received 20 ECT treatments, were considered ECT nonresponders. The same considerations applied to the patients who had not shown significant improvement (BPRS . 25) until their 20th ECT treatment. Thus, ECT responders were patients who completed the 3-week stabilization period during which the BPRS score assessed before each treatment was consistently 25 or less.
Measures used to assess study outcome were 1) BPRS as-sessed before each treatment, during the acute and stabilization periods, and end of the study (1 week after the last treatment); 2) Global Assessment of Functioning (GAF) assessed before each acute treatment and at the end of the study; and 3) the Mini-Mental State Exam (MMSE, Thai version; Kongsakon and Vanichtanom 1994) assessed at the same time as the BPRS. Three psychiatric nurses served as raters, masked to the treatment conditions. Each patient was rated by the same nurse. These raters underwent training for 12–24 months. Interrater reliability was assessed. Each rater provided ratings simultaneously on 10 patients. Each patient was interviewed by a psychiatrist for 20 min. The correlations of BPRS scores between each rater and the psychiatrist indicated strong reliability (0.93, 0.96, and 0.97).
Statistical Analyses
The results are expressed as mean6SD. Seizure threshold data were analyzed after logarithmic transformation to improve the normality of the data distribution (Sackeim et al 1987b). For discontinuous data, chi-square tests were used to test for differ-ences among the groups. One-way analyses of variance (ANOVA) were used to compare the three treatment groups in continuous demographic and clinical variables. Significant main effects of treatment group were followed by Scheffe´ post hoc comparisons to identify pair-wise differences. The groups were compared in speed of response by conducting ANOVAs on the number of ECT treatments and the number of days in ECT treatment for patients who met initial and final remitter criteria. In addition, across the total sample, Kaplan-Meier survival analyses were conducted on these measures, treating ECT nonresponders as censored observations.
Results
Sixty-seven patients with schizophrenia received ECT.
Five patients dropped out, leaving 62 patients in the study.
All 5 patients who dropped out withdrew consent,
report-ing fear of ECT, despite receivreport-ing basic information about
ECT and schizophrenia. Table 1 presents demographic and
clinical features of the 62 patients as a function of
treatment condition. There were no significant differences
among the three treatment groups in any variable.
Thirty-three patients maintained remitter status through
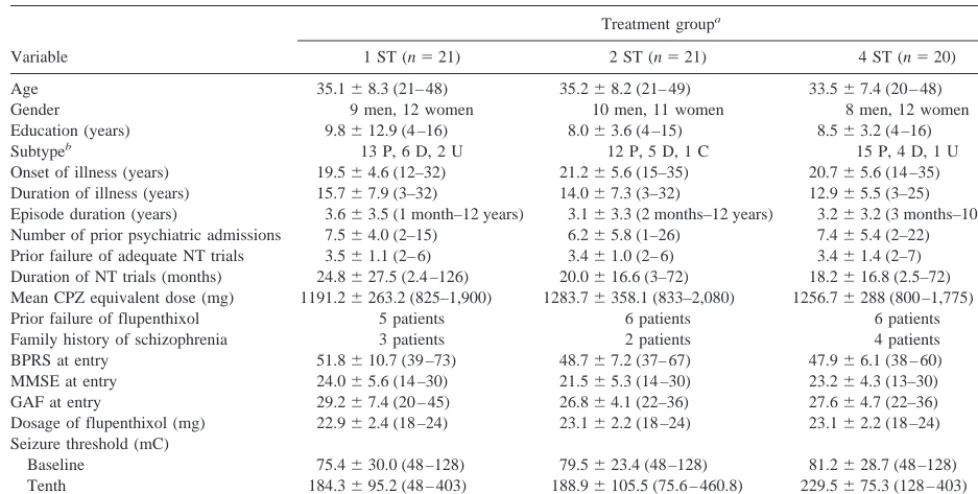
Table 1. Demographic and Clinical Features of the Sample
Variable
Treatment groupa
1 ST (n521) 2 ST (n521) 4 ST (n520)
Age 35.168.3 (21– 48) 35.268.2 (21– 49) 33.567.4 (20 – 48)
Gender 9 men, 12 women 10 men, 11 women 8 men, 12 women
Education (years) 9.8612.9 (4 –16) 8.063.6 (4 –15) 8.563.2 (4 –16)
Subtypeb 13 P, 6 D, 2 U 12 P, 5 D, 1 C 15 P, 4 D, 1 U
Onset of illness (years) 19.564.6 (12–32) 21.265.6 (15–35) 20.765.6 (14 –35) Duration of illness (years) 15.767.9 (3–32) 14.067.3 (3–32) 12.965.5 (3–25)
Episode duration (years) 3.663.5 (1 month–12 years) 3.163.3 (2 months–12 years) 3.263.2 (3 months–10 years) Number of prior psychiatric admissions 7.564.0 (2–15) 6.265.8 (1–26) 7.465.4 (2–22)
Prior failure of adequate NT trials 3.561.1 (2– 6) 3.461.0 (2– 6) 3.461.4 (2–7) Duration of NT trials (months) 24.8627.5 (2.4 –126) 20.0616.6 (3–72) 18.2616.8 (2.5–72) Mean CPZ equivalent dose (mg) 1191.26263.2 (825–1,900) 1283.76358.1 (833–2,080) 1256.76288 (800 –1,775)
Prior failure of flupenthixol 5 patients 6 patients 6 patients
Family history of schizophrenia 3 patients 2 patients 4 patients BPRS at entry 51.8610.7 (39 –73) 48.767.2 (37– 67) 47.966.1 (38 – 60) MMSE at entry 24.065.6 (14 –30) 21.565.3 (14 –30) 23.264.3 (13–30) GAF at entry 29.267.4 (20 – 45) 26.864.1 (22–36) 27.664.7 (22–36) Dosage of flupenthixol (mg) 22.962.4 (18 –24) 23.162.2 (18 –24) 23.162.2 (18 –24) Seizure threshold (mC)
Baseline 75.4630.0 (48 –128) 79.5623.4 (48 –128) 81.2628.7 (48 –128) Tenth 184.3695.2 (48 – 403) 188.96105.5 (75.6 – 460.8) 229.5675.3 (128 – 403) Increment 107.5684.8 (0 –323) 108.86111.0 (0 – 412.8) 147.8686.0 (48 –355.2)
aTreatment groups: 1 ST, dose just above seizure threshold (ST); 2 ST, 23ST; 4 ST, 43ST. bSubtype: P, paranoid; D, disorganized; C, catatonia; U, undifferentiated.
the stabilization period and were classified as ECT
re-sponders. There was no difference in response rate among
the three treatment groups [52%, 52%, and 55% for 1 ST,
2 ST, and 4 ST, respectively;
x
2(2)
5
0.04, ns]. There
were 11 patients in each treatment group. The three
treatment groups did not differ in any of the demographic
or clinical variables listed in Table 1.
Table 2 presents the treatment features for the remitted
patients as a function of the ECT conditions. Compared
with remitters in the low-dosage group, remitters in both
the 2 ST and 4 ST groups received fewer ECT treatments
at the time of first significant clinical improvement,
defined as a BPRS score of 25 or less [F(2,30)
5
18.70,
p
,
.0001; 1 ST vs. 2 ST, p
5
.002; 1 ST vs. 4 ST, p
,
.0001] and had fewer days in ECT treatment [F(2,30)
5
16.35, p
,
.0001; 1 ST vs. 2 ST, p
5
.003; 1 ST vs. 4 ST,
p
,
.0001]. The 2 ST and 4 ST groups did not differ
significantly in these measures (Table 2), although the
pattern of means suggested more rapid onset of
improve-ment in the 4 ST relative to the 2 ST group (p
5
.19).
At the end of study, the same effects were manifest.
Remitters in both the 2 ST and 4 ST groups received fewer
treatments [F(2,30)
5
18.70, p
,
.0001; 1 ST vs. 2 ST,
p
,
.002; 1 ST vs. 4 ST, p
,
.0001] and had fewer days
in treatment than remitters in the low-dosage group
[F(2,30)
5
16.29, p
,
.0001; 1 ST vs. 2 ST, p
5
.003; 1
ST vs. 4 ST, p
,
.0001]. The 2 ST and 4 ST groups did not
differ in these measures (Table 2), although again the 4 ST
group averaged three fewer treatments than did the 2 ST
group (p
5
.13). Including the stabilization phase,
remit-ters in the 1 ST group on average received 18.64 (SD
5
4.95) treatments, whereas the 2 ST remitters averaged
12.46 (SD
5
3.75) treatments and the 4 ST remitters
averaged 9.18 (SD
5
1.47) treatments.
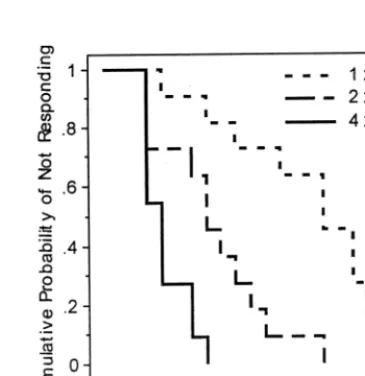
These effects were reexamined using survival analysis
on the total sample of 62 patients, with ECT
nonre-sponders treated as censored observations. The survival
analysis on number of ECT treatments administered
yielded a significant effect of treatment group [Figure 1;
log-rank
x
2(2)
5
25.00, p
,
.0001], as did the analysis of
the number of days in treatment [log-rank
x
2(2)
5
24.38,
p
,
.0001].
The extent of clinical improvement was comparable
among the three remitter groups. At the end of the study,
the 1 ST, 2 ST, and 4 ST remitter groups, respectively, had
62.6%, 60.8%, and 65.6% reductions in BPRS scores and
55.5%, 66.7%, and 55.8% increases in GAF scores. In the
total sample, ANOVAs were conducted on the percentage
change from baseline to study exit in BPRS, GAF, and
MMSE scores, with treatment group and remitter status as
Figure 1. Kaplan–Meier survival plot for cumulative probability of nonresponse as a function of the number of electroconvulsive therapy (ECT) treatments. Groups were treated just above seizure threshold (13ST), two times above threshold (23ST), or four times above threshold (43ST).
Table 2. Treatment Features of Electroconvulsive Therapy (ECT) Remitters
Variable
Treatment Groupa
1 ST (n511) 2 ST (n511) 4 ST (n511) Seizure threshold (mC)
First 68.4616.1 (48 – 80) 83.2623.3 (48 –128) 77.3629.1 (48 –128) Tenth 179.5697.3 (80 – 403) 222.5693.8 (126 – 403) 224655.4 (192–288) Average charge (mC per session)
Sessions 2–9 118.4647.1 (68 –196) 195.6632.9 (151–259) 372.2666.5 (245– 461) Sessions 11– end 254.66131 (128 –533) 413.16139 (230 –576) 576 all At first improvement
No. of ECT treatments 13.665.0 (4 –18) 7.563.8 (3–15) 4.261.5 (3–7) Days of treatment 35.4616.3 (7–58) 16.4611.9 (4 – 42) 7.063.6 (4 –14) At the end of study
No. of ECT treatments 18.665.0 (9 –23) 12.563.8 (8 –20) 9.261.5 (8 –12) Days of treatments 56.4616.3 (28 –79) 37.5612.0 (25– 63) 28.063.6 (25–35)
Valus are given in mean6SD (range). mC, millicoulombs.
between-subject factors. In the ANOVA on change in
BPRS scores there was only a main effect of remitter
status [F(1,56)
5
102.37, p
,
.0001]. Similarly, there was
only a main effect of remitter status in the ANOVA on
change in GAF scores [F(1,56)
5
18.23, p
,
.0001]. This
indicated that the treatment groups were equivalent in the
degree of change in symptoms and functional status. The
ANOVA on MMSE scores produced no significant
ef-fects, indicating that change in global cognitive status was
independent of treatment condition and remitter status.
In the total sample, the three treatment groups did not
differ in initial seizure threshold [F(2,59)
5
0.40, p
5
.67]
or in the percentage increase in threshold from the first to
tenth treatment [F(2,47)
5
0.79, p
5
.46; Table 1].
Patients who did or did not achieve remitter status did not
differ in initial seizure threshold [t(60)
5
0.45, p
5
.65] or
in the increase in threshold [t(48)
5
0.41, p
5
.68].
Although there is some evidence that response to ECT in
major depression is associated with a larger cumulative
seizure threshold increase (Sackeim 1999; Sackeim et al
1987b), this does not appear to be the case in
schizophrenia.
Discussion
This study found that speed of clinical response to bilateral
ECT in patients with schizophrenia is influenced by the
degree to which stimulus dosage exceeds seizure
thresh-old. Both of the high-dosage groups had more rapid
improvement than did the low-dosage group. The findings
parallel the results reported in patients with major
depres-sion ( Nobler et al 1997; Ottosson 1960; Robin and De
Tissera 1982; Sackeim et al 1993, in press). This is of
clinical importance because ECT is frequently used when
rapid improvement is needed. This was the first ECT study
examining the effect of stimulus intensity in patients with
schizophrenia.
Dosage factors, which are known to have greater impact
on the efficacy of right unilateral ECT, may also influence
the therapeutic properties of bilateral ECT. In major
depression, Ottosson (1960) examined the effects of
stim-ulus dose intensity on the efficacy of bilateral ECT.
Although there was no effect of dosage on response rate,
speed of symptom reduction was faster in patients who
received a stimulus intensity that was markedly
suprath-reshold. Cronholm and Ottosson (1963) reported that
low-intensity, ultra-brief pulse, bilateral ECT was less
effective than higher intensity, sinusoidal waveform,
bi-lateral ECT. Robin and De Tissera (1982) found that with
bilateral ECT, high-intensity brief-pulse or chopped sine
wave stimulation resulting in faster clinical improvement
than did low-intensity brief-pulse stimulation. Sackeim et
al (1993) randomly assigned patients to four treatment
conditions: low-dosage right unilateral ECT, high-dosage
right unilateral ECT, low-dosage bilateral ECT, and
high-dosage bilateral ECT. They found that regardless of
electrode placement, the high-dosage groups had more
rapid improvement than did the low-dosage groups. In
addition, low-dosage right unilateral ECT had poor
effi-cacy. In another four-group study, Sackeim et al (in press)
found that markedly suprathreshold (6
3
ST) right
unilat-eral ECT and moderate dosage (2.5
3
ST) bilateral ECT
were more effective and resulted in more rapid
improve-ment than lower intensity (1.5
3
ST and 2.5
3
ST) right
unilateral ECT. Thus, in studies of major depression, there
have been repeated findings that stimulus intensity
im-pacts on speed of clinical response. This study extends this
observation to patients with schizophrenia.
Analyses restricted to the remitter sample and survival
analyses of the total sample both supported the conclusion
that higher stimulus intensity resulted in more rapid
improvement in patients with schizophrenia. The analyses
in the remitter sample were particularly critical because
comparisons of speed of clinical improvement are most
relevant when restricted to patients who actually respond
(Laska and Siegel 1995; Nobler et al 1997). This approach
also avoids confounding likelihood of response (efficacy)
with speed of response (efficiency). There was no
evi-dence in this study that the stimulus-intensity conditions
differed in likelihood of response or degree of
symptom-atic improvement.
An unusual aspect of this study was the use of a 3-week
stabilization period as a screening method to identify
remitters (Chanpattana 1998, 1999; Chanpattana et al
1999a, 1999b). The stabilization period started
immedi-ately after the first sign of significant clinical improvement
(BPRS
#
25). Stability of clinical symptoms across these
3 weeks was required for classification as an ECT remitter.
Therefore, ECT remitters in this study maintained clinical
improvement for a substantial period of time.
This preliminary study had a number of limitations. A
washout period for previous antipsychotic medications
was not used because all study patients were in psychotic
exacerbations, and many patients were difficult to manage.
Lack of a washout may have had a substantial effect on the
patient’s initial seizure threshold because many patients
had prior treatment with low potency neuroleptics
(Sack-eim et al 1991). In contrast, all patients were treated with
flupenthixol, a typical neuroleptic with a potency about
1.5 times that of haloperidol. A fixed titration schedule of
flupenthixol dosage was used to minimize variability in
effects on seizure threshold.
the episode of illness. Theoretically, the inclusion in the
sample of patients with depressive or manic disorders
could account for the concordance of the results with
similar studies major depression. The relatively high
representation of female patients might suggest this
pos-sibility. That the patients presented with schizophrenia is
supported by the average episode duration of
approxi-mately 3.5 years (Table 1) and relatively low baseline
scores on the BPRS items of depressive mood,
grandios-ity, and excitement, coupled with high scores on
tradi-tional BPRS positive symptoms of psychosis (data not
shown).
It is well documented that ECT produces cognitive
impairment (American Psychiatric Association Task
Force, in press; Sackeim 1992). There were no differences
among the treatment groups or among ECT responders
and nonresponders in change in MMSE scores. Although
this suggests that the dosage conditions did not differ in
changes in global cognitive status, the MMSE is a measure
insensitive to the characteristic effects of ECT on
antero-grade and retroantero-grade memory (Sobin et al 1995). In major
depression, there is evidence that higher stimulus intensity
and longer courses of ECT treatment can result in more
severe transient cognitive side effects (Sackeim 1992;
Sackeim et al 1993). It will be important in future research
to use more sensitive neuropsychologic measures and
determine in patients with schizophrenia whether the
enhancement in speed of response due to higher stimulus
intensity offsets a detrimental effect of cognitive function.
In summary, this preliminary study indicates that in
patients with schizophrenia, electrical dosage impacts on
the speed of clinical response to bilateral ECT. Both the
groups treated at twice and four times seizure threshold
had more rapid improvement than did the low-dosage
group, who were treated just above seizure threshold.
Future research should examine the effects of high-dosage
bilateral ECT on cognitive functions, which should be
assessed
with
a
comprehensive
neuropsychological
battery.
Supported in part by Grant No. BRG 3980009 from the Thailand Research Fund (WC) and Grant No. R37 MH35636 from the National Institute of Mental Health (HAS). The authors thank Wiwat Yatapoot-anon, M.D., for technical support and the nursing staff of the Srithunya mental hospital.
References
American Psychiatric Association (1994): Diagnostic and
Sta-tistical Manual of Mental Disorders, 4th ed. Washington, DC:
American Psychiatric Press.
American Psychiatric Association Task Force (in press): The
Practice of ECT: Recommendations for Treatment, Training
and Privileging, 2nd ed. Washington, DC: American
Psychi-atric Press.
Baudis P (1992): Elektrokonvulsini lecba v Ceske republice v letech 1981–1989. Cesk Psychiatr 88:41– 47.
Chanpattana W (1997): Continuation ECT in schizophrenia: A pilot study. J Med Assoc Thai 80:311–318.
Chanpattana W (1998): Maintenance ECT in schizophrenia: A pilot study. J Med Assoc Thai 81:17–24.
Chanpattana W (1999): The use of the stabilization period in electroconvulsive therapy research in schizophrenia: I. A pilot study. J Med Assoc Thai 82:1193–1199.
Chanpattana W, Chakrabhand S, Kongsakon R, Techakasem P, Buppanharun W (1999a): The short-term effect of combined ECT and neuroleptic therapy in treatment-resistant schizo-phrenia. J ECT 15:129 –139.
Chanpattana W, Chakrabhand S, Sackeim HA, Kitaroonchai W, Kongsakon R, Techakasem P, et al (1999b): Continuation ECT in treatment-resistant schizophrenia: A controlled study.
J ECT 15:178 –192.
Coffey CE, Lucke J, Weiner RD, Krystal AD, Aque M (1995): Seizure threshold in electroconvulsive therapy: I. Initial seizure threshold. Biol Psychiatry 37:713–720.
Cronholm B, Ottosson JO (1963): Ultrabrief stimulus technique in electroconvulsive therapy, II. Comparative studies of therapeutic effects and memory disturbance in treatment of endogenous depression with the Elther ES electroshock ap-paratus and Siemens Konvulsator. J Nerv Ment Dis 137:268 – 276.
Fink M, Sackeim HA (1996): Convulsive therapy in schizophre-nia? Schizophr Bull 22:27–39.
Kongsakon R, Vanichtanom R (1994): Assessment of the differ-ences in MMSE between neurological, psychiatric patients and normal population. J Rajwithi Hosp 5:99 –106.
Krueger RB, Sackeim HA (1995): Electroconvulsive therapy and schizophrenia. In: Hirsch S, Weinberger D, editors.
Schizophrenia. Oxford, UK: Oxford University Press,
503–545.
Krystal AD, Coffey CE, Weiner RD, Holsinger T (1998): Changes in seizure threshold over the course of electrocon-vulsive therapy affect therapeutic response and are detected by ictal EEG ratings. J Neuropsychiatry Clin Neurosci 10:178 –186.
Laska EM, Siegel C (1995): Characterizing onset in psychophar-macological trials. Psychopharmacol Bull 31:29 –35. McCall WV, Reboussin DM, Weiner RD, Sackeim HA (in
press): Titrated, moderately suprathreshold versus fixed, high dose RUL ECT: Acute antidepressant and cognitive effects.
Arch Gen Psychiatry.
Nobler MS, Sackeim HA, Moeller JR, Prudic J, Petkova E, Waternaux C (1997): Quantifying the speed of symptomatic improvement with electroconvulsive therapy: Comparison of alternative statistical methods. Convuls Ther 13:208 –221. Ottosson JO (1960): Experimental studies of the mode of action
of electroconvulsive therapy. Acta Psychiatr Scand Suppl 145:1–141.
Overall JF, Gorham DR (1962): The Brief Psychiatric Rating Scale. Psychol Rep 10:799 – 812.
comparison of the therapeutic effects of low and high energy electroconvulsive therapies. Br J Psychiatry 141:357–366. Sackeim HA (1992): The cognitive effects of electroconvulsive
therapy. In: Moos WH, Gamzu ER, Thal LJ, editors.
Cogni-tive Disorders: Pathophysiology and Treatment. New York:
Marcel Dekker, 183–228.
Sackeim HA (1999): The anticonvulsant hypothesis of the mechanisms of action of ECT: Current status. J ECT 15:5–26. Sackeim HA, Decina P, Kanzler M, Kerr B, Malitz S (1987a): Effects of electrode placement on the efficacy of titrated, low-dose ECT. Am J Psychiatry 144:1449 –1455.
Sackeim HA, Decina P, Portnoy S, Neeley P, Malitz S (1987b): Studies on dosage, seizure threshold, and seizure duration in ECT. Biol Psychiatry 22:249 –268.
Sackeim HA, Decina P, Prohovnik I, Malitz S (1987c): Seizure threshold in electroconvulsive therapy. Arch Gen Psychiatry 44:355–360.
Sackeim HA, Devanand DP, Prudic J (1991): Stimulus intensity, seizure threshold, and seizure duration: Impact on the efficacy
and safety of electroconvulsive therapy. Psychiatr Clin North
Am 14:803– 843.
Sackeim HA, Prudic J, Devanand DP, Kiersky JE, Fitzsimons L, Moody BJ, et al (1993): Effects of stimulus intensity and electrode placement on the efficacy and cognitive effects of electroconvulsive therapy. N Engl J Med 328: 839 – 846.
Sackeim HA, Prudic J, Devanand DP, Nobler MS, Lisanby SH, Peyser S, et al (in press): A prospective, randomized, double-blind comparison of bilateral and right unilateral ECT at different stimulus intensities. Arch Gen Psychiatry. Shukla GD (1981): Electroconvulsive therapy in a rural teaching
general hospital in India. Br J Psychiatry 139:569 –571. Sobin C, Sackeim HA, Prudic J, Devanand DP, Moody BJ,
McElhiney MC (1995): Predictors of retrograde amnesia following ECT. Am J Psychiatry 152:995–1001.