Conference Proceedings
December 2013
Hong Kong
HKI CEAS
EECS
International Conference on Electrical Engineering and Computer Sciences
ISBN:978-986-88450-3-9
Contents
General Information for Participants ... 6
Conference Organization ... 8
Conference Schedule ... 12
Conference Venue Information ... 16
Oral Session ... 18
Electrical and Electronics Engineering I/ Photonics and Optoelectronics ...18
EECS-730 ...20
EECS-747 ...27
EECS-711 ...34
EECS-716 ...38
EECS-700 ...44
EECS-818 ...46
Fundamental and Applied Sciences I...47
HKICEAS-622...49
HKICEAS-877...55
HKICEAS-884...61
HKICEAS-632...68
HKICEAS-783...72
HKICEAS-909...77
HKICEAS-819...83
Natural Science Keynote Speech ...89
Civil Engineering I...91
HKICEAS-857... 100
Computer Science and Information Engineering I ... 138
HKICEAS-743... 139
HKICEAS-903... 147
EECS-721 ... 153
EECS-722 ... 160
Environmental Science I... 169
HKICEAS-788... 171
Biomedical Engineering I/ Civil Engineering ... 218
HKICEAS-657... 220
Biomedical Engineering II ... 280
HKICEAS-771... 282
Material Science and Engineering I ... 333
HKICEAS-908... 335
Computer Science and Information Engineering II ... 393
EECS-740 ... 395
Environmental Science II/ Civil Engineering II ... 446
HKICEAS-800... 486
Chemical Engineering ... 492
HKICEAS-770... 494
Computer Science and Information Engineering III/ Wireless or Mobile Communication & Computing ... 524
EECS-748 ... 526
Material Science and Engineering II ... 565
HKICEAS-569... 567
HKICEAS-851... 575
HKICEAS-852... 581
HKICEAS-853... 589
HKICEAS-854... 596
Electrical and Electronic Engineering II ... 602
HKICEAS-718... 604
Fundamental and Applied Sciences II ... 648
HKICEAS-821... 649
HKICEAS-665... 658
HKICEAS-906... 665
HKICEAS-764... 670
HKICEAS-904... 688
Mechanical Engineering ... 694
HKICEAS-580... 696
Material Science and Engineering III... 733
HKICEAS-860... 735
Material Science and Engineering ... 749
HKICEAS-717... 752
HKICEAS-556... 759
HKICEAS-861... 763
HKICEAS-733... 769
HKICEAS-680... 781
Electrical and Electronic Engineering... 823
HKICEAS-876... 824
HKICEAS-939... 828
Biomedical Engineering I/ Biomedical Electronics and Bioinformatics ... 837
HKICEAS-650... 842
Civil Engineering ... 896
HKICEAS-735... 898
HKICEAS-542... 906
HKICEAS-672... 919
HKICEAS-667... 920
HKICEAS-889... 927
Mechanical Engineering ... 930
HKICEAS-744... 931
HKICEAS-652... 938
HKICEAS-782... 946
Chemical Engineering ... 951
HKICEAS-663... 952
HKICEAS-559... 956
HKICEAS-552... 972
Wireless or Mobile Communication & Computing/ Nano-Electro-Mechanical System, Networking ... 973
EECS-670 ... 974
EECS-735 ... 981
Fundamental and Applied Sciences ... 982
HKICEAS-836... 984
HKICEAS-837... 985
HKICEAS-607... 986
HKICEAS-891... 987
HKICEAS-893... 989
Electrical and Electronic Engineering/Photonics and Optoelectronics ... 990
EECS-736 ... 993
EECS-701 ... 999
EECS-713 ... 1007
EECS-718 ... 1015
EECS-743 ... 1021
EECS-761 ... 1028
EECS-809 ... 1034
EECS-799 ... 1043
EECS-717 ... 1050
Computer Science and Information Engineering/ Environmental Science .... 1051
HKICEAS-673... 1053
HKICEAS-773... 1058
HKICEAS-711... 1065
EECS-712 ... 1066
EECS-731 ... 1071
EECS-738 ... 1078
EECS-746 ... 1085
HKICEAS-887... 1086
General Information for Participants
Information and Registration
The Registration and Information Desk will be situated in the below time and location at Regal
Airport Hotel:
2F, December 19, 2013 (15:00-17:00)
B1, December 20, 2013 (08:15-16:00)
B1, December 21, 2013 (08:15-16:00)
Parallel Sessions
Parallel Sessions will run on December 20 and 21. Sessions are usually 90 minutes in length.
Presentations and Equipment
All presentation rooms are equipped with a screen, an LCD projector, and a laptop computer
installed with PowerPoint software. You will be able to insert your USB flash drive into the
computer and double click on your presentation to open it in PowerPoint. We recommend that
you bring two copies of your presentation in case of one fails. You may also link your own
laptop computer to the projector cable, however if you use your own Mac please ensure you
have the requisite connector.
A Polite Request to All Participants
Participants are requested to arrive in a timely fashion for all addresses, whether to their own,
or to those of other presenters. Presenters are reminded that the time slots should be divided
fairly and equally between the number of presentations, and that they should not overrun. The
session chair is asked to assume this timekeeping role.
6
Poster Sessions & Poster Requirements
Materials Provided by the Conference Organizer:
1. X-frame display & Base Fabric Canvases (60cm×160cm)
2. Adhesive Tapes or Clamps
Materials Prepared by the Presenters:
1. Home-made Poster(s)
Requirement for the Posters:
1. Material: not limited, can be posted on the canvases
A 60cm*160cm Poster Illustrates the research findings.
1.Wider than 60cm (left)
2.Copy of PowerPoint Slides in A4 papers (right)
Conference Organization
HKICEAS Committee Board
Linda Osman-Schlegel Deakin University (Geelong Waterfront Campus)
Khalid M. Mosalam University of California, Berkeley
Chueerat Jaruskulchai Kasetsart University
M. Cheralathan SRM University
J N Bandyopadhyay Indian Institute of Technology Kharagpur
S. Dhar University of Calcutta
Poongothai Shankar Annamalai University
Amit Agrawal Indian Institute of Technology Bombay
Cheng Li The Hong Kong Polytechnic University
T.M. Indra Mahlia University of Malaya
Kunal Ghosh Indian Institute of Technology Kanpur
Narayanan Kulathuramaiyer University of Sarawak Malaysia
Arup K. Sarma Indian Institute of Technology Guwahati
Suresh K Bhargava School of Applied Sciences
Banerji P Indian Institute of Technology Kharagpur
P.K. Ghosh Indian Institute of Technology Poorkee
E George Dharma Prakash Raj Bharathidasan University
R.P.Bhatnagar Birla Institute of Technology
V. Vijayagopal Annamalai University
Amit Awekar Indian Institute of Technology Guwahati
Gustavo Carneiro University of Adelaide
Pui-In Mak University of Macau
E, Rathakrishnan Indian Institute of Technology Kanpur
Bassim H. Hameed University of Science Malaysia
Sudhirkumar Barai Indian Institute of Technology Kharagpur
S. N. Sarkar Calcutta University
Samit Bhattacharya Indian Institute of Technology Kanpur
A. P. Shashikala Birla Institute of Technology
RM. Senthamarai Annamalai University
Arnab Bhattacharya Indian Institute of Technology Kanpur
Zbigniew Michalewicz University of Adelaide
B. Bhattacharya Indian Institute of Technology Kharagpur
Amin Heidarpour Monash University
Faizal Mustapha Universiti Putra Malaysia
Susanta Banerjee Indian Institute of Technology Kharagpur
M.V.L.R Anjaneyalu Birla Institute of Technology
P. Balasubramanian Universiti Teknologi PETRONAS
Hui Tong Chua University of Western Australia
Andrew Whyte Curtin University
Johnson Agbinya La Trobe University
Yuen Ka Veng University of Macau
ShahNor Basri Universiti Putra Malaysia
T. V. Gopal SRM University
Surendra Kumar Indian Institute of Technology Poorkee
K. K. Saju Cochin University of Science and Technology
HKICEAS-906
Percentage of Free Fatty Acid and Characteristics of Biodiesel Fuel in Waste
Coconut Oil (WCO)
N M. Suanitia, I W. B. Adnyanab
a
Chemistry Department, Faculty of Mathematic and Natural Sciences, Udayana University, Bali
b
Mechanical Engineering, Faculty of Engineering, Udayana University, Bali E-mail: suanitisr@yahoo.com
Abstract
Various commercial product in tropical regions has a wide impact in many field. Coconut as one of the export scale product often processed becoming coconut oil to fulfill daily life matter. Coconut oil usually be used as a media to fry foodsttufs such as chicken and residual oil during frying process named Waste Coconut Oil (WCO). Over the past year WCO serve as a waste product and known less advantage. Nowdays, WCO be treated become biodiesel as an alternative fuel of diesel engine to reduce utilization fosil fuels. The aims of this study were to analyze some characteristics as biodiesel from WCO which undergo esterification or transesterification process. Esterification process obtained a specific purpose to convert fatty acid in coconut oil that is consisting with lauric
acid. Esterification process to produce methyl ester from WCO using catalyst H2SO4 and Toluene
sulfonate where upon the yield of esterification process subsequently analyzed contents of free fatty acid (FFA) from acid value. Synthesis of biodiesel from fatty acid methyl ester (FAME) from WCO ester ongoing process of frying chicken during chicken 1, 3, 5, and 7 hours. Percentage (%) of FFA
has a variation under the influence both of catalyst H2SO4 and Toluene sulfonate and lengthy of
frying at oil. % FFA with H2SO4 catalyst obtained each 1.01 to 2.02 % and with toluene-sulfanote
catalyst produced each 0.68 to 1.02 %. Characteristics of biodiesel fuel analyzed of density,
viscosity, and flash point value were 1.91 to 2.03, 6.43 to 17.25, 38 to 46 respectively with H2SO4
catalyst and 1.94 to 2.00, 10.73 to 12.97, 35 to 60 respectively with toluene-sulfonate catalyst. WCO obtained after frying during 5 and 7 hours eligible as a Biodiesel because as avaluable result.
Determination of flash point has been done under temperature 50oC which and the oil should not
use for frying again some values of density and viscosity were getting high. Keyword: Fatty acid, Biodiesel, WCO
1.Introduction
Coconut oil especially used in frying can undergo an oxidation process because of the high temperature, that condition can reduce the quality of oil such as physic-chemical properties, odor and resulting rancidity of coconut oil which reported as one of the toxic substance that can promote a serious malignant disease. Coconut oil heated for every different duration of time at present can produce a renewable product namely biodiesel and the process called esterification where the fatty acid from coconut oil reacted with methanol to produce ester with the side product like glycerol as known still have a high economical value.
The development of human civilization along with advances in technology demand a large scale of energy sources from time to time. Enhancement needs of energy sources caused the depletion of petroleum energy sources. According to the Department of Energy and mineral resources, reserve petroleum in Indonesia estimated as a fosil fuel which can’t refurbished with the result that an average production rate around 500 million barrels per year will run out in 18 years due to increased fuel consumption.
One of the fuel energy sources that are not derived from petroleum is coconut oil, more than that used cooking oil can be used as a biodiesel. WCO can be processed into biodiesel that environmentally friendly form and usually obtained by esterification and transesterification process.
Esterification method according to IUPAC (1987) often used these two methods than the other, namely Christie and Laureles to produce methyl esters from Virgin Coconut Oil (VCO). This Process undergo some step consist with added n-hexane, vortex the oil, and added 1.5 mL NaOH (Pontoh and Makasoe, 2011). Esterification is a reaction between ester and methanol with an acid catalyst, whereas ester transesterification obtained in the esterification reaction which methanol is reacted again but using a base catalyst. This result known as esterification and transesterification of Fatty Acid Methyl Ester (FAME). In principle, all strong inorganic acids catalyst can be used such as sulfuric acid, phosphoric acid, hydrochloric acid or organic acids such as catalyst toluene- sulfonate (Hamelinck, et al., 2007).
Based on the background above, the series of conducted research has been done to analyze the characterization of biodiesel made from waste coconut oil (WCO) after frying chicken during 1, 3, 5, and 7 hours.
2.Materials and Method
Waste Coconut Oil after frying chicken 1, 3, 5, and 7 hours, n-hexane, 95-98% sulfuric acid (E.
Merck), followed by toluene sulfonic acid, NaOH, methanol p.a 99%, and anhydrous CaCl2.
Esterification: first of all methanol added to WCO, followed by hexane, and stirred to 5 minutes and
then added H2SO4 catalyst or toluene sulfonate catalyst, solution with a mixture of heated 1 hour.
Transesterification: the cold solution added Na methoxide and hexane, then heated at temperature
55oC and stirred for 2.5 hours which placed into a separating funnel. Process of separating begun
when the top layer of solution washed with distilled water until the washing water distillation clear.
Solution added CaCl2 anhydrous, filtered, collected a number of biodiesel and analyzed.
3.Results and Discussion
Percentages of free fatty acids (% FFA), density, viscosity, and flash point in Figure 1 described bellow with sulfuric acid catalyst and Figure 2 with toluene sulfonat catalyst. %FFA, in this research the maximum result obtained after frying chicken during 5 hours and still have high value after 7 hours of frying. However %FFA decreased after heating at frying chicken for 1 and 3 hours. Subsequently after heating at 5 and 7 hours the value of % FFA and acid number increased, it indicates that pure coconut oil needs to be heated to reduce free fatty acids content in the oil, but when the heating of the used oil did continuously until 7 hours there was a slight increased value of fatty acids. It can occur after process of frying chicken without skin obviously produce fatty acids and increased %FFA, moreover the diffferent result obtained from the frying process of other chicken part like skin, viscera, and so on. This study can be used as a reference in the field of health, because high fatty acid content is not good for health. Therefore we recommended utilization of cooking oil more than 5 hours after frying chicken, it must be used to be a fuel, and it can not be used for processing food furthermore but can be used to supply needs of motor vehicles.
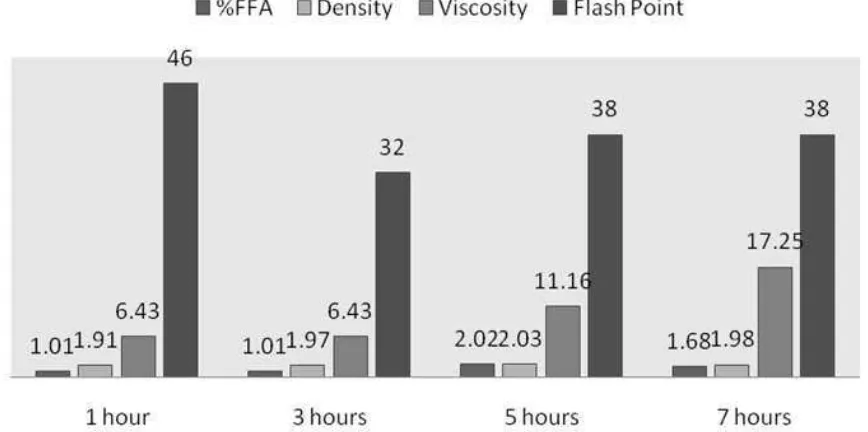
Figure 1: %FFA, density, viscosity, and flash point values of FAME with sulfuric acid catalyst
Measurements of density, viscosity, and flash point of FAME have been done using Ostwald method as shown in Figure 1 above. Density value ranged from 1.91 to 2.03 (5 hours) and then decrese to 1.98 (7 hours). The viscosity of FAME ranged from 6.43 to 17.25, and flash point ranged from 32 to 46 (1 hour). The European biodiesel quality standard (EN 14214) ranged from 0.86 to 0.90 and the Indonesian National Standard (SNI: 04-7182 -2006), ranged from 0.850 to 0.890. Density of FAME in this research is still higher than Indonesia standard and European, after all it needs to be optimized in the process manufacturing of oil mixture.
Figure 2: %FFA, density, viscosity, and flash point values of FAME with toluene-Sulfonate catalyst
Measurements of % FFA has been done were 0.68 to 1.01 as shown in Figure 2 above, value also accomplished with the same method, while value of density, viscosity, and flash point of FAME wth the density ranged around 1.96 to 2 (5 hours) and decreased until 1.97 (7 hours). The viscosity of FAME ranged from 12.97 to 17.06, and flash point ranged from 35 to 60 (3 hours). The European biodiesel quality standard (EN 14214) ranged from 0.86 to 0.90 and the Indonesian National Standard (SNI: 04-7182 -2006), ranged around 0.850 to 0.890. Density at FAME is still higher than the standard Indonesian and European, eventually it needs to be optimized in the process manufacturing of oil mixture. Viscosity FAME with toluene-sulfonate catalyst obtained high value compared with sulfuric acid, this suggests that sulfuric acid catalyst is still better.
% FFA obtainable around ≤ 2 according to the research Yuliani et al., There high fatty acid content
(> 2), represent the impact of catalyst alkaline in the transesterification because the fatty acid reacts with the catalyst to form soap through a saponification reaction. This resulted ineffective catalyst role because the catalyst reacts with the fatty acids that can degrade ester formed and separation process more difficult to be solved.
Density values according to the standards of Indonesian biodiesel (SNI) :04-7182-2006 (Supardan,
et al., 2011) indicating the value of used cooking oil viscosity at 5 and 7 hours with sulfuric acid and toluene sulfonate catalyst however still high so it needs to be reviewed for the optimum conditions of fat in the process of mixing oil with alcohol while flash point value has been reached fulfill as biodiesel.
4.Conclusion
Percentages of FFA (% FFA) were obtained from 1.01 to 2.02 with H2SO4 catalyst, and 0.68 to 1.02
with toluene-sulfonate catalyst. Characteristics determination of biodiesel such as density, viscosity,
and flash point were 1.91 to 2.03, 6.43 to 17.25, 38 to 46 respectively with H2SO4 catalyst and 1.94
to 2.00, 10.73 to 12.97, 35 to 60 respectively with toluene-sulfonate catalyst. WCO after frying 5 and 7 hours with sulfuric acid catalyst because a developed good result as Fuel. Flash point under
50oC has been done and analyzed for further processing like frying it should be discontinued by
reason of some values of density and viscosity were getting high.
Acknowledgement
Authors express their gratitude and appreciation to the Directorate General of Higher Education (DIKTI) for the opportunity to conduct the process of research in a laboratory scale gradually. Thanks as well given to LPPM Udayana University and all those who helped this study achieved success.
References
[1] Hamelinck, C., Schober, S., Mittelbach, M., Verolet, J., dehue, B., Bioscopes, Fatty acid
Ethyl Ester. Ecofys Uni Craz, 2007, 3-5
[2] Pontoh, J., makasoe, L., Comparison of several methods of making the analysis of methyl
esters of fatty acids of Virgin Coconut Oil ( VCO ) . Scientific journal Science , Chemistry courses F.MIPA Univ . Sam Ratulangi Manado, 2011. 11, 241-247
[3] Suirta, IW. Preparation of Biodiesel from Palm Oil Jelantah . Journal of Chemistry
F.MIPA Unud , 2009, 3 , 1 , 1-6 . Bukit Jimbaran . Badung . Bali . ISSN 1907-9850
[4] Supardan, M.D. Use of Ultrasonic for Used Cooking Oil Transesterification .
Journal of Chemical and Environmental Engineering , 2011, 8 , 1 . 11-16 . Department of Chemical Engineering , FT . Syah Kuala University . Banda Aceh . ISSN 1412-5064 .
[5] Yuliani, F., Primasari, M., Rachmaniah, O., Rachimoellah, M., Effect of acid catalyst (
H2SO4) and reaction temperature on the esterification reaction Seed Oil rubber (Hevea
brasiliensis) into Biodiesel. Biomass and energy laboratory ITS Surabaya , 2003