THE FLORISTIC STUDY OF
HERBACEOUS GRASSES IN SULAWESI
MARTHEN THEOGIVES LASUT
DEPARTMENT OF BIOLOGY
THE GRADUATE SCHOOL
BOGOR AGRICULTURAL UNIVERSITY
BOGOR
STATEMENT OF RESEARCH ORIGINALITY AND
INFORMATION SOURCE
This is to verify that my dissertation entitled: The Floristic Study of Herbaceous Grasses in Sulawesi is my own work and never been submitted to any institution before. All of the incorporated data and information here are valid and stated clearly in the text, and listed in the references.
Bogor, November 2008
ABSTRACT
MARTHEN THEOGIVES LASUT. The Floristic Study of Herbaceous Grasses in Sulawesi. (Under direction of Dr. Sri S. Tjitrosoedirdjo, Prof. Dr. Edi Guhardja, Prof. Dr. Mien A. Rifai, and Dr. Jan F. Veldkamp).
Grasses is the one of the largest plant family in the world, it consist of about 657 genera comprising of 9379 species occur in the tropic region to the sub-tropic region. In Malesia there are about 150 genera; Papua New Guinea 130 genera; Malaysia 80 genera; Java 116 genera. Taxonomically, herbaceous grasses are very large, difficult and poorly known in Sulawesi. Therefore the floristic study of herbaceous in Sulawesi was conducted by using explorative and descriptive methods, to which the data was taken from morphological point of view. Based on gross morphological study on 764 number specimens, it was concluded that Sulawesi herbaceous grasses comprises of 17 tribes, 93 genera, 212 species, and four varieties. There are one endemic variety and seven endemic species in this area. One species is described as a new for science and one other species is proposed to be a new species. Twenty five new records are reported also in Sulawesi.
ABSTRAK
MARTHEN THEOGIVES LASUT. Kajian floristik rumput herba Sulawesi. (Dibimbing oleh Dr. Sri S. Tjitrosoedirdjo, Prof. Dr. Edi Guhardja, Prof. Dr. Mien A. Rifai, dan Dr. Jan F. Veldkamp).
Rumput herba adalah salah satu keluarga tumbuhan terbesar di dunia, anggotanya sekitar 657 marga dengan kurang lebih 9379 jenis yang tersebar di daerah tropis dan sub tropis. Kawasan Malesia memiliki 150 marga, Papua New Guinea 130 marga, Malaysia 80 marga, Jawa 116 marga. Secara taksonomi, rerumputan sangat besar, sulit dan kurang dipahami di Sulawesi. Karena itu maka kajian flora rumput Sulawesi dilakukan. Kajian ini menggunakan metode eksplorasi dan deskripsi dengan karakter morfologi sebagai cara identifikasinya. Berdasarkan pengamatan yang dilakukan pada 764 nomor koleksi spesimen rumput herba Sulawesi, ditemukan 17 puak, 93 marga, 212 jenis dan 4 varietas. Terdapat satu varitas endemik dan tujuh jenis endemic. Satu jenis rumput telah di pertelakan sebagai jenis baru dan satu lagi diusulkan sebagai jenis baru. Ditemukan pula dua puluh lima catatan baru rumput Sulawesi dalam kajian ini.
SUMMARY
MARTHEN THEOGIVES LASUT. The Floristic Study of Herbaceous Grasses in Sulawesi. (Under the supervision of Dr. Sri S. Tjitrosoedirdjo, Prof. Dr. Edi Guhardja, Prof. Dr. Mien A. Rifai, and Dr. Jan F. Veldkamp).
The grass family is one of the largest flowering plants next only to the Compositae, Orchidaceae, and Leguminosae. However, in terms of individual numbers, the grasses are most abundant and most widely distributed seed families. There are few habitats in which grasses do not occur naturally, and their range is only restricted by alpine or arctic cold on one hand and the intense shade of some tropical forests and drought of desert areas on the other. It is not surprising that the grass family has diversified into a rich array of variously modified and adapted forms. Grasses are so different from most other plants that special of terms have been devised to their vegetative and generative structures. The vegetative parts of the grass plant are superficially rather uniform. Moreover, within certain limits, the most obvious differences of size, colour, and hairiness are strongly influenced by environmental factors. As a result the use of vegetative characters for the identification of grasses is rather limited
The flowering parts (inflorescence) of the grass plant are a composite structure comprising various patterns of aggregation of the basic floral unit, the spikelet. Most commonly the inflorescence is a panicle of more or less open-branched structure. Less commonly the inflorescence is a spike (a single axis upon along which are borne sessile or stalkless spikelets). However, some panicles have their branches so reduced that they may appear to be spikes. Racemes (a single axis upon which are borne single pedicelled or stalked spikelets) are less common. However, a multitude of combinations of these various forms exists.
For Malesian area so far, no comprehensive floristic account is available. Partial floras have been published for Java (1968) by Backer and Bakhuizen, Papua New Guinea (1969) by Henty, and Malay Peninsula (1971) by Gilliland. Accordingly an attempt is made to undertaken floristic study of herbaceous grasses in Sulawesi.
Based on the notes on specimen kept in Herbarium Bogoriense, exploration of grasses in Sulawesi was began by the work of Koorders in 1895. The first grass collection in Sulawesi was made by him from Singkil – near Buha Manado, at 2 January 1895. After that, it followed by many other explorers and collectors from several areas.
distribution, along with pertinent synonymy used in others floras, and references to the taxonomy of the included taxa. All species of Sulawesi herbaceous grasses found in the wild – not cultivated are described and provides a source of information of those taxa.
The results of this study indicate that, there are 17 tribe with 93 genera from 212 species and 4 varieties of herbaceous grasses existing in Sulawesi island. The tribes are Andropogoneae, Aristideae, Arundineae, Arundinelleae, Aveneae, Brachypodieae,
Centotheceae, Cynodonteae, Ehrharteae, Eragrostideae, Eriachneae, Isachneae,
Oryzeae, Paniceae, Phareae, Poeae, and Thysanolaeneae.
Based on the size of taxa recognized in Sulawesi, the Andropogoneae is the largest one with 35% of genera, followed by the Paniceae 25%, Cynodonteae 8%,
Eragrostideae 6%, Aveneae and Poeae 3%, Arundineae, Arundinelleae, Centotheca,
Oryzeae, and Phareae 2%, the others tribes with 1%.
South Sulawesi exhibit the highest degree of endemic taxa if compared with the other parts of Sulawesi, apparently due to the considerable areas of high mountains. There are one endemic variety and five endemic species in this area –
Anthoxanthum horsfieldii (Kunth ex Bennet) Mez. var. celebicum Y. Schouten;
Hyparrhenia pratermissa Veldk., Ischaemum celebicum P. Jansen, Ischaemum veldkampii Lasut, Poa celebica Ohwi ex Veldk., and Rytidosperma bonthainicum (P. Jansen) Veldk. All endemic taxa above concentrated in Latimojong ridge (Mts. Rantemario) and Lompobatang mountain. North Sulawesi have only one endemic species – Eragrostis dyskritos Lasut, which is found in Mounts Soputan. This species is a new for science (in preparation to publish). Central Sulawesi also have one endemic species – Cymbopogon minutiflorus S. Dransf., found in Donggala area on the slopes of limestone hills.
Copyright © 2009, Bogor Agricultural University
Copyright are protected by law
1. It is prohibited to cite all or part of this dissertation without referring to and mentioning the source.
a. Citation only permitted for the sake of education, research, scientific writing, report writing, critical writing or reviewing scientific problems. b. Citation does not inflict the name and honor of Bogor Agricultural
University.
THE FLORISTIC STUDY OF
HERBACEOUS GRASSES IN SULAWESI
MARTHEN THEOGIVES LASUT
A dissertation submitted to fulfill one of the requirements for the Doctorate Degree at the Study Program of Biology,
Graduate School, Bogor Agricultural University
DEPARTMENT OF BIOLOGY THE GRADUATE SCHOOL
BOGOR AGRICULTURAL UNIVERSITY BOGOR
EXTERNAL EXAMINERS
Examination I : Prof. Dr. Elizabeth A. Widjaja
Herbarium Bogoriense, Botany Division, Research Centre for Biologi, Indonesian Institute of Science Bogor, Indonesia.
Examination II : Prof. Dr. Johanis P. Mogea
Herbarium Bogoriense, Botany Division, Research Centre for Biologi, Indonesian Institute of Science Bogor, Indonesia.
Dr. Nunik Sri Ariyanti.
Dissertation title : The Floristic Study of Herbaceous Grasses in Sulawesi Name of student : Marthen Theogives Lasut
Number of Student : G361030061 Study Program : Biology
Approved by
Advisory Committee
Dr. Sri S. Tjitrosoedirdjo, MSc. Prof. Dr. Ir. Edi Guhardja, MSc.
(Chairman) (Member)
Prof. Dr. Mien A. Rifai Dr. Jan F. Veldkamp
(Member) (Member)
Head of Study Program of Biology Dean of Graduate School
Dr. Ir. Dedy Duryadi Solihin, DEA Prof. Dr. Ir. Khairil Anwar Notodiputro, MS
FOREWORD
Research on the herbaceous grasses in Sulawesi under the title “The Floristic Study of Herbaceous Grasses in Sulawesi” was conducted from 2004-2008. Field study was carried out at 35 localities in Sulawesi. Specimen examination was conducted at Herbarium Bogoriense (BO), Herbarium Leiden (L), BIOTROP (BIOT) and Herbarium Wallaceana (WALL).
Some part of this dissertation have been published under the title: “A New Species of Ischaemum from Sulawesi”, that published in Reinwardtia 12(3): 257-259 (2006); and “The List to Genera of Herbaceous Grasses in Sulawesi”, published in
Eugenia 14(4): 387-396 (2008).
I am greatly appreciate to all of my advisor: Dr. Sri S. Tjitrosoedirdjo, Prof. Dr. Edi Guhardja, Prof. Dr. Mien A. Rifai, and Dr. Jan F. Veldkamp, for their advice, guidance, and encouragements throughout this study. I would like to sincerely thank to Prof. Dr. Elizabeth A. Widjaja and Prof. Dr. Johanis P. Mogea for their advice and technical support in the fields. I am grateful to Dr. M.M.J. van Balgooy for discussion and support when I was studying grasses specimens in Leiden. My special thank to Dr. Arie Keim, Drs. Tahan Udji, Dra. Himmah Rustiami, M.Sc., Ir. Emil S., M.Si for fruitful discussion and technical support in the field. I also thank to Mr. Onong Sabintoe, Mr. Uping Papa, Mr. Kurniawan Syam, Mr. Reinjel Manitik, Mr. Christian Sumampouw, and Mr. Bueng Kananga for collecting and helping in the field. I am also grateful to Mr. Sobari for line drawing the new specimen. I also thank to Mr. Alex Sumadijaya for typing the preliminary grasses list of Sulawesi.
My sincerely thanks to the following institutes for allowing me to used their facilities: 1) Herbarium Bogoriense (BO), Botany Division, Research Centre for Biology – Indonesian Institute of Science, 2) Nationaal Herbarium Leiden (L), Leiden Universiteit Branch, 3) Herbarium Wallaceana (Wall), Department of Agronomy, Faculty of Agriculture, UNSRAT Manado, and 4) Herbarium Biotrop (BIOT).
My special thanks to the BPPS scholarship from Directorate General of Higher Education (DIKTI), Department of National Education Republic Indonesia and Masarang Foundation (“Yayasan Masarang”) Tomohon, Indonesia for financing the field works.
CURRICULUM VITAE
Marthen Theogives Lasut was born on 28 March 1963 in Tomohon, North Sulawesi, as the second son from four children from the late Alexander N. Lasut and the late Carolina S. Mukuan. He was graduated from Sam Ratulangi University (UNSRAT) Manado in 1987. Since 1989 he was employed as a lecture at Faculty of Agriculture, Sam Ratulangi University (Unsrat) Manado. In 15July 1995 was married with Dra. Hilda V. Oroh. In 23 Desember 1996 he admitted the degree of Magister Science at Bogor Agricultutal University (IPB).
CONTENTS
Page
TABLE ………...XII LIST OF FIGURES ……….XIII
HISTORICAL INTRODUCTION ...1
The Grass Plant ………..1
The Explorations of Grasses in Sulawesi ………..……….. .4
MATERIAL AND METHODS ...6
Procedure .………..6
Locations ...6
Description of taxa ………...7
RESULTS AND DISCUSSIONS ...8
THE CLASSIFICATION OF SULAWESI GRASSES ………...8
ENDEMIC TAXA AND THEIR DISTRIBUTION IN SULAWESI ...10
NEW RECORDS OF HERBACEOUS GRASSES IN SULAWESI …………..11
TAXONOMY ...12
POACEAE Barnhart ...12
Key to the tribes ...13
Tribe 1. ANDROPOGONEAE Dumort ...14
Key to the genera ...15
Genus description and key to the species ...17
5. APLUDA L. ...17
5.1. Apluda mutica L. ...18
7. ARTHRAXON P. Beauv. ...19
Key to the species ...20
7.1. Arthraxon hispidus (Thunb.) Makino ...20
7.2. Arthraxon lancifolius (Trin.) Hochst. ...21
9. ASTHENOCHLOA Buse ...22
9.1. Asthenochloa tenera Buse ...22
11. BOTHRIOCHLOA Kuntze ...23
11.1. Bothriochloa bladhii (Retz.) S.T. Blake ...24
14. CAPILLIPEDIUM Stapf. ...25
Key to the species ...26
14.1. Capillipedium assimile (Steud.) A. Camus ...26
14.2. Capillipedium parviflorum (R. Br.) Stapf. ...27
17. CHIONACHNE R. Br. ...28
17.1. Chionachne punctata (R. Br.) Jannink ...28
Key to the species ...31
22.1. Cymbopogon citratus (DC.) Stapf. ...38
22.2. Cymbopogon tortilis (J. Presl.) A. Camus ...39
22.3. Cymbopogon flexuosus (Nees ex Steudel) J.F. Watson ...40
22.4. Cymbopogon minutiflorus S. Dransf. ...41
22.5. Cymbopogon procerus (R. Br.) Domin. ...42
27. DICHANTIUM Willemet ...44
Key to the Species ...44
27.1. Dichantium annulatum (Forssk.) Stapf. ...44
27.2. Dichantium aristatum (Poir.) C.E. Hubb. ...46
27.3. Dichantium caricosum (L.) A. Camus ...47
28. DIECTOMIS Kunth. ...48
45.1. Heteropogon contortus (L.) P. Beauv. ex Roem. & Schult. ...58
45.2. Heteropogon triticeus ( R. Br.) Stapf ex Craib ...59
47. HYPARRHENIA Andersson ex E. Fourn. ...60
Key to the species ...61
47.1. Hyparrhenia diplandra (Hack.) Stapf ...61
47.2. Hyparrhenia praetermissa Veldk. …...62
Key to the species ...63
49.1. Imperata conferta (J. Presl) Ohwi ...64
49.2. Imperata cylindrica (L.) Raeusch ...64
51. ISCHAEMUM L. ...66
Key to the species ...66
51.1. Ischaemum aristatum L. ...67
51.2. Ischaemum barbatum Retz. ...68
51.3. Ischaemum beccarii Hack. ...70
51.4. Ischaemum celebicum P. Jansen ...70
51.5. Ischaemum ciliare Retz. ...71
51.6. Ischaemum digitatum Brongn. ...73
51.7. Ischaemum muticum L. ...74
51.8. Ischaemum rugosum Salisb. ...75
51.9. Ischaemum timorense Kunth. ...76
51.10. Ischaemum velkampii Lasut ...77
58. MICROSTEGIUM Nees. ...79
72. POGONATHERUM P. Beauv. ...89
72.1. Pogonatherum paniceum (Lam.) Hack. ...89
79.2. Saccharum barberi Jeswiet ...96
79.3. Saccharum fastigiatum Ness ex Steud. ...97
79.4. Saccharum spontaneum L. ...98
81. SCHIZACHYRIUM Nees. ...99
Key to the species ...99
81.1. Schizachyrium brevifolium (Sw.) Nees. ....100
81.2. Schizachyrium fragile (R. Br.) A. Camus ..100
81.3. Schizachyrium pseudelalia (Hosok.) S.T. Blake ...102
82. SEHIMA Forssk. ...103
82.1. Sehima nervosum (Rottl.) Stapf. ...103
85. SORGHUM Moench. ...104
Key to the species ...105
85.1. Sorghum bicolor (L.) Moench. ...105
85.2. Sorghum laxiflorum F.M. Bailey ...106
85.3. Sorghum nitidum (Vahl.) C.E. Hubb. ...107
85.4. Sorghum propinquum (Kunth) Hitchc. ...108
89. THEMEDA Forssk. ...109
Key to the species ...109
89.1. Themeda arguens (L.) Hack. ...110
89.2. Themeda caudata (Nees) A. Camus ...111
89.3. Themeda gigantea (Cav.) Hack. ...112
89.4. Themeda intermedia (Hack.) Bor ...113
89.5. Themeda triandra Forssk. ...114
89.6. Themeda villosa (Poir.) A. Camus ...115
Tribe 2. ARISTIDEAE C.E. Hubb. ...116
Genus description and key to the species ...116
6. ARISTIDA L. ...116
Genus description and key to the species ...120
70. PHRAGMITES Trin. ...120
70.1. Phragmites vallatoria (L.) Veldk. …...120
78. RYTIDOSPERMA Steud. ...121
Key to the species ...122
78.1. Rytidosperma bonthainicum (P. Jansen) Veldk. ...122
Tribe 4. ARUNDINELLEAE Stapf. ...123
Key to the genera ...124
Genus description and key to the species ...124
8. ARUNDINELLA Raddi. ...124
Key to the species ...124
8.1. Arundinella goeringii Steud. ...125
8.2. Arundinella pumila (Hochst. ex A. Rich.) Steud. ...126
8.3. Arundinella setosa Trin. ...127
43. GARNOTIA Brongn. ...128
26. DEYEUXIA Clarion ex P. Beauv. ...133
18.1. Chloris barbata (L.) Sw. ...142
23. CYNODON L. C. Richard. ...144
Key to the species ...144
23.1.Cynodon dactylon (L.) Pers. ...145
23.2.Cynodon radiatus Roth. ...146
32. ECTROSIA R. Br. ...147
93.2.Zoysia pasifica (Goudswaard) M. Hotta& Kuroki ...155
34.1.Eleusine coracana (L.) Gaertn. ...161
34.2.Eleusine indica (L.) Gaertn. ...162
36. ERAGROSTIS N.M. Wolf. ...163
Key to the species ...164
36.1.Eragrostis amabilis (L.) Wight & Arn. ex Nees ...165
36.3.Eragrostis brownii (Kunth) Nees ex Steud
...167
36.4.Eragrostis cilianensis (Bellardi) Vignolo ex Janch. ...168
36.5.Eragrostis cumingii Steud. ...169
36.6.Eragrostis dyskrytos Lasut, sp. nov. ...170
36.7.Eragrostis japonica (Thunb.) Trin. ...172
36.8.Eragrostis lasioclada Merr. ...173
36.9.Eragrostis minor Host. ...174
36.10. Eragrostis pilosa (L.) P. Beauv. ...175
36.11. Eragrostis tenuifolia (A. Rich) Hochst. ex Steud. ...175
36.12. Eragrostis unioloides (Retz.) Nees ex Steud. ...176
36.13.Eragrostis warburgii Hack. ...177
54. LEPTOCHLOA P. Beauv. ...178
Key to the species ...178
54.1. Leptochloa chinensis (L.) Nees ...179
54.2. Leptochloa panicea (Retz.) Ohwi …...179
61. MUHLENBERGIA Schr. ...181
61.1. Muhlenbergia huegelii Trin... 181
87. SPOROBOLUS R. Br. ...182
Key to the species ...183
87.1. Sporobolus humilis J. Presl. ...183
87.2. Sporobolus indicus (L.) R. Br. ...183
87.3. Sporobolus tenuissimus (Mart. ex Schrank) Kuntze ...184
87.4. Sporobolus virginicus (L.) Kunth. ...185
50.7. Isachne minutula (Gaudich.) Kunth ...194
50.8. Isachne myosotis Nees ...195
50.9. Isachne pulchella Roth ex Roem. ...196
50.10. Isachne pangerangensis Zoll. & Mor. ..197
50.11. Isachne stricta Elmer ...198
50.12. Isachne surgens P. Jansen ...198
Tribe 13. ORYZEAE Dumort. ...199
24.1. Cyrtococcum accrescens (Trin.) Stapf. 211 24.2. Cyrtococcum oxyphyllum (Hochst.) Stapf. ...212
24.3. Cyrtococcum patens (L.) A. Camus ...213
29. DIGITARIA Haller. ...214
Key to the species ...215
29.1. Digitaria abludens (Roem. & Schult.) Veldk. …...216
29.2. Digitariabicornis (Lam.) Roem. & Schult ...217
29.3. Digitaria ciliaris (Retz.) Koeler ...218
29.4. Digitaria fuscescens (Presl.) Henrard ...219
29.6. Digitaria longiflora (Retz.) Pers. ...220
29.7. Digitaria milanjiana (Rendle) Stapf. ...221
29.8. Digitaria radicosa (J. Presl) Miq. ...222
29.9. Digitaria sanguinalis (L.) Scop. ...223
29.10. Digitaria setigera Roem. & Schult. ....224
29.10.1. Digitaria setigera Roth. var. callible- pharata (Henrard) Veldk. …...225
29.11. Digitaria ternata (A. Rich.) Stapf. ...226
29.12. Digitaria violascens Link. ...227
31. ECHINOCHLOA P. Beauv. ...228
Key to the species ...229
31.1. Echinochloa colona (L.) Link. ...229
31.2. Echinochloa crus-galli (L.) P. Beauv. ..230
31.3. Echinochloa stagnina (Retz.) P. Beauv 231 39. ERIOCHLOA Kunth. ...233
57.1. Melinis repens (Willd.) Zizka ...237
63. OPLISMENUS P. Beauv. ...238
Key to the species ...238
63.1. Oplismenus aemulus (R. Br.) Roem ex Schult. ...239
63.2. Oplismenus burmannii (Retz.) P. Beauv ...239
63.3. Oplismenuscompositus (L.) P. Beauv. 240 63.4. Oplismenus hirtellus (L.) P. Beauv. ....241
65. OTTOCHLOA Dandy ...242
65.1. Ottochloa nodosa (Kunth) Dandy ...242
66. PANICUM L. ...243
Key to the species ...244
66.1. Panicum auritum J. Presl. ex Nees ...244
66.2. Panicum bisulcatum Thunb. ...245
66.3. Panicum luzonense J. & C. Presl. ...246
66.4. Panicum notatum Retz. ...247
66.5. Panicum paludosum Roxb. ...247
66.6. Panicum repens L. ...248
66.7. Panicum sarmentosum Roxb. ...249
67. PASPALUM L. ...251 Key to the species ...252
67.1. Paspalum conjugatum P.J. Bergius ....252 67.2. Paspalum longifolium Roxb. ...253 67.3. Paspalum orbiculare G. Frost. ...254 67.4. Paspalum scrobiculatum L. ...254 67.5. Paspalum vaginatum Sw. ...255 68. PENNISETUM Rich. ...256 Key to the species ...257
68.1. Pennisetum ciliare (L.) Link. ...257 68.2. Pennisetum macrostachys (Brongn.)
Trin. ...258 68.3. Pennisetum purpureum Schum. ...259 75. PSEUDECHINOLAENA Stapf. ...260
75.1. Pseudechinolaena polystachya (Kunth) 80.1. Sacciolepis indica (L.) Chase ...263 80.2. Sacciolepis myosuroides (R. Br.) Chase
...264 84. SETARIA P. Beauv. ...264 Key to the species ...265 84.1. Setaria barbata (Lam.) Kunth. ...265 84.2. Setaria clivalis (Ridl.) Veldk. ...266 84.3. Setaria flavida (Retz.) Veldk. …...267 84.4. Setaria palmifolia (J. Koenig) Stapf. ..268 84.5. Setaria parviflora (Poir.) Kergeulen ...269 84.6. Setaria punctata (Burm.f.) Veldk. …. 270 84.7. Setaria verticillata (L.) Beauv. ...271 86. SPINIFEX L. ...272 Key to the species ...272
86.1. Spinifex littoreus (Burm.f.) Merr. ...272 86.2. Spinifex longifolius R. Br. ...273
92.1. Urochloa glumaris (Trin.) Veldk. ….. 277 92.2. Urochloa maxima Jacq. ...278 92.3. Urochloa mutica (Forssk.) T.-Q Nguyen
...279 92.4. Urochloa reptans (L.) Stapf. ...280 92.5. Urochloa subquadripara (Trin.) R.D.
Webster ...281
Tribe 15. PHAREAE Stapf. ...282 Key to the genera ...282 Genus description and key to the species ...282 53. LEPTASPIS R. Br. ...282 Key to the species ...283
53.1. Leptaspis banksii R. Br. ...283 53.2. Leptaspis zeylanica Nees ex Steud. ...284 83. SCROTOCHLOA Judz. ...285 83.1. Scrotochloa urceolata (Roxb.) Judz. 286
Tribe 16. POEAE R. Br. ...287 Key to the genera ...288 Genus description and the species ...288 13. BRIZA L. ...288 13.1. Briza minor L. …...288 42. FESTUCA L. ...289 42.1. Festuca leptopogon Stapf. ...290 71. POA L. ...290 Key to the species ...291 71.1. Poa celebica Ohwi ex Veldk. …...291 71.2. Poa epileuca (Stapf.) Hitch. ...292 71.3. Poa papuana Stapf. ...293
Tribe 17. THYSANOLAENEAE C.E. Hubb. ...294 Genus description and the species ...294 91. THYSANOLAENA Nees ...294 91.1. Thysanolaena latifolia (Roxb. ex
Hornem) Honda ………...295
TABLE
Page
1. The number of tribes, genera, species, and endemic species/variety
LIST OF FIGURES
page
HISTORICAL INTRODUCTION
The grass family is one of the largest flowering plants next only to the Compositae, Orchidaceae, and Leguminosae (Brummitt, 1992). However, in terms of individual numbers, the grasses are most abundant and most widely distributed seed families (Keng, 1978). There are few habitats in which grasses do not occur naturally, and their range is only restricted by alpine or arctic cold on one hand and the intense shade of some tropical forests and drought of desert areas on the other. It is not surprising that the grass family has diversified into a rich array of variously modified and adapted forms (Tothill and Hacker, 1973).
According to Brummitt (1992), the grass family in the world consists of 657 genera. Almost all of these genera are widespread from the tropic region to the
subtropic region. The Malesian Key Group (2004) have noted that in Malesia the family consists of about 150 grasses genera which occurred from sea level to alpine
zone. For Malesian area so far, no comprehensive floristic account is available. Partial floras have been published for Java (1968) by Backer and Bakhuizen, Papua New Guinea (1969) by Henty, and Malay Peninsula (1971) by Gilliland. Accordingly an attempt is made to undertaken floristic study of herbaceous grasses in Sulawesi.
Based on present study – the preparation of the herbaceous grasses flora for Sulawesi, it is concluded that the grasses of Sulawesi should be accommodated in 93 genera with 212 species and 4 varieties. Sulawesi comprises of about 182.870 km2 of land (Keßler, et al., 2002) and fewer botanical specimen – 23 specimens each 100 km2 have been placed in herbaria whereas over 200 specimens each 100 km2 are known from Java (Whitten, et al., 1987). A density of 100 specimens each 100 km2 would be represents an adequately-known flora.
The Grass Plant
environmental factors. As a result the use of vegetative characters for the identification of grasses is rather limited (Tothill and Hacker 1973). In ordinary flowering plants, the unit of floral structure is the flower – its sepals, petals, stamens and ovary. Such flowers are either borne singly, or more usually on special branches or branch-system known as inflorescences. Each flower is in the axil of leaf; and if the leaf is small, it is called a bract. Grasses also have inflorescences, but the units are not ordinary flowers. They are seen as little groups of small overlapping greenish, brownish or purplish bracts (sometimes hairy) from between which the stamens and stigmata emerge. Each group of bracts is separately stalked, and is called a spikelet, and they arranged very much in the same way as flowers are arranged in an ordinary inflorescence (Gilliland 1971).
The vegetative parts of a typical grass plant are comprising the following:
Culm: This is the elongated stalk which bears leaves and eventually at its apex, the inflorescense. It consists of a series of nodes and internodes, each node subtending a leaf alternately on either side of the culm. Nodes: In tufted grasses these only become apparent after the commencement of culm elongation when they are seen as small annular lumps or sometimes concavities, often pigmented and sometimes with a collar of hairs associated with them. Many grasses have creeping stems, called rhizomes if below ground or stolon if above, and on such stems nodes are apparent all the time.
The leaf is conveniently separated into three parts, the leaf sheath, ligule, and
Ligule: This structure lies at the point of junction of the sheath with the blade. It may be absent or present in various degrees as a collar of hairs or a membranous rim. Although it is of considerable value in separating broads groups of grass genera, it is rarely useful in distinguishing between different species of the same genus. The long membranous ligules is found predominantly in temperate grasses and is less often found in the predominantly tropical grasses. Leaf blade: This is generally a parallel-nerved, linear structure although in some very short-leaved species the blades may be relatively broad. The blade may be flat, folded, or rolled, hairy or hairless, rough or smooth, green or bluish, and of various shapes from narrow to broad in relation to length, parallel-sided or tapering, with points long and slender, abrupt or boat-shaped.
The flowering parts (inflorescence) of the grass plant are a composite structure comprising various patterns of aggregation of the basic floral unit, the spikelet. Most commonly the inflorescence is a panicle of more or less open-branched structure. Less commonly the inflorescence is a spike (a single axis upon along which are borne sessile or stalk-less spikelet). However, some panicles have their branches so reduced that they may appear to be spikes. Racemes (a single axis upon which are borne single pedicelled or stalked spikelets) are less common. However, a multitude of combinations of these various forms exists. The gross form of the inflorescence is popularly used in recognizing many different grass species or groups of species.
Spikelet: This is the basic floral unit which incorporates one or more flowers or florets.
usually with two nerves, though sometimes 1-nerved or nerveless, occasionally 3- or 4-nerved. These show much the same series of modifications as the glumes. When a spikelet basically contains more than one floret, the florets alternate along the axis in a continuous sequence above the glumes and may be relatively and variously modified in their sexual parts and also display a wide range of variation in the relative modifications of the lemmas and paleas. A fertile floret contains an ovary, which is best seen by the presence of stigmas, with or without male parts or anthers. A male floret is devoid of ovary and is reduced only to the male parts or anthers. In both cases the usual anther number is 3 but may occasionally be 1, 2, 4, or 6. A sterile floret is devoid of all sexual parts and both lemma and palea may be severely reduced and modified. Just as spikelets differ in form and function and are often characteristically associated in pairs, of dissimilar florets occur in the same spikelet, for example with a lower male or neuter floret and an upper female or bisexual floret, the florets being modified according to their differing functions. When this modification takes the form of extreme reduction or loss of paleas combined with the loss of one glume, care is needed in the identification of the various parts of the spikelet (Tothill and Hacker 1973).
The Explorations of Grasses in Sulawesi
Based on the notes on specimen kept in Herbarium Bogoriense, exploration of grasses in Sulawesi was began by the work of Koorders in 1895. After that, many other explorers and collectors from several areas. Notable among them are:
1. J. Elbert. Worked at South Sulawesi in 1909.
2. L. van Vurren & Noerkas. Worked at Pare-Pare in 1912. 3. W. M. van Leeuwen. Worked at “Seleier Island” in 1913. 4. V. M. A Beguin. Worked at Manado in 1920.
5. H.A.B. Bunnemeijer. Worked at Mts. Bonthain in 1921. 6. S. Leefmans. Worked at North Sulawesi in 1924.
9. O. Posthumus. Worked at Donggala, Palu, and Mt. Bonthain in 1930 10. A. Rant. Worked at Malino in 1931
11. L. J. Toxopeus. Worked at South Sulawesi in 1936
12. C. G. G. J. van Steenis. Worked at South Sulawesi in 1937. 13. P. J. Eyma. Worked at South Sulawesi in 1937.
14. S. Bloembergen. Worked at South Sulawesi in 1939. 15. A. H. G. Alston. Worked at Tomohon, Minahasa in 1954. 16. S. Soenarko. Worked at Maros in 1974.
17. W. Meijer. Worked at Palu in 1975.
18. E. A. Widjaja. Worked at Pare-pare in 1977.
19. M. M. J. van Balgooy and E. F. de Vogel. Worked at Central Celebes in 1979.
MATERIAL AND METHODS
This study was conducted from 2004 – 2008. The research materials are herbarium specimen, kept at the following herbaria: Herbarium Bogoriense (BO), Herbarium Leiden (L), Herbarium Wallaceana (WALL), Herbarium Biotrop (Biot), and collected fresh specimens from the field. The equipment used in collecting, preserving, and studying specimens are: scissors and scateurs, double flimsy (‘specimen folder’), press frames, press straps, corragurated sheets, paper or mesh
bags, cardboard supports, plastic bottles, polytene bags, adhesive tape and string, alcohol, hand lens, compass, altimeter, fine mounted needles, fine forceps, sharp
scalpel with fine point, small paint brush, and watch-glass or petridish (Forman and Bridson 1989).
Procedure
This research used explorative and descriptive method, according to the following steps: 1) collecting the specimens from the fields; 2) study of existing grasses already available in the herbaria; 3) identify and describe examined specimens;
4) defining the taxa accepted; 5) validation names and determined the status of taxon; 6) writing descriptions and illustrations; 7) and preparing the final draft flora.
Locations
The areas especially visited in: Tomohon and adjacent area (2004), including Mts. Mahawu (Alt. 1400 m), Mts. Masarang (Alt. 1300 m); Langoan and adjacent area, including Mts. Kawatak (Alt. 1450 m), Mts. Soputan (Alt. 1600 m); Tondano and
Tangkoko Nature Reserve; Modoinding and adjacent area, including Mts. Ambang (Alt. 1350 m).
Gorontalo (2005), Suwawa and adjacent areas, Pinogu enclave in Bogani Nani Wartabone National Park, including Mts. Gambuta (Alt. 1950 m); Paguyaman and adjacent area, including Nantu Adudu Nature Reserve.
Palu and adjacent areas (2005), including Mts. Nokilalaki (Alt. 2260 m); Toro and adjacent area; Doda valley and adjacent area, including Mts. Pohupaia (Alt. 1950 m).
Makassar and adjacent area (2005), including Bantimurung Bulusaraung National Park; Rantelemo and adjacent area, including Mts. Rantemario (Alt. 3465 m); Lembangbune and adjacent area, including Mts. Lompobatang (Alt. 2900 m).
Kendari and adjacent area (2005), including Rawa Aopa Watumohai National Park; Kolaka and adjacent area, including Mts. Mekongga (Alt. 2750 m).
Description of taxa
RESULTS AND DISCUSSIONS
The classification of Sulawesi grasses
The results of this study indicates that, there are 17 tribes with 93 genera from 212 species and 4 varieties of herbaceous grasses existing in Sulawesi island (Table 1). The tribes are Andropogoneae, Aristideae, Arundineae, Arundinelleae, Aveneae,
Brachypodieae, Centotheceae, Cynodonteae, Ehrharteae, Eragrostideae, Eriachneae,
Isachneae, Oryzeae, Paniceae, Phareae, Poeae, and Thysanolaeneae. As a comparison the Malay Peninsula and Papua New Guinea have 13 tribes and 19 tribes; China, Taiwan, and Australia have 28, 27 and 26 tribes respectively.
Based on the size of taxa recognized in Sulawesi, the Andropogoneae is the largest one with 35% of genera, followed by the Paniceae 25%, Cynodonteae 8%,
Eragrostideae 6%, Aveneae and Poeae 3%, Arundineae, Arundinelleae, Centotheca,
Oryzeae, and Phareae 2%, the others tribes with 1%.
In the world there are 87 genera of the Andropogoneae, distribution throughout the tropics into warm regions. The largest tribes in Sulawesi is Andropogoneae, which is consists of 33 genera and 70 species.
The Paniceae consists of 86 genera, throughout the tropics, extending into warm temperate regions. This is the second largest tribe in Sulawesi with 24 genera and 60 species.
The Eragrostideae consists of c. 26 genera; throughout the tropics. The fourth largest tribe in Sulawesi with 6 genera and 23 species.
The Aveneae and Poeae, each has 3 genera, with 2 species and 1 variety in the first, and the second with 5 species and 1 variety. The Aveneae consists of 60 genera, temperate and cold regions of the worlds, extending to the mountains in the tropic. The Poeae consists of 50 genera, temperate and cold regions in the worlds, also found in tropical mountain.
Table 1. Number of tribes, genera, species, and endemic species/variety of herbaceous grasses in Sulawesi. 1 Andropogoneae Apluda, Arthraxon, Asthenochloa,
Bothriochloa, Capillipedium,
8 Cynodonteae Chloris, Cynodon, Ectrosia, Enteropogon, Eustachys, Lepturus, Perotis, Zoysia
8 11
9 Ehrharteae Ehrharta 1 1
10 Eragrostideae Dactyloctenium, Eleusine, Eragrostis,
Leptochloa, Sporobolus, Muhlenbergia 6 23 1*
Notes: * Endemic species and ** Endemic variety
The Arundineae, Arundinelleae, Centotheceae, Oryzeae, and Phareae are the tribes with 2 genera and have respectively 3, 5, 2, 2, and 3 species. The Arundineae
consists of 25 genera, is found as a tribe with cosmopolitan distribution; the
Arundinelleae consists of 9 genera, found in the tropics, often favouring infertile soils; the Centotheceae consists of 10 genera, mainly in the tropics, in shading places; the
Oryzeae consists of 7 genera, found in tropical and warm temperate region throughout the world; the Phareae consists of 2 genera, found on both hemisphere.
The Aristideae, Brachypodieae, Ehrharteae, Eriachneae, Isachneae, and
Thysanolaeneae are the tribes with only have 1 genera in Sulawesi. The Isachneae
have 12 species, the Eriachneae and Thysanolaeneae just have 1 species in Sulawesi. In the world Aristideae have 2 genera, found in tropic and subtropic, often in dry climate, or on poor soils; Brachypodieae have 1 genus, found in Europe and Asia, African highlands, and Central America; Ehrharteae have 3 genus, found on warm temperate regions of the Old World, mainly in Australia and South Africa; Eriachneae
have 2 genera, mainly in Australia, a few species extending into South and Southeast Asia; Isachneae have 5 genera, found in the tropics, mainly in Asia; Thysanolaeneae
have 1 genus, found in tropical Asia, including China.
12 Isachneae Isachne 1 12
13 Oryzeae Leersia, Oryza 2 1 1
14 Paniceae Acroceras, Alloteropsis, Axonopus, Cenchrus, Cyrtococcum, Digitaria, Echinochloa, Eriochloa, Hymenachne, Ichnanthus, Melinis, Oplismenus, Ottochloa, Panicum, Paspalum, Pennisetum, Psedechinolaena, Pseudoraphis, Sacciolepis, Setaria, Spinifex, Stenotaphrum, Thuarea, Urochloa
24 67 1
15 Phareae Leptaspis, Scrotochloa 2 3
16 Poeae Briza, Festuca, Poa 3 5 1*
17 Thysanolaeneae Thysanolaena 1 1
Endemic taxa and their distribution in Sulawesi
The Poaceae is one of the largest and most widespread of the families of flowering plants. There are only a few places or habitats in which grasses do not occur naturally, and their range is only restricted by alpine or arctic cold on the one hand and the intense shade of some tropical forests and drought of desert areas on the other (Tothill and Hacker 1973). It is hence not surprising that the grass family has diversified into a rich array of variously modified and adapted forms.
Based on 212 species and 4 varieties occurring in Sulawesi, there are 7 endemic species and 1 endemic variety. These mainly montane endemics belonging to the temperate genera, supporting the opinion of Steenis (1933) that in the tropics such as Malesia these are restricted to the mountains and represent only small offshoots from
their generic centers. If compared with China, with 1795 species of grasses and 809 species are endemic, the number of endemic taxa in Sulawesi is very low, this is
because of many endemics are confined to the temperate regions of the mountains in China (FOC 2006).
South Sulawesi exhibits the highest degree of endemic species if compared with the other parts of Sulawesi, apparently due to the considerable areas of high mountains. There are one endemic variety and five endemic species in this area –
Anthoxanthum horsfieldii (Kunth ex Bennet) Mez. var. celebicum Y. Schouten;
Hyparrhenia pratermissa Veldk., Ischaemum celebicum P. Jansen, Ischaemum veldkampii Lasut, Poa celebica Ohwi ex Veldk., and Rytidosperma bonthainicum (P. Jansen) Veldk. All endemic taxa above concentrated in Latimojong ridge (Mts. Rantemario) and Lompobatang Mountain.
North Sulawesi has only one endemic species – Eragrostis dyskritos Lasut, which is found in Mounts Soputan. This species is a new for science (in preparation to publish).
Central Sulawesi also has one endemic species – Cymbopogon minutiflorus S. Dransf., found in Donggala area on the slopes of limestone hills.
This study found that about 11 percent of herbaceous grasses in Sulawesi are new records to Sulawesi. From 212 species and 4 varieties found in this island, there are 25 species as new records to this island.
Ichnanthus pallens (Sw.) Munro ex Benth., found in North and Central Sulawesi at secondary forest and opened area. Usually found at more than 1000 m above sea level. This species is found in South America, North America, India, China, Vietnam, Thailand, Burma, Taiwan, Malaysia, Philippine, Papua New Guinea, Papua, and Java.
The Isachne have 4 species as a new records. Those are Isachne brassii Hitch.,
Isachne confusa Ohwi, Isachne kunthiana (Wight & Arn. ex Steud) Nees ex Miq., and
Isachne pangerangensis Zoll. & Mor. This genus found in Asia tropical.
The other new records of herbaceous grasses in Sulawesi are as following:
Oplismenus aemulus (R. Br.) Roem. ex Schult., this species found in America, Australia, New Guinea, Taiwan,and Malesia; Muhlenbergia huegelii Trin., found in India, China, Taiwan, Nepal, Japan, and Malesia, usually found at high altitude;
Panicum luzonense J. & C. Presl., and Panicum curviflorum Hornem, found in Malesia, Indo-China, Sri Lanka, India, China, Africa, and United states of America;
Paspalum orbiculare G. Frost, found from India, Australia, Hawaian islands, Taiwan, America, Borneo, and Sumatera; Perotis indica (L.) Kuntze, and Perotis hordeiformis
Nees, found in India, Nepal, Indo-China, China, Philippine, and Pakistan; Eragrostis minor Host, have a wide range of distribution, from Africa to Europe, and from Asia
to America; Melinis repens (Willd.) Ziska, it is another widespread species, found in Africa, Asia, Australia, Caribean, and America; Setaria clivalis (Ridl.) Veldk., found in Christmas island, Java, Lesser Sunda Islands, Philippines, and Sumatra; Digitaria bicornis (Lam) Roem. & Schult., found in America, India, Australia, and Asia;
Polytrias indica (Houtt.) Veldk., found in Carribean, Colombia, Costa Rica, Guatemala, India, Timor, Java, Panama, Suriname, United States of America, Australia, China, and Africa; Saccharum fastigiatum Nees ex Steud., found in India, and Malesia; Sorghum laxiflorum F.M. Bailey, found in Australia, Papua New Guinea;
Spinifex longifolius R. Br., found in Australia, Malaysia, Papua New Guinea;
Carribean, and Paraguay; Cymbopogon procerus (R. Br.) Domin, found in Papuasia, and Australia; Cyrtococcum oxyphyllum (Hochst.) Stapf., found in Australia, Burma, India, Malaysia, Sri Lanka, Africa, China, Indo-China; Ectrosia leporina R. Br., found in Papua New Guinea, and Australia; and Microstegium eucnemis (Nees ex Steud.) A. Camus, found in Indo-China, and Burma.
Taxonomi
POACEAE Barnhart. nom. cons., Bull. Torrey Bot. Club 22: 7. 1895. nom. alt., Gramineae Juss, Based on Tribe Poeae R. Br. Voy. Terra Austr. 2: 582. 19 Jul
1814.
Annual or perennial herbs, sometimes with rhizomes or stolons. Culms erect, ascending, or creeping, usually branched at the base, in perennials with sterile shoots and flowering culms mixed, in annuals only the latter present; cylindrical, rarely flattened, jointed, usually hollow in the internodes; branches arises singly from nodes and subtended by a leaf sheath and 2-keeled prophyll. Leaves arranged alternately in 2 ranks, differentiated into sheath, blade and an adaxial erect appendage at sheath/blade junction – ligule; leaf sheath surrounding and supporting culm-internode, split to base
or fleshy; stamens 3, rarely 1, 2, 6, hypogenous, filaments capillary, anthers versatile; ovary 1-celled, styles 1, or 2, rarely 3, free to united at the base, topped by feathery stigmas, exserted from sides or apex of floret. Fruit mostly caryopsis with a thin pericarp adnate to the seed, rarely with a free seed; caryopsis commonly combined with various parts of the spikelet, or less often the inflorescence, to form a false fruit; seed with starchy endosperm, an embryo at the base of the abaxial face, and a point or line – hilum on the base of adaxial face marking the connection between pericarp and seed.
Key to the tribes
1.a. Spikelets paired, sessile and pedicellate spikeletes………..2 b. Spikelets singly in panicles or racemes………4 2.a. Spikelets persistent ...4. Arundinelleae b. Spikelets deciduous ………...3 3.a. Glumes thinner than fertile floret; lower glumes short or suppressed; fertile floret
papery to crustaceous or leathery, without awn …………14. Paniceae
b. Glumes as long as spikelets, tougher and enclosing hyaline florets; fertile floret usually with geniculate awn ………...……….1. Andropogoneae
4.a. Spikelets with 2 or more fertile florets ………5 b. Spikelets with 1 fertile florets, sometimes with additional staminate or barren
florets ……….11 5.a. Inflorescence of one or more racemes ……….6 b. Inflorescence a panicle, open, contracted, dense, and spikelike ……….8 6.a. Ligule a line of hairs; lemmas 1-3-nerved ……….10. Eragrostideae b. Ligule membraneous; lemmas 5 or more nerved ………7 7.a. Spikelets placed broadside to rachis; both glumes present; ovary with hairy
apical appendage ……….6. Brachypodieae b. Spikelets placed edgeways to rachis; lower glume absent (except terminal
spikelet); ovary glabrous ………...16. Poeae 8.a. Spikelets with 2 florets, without rachilla extension ....………...9 b. Spikelets with several florets, with rachilla extension ………...10 9.a. Lemmas entire, without awn, glabrous or puberulous ………...12. Isachneae b. Lemmas bi-toothed, awn from sinus, densely hispid………...11. Eriachneae 10.a. Culms reed-like, usually tall; panicle large, plumose; blades articulated with with
13.a. Spikelets unisexual; female spikelet inflated, shell- or urn-shaped …….…. ………...15. Phareae b. Spikelets bisexual ………..14
14.a. Blades with cross nerves, linear to ovate ………17. Thysanolaeneae b. Blades without cross nerves, narrow ……….15
15.a. Inflorescence composed of one or more racemes; rachis tough; glumes opposite ………...8. Cynodonteae
b. Inflorescence an opened, contracted, or spikelike panicle ………16 16.a. Spikelets with 1 to several floret; lemma with 3 awns …………2. Aristideae b. Spikelets with 3 florets; lower lemma enclosing fertile floret …9. Erharteae
Tribe 1. ANDROPOGONEAE Dumort. Observ. Gramin. Belg. 84, 90, 141. 1824. Type: Andropogon L. Sp. Pl. 2: 1045. 1753.
Coiceae Nakai. Ord. Fam. Prof. Nakai, Edita App. 223. 1943; Contr. U.S. Natl. Herb. 41: 7-8. 2003.
Imperateae Gren. & Godr. Fl. France Prosp. 3: 471. 1855.
Rottboellieae Kunth. Revis. Gramin. 1: 150. 1829.
Sacchareae Rchb. Consp. Regn. Veg. 48. 1828; Contr. U.S. Natl. Herb. 48: 12-14. 2003.
Plants annual, or perennial. Blades linear, rarely lanceolate or filiform. Ligule
membraneous, rarely ciliate. Inflorescence composed of fragile (infrequently tough) racemes, these arranged in a terminal panicle with elongate central axis, or more frequently sub-digitate, paired or solitary, often axillary, subtended by spathes and spatheoles and gathered into a compound panicle. Racemes usually bearing paired spikelets, rarely spikelets single or in threes, usually one spikelet of a pair sessile and
2-keeled; upper glume boat-shaped, fitting between internode and pedicel; lower floret male or barren, lower lemma hyaline, 2-keeled, lower palea suppressed when floret barren; upper floret fertile, upper lemma hyaline, narrow, entire or bi-toothed, awnless or bearing a geniculate awn with twisted column, upper palea short or absent.
Pedicelled spikelet usually lanceolate, papery, often smaller than pedicelled spikelet; pedicel resembling rachis internode, rarely absent or fused to internode.
Key to the Genera
1.a. Spikelets unisexual, separated in different inflorescences, or in parts of the same inflorescence ………...2
b. Spikelets bisexual, or at least the sessile spikelet of a pair, male and female not separated ………5
2.a. Annual; male and female spikelets mixed in the inflorescence ………...
………....28. Diectomis
b. Perennial; male and female spikelets in different parts of the same inflorescence ………3
3.a. Female spikelets enclosed in a beadlike, bony utricle ………....21. Coix b. Female spikelets not enclosed in a bony utricle ………..4 4.a. Female spikelets conspicuously transversely constricted; racemes solitary in
spatheate axillary clusters ………...17. Chionachne b. Female spikelets not conspicuously transversely constricted; racemes digitate in
spatheate axillary clusters ………73. Polytoca 5.a. Spikelets single, without an accompanying vestigial spikelet or pedicel ……6 b. Spikelets paired, but sometimes pedicelled spikelet vestigial or represented only
by the pedicel ………...7 c. Spikelets paired, or in three’s ...34 6.a. Racemes rachis tough; blades linear ……….30. Dimeria b. Racemes rachis fragile; blades lanceolate ………..7. Arthraxon 7.a. Rachis internodes and pedicels slender, filiform to linear, or if widened upward,
the upper lemma awned ………8
b. Glumes subequal; lower glumes 3-5-nerved ……...……..72. Pogonatherum 14.a. Inflorescence terminal; raceme solitary, subdigitate, or spread along an elongate
axis ………..15
b. Inflorescence of racemes from the upper leaf axils ………...16
15.a. Sessile spikelet with pectinate, spinose margin, or tuberculate; racemes solitary ………37. Eremochloa
b. Sessile spikelets without pectinate margin; racemes solitary to many ……….. ………60. Mnesithea 16.a. Sessile spikelets alternating on raceme in 2 opposite rows; pedicelled spikelets
absent; pedicel joined to rachis internode ………62. Ophiuros
b. Sessile spikelets all on one side of racemes; pedicelled spikelets well developed to vestigial; pedicel free or partially or completely joined to rachis internode ………17
17.a. Spikelets of a pair similar; racemes tough or tardily disarticulating; rachis articulation line oblique without central peg ………..44. Hemarthria b. Spikelets of a pair different; racemes easily disarticulating ; rachis articulation
line straight with central peg ………..18
18.a. Lower floret of sessile spikelet staminate, its palea well developed; spikelets smooth ………..77. Rottboellia b. Lower floret of sessile spikelet barren, its palea reduced or absent; spikelets
grooves ……….……….60. Mnesithea 19.a. Pedicellate spikelet reduced to a minute pedicel and hidden in the callus hairs
………9. Asthenochloa b. Pedicellate spikelet not hidden in callus hairs ………...20 20.a. Racemes borne on an elongate central axis or its branches, axis longer than
lowest raceme, not supported by spathes ………...21 b. Racemes solitary , paired, or subdigitate, supported by spathes ………24 21.a. Rachis internodes and pedicels without a purple translucent median line …22 b. Rachis internodes and pedicels with a purple translucent median line …….23 22.a. Lower glume of sessile spikelet laterally compressed; racemes reduced to a triad
………..19. Chrysopogon
b. Lower glume of sessile spikelet dorsally compressed; raceme of several spikelet pairs below terminal triad ………..85. Sorghum
23.a. Racemes of 1-5 spikelet pairs ……….14. Capillipedium b. Racemes of more than 8 spikelets pairs ………11. Bothriochloa 24.a. Lower floret of sessile spikelet staminate, with well-developed palea ……..25 b. Lower floret of sessile spikelet barren, reduced to a lemma; palea absent …28 25.a. Ligule a line of hairs; raceme solitary; lower glume of sessile spikelet deeply
grooved, apex elongate, scarious ………...82. Sehima b. Ligule membraneous; racemes more than one; lower glume of sessile spikelet
not deeply grooved ………26 26.a. Sessile spikelet laterally compressed, smooth; raceme solitary, reduced to
b. Sessile spikelet dorsally compressed; racemes (1) 2- or more, terminal or axillary ………27
27.a. Rachis internodes and pedicels stoutly linear to thickly clavate; sessile spikelet rugose ……….51. Ischaemum
b. Rachis internodes and pedicels filiform to linear; sessile spikelet smooth, grooved along midline……….58. Microstegium 28.a. Awn arising from low down on lemma back; culms slender, trailing; blades
lanceolate ………7. Arthraxon b. Awn arising from apex of lemma, or from sinus of bi-lobed apex …………29 29.a. Lower glumes of sessile spikelet 2-keeled; callus inserted into hollowed
internode apex ………30 b. Lower glumes of sessile spikelet convexly rounded without keels; callus attached
obliquely ………..31
30.a. Racemes paired or digitate; leaves aromatic ………22. Cymbopogon b. Racemes solitary; leaves not aromatic ………..81. Schizachyrium 31.a. Upper lemma bi-toothed, awned from sinus ………47. Hyparrhenia b. Upper lemma entire, awned from apex ………..32 32.a. Raceme with 2 homogamous spikelet pairs at base, forming an involucre
……….89. Themeda b. Raceme with or without homogamous spikelet pairs, but not forming an
involucre ………33 33.a. Sessile spikelet with pungent callus ……….49. Heteropogon b. Sessile spikelet with obtuse callus ………27. Dicanthium 34.a. Spikelets in pairs or in three’s; upper glume 3-keeled ...40. Eulalia b. Spikelets in three’s; upper glume without keeled ...74. Polytrias Genus description and key to the species
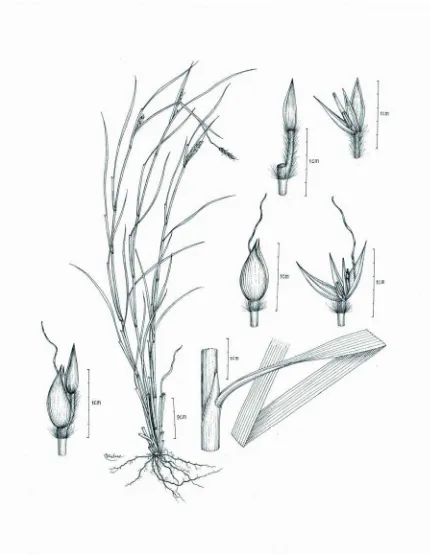
5. APLUDA L. Type: Apluda mutica. Sp. Pl. 1: 82. 1753.
Calamina P. Beauv. Ess. Agrostogr. 128, 157. 1812.
From the Latin name apluda (appl-) ae for chaff or bran, alluding to the involucres, or to the spikelets.
One species, Saudi Arabia, Mauritus, through India to Southeast Asia, Australia and New Caledonia. Plants perennial, rambling, leafy, more or less glaucuos, often scrambling or decumbent and rooting at the base. Auricle absent. Sheath short.
Ligule an unfringed membrane. Blades linear-lanceolate and often falsely petiolate.
sessile spikelets awned or not, pedicellate spikelets unawned; lower floret staminate; upper floret hermaphrodite. Glumes similar , and more or less leathery or thin.
Lemmas membraneous. Palea reduced to a membraneous scale. Lodicules 2, free and fleshy. Stamens 2-3. Ovary glabrous. Stigmas 2.
A good fodder when young suitable for all classes of animals, good hay and silage, stalks used for making hats, growing at the edge of woodlands and in hedges, thickets, on moist or dry stony soils, forest margins.
5.1. Apluda mutica L. Sp. Pl. 1: 82. 1753.
Apluda aristata L. Cent. Pl. II: 7. 1756.
Calamina gigantea P. Beauv. Ess. Agrostrgr. 128, 157. 1812.
Apluda gigantea (P. Beauv.) Spreng. Syst. Veg. 1: 290. 1825.
Calamina humilis J. Presl. Reliq. Haenk. 1(4-5): 344. 1830.
Apluda humilis (J. Presl.) Kunth. Revis. Gramin. 1: Suppl. XLI. 1830.
Apluda communis Nees. Gramineae 62. 1841.
Apluda inermis Regel. Trudy Glavn. Bot. Sada 7: 658. 1881.
Plants perennial. Culms 50-350 cm long, tufted, slender, straggling, creeping or scandent, base decumbent, rooting from lower nodes, branched in upper part. Sheaths
usually glabrous. Ligule 1-2 mm, a ciliolate membrane. Blades 8-30 cm by 2-20 mm, flat, apex attenuate to a setaceous. Synflorescence compound, paniculate, 5-35 cm long. Inflorescence a solitary raceme tightly embraced by a spatheole, terminal and axillary. Spatheole 3-9 cm long, ovate, membraneous. Racemes single, bearing a triad of spikelets. Rachis obsolete. Spikelets in threes: Fertile spikelets sessile; 1 in the cluster. Sterile spikelets: companion sterile spikelets rudimentary and well-developed, male, lanceolate, dorsally compressed, 2-5 mm long, as long as fertile spikeletes; glumes herbaceous, acute, muticous; lemmas 2, enclosed by glumes, muticous. Fertile spikelets comprising 1 basal sterile floret, 1 fertile florets, without rachilla extension, oblong, laterally compressed, 2-6 cm long. Glumes dissimilar, exceeding apex of florets, firmer than fertile lemma. Lower glume lanceolate, as long as spikelets,
Fertile lemma oblong, 4 mm long, hyaline, without keel, 3-nerved, apex entire, or lobed, bifid, muticous, or awned, principal lemma awn from a sinus, straight, or geniculate, 4-12 mm long. Palea hyaline, without nerved, no keels.
Notes
Found in Africa, Asia, Australia, and Pasific. This is a polymorphic species widespread in tropical Asia. Good fodder value, palatable to stock when young, young grass eaten by buffaloes.
Distribution at Sulawesi
Central: Between Palu and Palolo, Alt. 550 m. South: Maros. Habitat
Ditch and river banks, wet places, along roadsides, on poor soil, in waste grounds.
Specimens examined
Lasut 971 (WALL); Chin 3539 (L); Darnaedi s.n. (L); Eyma 3492(L); Meijer 10674 (BO).
7. ARTHRAXON P. Beauv. Type: Arthraxon ciliaris P. Beauv. Ess. Agrostogr. 111, pl. 11, f. 6. 1812.
Alectoridia A. Rich. Tent. Fl, Abyss. 2: 447. 1850.
Batratherum Nees. Edinburgh New Philos. J. 18: 180-181. 1835.
Lasiolytrum Steud. Flora 29: 18. 1846.
Lucaea Kunth. Revis. Gram. 2: 489. 1831.
Pleuroplitis Trin. Fund. Agrost. 174, t. 16. 1820.
Psilopogon Hochst. ex A. Rich. Tent. Fl. Abyss. 2: 447. 1852.
From the Greek arthron “a joint” and axon “axis”, referring to the rachis, to the jointed rachides.
About 25 species, Old World Tropics, mainly in India. Plants annual or perennial, herbaceous, slender, often trailing, decumbent or rooting on the lower nodes. Auricles absent. Ligule a ciliate membrane. Blades linear-lanceolate to ovate-lanceolate. Plants bisexual; terminal and axillary slender subdigitate racemes.
Spikelets solitary and sessile or pairs of sessile and pedicellate spikelets; sessile spikelets awned and with 2 florets; lower floret reduced to a lemma; upper floret bisexual. Glumes equal or subequal. Lower glume convex or laterally 2-keeled;
Shade or open habitats, rainforest, wet places, moist pastures, rocky slopes, old cultivated fields.
Key to the species
1.a. Sessile spikelets strongly scabrid to spinulose……...7.1. Arthraxon hispidus b. Sessile spikelets smooth to scaberulos …………..7.2. Arthraxon lancifolius
7.1. Arthraxon hispidus (Thunb.) Makino. Bot. Mag. (Tokyo) 26(307): 214. 1812.
Phalaris hispida Thunb. Syst. Veg. (ed.14): 104. 1784; Webbia 49(2): 265-329. 1995.
Arthraxon ciliaris P. Beauv. Ess. Agrostogr. : 111, pl. 11, f.6. 1812; Key Austral Grasses 1-150. 1990.
Alectoridia quartiniana A. Rich. Tent. Fl. Abyss. 2: 448, pl. 102. 1851.
Plants annual, mat forming. Culms 10-55 cm long, less than 1 mm diameter, ascending, rooting at the lower nodes. Nodes pubescent; internodes up to 6 cm long.
Sheaths glabrous on surface, or pilose, with tubercle-based hairs. Ligule 1-4 mm long, a ciliolate membrane. Blades 2-10 cm by 5-30 mm, margins tuberculate-ciliate, apex
acute, the surface glabrous, or pilose. Inflorescence composed of racemes. Racemes
2-5, 2-6 cm long, digitate. Rachis fragile at the nodes, hair less than 1 mm long, internodes filiform to linear 2-5 mm long. Spikelets in pairs: fertile spikelets sessile, 1 in cluster; companion sterile spikelets pedicelled, 1 in cluster. Pedicels linear, up to 2 mm long, glabrous or ciliate. Fertile spikelets comprising 1 basal sterile florets, 1 fertile florets, without rachilla extension, lanceolate, laterally compressed 4-9 mm long, falling entire; callus glabrous, or pubescent, base truncate, attached transversely.
Glumes dissimilar. Lower glume as long as spikelet, lanceolate, chartaceous, much thinner on margins, without keels, 7-9-nerved, surface convex, spinose, rough on nerves, apex obtuse. Upper glume lanceolate, chartaceous, 1-keeled aboved, 3-5-nerved, primary nerve scaberulous, apex acute, or obtuse. Florets: basal sterile florets barren, without significant lemma. Lemma oblong, 0.6 times length of spikelet, hyaline, 2-nerved, obtuse. Fertile lemma oblong, 2-6 cm long, hyaline, without keel, 1-nerved, apex acute, muticuous. Principal lemma awn dorsal, arising back of lemma, geniculate, up to 20 mm long, with a straight or slightly twisted column. Palea absent or minute. Anthers 2, up to 1 mm long. Caryopsis fusiform, or oblong, 3-5 mm long.
Found in Africa, Asia, Indo-China, Malesia, Papuasia, Australasia, Pasific, North and South America. It is now a widespread weed, occurring in many warm-temperate and tropical parts of the world.
Distribution at Sulawesi
Central: Palu. Habitat
Streamsides, moist places Specimen examined
Bunnemeijer 11326 (BO), 11330 (BO, L); Kjellberg 3033 (BO).
7.2. Arthraxon lancifolius (Trin.) Hochst. Flora 39: 188. 1856.
Andropogon lancifolius Trin. Mem. Acad. Imp. Sci. St.-Petersbourg, Ser. 6, Sci. Math. 2(4): 271-272. 1832; Blumea 27(1): 255-300. 1981.
Pleuroplitis ciliata J.A. Schmidt. Beitr. Fl. Capverd. Ins. 152. 1852.
Lucaea ciliata (J.A. Schmidt) Steud. Syn. Pl. Glumac. 1: 414. 1854.
Andropogon multicaulis Steud. Syn. Pl. Glumac. 1: 383. 1854.
Pogonatherum tenue Edgew. Flora 39: 188. 1856.
Batratherum schimperi Nees ex Hochst. Flora 39: 179. 1856.
Pleuroplitis microphylla (Trin) Regel. Bull. Acad. Imp. Sci. Saint-Peterbourg 10(3): 370. 1866.
Arthraxon figarii (De Not.) Asch. & Schweinf. Beitr. Fl. Aethiop. 310. 1867.
Batratherum lancifolium (Trin.) W. Watson. Gaz. N.W. Prov. India 10: 392. 1882.
Arthraxon linifolius Henrard. Blumea 4(3): 525. 1941.
Arthraxon comorensis A. Camus. Bull. Soc. Bot. France 98: 36. 1951.
Plants annual, mat forming. Culms up to 30 cm long, loosely tufted, very slender, decumbent, much branched. Sheaths loose, margins ciliate. Ligule 1.3 mm long, a ciliate membrane. Blades 0.5-4 cm by cm 2-9 mm, elliptic to narrowly ovate, thin, flaccid, puberulent to densely pubescent, scattered tubercled-based hairs, margin pectinate-setose, base cordate, apex setaceously acuminate. Inflorescence composed of racemes. Racemes 2-9, 1-2 cm long. Rachis fragile at the nodes, ciliate on margins, internodes linear 1 mm long. Spikelets in pairs: fertile spikelets sessile, 1 in cluster; companion sterile spikelets pedicelled, 1 in cluster; pedicells 1.5 mm long, filiform, ciliate. Fertile spikelets comprising 1 basal sterile floret, 1 fertile florets, without rachilla extension, linear, laterally compressed, 2-5 mm long, falling entire, callus pubescent, base truncate, attached transversely. Glumes dissimilar. Lower glume as long as spikelets, lanceolate, chartaceous, without keels, surface convex, asperulous.
Florets: Basal sterile florets barren, without palea. Lemma of lower sterile oblong, hyaline, 2-nerved, obtuse. Fertile lemma lanceolate, hyaline, without keel, 1-nerved, apex acute, 1-awned; principal lemma awn dorsal, arising from back of lemma, 5-9 mm long, geniculate, with twisted column. Palea absent or minute. Anthers 2, 0.5-0.7 mm long.
Notes
Found in China, Bhutan, India, Indonesia, Myanmar, Nepal, New Guinea, Pakistan, Philippines, Sri Lanka, Thailand, Vietnam, East Africa, South Arabia.
Distribution at Sulawesi Celebes.
Habitat
Growing among rocks, damp situation, exposed hill slopes, road banks, steep stony slopes, rocky sites, steep banks, open places, plains, riverbanks, cliffs, gullies. Specimen examined
Yoshida 989 (L); Bunnemeijer 11558 (L).
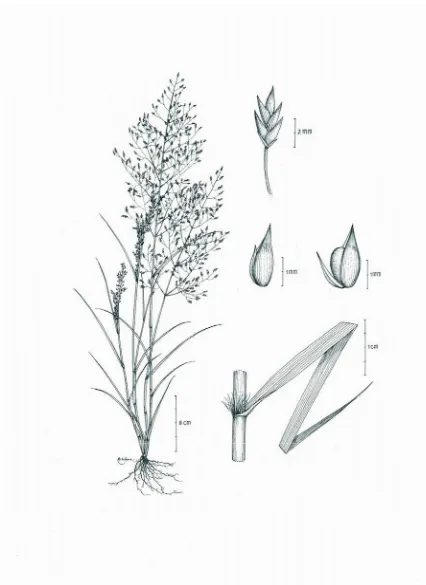
9. ASTHENOCHLOA Buse. Type: Asthenochloa tenera Buse. Pl. Jungh. 367. 1854.
Garnotiella Stapf. Hooker’s Icon. Pl. 25: t. 2494. 1896.
From the Greek asthenes “weak, feeble” and chloe, chloa “grass”.
One species, Indonesia, Southeast Asia. Plants annual or perennial; decumbent, herbaceous, slender, branched. Plants bisexual. Inflorescence opened, paniculate.
Spikelets paired. Lower floret suppressed; pedicellate spikelets reduced to barren pedicels. Glumes 2 subequal. Upper glume keeled. Lower glume convex. Upper lemma bilobed and awned. Palea absent. Lodicules absent. Stamens 2. Ovary
glabrous. Stigma 2.
9.1. Asthenochloa tenera Buse. Pl. Jungh. 3: 367-368. 1854; Kew Bull. 27: 515-562. 1972
Garnotiaphillipinensis Stapf. Hooker’s Icon. Pl., t. 2494. 1896.
Garnotia leptos (Steud.) Stapf. Bull. Misc. Inform. Kew 1910: 302. 1910.
Plants annual. Culms up to 100 cm long, rooting from the lower nodes. Ligule