lnternational Journal of Engineering & Technology IJET-UENS
vol;
1l
No; 04PROVIDING VEGETABLE
STEARIC
ACID
SUPER
V
1895 S FROIVI
HYDROGENATED
OF
CRUDE
VEGETABLE
STEARIC
ACID HCV
1895
SWITH A
SINGLE
FRACTIONAL DISTILI-ATION COLUMN
Muhammad
yusuf.
Ritongal
l,Chemical Engineering Department, Faculty of Engineering, University of Sumatera Utara, Jl. Almamater Kampus USU, Medan 20155, Indonesia
E-mail : yu s ufrit @ gmail. com
Abstract
ln 2005 stearic acid
V
1895 S is manufactured for l*ttime in Indonesia base on the natural vegetable oil, Crude palm Cil to apply Twin Rivers's demand. One of it specific quality (iodine value 0.15 gll00 g maximum) is very influence to,re purifying steps, one
of
the steps is purifying of hydrogenated crudeV
1895 S (HCV 1895 S) feed by a single :olumnof
fractional distillationto
produceV
1895 S with the lowest possibleof
it
iodine value, lower than thenaximum value. The results of application and small adjustments of determination operating conditions of the single ;olumn priority based on the feed composition, product composition of
V
1895 S andit
pressure drop character are ;ompared based onit
quality standards. This is the method used in this research to provideV
1895 S and conducted in ihe plant scale of PT.XXX
factory with production yield 88-
89 Vo of 0.90mtHCV
1895 S, unsarurated content is ioiver than 0"15 g/100 g and higher than 98 7o wtw Cl8 purity. In general the specific quality standards ofV
1895 S can be met"Keywords: quality standards, fractionation, composition, unsaturated contenl, adjustments
1.
Introduction
Fany acid
ofV
1895 Sis meansuperfatty
acidbasedonnatural
vegetableoilslfotswith
stearic
acid C17I{35COOHor
C1s compositionisminimw 95
Vowlw. Ithas been never manufactured at
all,
although oleo chemical industryhad been growth in Indonesia inthe
period
1989-2004.This fatty
acid had
beenproduced in America and Europeto consume for:one of the consumption asthe raw material
of
the cosmetic manufacturing that have the lowest skin effect, for exampleto manufacturethe
premium
shampoo Pantene.Thts stearic acidhasvery
low
unsaturated content.Sincethe beginning of the year 2005,South East Asia's begunto see as aV
1895 S producer. In October 2005Twin Riversas a buyer gave an idea, a standardquality that had
to
be
followed and
produceit
according
to
the
process sequenceswhich
were determined and doby
PT.XXX
alone(Figure 1), acompany
that had
beenexist
in
operation inoleo chemical industry, since the lastof
the year 1998.Quality of
V
1895 S produced had to meet the quality standard which is shownin
Table 2 below (settled by Twin Rivers).The same quality standard which wasproduced
in
America and Europe by the method wasdifferent at all.This was because of the different raw material and the used technology"
In
this research is useda natural vegetableoil,
Crude Palm Oil (CPO) as raw material for the manufacturingand refining of stearic acid super
V
I 895 S as shown in Figure lbelow. Manufacturing and refining technique of CPO to produce stearic acid superV
1895 S (Figurel)is
so
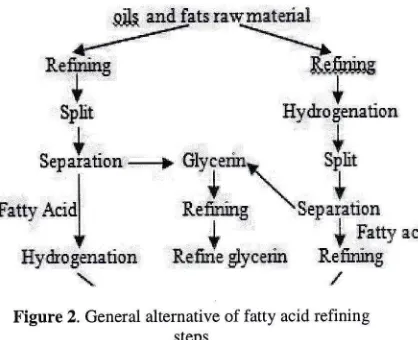
different compare tothe general alternative manufacturing and refining techrique of fatty acid in Figwe 2 below [12]. Figure 2 is modified to be FigureI
to produceV
1895 S to apply Twin Rivers's demand with the lowest possibleit
unsaturated content, as thefirst technique in the world. Refining
of
raw material onleft
sideof
Figure land Figure2
can be done by degummingonly
and/or
combined together with bleaching andor
deodorization process alternatively according to raw material used, product quality have to be met and equipment capability. Refining of fatty acid on theleft
sideof
the both figures can be proceed through distillation and./or combinedwith
fractional distillation alternatively, againit's
very depend on theI 10404-3737 IJE'I-IJENS (d Augusr 2011 I.lflNS
lntemational Journal of Engineering & Technology IJET-IJENS Vol: 11 No: 04
raw material specs, product quality have to be met and
present equipment capability.
oils and fats ra$'meterial
;-
-=\_-r
Returing
Br.Eflat
+t
$plit
Hvdrogenationr+
S.pJrtior,
+
Gl1'ceru:ra
t{rt
I
r*uu
e.,al
R.t*s \*.,*Luo'
+
J
{Fanv
Hy&ogenation
Refineglycerin
Refimng\/
---*CPo'.-+-
\-*
Refrning
eqfiu$g{+
Split
HvdrogenauonSepaiation
-r
Glycerin,*
Sil,,r*tty
roal
R,***
\*rri*r.,,
+
+
{Fauvaud
H1'drogenahon Refine
glvcenn
Refitting H1'drogenahon Refineglvcenn
RefinurgHytuse-
I
+pfllcLfilUl
I
Hydrogenated fattv acicflfr\4
{
Distillahon
Frttyrdd
tyrcid
I
I
_l
_l
1850
I
L+
ResldueFnctiopation
-|
pl"lmid.q acid
{J
flv
1$9J S [image:2.612.385.594.115.285.2]Table 1 .Thespecific quality of CPO to provide
V
1 895 SFigure 2. General alternative of fatty acid refining
steps
Palm
oil is
one
of
the
sourceof
stearic acid (CrzH:aCOOH) or Crs also as a source of unsaturatedfatty
acid oleic acid
(CtrHgsCOOH)or
C1s-1 andlinoleic acid (CrzHgzCOOH)
or
C1s-2 that could be hydrogenatedto
be
stearicacid
as also stated byt7ltl0lll5l
till
the total amount of Crs is around 50 7ow/w. See the raw material quality CPO that used in this study. The
fatty
acid distillate whichis
obtained of splittedCPOis
called fatty acid 1850[7]. Crude Palm Oil has the highest C1s content than the others natural vegetableoils
such as: crude palm kerneloil
andcoconut oil. This's the reason the raw material CPO is
used in this research.
ecid
iuci;issfrf
-
,I
Fractiohation---* \'1895S
I
ll,
S.awrrai+rial
A\tr
SY E?Ocru
c14 8tr6 *13
f,!&-X
C:8-!
c]0
"u-Ilkcro
?.6
101:
0:9
0.:1.1 43.l
,1.439.1
10.3 0.4 0.8io rrcg ." F?ora $4Xg1f4 C*emirudo,J
0fi
{-qLS-:,
AV= aeidvrlue;
SV =saponificationvelue;I];ftri=unkno*ncomponenUmsterialRefining steps that are started from CPO refinery
till
distillation stepin
FigureI
above are intended to separatethe
impurities of CPO(gum,
trace metals), reduce of ; pigments, odors, short carbon chain of C6till
C14 fatty acid (as the experience CPO contain trace C6-10 in the light end product of distillation process), Fractional distillationIn this manuscript is discussed priority the results of this study'in the end step of the refining Hydrogenated Crude
V
1895 S (HCV 1895 S) to produce stearic acid superV
1895 S through a single fractional distillation columnin
Figure 3whichis
equippeda
packed of structured packing and fallingfilm
reboiler [6]. The using this column rvill be obtained the better saving ofaldehydes, ketones,
iodine value
(through hydrogenation and distillation) to provide of fatty acid feed quality(HCv
1895S in
Table2
below) that refined in the end step, in a single fractional distillation column (Figurel).
Each step affect to the each others, agreed also by the researcher [8].energy
consumption, equipment, operating cost, flexible and product quality compared to the using oftwo
fractional distillation columnsthat utilized trays andlive
steam (Figure4)
tl3l.
This technology in Figure 4 was mostly used in Indonesia since 1989, in oleo chemical industries.I 10.104--i7J7 IJDT-IJENS {!) Augast 20ll UENS
ITN
[image:2.612.147.361.145.455.2]--lntemational Journal of Engineering & Technology UET-UENS Vol: I
I
No: 04\recuum rystem Cl6purge rslight end
V 1895 S asside product
Frsn* akier of
Sottorepro*mt
orresid*e
[image:3.612.46.240.113.268.2]D
Figure 3. The refining of HCV 1895 S by a single fractional distillation column
Please compare this new technology in Figure 3 above
with
the
old
onein
Figure4
below. Refining of blended fatty acid that contentof
at leasttwo
mainfatty
acid
componentswasusually used
twofractionation columns.
The
l't
colurnnis
used to separate light end and 2od colurnn is used to separatethe top product with bottom product. Light end contain any substances
or
impurities which have the lower boiling point than boi-ling point of the main product in 2'o column (the side or bottom product). The bottom product has the higher boiling point than the boilingpoint
of
the
side
productin
the
same column. Separationof
the light endin I't
column and bottom productin
2od column affect much better to the main products quality..[ehlCJ" Quality of feed
]ICV
1E95 SVacuum pressure fractional distillation
is
applied in this study as done for distillation because of fatty acid included unsaturated bond inside are so sensitive for oxidationtl3ltl4l
(whichis so influence into the bed of fatty acid color and odor). This is also according tothe reseachers
tl-3lt9l.
Unsaturated bond in fatty acid is more sensitive. Ifunsaturated bond is higher in fatty acid,it's easier to oxidize [10]. Prevention of fatty acid oxidationis mean to prevent the increment of fatty acid iodine value or unsaturated content, colors and odors.It
is mean also control closely fatty acid quality which is made"In
this
researchwasV
1895S
quality" A researcher explainedin
his disertasion: reduction of bottom temperature of column can reduce stearic acid iodine value or unsaturated content or vice versa [18]. Fractional distillationis
shownin
Figure3
above is intended to reduce unsaturated content of stearic acidC16" Separation
of
oleic acid Cls-1in
the residue of single fractional distillation columnwill
reduce oleic acid (C1s.1) content in stearic acid Clsas a side product"It
is
mean the lower color and odorsin
stearic acid ClsorV
1895 S. Reduction of fattyacid
iodine value,color
and
odors
is
not only
can
be
done by hydrogenation priority, but can be done also through fractional distillation processto
achievethe
lower point. How it can be done?Tab!* fu Quality :*aadsrd o f *gg{i$ ecid. s up e* V I B 9 5 $
FattS' o*id comy o s itioa,
$i'+-Jw)
Arr]lrditr'rane*
< ctr4
clf
c:6-t
c1?
c!8
cl8-I
C}8.]
C:O
0;5
lorrimus
I0.0 msriuurr9fi.{ luinillu.m
s,44
artlmuu
cI0-l
fiqgs6; flo I o fi fir[ilfl $:andu da, J 005 Figure 4. The refining of blended fatty acid through 2
fractionation columns
Met*iaX(ltl
C14 CI6 q:8
Clg-l
C30
Seh
Remark*
Qualityrtatus
Status-$qur*E;
fforo
$flslIs
C*aml *da,261 5Unsaturated content of HCV 1895 S is higher than the maximum standard (see Table 3 above), because ofit value is over than O.l5 Vo. So
it
has to be reducedtill
meet the standard, Reduction of C1s-lamountin
C1s is also decrease of arachidic acid (C26) composition and the others impurities that have the same or higher thanstearic acid C18 boiling
point
whichis
so positive influenceto
theV
1895 S iodine value, unsaturated content, color and odors,to
be lower. Thiswill
beinfluence to the lower production yield
of
V
1895 Sabsolutely, but hopefully with the better quality
V
1895Check tl.e troruogenous feed tank
sample; GC, !\r and rclor
Appliad tlle main caemting co*&trs** t* *.:in5!e tellmr::o:erd remallised om 8O 96 cf Y 1895 S
gm&xt*teis de.ters*red
Ttre rtain oPer=iing cenditia* are detemine* and Fredict ba3€d on tl.e ccmPo:iticn (GC) of feed. pu.ge, side and bottamProdircts
*e Pr*dicted
Dislillate a' 189f $ PuitY conttol
Reduce side stream &aw off rste
Stored as ttrc in rpe* V 1895 S in fhe fiael
lntemational Journal of Engineering & Technology IJET-IJENS Vo1:
1l
No: 04S
thanHCV
1895S will
be
achieved' The above question is answered'Operating conditions
of
single fractional distillati,on co'lumnias
tobe determined and predict carefully beforeby Roult's Law [5][16]. Oleic acid content.Cls-thadtoie
a main considerationin
this study'It
is so sensitiveto
the oxidation and heating' Increment of unsaturatedcontent
or
iodine value
should
beprotected, should be lower in
V
1895 S' The predictionLf
the single fractional distillation column operating conditionsis done base on the capacity and column characters (the pressure drop on the top, bottom.and feed tray locationof
the column), feed compostuonHCv
friSS S, product compositionof
V
1895 S and residueproduit
compositionis
predicted'-.{ctual applicationof
this predictionwill
change alittle
bit depend on the actual product qualityis
made' The2" Method
Semi
trial
and error methodis
usedin
this researctr'Operating conditions are determined, predict first-and uppty
to-*".t
V
1895 S specs' The glalClnC.is fone u'"lorOlng to the actualtetoltt
V
1895 Sis
obtained' basedoi
it
quality standard (priorityit
composition'changing
have
to
be
predictedand
make better;;ft"?
to the quality standard' The understanding of fru.iionuf distillation, physical and chemical properties oi th" tobrtur.e, e*pJti"nc" in this process and fillingt"urr-ftur"
the big conffibution to do the changing oftfr"
operating conditions
tl{'
The-. operati^ng "orOl,iont arJ determined
and predict
1" for
807oproJr"ri",
yield ofV
1895 S according to the specs- ofV
rasS S composition to getin
specs and the higher reflux ratein
ihe
single column, accordingto
C13-1ptop.tti"t
that so sensitive to theoxidation and heating'it.'trigt
reflux rate hasto
be made asit
value is;;hi";."d
on the actual 807o productionleld
of V 1895S or higher, that's limited by
V
1895-S composttion'
*rut*u',"4'"ontent,
colorar; achieved on the actual operating conditions are applied., to^ get the higher yield. What are the suitable conditions?unsaturated content) and production yield' See Figure 5
b"lo,". Proc"dure of anaiysis refers to AOCS Ce l3e-Oiana
fg
l-64 to do iodine value analysis of fatty acid"f."
eO6S
1-62 to do fatty acid compositionanalysis t4t.
Product:V1895S Ilurit]' : 90 i'b minirrum
C:s-t : fi,15 0,6maximum Crc : 1O -qio maxirctr'
Figor.
s=u.ti;;uto#
"or
and qualityv
1895 s as it standardIntemational Journal of Engineering & Technology IJET-UENS
vol:
II
No: 043.
Results and Discussions
T-r_rPp
,l
intended to separate stearicacid (C1s) superuV
1895 Sandfarty acid residue C1s_2s of the feedUiV
1895 S to reduce unsaturated contentof
V
lg95
S. lhere is no light end draw off in this study because of color (not shown here), C16 and C1s of HCV 1g95 S isin
specs. The final qualityof
V
1895 Sis
shown in Table 4 below. The off specs oneV
1895 S in the final storage tank is caused of by production yield increment over than 87 Vo (see Table 5 and Figure 7 below).1S
I+h}fl.*" Unsaturated content o f HCV I I 9 5 S and.
\r
1 895 S (96:*rsr)9S,06 \r1s95
S
0.s9
1"12
!S
.Qgs.r.;5g;
f'lora
$mCr{s S&fil{n{dg, .?00iThe results of,
this
research show that unsaturated contentin
this step came down to beaverageof
0.19Vo(based on Twin River's analysis) or iodine value of
\/
1895 S came down ro be average of 0.12gr l2fi00
g. Comparison
of
unsaturated contentClg-l
of HCV 1895 S andV
1895 S can be seen in Table 4 above"Actual color of
V
1895 S is in specs, but not shown in Table 4 above, discussionis centered to the unsaturatediEl-l
rrra,
34.r-r45
i$
F€€d"
HgV
1895 SCI4
O,cl6
?",6
fl.?6cI
I 980.5
98.S9ci&l
].-l
0-11c:0
9,:
0.9?total 999.7
l{0.0
iliii
iir*
*t*
:63-X64:9.
trB-t
>ff,
tr$-1. C30 are .lPees Feedcl8-1
> C18-l iscontent which is off specs. See inTable 3 and Table 4 above.
The operating conditions are determined and predict,
can bee seen in Figure 6 below.This prediction is made base
on
the fatty acid
compositiononly. please, compare this one with the actual operating conditions in Table 6, in discussion 3.3.eoffdsrr:rer
Li_Eht Eud
lnol
rs'ei-e]rt1',) e
35f
184
c14
0.{0
cx6
]..05ct
E
0.4_iC,.0S
76.S0 3fl.08
total
1,,50to0.0s
ri.r
l895
S ?i,f*ad]s.8 x9 98.S4I s.s4 o,30
totstr
?99.?6
I00.0tl
cI6
6_55cl8
?!1.}0
cl8-r
ff.33c30
*.53
cls
c!3
ct8-1
ct0
o 18s.86
I.9S ?.6$
s.g5 95.03
x.ffo
3_ E3
tot,sl
EgS.4O
IflS.8Figure 6.The operating conditions design forthe providing
ofHCV
1895 S by a single fractional distillationcolumnIl{)40.+-3737 tJEl,ltUNS @ Augusr20II IJENS
[image:5.612.60.524.375.687.2]lnternational Joumal of Engineering & Technology UET-IJENS Vol: 11 No: 04
3.1.
How
to
minimize unsaturated
content
in
V1895
S?10
Based on Table 5 and Figure 7 below no doubt at all that
Cl8
purityis over than 95 7o, althoughv 1895 Sproduction yield are (80.0 - 95'0 %). This is caused by
bt6
feed is lower thanI
Va (Table4
above).If C16 amountin
the feedHCV
1895 Sis
kept asis
in
V 1895 S feed and nolight
end draw offor total reflux in this system./study, ClS puritywill
not be lower than90 7o wlw. This
also affect
to
the
bigger C16concentration
in V
1895
S
that can
minimize unsaturated(Cl8-1)
contentin
V
1895 S, the others components too, C20 and unknown (Unk) (in Table 5)' The higher S production yield ofV
1895 affect to the smaller C16 content and the higher C20 and unknown relatively.It
affectedto
the
off
specs unsaturated contentin
V
1895 S.It
can't be allowed. Please, seealso
both
figures, Figure7
and Figure8
below's7
0,35
99,.131.1
5
9?,580,16
0,11 0,f s111:
0s-1.0 6.8-0-9
In *pecs
3rr specs
$ff
sprcr0"04
0,0:CIJ6
oJ3
8?"0
s3$
s*foh+Eff.ectof,*B*if,:oati:omgreductitr:,xi€K*]s#wr,ra{a.ratesccnlerr1oflxlEEs$99
fl
ss.:E99
:
3 91"5@
96.J
4
fl"39
1,Xl
97 Sawqe: F/ara Sqrtt'ifsC]e*ind+. lS$J
100
99"J
9d
95"5
0.11 95
off
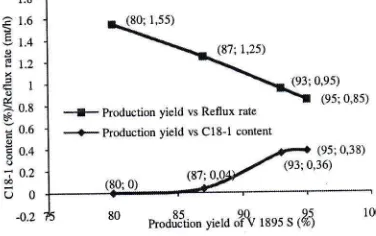
[image:6.612.389.577.516.634.2]The influence of the reduction of
V
1895 S production yield on C18-l content inV
1895 S and reflux rate in Figure7
can be used as the evidenceto
support to state; reductionof
V
1895S
production yieldwill
reduce unsaturated contentin
the
main
product' Reduction ofV
1895 S production yield is cause of theincreasing
of
reflux
rate which reduced theBT
of fractionation column(in
Figure9
at discussion 3.4)"The same thing that had been done by a researcher to reduce stearic acid iodine value or unsaturated content in stearic acid distillate; by reducing BT of distillation column
[18].
The
reductionof
BT
affect
to
the reductionof
evaporation rateof
unsaturated content, reduceit
amountinto
V
1895 S,of
course also the other components. This is according to the distillation principlest5ltl llt17l.
Figure 8. The influence of
V
1895 S production yieldon C18-lcontent in
V
1895 S and reflux rate*o rbditryfifd tJ lti [!gJE $ii
Figure 7. The influence
ofV
1895 S production yieldon C18 purity in
V
1895 SSeparation of amount C18 into fatty acid residue (see
Figure 5 above) affect to the lower amount ofV 1895 S.
Reduction the production yield
of
V
1895 S. This is cause of C16 amount ratio to the total amount of side productV
1895 S is bigger. Finally although without iight end draw off, color ofV
1895 S met the quality standard (not shown in discussion). So the decision not to draw off light end is the exact consideration in the beginning of this study, one technique to minimize the unsaturated content or not reach 0.15 Vo. The ratio canbe
changed dependon
the changingof
V
1895 Sproduction yield. So in this study reduction of
V
1895S production yield is also the other technique to control or reduce unsaturated (C18-1) content as
it
specs (seeFigure 8).
1.8
? 1.6
c
cl
> 0.8
:
0.6E 0.4
UO
-0.2 91.J8)
*.l* Production yield vs Reflux rale
---*- Production yield vs C18-l conrcnt
p.od,iJtion yi"ro ,li$ rsss s i&i)
I I 0 104--1737 Ll U I''lJ IjNS (cl,\u gu sl l0 I I IJ E^-S
@
International Journal of Engineering & Technology IJET-IJENS Vol: I 1 No: 04
-1,i.
Prediction
versus
actual
V
1895 Sproduction yield
;::-:uon
ofV
18955 unsaturated content was done in,: . ,:.rdy.Theproduction yield of
V
1895 S is tried firstr.
'-:
asthe design and predictionof
the operating. ,::::rons(Figure 5 above). ReJlux rate is also made so
.:-,
rgh
in the beginning, on the intervall5OO-
1600.;
-,- Thisreflux rate
is
intendedto
suppress the:
.:,:iation rateof
unsaturated component as low as:..,::le
becauseof
the boiling pointof
unsaturated.
-.
.cid Cls-1is lower than C1s boiling point[16]. This-:
l-r
rate is made also because of the amount of C1s-1:
---: feed HCV 1895 S is so small (0.23 7o in Table 4-:,-,:r.
also the other components (Table 4). On the -'::
reflux rate,it's
moredifficult to
be separated:-l
,rnto the bottom productor
residue (Figure 5-;.,r3). Of course the amount of Cra
will
be bigger in--.
::sidue.It's
a riskof
a high reflux rate. The feed --.:,sried first
onl.000 kg/hr. These predictions are.::
accordingto
the experiencein
thefatty
acid'
.:..ation and fractionation in the previous"'-.
:roduction yieldof
V
1895 Sis
then increased:ii-)'(as
shownin
Table5
above) after fractional -.:-iation
conditions
werestablefor
80.0
7or-:,:jction
yield. This simulation is done for knowing--.
.;rfluenceof
the incrementof
V
1895S
on the-::.ment
of
unsaturated contentin
V
1895 S. The':,
-.:s rvithin Table 5 proved the prediction above. See..
Figure8
below.The higher possible production..:
can be achieved but at the same time unsaturated- t:,3nt is closer to the maximum point of unsaturated : --,ent and finally of specs on
9l
Vo,93 % and 95 Vo:: -,:.rction
yield
(Table
5
aboveand
Figure
8:t.:.r').The in specs one ofV 1895 S should be made in ,-
:
research.How
many
percentthe maximum:::'r.rction
yield
hasto
be
achieved?How
many:r::ent
the safest production yield?It
has to be met.-::
rendin
this Figure 9 can be used to predict the,
--"ble production yield ofV
1895 S to provide.:.
rodine value and unsaturated content ofV
1895 S -- Table 5 above) came up to be higher. This is cause:.
*nsaturated component is evaporated more together, :.
\/
1895 S as the impactof
the incrementof
V-.::
S production yield and/or decreasingof
reflux:r,:
see Figure 8 above). The compositionof
C20 is: :-ler
also, causedof
the
same reasons.This
is .: - :rding to the distillation principles t5l t I 1l I171.i.3.
Actual
versus
predicted operating conditions
-:.:
reduction ofV
1895 S production yield mean the-:
:3mperature of the fractionation column is adjusted.-.rer.
This
temperaturecould
be
determined and::::rct
practicallybyRoult's
Law but
haveto
be:
=l-red actually through this research. The predicted11
The
off
specs one stearic acidV
1895 Sin
Table 5 [image:7.612.299.502.415.536.2]above is sent back the other feed tank as described in Figure 5 above to avoid the higher unsaturated content in the same feed tank and in the next
V
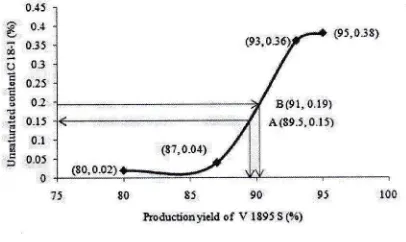
1895 S will be produced.Based on data in this table,it
is so clear the influence of the higher production yieldV
1895 S tothe higher it unsaturated content. See Figure 8 below, Base
on
this figure,V
1895S
production yield is predicted 89.5 Voto
achieveO.l5
7o unsaturatedcontent
in V
1895S
(point
A).
So the
safestproduction yield should be under 89.5 Vo. Based on this
predictionv
1895S
productionyield
should be adjusted to be lower than 89.5 % (see Figure 9 below).It
could be 88 Vo or 89 Vo. Whyit can be happened?It
has to be considered strongly the unsaturated content should be achieved first lower than 0.15 Vo.lt
is very importantto
meet the specsfirst.
If
the production yield is kept constantly as is (89.5 Vo), therc is a big possibility thatV
1895 S will be off specs.Beside
this
reasonthe
stabilityof
this
process is considered strongly.It is
not
guaranteedall
the instru:mentationshave
the
straightline
response,according to the experience it's normally has the small fluctuate on the same amplitude.
,5, 80 *5 90 9' t00
lro&rtionyi{d of Y lS95 S G6)
Figure 9. The influence of
V
1895 S production yield on unsaturated content inV
1895 SThe actual average production yield is achieved 85.0
Vo. Why is it different? The production yields in Table 5 are not straight since the increment is done, It is done slowly, step by step.It so logic the actual production yield is lower than the predicted one.
one
is
used as a main guide and very important to conduct this research. This prediction was limited by unsaturated contentor
double bondof
C13-1in
the quality standard.If
the consideration was based only of C18 purity (see Table 3 above),V
1895 S can be draw0.45
3 0.1
; 0 35
6
o.r6 0?5
6 ^-i n r{
:
0.r5
o.o5B (91, 0.19 !
a{s9.i,0.ri)
lnternirtional Journal of Engineering & Technology IJET-IJENS Vol: I
I
No: 04 t2off 98 Vo or
it
production yield 98 70 asit
composition in the feed but actually unsaturated content is off specswhen
it
production yield achieved 93 7o and95 Vo. Soit
must belower becauseof
timitedby
unsaturated contentin
the quality standard.It's
mean also theactual operating conditions
are
differentwith
the predicted operating conditions. See Table 6 until Table9 below.
The average conditions for the production yields 80 7o,
87 Vo and 93 Va (Table
6)
were the averageof
thesimulation data on the same yields from Table 7 below. The data for 93 7o and 95 Vo production yield didn't take more because
of
the
conditions wereup
setalready.
The average conditions for the production yields 88 7o,
89 Vo
in
Table 10 were the average of the simulationdata on the same yields from Table 11 below.
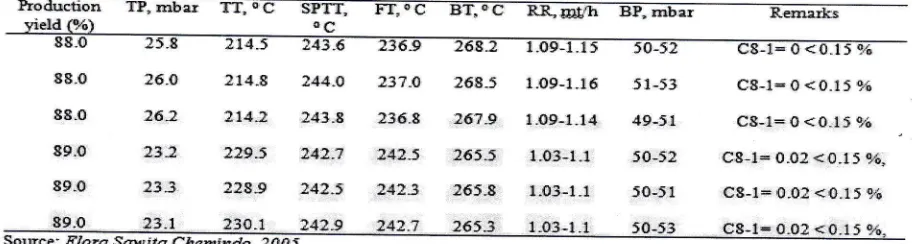
Table 6. The actual operating conditions to produce V 1895 S of l't test
IJroductron TP"
mbar
TT. {C
SPTT.
FT, oC
BT, oC
RR,U:t&
E},mbar
Remarksv'ipld l9,ir\ o fl
80.0
19.3
.I3X.S
l4j{.4 231"4 ?66"6 1.1-1.1 50-J4
Cg-1<015 9',;sl-o
:J.8
ll4-i
t43"S 136.8 :6S.0 1.09-1.1
50_Jf93.0
27.2 :03"8 ?45.1 138-9 t?4.0 0.Sl-0.9
50-51c8-1< 0-15 f.i C8-l> 0"15 o;';
fl8-1> 0"15 o]$
95-0
?5.5 lBl,I ?44.: ?t8.9
I?-1.$
0.7S,0"S3
50"0Source:
ffara {6g;ffi
C*s#i11&" JOUj
Notes: TP
:
top PreB$uE; TT = top temperatlue;SITT:
aide product tlsy temFerature;FT:
f*ed temperstue;ET:
fo1t1g;g1 tenlperature; RR
-
reflus mte ; BP:
bottom pres$ureTable 7. The actual simulation ofthe operating conditions to produce
V
1895 S ofl't
testProduction
TP,mbar
TT,DC
SPTT,
FI,{C
BT,sC
}-R,E*r,h
Bp,mh4rIroduction TP,mbar TT,DC SPTT, FI,{C BT,sC RR,E*iih BP,mhar
Remzrksyietlf"..t) ,..
oc80.0 l9J
t32_S
:44.4 :3?.4
256"6
1.1-11 5g:54
Cts-l< 015 0/6s0-0 80,0
The actual operating conditions on 80.0 7o production yield has a small different than the predicted one (see
Figure 5). The actual suitable prediction of production
yield
was 89.5 Vo (discussion3.2
above).It's
sodifferent also than the prediction was done only base
on the C18 composition in the feed. Feed temperature can't be reached because of the limitation Oil Thermal Heater system capacity
(on
850-900 kgs/trour feed rate), to conduct heatingin
a heater and a reboiler of fractionation column.Reducing of TT and SPTT trends that almost constant are according
to
the simulationof
Roult's Law (not shown here)I5l.
SPTT trendas
is
due
to
Cl8
concentrationis
almost constant around 98 Vo on the same pressure27
mbat. Reducingof TT is
due to increasingCl6
or
reducing C18 compositionin
thec.8-1{O-t5 9d
c.8-1{8-15 9/a
cB-:{
*-15 9t *8-:{0-1"5 *6 c*-14 0.1,J % c8-1> 0-15 $6cg-1> 0"15 BL
tr8-1> 0.15
}t
total reflux of light end on the same pressure, around 25 mbar, as affect
of
V
1895S
production yield incrementor
decreasingof
total reflux into
the column,This
incrementis
causeof
reducing C15 composition inV
1895 S that is added major from light end and also C18 composition increment that is added major from bottom product. This incrementis
also cause of the increment of BT that is effect to the higher C18 evaporation soit
compositionis
higherat
the productionyield
87
Vo (see Figure8 in
the next discussion 3.4). [ncrement of production yield (priority 93 Vo and 95 Vo) is cause of the increment of BT that is so effectto
the higher contentof
Cl8-1,
C20 andunknown components, unsaturated content is off specs
(0.36 and 0.38 Vo). This is also so affect to the lower C18 content in
V
1895 S. That is why both the last 2actual operating conditions in Table 6 can't be applied
19"5
]]J
-0 344,2 13?.6 ?67.0 1-1-1.:
51_5419-1 23t.6 :.{.1.6 ?3?3 266.2 1.1-1.19
49-5487,0
25.8 ?14.5 f,43.6 :36.S 268"0 i.g9_1"1
5S-53s7.0
21"6
X14-7
?44.0 J37.0
?68..+
1.0S-1.11
51_51B?"0
?6.0 214-3 t4t.I 236.6 ?67.6 1.09,1.1
49-5293,0
:?.:
203.$ 145.1 :38"9
2?4-1
0.8.1_0.9
50_5193,0
27.25 ?03"7 :44.9 :38.9
??3.9
0.8$_0"39
50-519t"0
ts.l
:04-5 J44-5 :38"9
]?i
.83
50-51Soruce: SIora SqU*;g SftS4i*f{k 3
I'{ote: IP
-
top prersurs;TT:
tcp t€nlperaturB; SPTT = side pro'duct ua}'trr.$erertue;r-I:
feed iemperaft$e;ET:
Itltgm
terRperature ; RR = r,eflux ret* ; BF = boitorn pre*:,r:reInternational Journal of Engineering & Technology UET-IJENS 11 No: 04
,he next test, but the higher yield should be pointed
.,
shown as discussion 3.2, based on Figure 7. The:
,"iuction yield 88 and 89 Vo are met graphically and89.0 ?3
J6
:1?- ?44-
246_ll& 145
24"7the
operating conditionsRoult's Law, next
hascompared how precise it.
have
to
be
predicted byto
be
tested actually, and-
":le
8. The predicted operating conditions to produce V lg95 S of l,ttestion
TP,mbar
TT, oC
SPTT,BT,{C
RR,,
mbar
Rr{d*lT)
==
..
,
,
oc
ss.0 2336 217-
?44118 ?45 ?47
264 9t (beee onligure
9of
I st tes$ C8-1= 0.14 +6
{
CI"15flo, (base on Frgule
I
?s3-
:.01-1.s.1+ 264cflsttest
\ote:T}=tgpPr*sure;TT:toFtempi[etue;S}T:=*i*uperEtue;BT=
hSffnS
tenper*true
; RR = reflti:r rate ; EP = bottompre:ssire ;;
base on the highest agtual 1.ecu11111Irregflrrel:ble
9" The predicted ofV
I 895 S quality on l,t tesr-i
c:6
18-1
C?S Reflux rate, ductio* yield s.
?
0,76
99.6? 0,06
0"32
0.30* -
1.0I-1.04+
89$
In
specsTable 10. The actual operating conditions to produce
V
1895 S of 2nd testProduction
TP,mbar
TT,oC
SPTT,
m
rield (Va)
25.8
214.5 243.6
236.9
268.2
LOg_Lts C8-1= 0 < 0.15 7p (baseon Table 12)
23.2
229.5 242.7
242.5
265.5
1.03-1.1 CS-l= 0.02 <0.15 Vo,base on Table 12
\ote: TP = top pressure ; TT = toP temperature; SPTT = side product tray temperature ; FT = feed temperature; BT = bottom temperature; RR = reflux rate ; Bp = bottom pressure
Table I L The actual simulation of the operating conditions to produce
v
1 g95 S of 2nd test 5A-5250-52
16-0
t14-S :44-0 :37,O :69-5
1.09_1-ld
51-5316l
I14.2 243.8 :36,8 t6?.9
1.O9-1,14
49-i1
88.0 83.0
89.O
89.0 s9.0
?3-l
iJ .J
C8-1:0
<0.15 9.5C8-1:O<O.15li)
2?9.i Xtr3-7 342"5 265,5
t-O3-1"1 50-it
CB_l:
O"0-?<fi_l5
9.6.:t$.g f4"?-: 24?"3 :65.8
t-03-1,l
50-11
CS-t:
0-01 < 0.15 9.a?3fr-1
t41"92.7 t65-3
1"03-J0-t3
CS-1: O.01 < 0-15 9nSouree; Floro fi4ggiitg "G&emUgdg ,OOJ
Note: ?P
:
toP Pre$u.re; TT:
top ternperehne; $PTT:
side product tra!' ternperature;FI:
fe.ed ternperature; BT:
l:ottomtemuer*ttre: RtrL: Teflux rete :}lp
: hcttomorsr.*.|
S ource: F lo ra S awit aChemindo, 2 00 5
1 I 0r10.+^J737 UEI'-IJENS @ Augusr 201 I LIE^-S
@
l3
50*
Remarks 88.0
89.0
vield
(%,)
o C [image:9.612.52.508.552.674.2]International Journal of Engineering & Technoiogy IJET-IJENS Vol: 1
I
No: 04Table 12. The acrual V 1895 S quality of 2nd test
cls
rate,1.09-1"15
TS
9S"9,7 1,CI3-1"I3'4' Mechanism of
the
changing of
the
main temperature in
the
manufacturing of
V
1g95 SC1
Ifuk
89 CI$0
0,ff1
180
,2
ztaEE
260t-E
250is
:ro t c 2i0T* rrn .e e :10
1 E roi:
ig
rco-
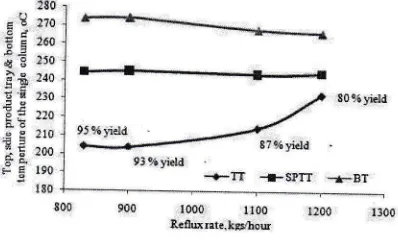
lsoThe changing of the top, side product hay and bottom temperatwe
versus
reflux
rate
in the
single fractionation column (in this research) can be seen-in [image:10.612.118.566.140.192.2]8qs
9*0
100& u!9
,!a00
13oo Reflq ntekE/hfl(Figure 10. Mechanism of the changing of the main temperature to produce
V
lg95 S in a singlefractionation column
4. Conclusions
Unsaturated content of
V
1895 S is higher than0.l5 Vo 11"_aft"
producrion yield ofV
l89ts
is higher than 90 Vo.Based on the actual operating conditions to meet the safest unsaturated content (lower than O.l5 Vo)
in
y
1895 S finally is found graphically on g9.5 Zo V iB95 S
production yield. Practically is found gg _ g9 Vo
V
lg95S production yield. The light end has to be total reflux and unsaturated content of
V
lg95 S is controlled by the adjustmenrof
V
1895 S production yield on rhefound practical or actual operating conditio;s
4.
Suggestions
[image:10.612.117.316.285.409.2]It's
suggested strongly to improve the OTHS capacity tg q:ttlg
higher FT/capacity andV
1895 S production yield. The incrementof
FTwill
be causeof
tfre Rn incrementthat
can
reducethe lower
unsaturated content.Figure
l0
below. The changing temperature above happen whtle total reflux of lisht endin
the single fractionation column as Figure 3 above.Reducing ofreflux rate or increment ofthe production yield
V
1895 S is causeof
the top temperature (TT) decreasing, relatively no changing of the side product ffay temperature (SPTT) and the bottom temperature(BT)
incrementof
the single column,on
the sameheating in the reboiler.
This mechanism
is
so different with the temperature mechanismoffatty
acid blended fractional disiillationas
Figure4
above. Accordingto
the
experience practically, the total reflux of light end is cauje of TT and BT decreasingin
lst column and the increment of productionleld
distillate productin
2nd column is causeof TT
and BT incrementin
2nd column on thesame heating in the reboiler. This is according also to
the reference
Il
l].
The actual operating conditions are found atTp (23.2 _ 25.8 mbar
), TT
(214.5-
229.5 "C), SPTT (242.7 _ 243.6"C),
FT
(236.9_
242.7.C), BT
(265.5 _ 268.2"C), RR (1.03-
l.I5
mt/hou0,Bp
(
'50_
52 mbar) and feed rate 850-
900 kg/hour. It is found baseon the predicted operating conditions
at
W
(23.36 mbar ), T TQfi -
218 "C), SPTT (244 -245 .C),FI
(246
-
247"C),BT
(263 -264C), RR(
l.0l
_
1.09 mVhour),BP
(50
mbar) and feed rare g50_
900 kg/hour.Feed temperature
is
actually found lower than the predicted one becauseof
the limitationof
the oil thermal heater system (OTHS) capacity.It's
suggested stronglyto
improve vacuum system capacity soit
can be achieved 3 -5 mbar Tp in of the single column to improve the total performance of the single columnfor
this
special product. The highest vacuum pressure 2 mbar is also stated by [9] to get the better resultsof
fractionation/distillation products.110i104-37-]7 III:1,IJINS @i Augusr 20lt IfjNS
@
International Journal of Engineering & Technology IJET-IJENS Vo1: I
I
No: 04 15-
!";I.rences
:::-:--:.::.
E.,
1985.,"Oii
Seeds Oils and Fats",-
l'd
Edition"B.E
Oil
Publishing House,-. ::-.:r
R and McPherson, W", 1979., "Fractional- -
.:-on", JAOCS,Vol.
56:743A-
745A :-:,::--.nn,
R."
Demmering,
G.,
Kreutzer,-
.-:3man,M.,
Plachinka,J.,
Steihrner, U., -:-
FattyAcid",
Henkel KGaA, Dusseldorf,;:=:-.
Republic of German.*
l
- .-. F.,
2006.,
"Official
Method
And;.-,rmended
Practicesof
the
American Oi1I-::--srs
Society",5thEdition,
2nd
Printing,-
-,-S Press, USA.::-
..-
8.L,, 1989., "Applied Process Design for-
-.
::;a1 and Petrochemical Plants", Volume -2,-'-
::riion,
Gulf Publishing Company, Houston.:. :
-nd
Hahn. GmbH,
1998.,"Fatty
Acid:-.:-onation",
Flora Sawita Chemindo, Medan, : -:-:sia-
-
-r"
Sawita
Chemindo,2005.,
"Production:
-:::rg
Control", Medan, Indonesia;-:.
E.. 1979., "Interrelationshipsin
Fatty Acid -*-,::ssing", JAOCS, Vo1.56; 7 45A-746A":r:irnann, A.
E.
S.,
1990.,::=:-:onation/Distillation
:
Improvement in-.- .:).
Efficiency, Energy and Environment-.::;ts",
Proceeding
World
Conference on.
..
Chemicalinto
21" Century, Kala Lumpur f.:.::3:'i. S., 1985., "Oils and Fatsof
Food", UI:::...
Jakarta.I
",::aphies
Muhammad Yuuf. Ritonga was bom in
Sipirok, Tapsel, North Sumatera province, lndonesia, on August 19,1962. He studied and graduate in North Sumatem University since 51 degree till obtaining his doctor.
He had being a lecture since 1989 in Chemical
Engineering Deptrtment, North Sumatera
University. He has 20 years of special field
;,ri::-.r: oleo chemical industry since l989in troubleshmting, u: : ;- : :i::i. start up, commissioning; oils and fats pretrcatment
;iL- :-:-:. glycerin purification, fatty acid dry fractionation,
' :-;::;:::. distillation md fractionation. His experiences related
"" : :l {ibhawana Utama (now called PT. Eco Green as a Fatty
[11] Muhammad.
Y. R.,
1996., "Practical Distillationtr',
Study
Progam
of
Chemical Engineer, Facultyof
Engineering, Universityof
Sumatera Utara, Medan, Indonesia.ll2l
Muhammad.Y.
R., 2009., 'Fatty Acidsof
OleoChemical Industry",
Proceeding
National Conference of Chemical Engineer, University of Sumatera Utara, Medan, Indonesia.t13l
Peter,W. Faesler.,
Karl
Kolmezt.,
Wang SengKek., 2004., "Revamp Strategiesfor
FattyAcid
Distillation
SectionIn
Oleo-Chemical Plants", Sulzer Chemtech, Singapore.[14]
SulzerChemtec. 2006., "Separation Technology for the Chemical Process Industry- Oleo chemicalStep
Into
a
Growing
Market
with
Sulzer Chemtech : 12".ll5l
Thomas, H.W. 1985., "Bailey's Industrial Oil andFat Product", Volume
3.
JhonWiley
&
Sons,New Yorlc
[16]
Unichema lntemational. 1998., "Fatty Acid DataBook"
2ndEdition,
Unichema, Republic of German.[17] Van Winkle. M., 1967., "Distillation", Mc. Graw -Hill Book Company, New
York
[8]
Yusuf Ritonga. M.,2010.,
"Optimization of the Providingof
StearicAcid
Basedon
Refined Bleached DeodorizedPalm
Stearin (RBDPS)Which
is
Stable Accordingto
the
Quality Standard",Doctor
Program,FMIPA,
USU, Medan, Indonesia.Alcohol and Glycerin Plant) md PT. Flora Sawita Chemido (as Fatty
Acid and Glycerin Plant), He had been done many resetrches relate
to oleo chemical industry ; fatty acid and glycerin products
diversification and modification and design and modification of
operating condition of the specific process in oleo chemical industry, on the plant scale. He also released many mmuscripts on the various science and technical journals relate to fatty acid and glycerin in the terrns splitting of various natural oils md fats, hydrogenation, distillation, fractionation and dry ftactionation of various fatty acid base on various natuml oils and fats. He is the author of book
Distillri Praktis. He planned to release two books (had being edited
till now on) ;"Practical Distillation Prirciples" and "Hydrolysis
Oik and Fats in Oleo Chemical Industy".
/
LEMBAR
HASIL PENILAIAN SEJAWAT
SEBIDANG
ATAU
PEER
REWEW
KARYA
ILMIAH
:
JURNAL
ILMIAH
Judul Jurnal
Ilmiah
(Artikel)
:
Providing
Vegetable Stearic
Acid
SuperV
1895 SFrom Hydrogenated
Of
Crude Vegetable Stearic
Acid
HCV
1895 SWith
A
Single
Fractional Distillation Column
:
Dr.Ir.
Muhammad Yusuf. Ritonga,
MT
:
a.Nama
Jurnal
:International
Journal
of
Engineering
&
Science
IJET/IJENS
Penulis Jurnal
Ilmiah
Identitas Jurnal
Ilmiah
(beri
/pada
kategori yang tepat)
Hasil
Penilaian Peer Review
:b.
Nomor/Volume
c.
Edisi (bulanltahun)
d. Penerbit
e.
Jumlah halaman
04111
Agustus
201I
1JENS
Publisher
-Pakistan
167KategariPublikasi
Jumal
Ilmiah
, d
Jurnal
llmiah
Internasional
f]
Jurnal
Ilmiah Nasional Terakreditasi
I
Jurnal
Ilmiah Nasional Tidak
Terakreditasi
Komponen
Yang
Dinilai
Nilai
Maksimal
Jurnal llmiah
Nilai
Akhir
Yang
Diperoleh
Internasig/al
TE
Nasional
Terakreditasi
n
Nasional
Tidak
Terakreditasi
tl
a.
Kelenskapafl
unsur
isi buku (10%)
sF
ge
b.
Ruang
lingkup
dan kedalaman
pembahasan{30%)
70
Jr,
/
c. Kecukupan
dankemutahiran
data/informasi
danmetodolosi (30%)
s&
2t,
?
d.Kelengkapan unsur
dankualitas
penerbit
(30%)
te
Je,?
Total
=
(100o/o)g/,9
Medan,
...(A.
:....!.*.:...*.Qf.*...
Nama
: Prof.
Dr. Harry
Agusnar.
M.
Sc.Phil
NIP
:
19530817
i98303
1002
tJnit
kerja
:
FMIPA
-
USU
i..i i1
Reviewer, _-
./
-A
g
Judul
Jumal Ilmiah
(Artikel)
Penulis Jurnal
Ilmiah
Identitas Jurnal
Ilmiah
Kategori Publikasi
Jurnal
Ilmiah
(beri
/pada
kategori yang
tepat)
Hasil
Penilaian Peer Review
:LEMBAR
IIASIL
PEMLATAN SEJAWAT SEBIDANG
ATAU PEER
REYIEW
KARYA
ILMIAH
:
JURNALILMIAH
:
Providing
Vegetable Stearic
Acid
SuperV
1895 SFrom Hydrogenated
of
Crude Vegetable Stearic
Acid
HCV
1895 SV/ith
A
Single
Fractional Distillation
Colurnn
.:
Dr.
h.
Muhammad
Yusuf. Ritonga,
MT
:
a.Nama
Jurnal
b.
Nomor/Volume
c.
Edisi (bulariitahun)
d.Penerbit
e.
Jumlah halaman
:
International
Journal
of Engineering
&.zc ScienceIJET/IIENS
04/11
Agustus
20i
i
IJENS
Publisher
-Pakistan
16'7
, lV
Jumal Ilmiah
Internasional
I--l
Jumal
Ilmiah Nasional Terakreditasi
t]
Jurnal
IlmiahNasional Tidak Terakreditasi
Medan,..4
Reviewer
2Nama
NIP
,r,,%,s,auan.u.
si
:
19600618198703 1002Unit
kerja
:FMIPA
-
Guru Besar
Biokirnia
-Universitas
Negeri Medan
Nilai
Maksimal
Jurnal Ilmiah
Internasioual
d
Nasional
Tidak
Terakreditasi
E
unsur
isi buku (10%
b.
Ruang
lingkup
dankedalaman
pembahasanc. Kecukupan
dankemutahiran
data/informasi
dand.Kelengkapan unsur
dankualitas penerbit
(30%)
:
l